
A reduction approach to prepare ZnSe nanocrystallites
JIANG Hai-qing(姜海青)1, 2, CHE Jun(车 俊)2, LI Zhi-min(李智敏)1, YAO Xi(姚 熹)2
1. School of Technical Physics, Xidian University, Xi’an 710071, China;
2. Key Laboratory of Electronic Material Research, Ministry of Education,
Xi’an Jiaotong University, Xi’an 710049, China
Received 10 April 2006; accepted 25 April 2006
Abstract: ZnSe nanocrystallites powders were successfully prepared by reduction method with H2SeO4 or H2SeO3 as selenium source and Zn(Ac)2・H2O as zinc source. The reaction mechanism was also discussed through XRD and DTA/TG analysis. In the process, ZnSeO4・H2O or ZnSeO3・H2O crystallites precipitated in organic solvent was prepared at first. Then the crystallites were reduced with carbon monoxide atmosphere. The XRD analysis reveals that the structure of ZnSe phase is sphalerite (cubic ZnS). The XPS results show that the atomic ratio of Se to Zn is 1?1 approximately. The TEM shows that the size of ZnSe crystallite is 30-50 nm. This method of synthesis of ZnSe nanocrystallites has the potential application in the preparation of optical composite thin films from sol-gel process.
Key words: ZnSe; nanocrystallites; reduction method; carbon monoxide
1 Introduction
The wide band gap Ⅱ-Ⅵ semiconductor materials ZnSe which have the unique chemical and physical properties have been extensively used in optical material, sun energy material and sensor material, especially in blue-green diode lasers. In recent years, much attentions [1-10] were paid on the preparation methods, performances and applications of ZnSe nanocrystallites and thin solid films, and a lot of important accomplishments have been obtained. Nowadays, researchers were focused on the improvement of properties and the preparation technology of thin films and nanocrystallites.
WANG et al [1] synthesized ZnSe using ZnCl2, selenium powder and aluminium foil as the starting materials under 1 mol/L alkali condition. SU and CHOK [2] synthesized ZnSe in aqueous conditions using ZnCl2, selenoure as the zinc and selenium source, respectively. NODA et al [4] prepared ZnSe films by MOCVD system on GaAs substrate, using diethylzinc(DEZn) and diethylselenide(DESe) as source materials with the carrier gas NF3. LOKHANDE et al [4] prepared ZnSe films through the chemical bath deposition method(CBD) using selenoure as selenium source and ZnSO4 as zinc source. Selenoure will releases Se2- ions in an alkaline medium which react with zinc ions. ZnSe will deposit on the surface of substrates. Then the films were washed with deionized water and dried under N2 atmosphere. LI and NOGAMI [7] prepared ZnSe/SiO2 thin films. They prepared ZnSe/SiO2 thin composite films using H2SeO4 and ZnCl2 as source materials by sol-gel process adding ZnSeO4・H2O into TEOS sol-gel system under nitrogen and hydrogen reduction atmosphere. But our research work revealed that selenium element would escape from the sample heavily because of the reaction between selenium and hydrogen at 215 ℃, which is the reason of emergence of ZnO phase and H2Se in their samples. In this paper, we successfully synthesized ZnSe nanocrystallites using relatively cheap H2SeO4 or H2SeO3 as selenium source and Zn(Ac)2・2H2O as zinc source in the carbon monoxide atmosphere at 500 ℃. The synthesis process of ZnSe can solve the problems of emergence of ZnO phase and it is very convenient to use in the process of preparing the composite thin films.
2 Experimental
The white ZnSeO4・H2O or ZnSeO3・H2O crystallites precipitated in organic solvent were prepared firstly by mixing 0.1 mol/L ethanol solution of H2SeO4 or H2SeO3 with the ethanol solution of 0.1 mol/L Zn(Ac)2・H2O at the atomic ratio of Se to Zn 1?1. For promoting the solubility of Zn(Ac)2・H2O in ethanol solvent, 0.2 mL NH3・H2O was added in the ethanol solution. The ZnSeO4・H2O or ZnSeO3・H2O crystallites can be obtained after stirring the mixture for 2 h and dried in revolving evaporator at low pressure at 60 ℃ for 3 h. Finally, ZnSeO4・H2O or ZnSeO3・H2O crystallites in quartz crucible were reduced under carbon monoxide atmosphere at 500 ℃. To ensure the partial pressure of selenium element, small amounts of metal selenium were also sealed in quartz tube stove. In the reduction course, oxygen must be removed as far as possible at low temperature. That is the important step to ensure the samples without the emergence of ZnO phase. For preparing the samples of ZnSeO4・H2O or ZnSeO3・H2O for DTA-TGA analysis, 0.1 mol/L Zn(Ac)2・H2O water solution and 0.1 mol/L H2SeO4 or H2SeO3 water solution were prepared firstly, then they were mixed without adding NH3・H2O so as to decrease the influence of NH3・H2O.
The phase structures of samples were characterized by XRD (Rigaku D/MAX-2400, Cu Kα). DTA-TG (TA- 2000) analysis was used to record the thermal effect in the process of heat-treatment. The atomic ratio of Se/Zn was analyzed by XPS(VG ESCALAB MKⅡ,MgKα). The size of ZnSe crystallite was investigated by TEM(JEM-200cx).
3 Results and discussion
3.1 DTA-TG analysis
3.1.1 DTA-TG analysis of ZnSeO4・H2O
The DTA-TG analysis of ZnSeO4・H2O prepared by the above-mentioned method was performed from room temperature to 700 ℃ with the heating rate of 10 ℃/min under air atmosphere. Although that condition is not consistent with that of ZnSe prepared, it can give us useful instruction in the preparation of ZnSe. The results of DTA-TG are shown in Fig.1. The curves indicate that there are three endothermic peaks. An endothermic peak at about 103 ℃ was associated with the losing of physical adsorption water. The endothermic peak at about 216 ℃ was associated with the losing of chemical adsorped water. The third endothermic peak at about 675 ℃ was associated with the decomposing reaction of ZnSeO4. The TG curve reveals that the mass ratio of ZnO/SeO3 is 39.3% which is associated with the mass

Fig.1 DTA-TG curves of ZnSeO4・H2O
loss from 76.61% to 29.56% at 674.51 ℃.
3.1.2 DTA-TG analysis of ZnSeO3・H2O
The DTA-TG analysis of ZnSeO3・H2O prepared by above-mentioned method was performed from room temperature to 600 ℃ under the condition same as the sample of ZnSeO4・H2O. The results of DTA-TG are shown in Fig.2. The curves indicate that ZnSeO4・H2O has an endothermic peak at 213.5 ℃ and an exothermic peak at 435.7 ℃.The endothermic peak at 213.5 ℃ was associated with the losing of chemical adsorbed water. The exothermic peak at 435.7 ℃ was associated with the creation of new phase of ZnSeO3.

Fig.2 DTA-TG curves of ZnSeO3・H2O
3.2 XRD analysis
3.2.1 XRD analysis of ZnSeO4・H2O
The phase structures of ZnSeO4・H2O with different heat-treatment temperatures are shown in Fig.3. The ZnSeO4・H2O was heat-treated at 100, 200, 300, 320 and 410 ℃ for 1 h under air condition, respectively. The XRD results indicate that ZnSeO4・H2O was compara- tively stable under 200 ℃, which is consistent with the results of DTA-TG curves. With the increasing of temperature, ZnSeO4・H2O becomes amorphous at 300 ℃, which means a new phase formed in this period. ZnSeO4 will form at 410 ℃. The XRD pattern of the sample (not shown in Fig.3) heat-treated at 650 ℃ indicated the emergence of ZnO wurtzite phase. Even though there are some differences between that under air condition and that under carbon monoxide atmosphere, the results could give us some valuable hints. The temperature of reaction between ZnSeO4 and carbon monoxide is much lower than the temperature of decomposing reaction of ZnSeO4. The reaction equation between ZnSeO4 and carbon monoxide also is
ZnSeO4+4CO == ZnSe+4CO2
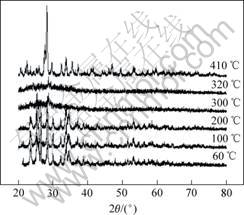
Fig.3 XRD patterns of ZnSeO4・H2O heat-treated at different temperatures for 1 h
The XRD pattern of ZnSe nanocrystallites synthesized at 500 ℃ under carbon monoxide atmosphere using ZnSeO4・H2O is shown in Fig.4. The XRD pattern indicates that the phase structure of ZnSe is sphalerite (cubic ZnS) without the appearance of ZnO phase.
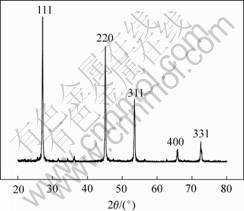
Fig.4 XRD pattern of ZnSe using ZnSeO4・H2O crystallites synthesized at 500 ℃ under carbon monoxide atmosphere
3.2.2 XRD analysis of ZnSeO3・H2O
The structure change of ZnSeO3・H2O with different heat-treatment temperatures is shown in Fig.5. The ZnSeO3・H2O was heat-treated at 100, 213, 300 and 438 ℃ for 1 h under air condition, respectively. The XRD results indicate that ZnSeO3・H2O is stable under 200 ℃, which is consistent with the results of DTA-TG curves. With the increasing of temperature, ZnSeO3・H2O will transform to ZnSeO3 above 300 ℃. The XRD pattern of ZnSe nanocrystallites synthesized at 500 ℃ under carbon monoxide atmosphere using ZnSeO3・H2O is shown in Fig.6. The XRD pattern indicates that the structure ZnSe is sphalerite (cubic ZnS) without the existence of ZnO phase.

Fig.5 XRD patterns of ZnSeO3・H2O heat-treated at different temperatures for 1 h

Fig.6 XRD pattern of ZnSe using ZnSeO3・H2O crystallites synthesized at 500 ℃ under carbon monoxide atmosphere
3.3 XPS analysis of ZnSe
The XPS results of ZnSe prepared from ZnSeO4・H2O and ZnSeO3・H2O are shown in Fig.7. The XPS results also indicate that the atomic ratio of Se to Zn of the two samples is 1?1 approximately.
3.4 TEM analysis of ZnSe nanocrystallites
The TEM images of ZnSe nanocrystallites are shown in Fig.8. It can be seen that the size of ZnSe
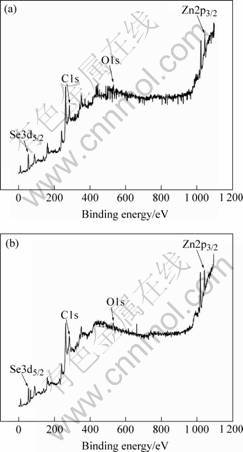
Fig.7 XPS spectra of ZnSe synthesized from ZnSeO4・H2O(a) and ZnSeO3・H2O(b)

Fig.8 TEM images of ZnSe synthesized from ZnSeO4・H2O(a) and ZnSeO3・H2O(b)
nanocrystallites is 30-50 nm.
4 Conclusions
ZnSe nanocrystallites without the emergence of ZnO phase has been synthesized by the reduction method with H2SeO4 or H2SeO3 as selenium source and Zn(Ac)2・H2O as zinc source under carbon monoxide atmosphere. The size of ZnSe crystallite is 30-50 nm. This method has potential applications for preparing ZnSe/SiO2 composite thin films.
References
[1] WANG C, ZHANG W X, QIAN X F, ZHANG X M, XIE Y, QIAN Y T. Aqueous approach to ZnSe and CdSe semiconductor nanocrystals[J]. Materials Chemistry and Physics, 1999, 60(1): 99-102.
[2] SU B, CHOY K L. Electrostatic assisted aerosol jet deposition of CdS, CdSe and ZnS thin films[J]. Thin Solid Films, 2000, 361: 102-106.
[3] NODA Y, ISHIKAWA T, YAMABE M, HARA Y. Growth of ZnSe thin films by metalorganic chemical vapor deposition using nitrogen trifluoride[J]. Applied Surface Science, 1997, 113/114: 28-32.
[4] LOKHANDE C D, PATIL P S, ENNAOUI A, TRIBUTSCH H. Chemical bath ZnSe thin films: Deposition and characterization[J]. Applied Surface Science, 1998,123/124: 294-297.
[5] MAEHASHI K, YASUI N, NOMA T, MURASE Y, NAKASHIMA H. Structural and optical properties of CdSe/ZnSe self-organized quantum dots[J]. Journal of Crystal Growth, 2001, 227-228: 1116-1120.
[6] SHUBINA T V, TOROPOV A A, SOROKIN S V, IVANOV S V, KOPEV P S, POZINA G R BERQMAN J P, MONEMAR B. Optical studies of carrier transport phenomena in CdSe/ZnSe fractional monolayer superlattices[J]. Thin Solid Films, 1998, 336(1-2): 377-380.
[7] LI G M, NOGAMI M, Preparation and optical properties of sol-gel derived ZnSe crystallites doped in glass films[J]. Journal of Applied Physics, 1994, 75(8): 4276-4278.
[8] BOLOGOROKHOV A I, BELOGOROHOVA L I, GAVRIOV S, Investigation of properties of porous silicon embedded with ZnSe and CdSe[J]. Journal of Crystal Gorowth, 1999, 197: 702-706.
[9] LEE H Y, KIM J W, CHUNG H B, Thermally and optically induced nanocrystallites in amorphous ZnSe thin films[J]. Japanese Journal of Applied Physics, Part 1: Regular Papers and Short Notes and Review Papers, 2003, 42(1): 202-207.
[10] JORKAALA H, STENNENEN H. Nonlinear optical properties of ZnSe nanocrystals incorporated within polyvinyl alcohol photopolymer matrices[J]. Journal of Optics A: Pure and Applied Optics, 2002, 4(3): 366-369.
(Edited by YUAN Sai-qian)
Foundation item: Project(2002CB613305) supported by the National Basic Research Program of China; Project supported by International Cooperation Research Project of China-Israel of the Ministry of Education of China
Corresponding author: JIANG Hai-qing; Tel:+86-29-82668679; Fax:+86-29-82668794; E-mail: hqjiang@mail.xidian.edu.cn