J. Cent. South Univ. (2012) 19: 1927-1931
DOI: 10.1007/s11771-012-1227-7
Pulse modulated microwave and infrared thermography for superficial hyperthermia
MA Guo-jun(马国军)1, 2, JIANG Guo-tai(江国泰)1, CHEN Zhi-hong(陈志宏)1
1. School of Life Science and Technology, Tongji University, Shanghai 200092, China;
2. School of Electronics and Information, Jiangsu University of Science and Technology, Zhenjiang 212003, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2012
Abstract: A 3D temperature field distribution of biological tissue for superficial hyperthermia using a pulse modulated microwave (PMMW) was presented. A 3D sliced homogeneous phantom was radiated by the PMMW and an infrared thermal imager was applied to image temperature distribution throughout the phantom. The period of the PMMW is 3 s and the output power is 35 W. The temperature rises by at least 3 ℃ in the phantom when the duty cycle varies from 1/3, 1/2, 2/3 to 1 (denoted by scenarios 1-4). Both the accumulative temperature-volume histogram and the relative depth-area ratio histogram show that the maximum temperature rise (MTR) is 6.6 and 8 ℃ in scenarios 2 and 3, and they are superior to scenarios 1 and 4. Furthermore, the PMMW can control temperature field distribution of biological tissue. It provides both preliminary basis for thermal volume control and new technology for temperature control and monitor in superficial hyperthermia.
Key words: 3D temperature field distribution; pulse modulated microwave (PMMW); infrared thermography; superficial hyperthermia
1 Introduction
Superficial hyperthermia is an application of heat to treat cancer diseases at surfaces of human body. Microwave hyperthermia is a minimally invasive technique extensively used for hyperthermia of liver tumors [1]. The microwave hyperthermia treatment of superficial tumors [2-3], such as breast cancer, is electively used in combination with chemotherapy and radiotherapy [4-5].
Previous studies have shown positive effect of hyperthermia used in tumor treatment [6-8]. However, the effectiveness of the hyperthermic treatment is strongly related to local temperature distribution of the tumor tissue in target volume [9]. The microwave output power is usually controlled continuously and too high temperature in biological tissue [10] will damage the normal tissue. Therefore, it is turned off in case that the maximum temperature in the healthy tissues is higher than 45 ℃, or patient complaints.
In addition, the effective microwave hyperthermia requires efficient temperature distribution [11] to destroy tumors and limit damage to normal tissue [12] or minimize the patient’s pain and discomfort. All these require microwave radiation with power distribution as homogeneous as possible over the target volume. A proper implementation of energy transport is essential for the temperature distribution in the tissue. To develop a process for hyperthermia treatment model based on microwave radiation, it is required to correctly describe the 3D temperature field distribution. The temperatures during the hyperthermia treatment should be measured by thermal infrared imager [13], which is used for the surface temperature monitor during the entire treatment.
The characteristic of the pulse modulated microwave (PMMW) and its heating effect compared with continuous microwave output power by using infrared thermography are demonstrated in this work. First, the 3D sliced phantom was radiated by PMMW. Second, infrared thermography was used to monitor the temperature distribution of 3D phantom and the heating effect of PMMW was analyzed with accumulated temperature-volume histogram and depth-area ratio histogram. Finally, a discussion of these results was presented.
2 Bio-heat transfer model
Electromagnetic energy radiated from external microwave system increases temperature in the target area to the therapeutic temperature level of 43-45 ℃. The achievement of such conditions depends not only on the power delivered by microwave, but also on physical mechanisms of biological tissue. The heat transfer problem inside the biological tissue can be described by using the following model known as the PENNES’ bio-heat transfer equation [14]:
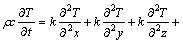
 (1)
(1)
where c, ρ and k represent heat capacity, tissue density and thermal conductivity, respectively; cb and wb are the specific heat capacity and perfusion of blood; Ta is arterial temperature; T is local temperature in biological tissue; Qm is the metabolic heat source term; Specific absorption rate (SAR), R, which represents the absorbed energy from electromagnetic field stimulation [15], is used as a source for thermal calculations.
Investigating the 3D homogeneous phantom instead of living tissues makes it possible to get rid of blood perfusion. Considering SAR generated by microwave radiation, the power delivered by microwave is the only contribution to the local energy balance and all the other terms in Eq. (1) being practically negligible. These assumptions simplify the implementation of the controller and analysis of heating effects, but preserve the essential features of the practical problem. The experimental system performance of hyperthermia treatment is evaluated using a 3D phantom system. Such model provides detailed information about the 3D temperature field distribution in the treated region which is monitored by thermal infrared imager.
The absorbed energy from the electromagnetic field was used as a source for thermal calculation. In the absence of blood perfusion and metabolic heat production, for instance, in a homogeneous phantom when the PMMW is applied, Eq. (1) is simplified as
 (2)
(2)
With the above assumptions, the power control term well represents the energy deposited by the PMMW. The control term c(t) is introduced here in order to change the pattern of microwave output power:
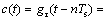
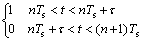 (3)
(3)
where gτ is the gate function; τ and Ts are the width and period of modulating pulse, respectively, and n is an integer number. The duty cycle of a pulse, which is defined as q=τ/Ts, is introduced to control the output power of microwave.
The temperature field distribution problem is characterized by microwave power that is delivered to the biological tissue. The PMMW is an attractive approach to control temperature distribution for tumor tissue, which radiates biological tissue for a period of time and switches off automatically by pulse.
In experiment, the performance of the PMMW is tested on simulated 3D phantom and the temperature distribution is monitored by the thermal infrared imager. The obtained infrared thermography sequences of the 3D sliced phantom are analyzed when the accumulated temperature-volume histogram and sliced depth-area ratio histogram are used.
3 Experiment systems
The PMMW superficial hyperthermia system is shown in Fig. 1. The temperature of the 3D phantom and the environment was 21 ℃. The size of the 3D phantom was 150 mm×150 mm×32 mm and the region of interesting (ROI) radiated by microwave antenna in horizontal direction was 100 mm×100 mm. The system consists of two parts: the microwave generator and the temperature measurement, which includes medical microwave apparatus (2 450 MHz), thermal infrared imager (NEC TH5108ME) and drive-control device. Cartesian coordinate system was used and microwave radiation antenna in spatial location relative to the phantom was assumed to be fixed at (0, 0, 2.4 cm) during the experiment. The thermal infrared imager was located at (0, 0, 35 cm). The 3D phantom was as homogeneous as possible. It had total 16 slices or equally 32 mm in thickness.
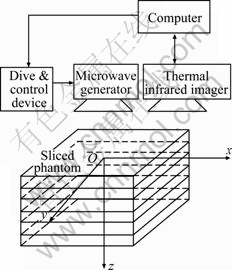
Fig. 1 Scheme diagram of PMMW tumor hyperthermia system
In the experiment, the medical microwave apparatus was used to radiate 3D sliced phantom for 8 min. It was moved away after the heating cycle had elapsed, and then the medical thermal infrared imager was used to image each slice of the 3D phantom to obtain infrared thermography sequences.
During the experiment, the PMMW generator output system was controlled by computer via the drive-control device. When the parameters of the PMMW changes, such as the duty cycle, the heating effect of the PMMW was analyzed including accumulated temperature-volume histogram and sliced depth-area ratio histogram. The duty cycle of the PMMW was 1/3, 1/2, 2/3 and 1, respectively, when four scenarios were associated with microwave output power of 35 W and the pulse period was 3 s.
4 Results and discussion
4.1 Maximum temperature rise (MTR) and accumulative temperature-volume histogram
In each scenario, there are 16 slices infrared thermographs of the 3D phantom. The infrared thermography of slice 7 (at the depth z=1.4 cm) in scenarios 1-4 is compared in Fig. 2.
To evaluate the heating effect of the PMMW, a maximum temperature rise (ΔT) is defined as ΔT=Tmax-Tinit, which reflects the maximum temperature compared with initial temperature of biological tissue. The optimal temperature to kill cancer cell in hyperthermia is 43-45 ℃ (compared with 37 ℃ of human body). The MTR in the 3D sliced phantom is 3, 6.6, 8 and 12.6 ℃ in scenarios 1-4, respectively (see Table 1). The MTR is 3 ℃ in scenario 1. It doesn’t meet the requirements of temperature rise in tumor hyperthermia. In scenario 4, the MTR is 12.6 ℃, which exceeds the temperature range of killing tumor cell and is very likely to do damage to normal tissue or bring patients’ complaints. The MTRs are 6.6 and 8 ℃ in scenario 2 and 3, respectively, which are proper pulse duty cycles for hyperthermia.
The accumulative temperature-volume histogram illustrates the temperature distribution of the ROI (Fig. 3). The distributions of the accumulative temperature- volume histogram are similar in scenarios 1-4, and there are 9.6 ℃ temperature difference between scenarios 1 and 4.
As far as the effective temperature range (6-8 ℃) is considered in tumor hyperthermia, the volume percentage of 3D phantom is 0, 2.15%, 5.532%, 8.326% in scenarios 1-4, respectively. But there is 4.091% of the volume in scenario 4 exceeding allowed maximum temperature rise of 8 ℃ (Fig. 3).
4.2 Relative depth-area ratio histogram
The pixels having temperature rise of each thermography slice are counted. The counted pixels are described as relative depth-area ratio histogram when they are compared to all the pixels of each slice.
The sliced depth-area ratio histogram described by relative area (pixel number) is higher than the setting temperature rise in each slice of 3D phantom. Increasing the duty cycle of pulse may increase the depth-area ratio. At the same time, the temperature rise is significant in the former 7 slices, but the temperature depth-area ratio decreases after the slice 7 (Fig. 4).
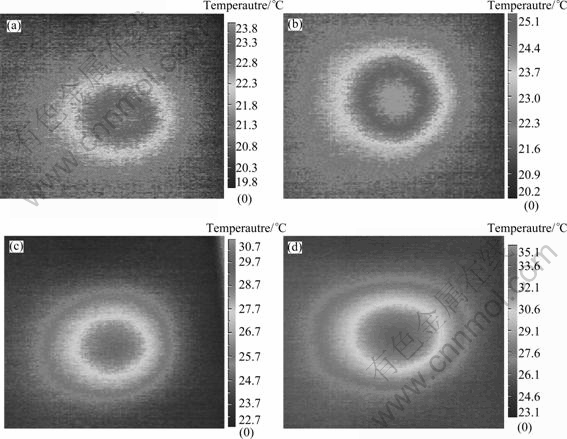
Fig. 2 7th infrared thermography with different duty cycles: (a) q=1/3; (b) q=1/2; (c) q=2/3; (d) q=1
Table 1 Maximum temperature rise of four scenarios
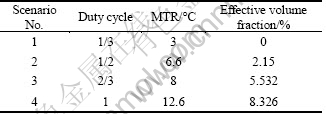
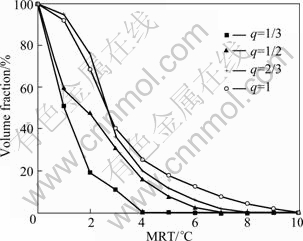
Fig. 3 Variation of volume fraction with MTR at different duty cycles
The scenario 4 has local high temperature in certain depth, for example, the MTR is 6 ℃ at the slice 7 (z=1.4 cm). Each duty cycle has corresponding relative area ratio (Table 2). The temperature rise of scenarios 2-4 is higher than 6 ℃. In scenario 3, the relative area ratio is twice than that of scenario 2 at temperature rise of 6 ℃.
4.3 Discussion
A novel approach to control the 3D temperature field distribution is proposed in this work. The experimental test of the developed PMMW radiation for the 3D phantom shows that it is suitable for superficial hyperthermia. To effectively describe the temperature distribution, the temperature of the phantom was measured by the thermal infrared imager. The comparison was done at temperature increment of 6-8 ℃ by changing duty cycle at certain output power and period of the PMMW.
There is temperature rise in the 3D phantom. The MTR is related to duty cycle of the modulating pulse. The larger duty cycle causes greater temperature rise in phantom, for example, the duty cycle at 2/3 has the temperature rise of 9.6 ℃. But the highest temperature rise is less than the temperature (12.6 ℃) of continuous microwave output and it avoids too high temperature in ROI.
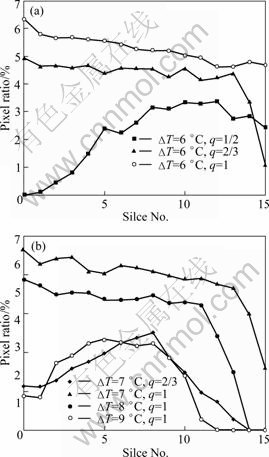
Fig. 4 Area ratio at each slice of different temperature difference: (a) ΔT=6 ℃; (b) ΔT=7, 8 and 9 ℃
Table 2 Depth-area ratios at different temperature rises of four scenarios (%)

However, the size and shape of the target volume are extensively dependent on the temperature profile setup in tissue. For this reason, a model that is implemented incorporating actual model. This is in accordance with reports of treatment planning optimization for superficial hyperthermia [16]. The method presented here will optimize the temperature distribution for microwave hyperthermia.
The comparison shows that all the four scenarios with different parameters of the PMMW have the similar 3D temperature distribution (Fig. 4). The MTR between scenarios 1 and 4 is up to 9.6 ℃. The positions of the maximum temperature points for all investigated scenarios are at slices 6-8 under the phantom surface [17]. A maximum temperature difference of 6 ℃ is found among the models with different duty cycles (scenarios 2 and 4) inside the phantom.
The dimensions of 3D phantom are large enough to avoid the influence of boundary calculations. The size of the effective region was 100 mm×100 mm×32 mm, to minimize the error of the experiment. All boundaries inside the model were set at 21 ℃. And only the temperature rise was considered in the temperature field distribution in all four scenarios. It is concluded that boundary has no influence on the temperature profile inside the ROI.
However, the effort toward direct control of the temperature in superficial hyperthermia [18] requires relative safety temperature distribution, which is a challenging problem because high temperature will damage normal tissue by continuous microwave [19]. This restricts the applicability with relatively larger temperature variability. The PMMW developed in this work overcomes these limitations and makes it applicable to a broad range of hyperthermia.
5 Conclusions
1) The conventional problem of microwave superficial hyperthermia treatments is formulated as a temperature field distribution problem by means of controlling temperature in the sliced 3D phantom. Therefore, an alternative approach of directly controlling the temperature by using the PMMW is applied. The method of the infrared thermal imaging is employed to monitor the temperature of each slice of 3D phantom.
2) The measurements allow an accurate assessment of the heating effect of the PMMW, in which the accumulated temperature-volume histogram and the depth-area ratio histogram are discussed. Duty cycle of modulated pulse affects the temperature distribution. Higher duty cycle of the PMMW radiated to biological tissue has higher temperature rise. But, the MTR is less than that of continuous microwave power. So, it will avoid the damage to normal tissue, and can form effective temperature rise and create appropriate temperature distribution in biological tissue.
3) The good performance of the PMMW procedure based on the infrared thermography and the 3D temperature field distribution suggests that the presented protocol is suitable for the superficial hyperthermia.
References
[1] BANSAL R. Battling cancer: The latest on microwave hyperthermia [J]. IEEE Microwave Magazine, 2005, 6(3): 32-34.
[2] ZASTROW E, HAGNESS S C, van VEEN B D. 3D computational study of non-invasive patient-specific microwave hyperthermia treatment of breast cancer [J]. Physics in Medicine and Biology, 2010, 55(13): 3611-3629.
[3] ZAGAR T M, OLESON J R, VUJASKOVIC Z, DEWHIRST M W, CRACIUNESCU O I, BLACKWELL K L, PROSNITZ L R, JONES E L. Hyperthermia combined with radiation therapy for superficial breast cancer and chest wall recurrence: A review of the randomised data [J]. International Journal of Hyperthermia, 2010, 26(7): 612-617.
[4] FEYERABEND T, WIEDEMANN G J, JAGER B, VESELY H, MAHLMANN B, RICHTER E. Local hyperthermia, radiation, and chemotherapy in recurrent breast cancer is feasible and effective except for inflammatory disease [J]. International Journal of Radiation Oncology Biology Physics, 2001, 49(5): 1317-1325.
[5] HAND J W. The current status of microwave induced hyperthermia and radiotherapy for the treatment of recurrent breast cancer [C]// IEE Electronics Division Colloquium on the Application of Microwaves in Medicine. London, IEE Press, 1995: 1-6.
[6] ZASTROW E, DAVIS S K, LAZEBNIK M, KELCZ F, VAN VEEN B D, HAGNESS S C. Development of anatomically realistic numerical breast phantoms with accurate dielectric properties for modeling microwave interactions with the human breast [J]. IEEE Transactions on Biomedical Engineering, 2008, 55(12): 2792-2800.
[7] SAITO K, YOSHIMURA H, ITO K, AOYAGI Y, HORITA H. Clinical trials of interstitial microwave hyperthermia by use of coaxial-slot antenna with two slots [J]. IEEE Transactions on Microwave Theory and Techniques, 2004, 52(8II): 1987-1991.
[8] DE BRUIJNE M, WIELHEESEN D H, van der ZEE J, CHAVANNES N, van RHOON G C. Benefits of superficial hyperthermia treatment planning: Five case studies [J]. International Journal of Hyperthermia, 2007, 23(5): 417-429.
[9] MACCARINI P, ROLFSNES H O, NEUMANN D, JOHNSON J, JUANG T, STAUFFER P. Advances in microwave hyperthermia of large superficial tumors [C]// IEEE MTT-S International Microwave Symposium Digest. Long Beach, USA. IEEE Press, 2005: 1796-1800.
[10] PLANCOT M. A new method for thermal dosimetry in microwave hyperthermia using microwave radiometry for temperature control [J]. International Journal of Hyperthermia, 1987, 3(1): 9-19.
[11] CRESSON P Y, DUBOIS L, PRIBETICH J. Determination of the optimal heating pattern obtained with external planar applicators used for 915 MHz microwave hyperthermia [C]// Annual International Conference of the IEEE Engineering in Medicine and Biology Society. Lyon, France: IEEE Press, 2007: 1160-1163.
[12] ARORA D, SKLIAR M, ROEMER R B. Minimum-time thermal dose control of thermal therapies [J]. IEEE Transactions on Biomedical Engineering, 2005, 52(2): 191-200.
[13] GAMAGAMI P, SILVERSTEIN M J, WAISMAN J R. Infra-red imaging in breast cancer [C]// Proceedings of the 19th Annual International Conference of the IEEE Engineering in Medicine and Biology Society. Chicago, USA. IEEE Press, 1997: 677-680.
[14] PENNES H H. Analysis of tissue and arterial blood temperatures in the resting human forearm [J]. Journal of Applied Physiology, 1998, 85(1): 5-34.
[15] NEUMAN D G, STAUFFER P R, JACOBSEN S, ROSSETTO F. SAR pattern perturbations from resonance effects in water bolus layers used with superficial microwave hyperthermia applicators [J]. International Journal of Hyperthermia, 2002, 18(3): 180-193.
[16] SIAUVE N, NICOLAS L, VOLLAIRE C, MARCHAL C. 3D modelling of electromagnetic fields in local hyperthermia [J]. EPJ Applied Physics, 2003, 21(3): 243-250.
[17] CORREIA D, KOK H P, DE GREEF M, BEL A, VAN WIERINGEN N, CREZEE J. Body conformal antennas for superficial hyperthermia: The impact of bending contact flexible microstrip applicators on their electromagnetic behavior [J]. IEEE Transactions on Biomedical Engineering, 2009, 56(12): 2917-2926.
[18] JOHNSON J E, MACCARINI P F, NEUMAN D, STAUFFER P R. Automatic temperature controller for multielement array hyperthermia systems [J]. IEEE Transactions on Biomedical Engineering, 2006, 53(6): 1006-1015.
[19] HE X, MCGEE S, COAD J E, SCHMIDLIN F, IAIZZO P A, SWANLUND D J, KLUGE S, RUDIE E, BISCHOF J C. Investigation of the thermal and tissue injury behaviour in microwave thermal therapy using a porcine kidney model [J]. International Journal of Hyperthermia, 2004, 20(6): 567-593.
(Edited by YANG Bing)
Foundation item: Project(50977064) supported by the National Natural Science Foundation of China
Received date: 2011-05-31; Accepted date: 2011-10-08
Corresponding author: MA Guo-jun, PhD; Tel: +86-21-65982589; E-mail: 2008mgj@tongji.edu.cn