Bioleaching of low-grade copper sulfide ore by extremely thermoacidophilic consortia at 70 °C in column reactors
来源期刊:中南大学学报(英文版)2020年第5期
论文作者:曾伟民 艾郴兵 梁昱婷 邱冠周
文章页码:1404 - 1415
Key words:extreme thermoacidophile; bioleaching; copper sulfide ore; copper resistance; microbial community
Abstract: The effects of introducing M. sedula derivatives having different Cu2+-resistance on bioleaching capacity of a defined consortium (consisting of A. brierleyi DSM1651 and M. hakonensis HO1-1) were studied in column reactors at 70 °C. Introducing M. sedula copA mutant, a copper sensitive derivative, only had negligible effects on bioleaching. While introducing M. sedula ARS50-2, a Cu2+ resistant strain, substantially consolidated bioleaching process, with 27.77% more copper recovered after 58 d of bioleaching. Addition of M. sedula ARS50-2 likely enhanced the sulfur oxidation capacity of consortium after the 24th day under the Cu2+ stress. The majority of extreme thermoacidophiles were attached on minerals surface as indicated by quantitative PCR (qPCR) data. Successions of microbial community of extremely thermoacidophilic consortia that attached on surface of minerals were different from those in leachate. M. hakonensis HO1-1 was the dominant species attached on minerals surface in each column reactor throughout bioleaching process. The sessile M. sedula ARS50-2 remained as a major species till the 34th day. A. brierleyi DSM1651 was the most abundant planktonic species in leachate of each column reactor. These results highlight that higher Cu2+-resistance is a beneficial trait for extreme thermoacidophiles to process copper minerals.
Cite this article as: AI Chen-bing, LIANG Yu-ting, QIU Guan-zhou, ZENG Wei-min. Bioleaching of low-grade copper sulfide ore by extremely thermoacidophilic consortia at 70 °C in column reactors [J]. Journal of Central South University, 2020, 27(5): 1404-1415. DOI: https://doi.org/10.1007/s11771-020-4376-0.

J. Cent. South Univ. (2020) 27: 1404-1415
DOI: https://doi.org/10.1007/s11771-020-4376-0
AI Chen-bing(艾郴兵)1, 2, 3, LIANG Yu-ting(梁昱婷)2, 4,QIU Guan-zhou(邱冠周)2, 4, ZENG Wei-min(曾伟民)2, 4
1. School of Metallurgy and Environment, Central South University, Changsha 410083, China;
2. School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China;
3. Chinese National Engineering Research Center for Control and Treatment of Heavy Metal Pollution, Central South University, Changsha 410083, China;
4. Key Laboratory of Biohydrometallurgy of Ministry of Education, Central South University,Changsha 410083, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2020
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2020
Abstract: The effects of introducing M. sedula derivatives having different Cu2+-resistance on bioleaching capacity of a defined consortium (consisting of A. brierleyi DSM1651 and M. hakonensis HO1-1) were studied in column reactors at 70 °C. Introducing M. sedula copA mutant, a copper sensitive derivative, only had negligible effects on bioleaching. While introducing M. sedula ARS50-2, a Cu2+ resistant strain, substantially consolidated bioleaching process, with 27.77% more copper recovered after 58 d of bioleaching. Addition of M. sedula ARS50-2 likely enhanced the sulfur oxidation capacity of consortium after the 24th day under the Cu2+ stress. The majority of extreme thermoacidophiles were attached on minerals surface as indicated by quantitative PCR (qPCR) data. Successions of microbial community of extremely thermoacidophilic consortia that attached on surface of minerals were different from those in leachate. M. hakonensis HO1-1 was the dominant species attached on minerals surface in each column reactor throughout bioleaching process. The sessile M. sedula ARS50-2 remained as a major species till the 34th day. A. brierleyi DSM1651 was the most abundant planktonic species in leachate of each column reactor. These results highlight that higher Cu2+-resistance is a beneficial trait for extreme thermoacidophiles to process copper minerals.
Key words: extreme thermoacidophile; bioleaching; copper sulfide ore; copper resistance; microbial community
Cite this article as: AI Chen-bing, LIANG Yu-ting, QIU Guan-zhou, ZENG Wei-min. Bioleaching of low-grade copper sulfide ore by extremely thermoacidophilic consortia at 70 °C in column reactors [J]. Journal of Central South University, 2020, 27(5): 1404-1415. DOI: https://doi.org/10.1007/s11771-020-4376-0.
1 Introduction
The excessive exploitation of copper ores leads to the gradual depletion of high-grade copper sulfide ores [1]. Thus, it is necessary to extract copper from the much more abundant reserves of low-grade copper sulfide ores to meet the demand of market [2]. Currently, low-grade copper ores could not be economically processed by the conventional energy-intensive comminution, floatation and pyrometallurgical technologies. On the contrary, heap bioleaching is a promising alternative to extract copper from low-grade ores ascribing to its characteristics of technical simplicity, low investments and appropriate high recovery yield [3, 4].
Since the bio-oxidation of sulfide ores is an exothermal process, temperature inside low-grade copper sulfide heap can increase gradually during bioleaching processes [5]. Accordingly, microbial community of the bioleaching consortium inhabiting the surface of minerals can be modulated [6-8]. The temperature inside large ore heaps can eventually reach 60-80 °C with the dissolution of sulfide minerals in the copper biomining industry [5, 9]. Such high temperature completely inhibits the growth of bioleaching mesophiles and moderate thermoacidophiles, but it is suitable for extreme thermoacidophiles (optimal growth Tm≥ 60 °C, pH≤ 3) [10-12].
Both mesophiles and moderate thermoacidophiles have been widely applied in the extraction of copper from secondary low-grade copper ore via heap bioleaching owing to their ferrous iron and sulfur oxidation capabilities [6, 13]. The successions of microbial community of mesophilic and moderate thermoacidophilic consortia have been extensively studied during the extraction of copper from low-grade copper ores in commercial bioleaching heaps and laboratory-scale isothermal column reactors [8]. However, little is known regarding on the copper recovery rates and the shifts of microbial community of extremely thermoacidophilic consortia during the bioleaching processes at high temperature above 60 °C [14]. Bioleaching low-grade copper sulfide ores in column reactor is a common way to simulate the industrial heap bioleaching practice [5]. Studying the bioleaching behaviors of low-grade copper ores by extremely thermoacidophilic consortia in isothermal column reactor at high temperature would evaluate their potentials for copper extraction and provide insights into the succession of microbial community inside the bioleaching heaps at industrial scale.
The Cu2+ together with other ions would be dissolved from minerals and accumulated in leachate to high concentration during bioleaching. The concentration of Cu2+ in the leachate of heap leaching at industrial scale usually accumulated to be higher than 6 g/L (around 94 mmol/L) before the leachate can be processed by downstream solvent extraction and electrowinning. Thus, they would severely inhibit the Fe2+/sulfur oxidation activities and the survival of extreme thermoacidophiles [12, 15]. Compared with bioleaching mesophiles and moderate thermoacidophiles, the isolated extreme thermoacidophiles mostly have much lower minimal inhibitory concentration (MIC) on Cu2+ [16]. Therefore, the availability of high resistance to cupric ion would be a beneficial trait for bioleaching extreme thermoacidophiles [17]. In our previous study, the Cu2+ resistant M. sedula ARS50-2 was isolated through adaptive laboratory evolution [18]. Introducing M. sedula ARS50-2 to a defined extremely thermoacidophilic consortium consolidated the bioleaching of refractory enargite in shaking flask at elevated pulp density [14]. Since the bioleaching of copper sulfide ores in shaking flask and the column reactor was operated with different physicochemical parameters in leachate, it is unclear whether the resistant M. sedula ARS50-2 can also consolidate the bioleaching of low-grade copper sulfide ores in column reactor.
Here, low-grade copper sulfide ore was bioleached in column reactors by different defined extremely thermoacidophilic consortia at 70 °C. The effects of introducing M. sedula derivatives with different Cu2+-resistance into a defined consortium (consisting of A. brierleyi DSM1651 and M. hakonensis HO1-1) on bioleaching were studied. Species-specific primer pairs for qPCR were designed for these bioleaching extreme thermoacidophiles. The successions of microbial community of consortia attached on surface of minerals and the planktonic in leachate throughout bioleaching process were quantified by qPCR.
2 Experimental procedures
2.1 Extreme thermoacidophiles and culture medium
All the extreme thermoacidophiles used in this study were isolated and characterized in previous studies: the Cu2+-resistant derivative M. sedula ARS50-2 [14, 18], the Cu2+ sensitive strain M. sedula copA mutant [19], M. hakonensis HO1-1 [20] and A. brierleyi DSM1651. The MIC of Cu2+ and the optimal growth pH and temperature of these extreme thermoacidophiles are listed in Table 1. They were grown in basic salt medium containing (NH4)2SO4 (3 g/L), K2HPO4 (0.5 g/L), MgSO4・7H2O (0.5 g/L), KCl (0.1 g/L), Ca(NO3)2 (0.01 g/L), which was adjusted to pH 2.0 using sulfuric acid, and supplemented with 0.1% (W/V) tryptone. A final concentration of 5 μg/mL uracil was added in medium for the growth of M. sedula copA mutant and M. sedula ARS50-2 since they are derivatives of an uracil auxotrophy ancestor [19]. All the cultures were incubated at 70 °C in flasks with aeration in orbital baths.
2.2 Mineral composition
The low-grade copper ore was obtained from Meizhou Copper Mine, Guangdong Province, China. The main chemical composition of this ore was:Cu 1.03%, Fe 4.61%, S 4.30%, Zn 0.30%, SiO2 83.98%, Al2O3 4.12%, CaO 0.25%. The copper phases of this low-grade copper ore were mainly in the forms of secondary copper sulfide (55% Cu) and primary sulfide (45% Cu). The ore was crushed and sieved to obtain a particle size between 3 to 10 mm.
2.3 Column reactor
Isothermal column reactor was designed for the bioleaching experiment. Schematic of this bioleaching apparatus is shown in Figure 1(a). The column reactor was made of 5 mm thick plexiglass and fixed to a scaffolding panel. The inner cylinder where ores were loaded was 50 cm in height and 3 cm in radius, containing a porous supporting plate ( 3 mm for each hole) on the bottom. This cylinder was embedded parallelly into a bigger cylinder in the same height and with a radius of 6 cm. The top and bottom sides between these two cylinders were sealed. The clearance between these two cylinders became the water jacket for the column reactor. During bioleaching processes, the hot water (72 °C) heated by a circulating thermostatic water bath was pumped through the bottom nozzle on the wall of the water jacket, filled it up and discharged from the nozzle on the top, and then recycled back to the circulating thermostatic water bath. A set of column reactors were connected sequentially by rubber tube to maintain the homogeneity of temperature inside each column reactor during bioleaching Figure 1(b).
3 mm for each hole) on the bottom. This cylinder was embedded parallelly into a bigger cylinder in the same height and with a radius of 6 cm. The top and bottom sides between these two cylinders were sealed. The clearance between these two cylinders became the water jacket for the column reactor. During bioleaching processes, the hot water (72 °C) heated by a circulating thermostatic water bath was pumped through the bottom nozzle on the wall of the water jacket, filled it up and discharged from the nozzle on the top, and then recycled back to the circulating thermostatic water bath. A set of column reactors were connected sequentially by rubber tube to maintain the homogeneity of temperature inside each column reactor during bioleaching Figure 1(b).
Table 1 Extreme thermoacidophiles used in this study (*: MIC of Cu2+ tested in this study)


Figure 1 Schematic of single column reactor (a) and photo of column reactors used in this study (b) (Temperature inside ore stack was maintained at 70 °C; leachate was recycled at a rate of 0.8 L/h; ore stack in column was aerated at a rate of 8 L/min)
2.4 Column bioleaching experiments
The original low-grade copper ore sample was crushed and ground, and then screened to obtain the particles with a size smaller than 0.8 cm for the following column bioleaching. Around 2.10 kg of ores were soaked into 2 L basic salt medium in plastic beaker for 2 d to eliminate the pH fluctuation in the following bioleaching process caused by acid-consuming gangue. The leachate was separated from minerals and collected for quantification of the amount of copper recovered in this pretreatment. Around 3.50% of copper was recovered from the pretreatment process.
Double layers of circular etamine were put on the porous supporting plate of the inner column. Then, some of the pretreated minerals were loaded into the column. An acid-resistant tube was put into the column for aeration when the height of the loaded ore was around 6 cm (relative to the porous supporting plate), then loaded all the remanent ores. The leachate in collected in glass container was continuously pumped through an acid-resistance plastic tube to the top of ore stack by a peristaltic pump at a rate of 0.8 L/h. The leachate dripped centrally on a filter paper that was located horizontally on the top of ore stack. The leachate percolated the ore particles by gravity and then collected in the glass container. The leachate in the container was aerated at a rate of 2 L/min. The ore stack in column was aerated inside at a rate of 8 L/min.
The biomining extreme thermoacidophiles adopted in column bioleaching were grown heterogeneously and supplemented with 30 mmol/L Cu2+. The cultures in middle log phase were harvested by centrifugation and washed twice with the basic salt medium. In order to study the effects of Cu2+-resistance on the column bioleaching of sulfide ores, different defined microbial consortia were constructed: consortium A (consisting of M. hakonensis HO1-1 and A. brierleyi DSM1651), consortium B (consisting of M. hakonensis HO1-1, A. brierleyi DSM1651 and M. sedula copA mutant), and consortium C (consisting of M. hakonensis HO1-1, A. brierleyi DSM1651 and M. sedula ARS50-2). Each consortium was added into an individual column respectively. An abiotic control column was also set up. For each column, identical amount of cultures were inoculated. Basic salt medium of pH 2.0 was added into each column to the final leachate volume of 1 L, and supplemented with 0.05% (W/V) tryptone. A final concentration of 5 μg/mL uracil was added respectively into column reactors inoculated with consortium B and consortium C. When the copper concentration accumulated to high concentration in leachate at the 24th and 54th day, all the leachate leached by each consortium was replaced with 1 L fresh basic salt medium (pH 2.0) supplemented with 0.05% (W/V) tryptone at the same time. A final concentration of 5 μg/mL uracil was also added respectively into column reactor inoculated with consortium B or consortium C respectively. Bioleaching leachate samples were taken for assay at an interval of two days increment. The evaporation of water was compensated daily with distilled water.
2.5 Analytical methods
Planktonic cell numbers were determined using a Thoma counting chamber [21]. The pH of the leachate samples was measured using a pH meter (PHSJ-4A). The concentrations of Fe2+ and Fe3+ ions were determined using the 1,10-o- phenanthroline [22]. The concentration of Cu2+ was measured spectrophotometrically using bis- cyclohexanone oxalydihydrazone [23].
2.6 Species specific qPCR primer pairs design
Neither the genome sequences nor the eligible house-keeping genes of the M. hakonensis HO1-1 and A. brierleyi DSM1651 were available for designing their species-specific primer pairs for qPCR when the initiation of this bioleaching experiment. The 16S rRNA genes of these extreme thermoacidophiles employed in this study were too similar to designed species-specific primer pairs (data not shown). Therefore, the house-keeping gene existing in these extreme thermoacidophiles and meanwhile containing adequate divergent fragments would be an ideal gene for designing their qPCR primer pairs. Comparative genomic study revealed that copA, a gene around 2.2 kb in length encoding a membrane-bound pump for Cu2+ efflux, was highly conserved in the phylogenetically related extreme thermoacidophiles belonging to the genera of Sulfolobus, Acidianus and Metallosphaera. The following degenerate primer pair was designed to amplify the proposed copA gene fragment from M. hakonensis HO1-1 and A. brierleyi DSM1651 respectively: forward-GGAATGCAYTGYGCHAC, reverse- GCRTCRTTNAYNCCRTCNCC. Primer pairs with specific amplification for each strain were designed based on the divergent regions in their copA genes (Table 2).
2.7 Genomic DNA extraction and microbial community analyses
The planktonic cells in leachate were collected by centrifugation at 10000g for 12 min. Irrigation of leachate was stopped to drain up the mobile leachate around 30 min before sampling the ores. The ore on the top of column (around 5 cm in depth) was taken out first. Then, around 50 g of ores were sampled to harvest the microbes attached on surface of minerals. The ores taken from the top of ore column were dump back to the column, and the irrigation started. For each time, around 10 g of ore was transferred into a 50 mL falcon tube, suspended with around 10 mL sterilized basic salt medium and thoroughly agitated using a bench-top vortex for 1 min. The slurry in falcon tube was stood still on bench for 1 min, and then the liquid phase was transferred and filtered through a filter paper. The residual ores were re-suspended with 10 mL sterilized basic salt medium and the washing step was repeated a total of 5 times. For the last three washing steps, 0.2% (V/V) Tween 20 was added to mobilize the tightly attached bioleaching microbes on the surface of ores. Repeat the overall washing steps to process remanent ores. The filtered liquid was combined and centrifuged at 2000g for 25 s. Transfer the supernatant gently to a clean tube and collect microbes by centrifuging at 10000g for 10 min. Transfer the cell pellet into a 2 mL tube. The genomic DNAs of these samples were extracted using TIANamp Bacteria DNA Kit (Tiangen Inc, Beijing, China) according to the manufacturer’s instructions.
Each real-time PCR mixture (total volume 25 μL) contained 12.5 μL of SYBR Green Real- time PCR Master Mix (Toyobo Co., Ltd., Osaka, Japan), 1 μL of 10 mmol/L forward and reverse primer, 2 μL of genomic DNA, and 8.5 μL of nuclease-free water. The real-time PCR was carried out with the iCycler iQ Real-time PCR detection system (Bio-Rad Laboratories, Inc., Hercules, CA) to quantify the numbers of each extreme thermoacidophile: 1 cycle at 95 °C for 5 min; and then 40 cycles at 95 °C for 20 s, 59.5 °C for 20 s, and 72 °C for 20 s. At the completion of each run, melting curves for the amplicons were measured by raising the temperature in increments of 0.5 °C from 60 to 95 °C while monitoring fluorescence. Technical repeats were set up for each sample.
Green Real- time PCR Master Mix (Toyobo Co., Ltd., Osaka, Japan), 1 μL of 10 mmol/L forward and reverse primer, 2 μL of genomic DNA, and 8.5 μL of nuclease-free water. The real-time PCR was carried out with the iCycler iQ Real-time PCR detection system (Bio-Rad Laboratories, Inc., Hercules, CA) to quantify the numbers of each extreme thermoacidophile: 1 cycle at 95 °C for 5 min; and then 40 cycles at 95 °C for 20 s, 59.5 °C for 20 s, and 72 °C for 20 s. At the completion of each run, melting curves for the amplicons were measured by raising the temperature in increments of 0.5 °C from 60 to 95 °C while monitoring fluorescence. Technical repeats were set up for each sample.
3 Results and discussion
3.1 Column bioleaching low-grade copper sulfide ore by extremely thermoacidophilic consortia
The effects of introducing M. sedula copA mutant or M. sedula ARS50-2 into the defined extremely thermoacidophilic consortium A (consisting of M. hakonensis HO1-1 and A. brierleyi DSM1651) on the bioleaching of low-grade copper ore in column reactors were studied. Figure 2(a) shows the concentration of Cu2+ in leachate during the bioleaching process. Cu2+ concentration in leachate was low in the first 6 d after the extremely thermoacidophilic consortia had been inoculated into each column reactor. Then, it started to increase till the 24th day. There was no significant difference regarding on Cu2+ concentration before the 16th day between the column reactors that inoculated with consortium A and consortium C respectively. Thereafter, the Cu2+ concentration in leachate of consortium C was higher than that of consortium A till the 24th day.
Table 2 Species-specific qPCR primer pairs for extreme thermoacidophiles based on their unique copA gene sequence

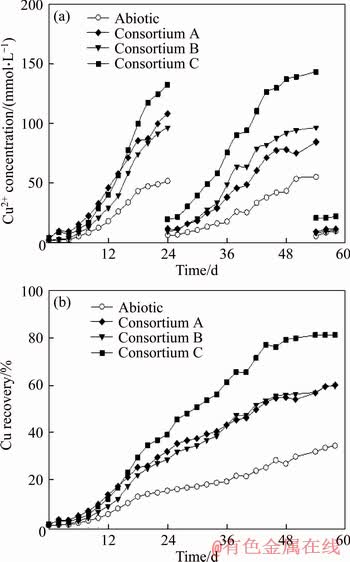
Figure 2 Copper concentration in leachate (a) and copper recovery yield (b) during bioleaching of low-grade copper ore with extremely thermoacidophilic consortia in column reactors at 70 °C (Consortium A consisted of M. hakonensis HO1-1 and A. brierleyi DSM1651; Consortium B consisted of M. hakonensis HO1-1, A. brierleyi DSM1651 and M. sedula copA mutant; Consortium C consisted of M. hakonensis HO1-1, A. brierleyi DSM1651 and M. sedula ARS50-2)
The Cu2+ concentration in the leachate of column inoculated with consortium B was always slightly lower than that in leachate of consortium A in the first 24 d. This observation might be ascribed to the M. sedula copA mutant strain existing in consortium B having the lowest MIC of Cu2+ (Table 1). The Cu2+ concentration in leachate of abiotic control increased gradually in the first 24 d with a much lower rate compared with other reactors that inoculated with extremely thermoacidophilic consortia. After the replacement of bioleaching leachate with fresh basic salt medium on the 24th day, the Cu2+ concentration in leachate of consortium C was much higher than that in column reactor inoculated with consortium A or consortium B. This observation is probably attributed to the fact high concentration of Cu2+in leachate before the 24th day severely inhibits the bioleaching capacities of consortium A and consortium B. However, the bioleaching capacity of consortium C was not inhibited owing to the containing of Cu2+ resistant strain M. sedula ARS50-2. The concentration of Cu2+ in consortium B was higher than that of consortium A after the 34th day, which was likely attributed to the increased bioleaching capacity resulting from mutations occurring in these strains in consortium B.
Figure 2(b) shows the trend of copper recovery yield obtained in each column reactor. After 58 d of bioleaching, 60.01% and 59.77% copper were recovered by consortium A and consortium B, respectively. A significant increase of 27.77% in copper recovery yield was achieved by consortium C, with 87.78% copper achieved. While, only 34.36% copper was recovered from copper sulfide in the abiotic control column reactor. These data indicate that extremely thermoacidophilic consortia accelerated the bioleaching of copper sulfide ore. Introducing the copper sensitive derivative M. sedula copA mutant only had negligible effect on bioleaching; while introducing the Cu2+ resistant strain M. sedula ARS50-2 substantially enhanced the bioleaching process.
The variations of pH and total iron concentration of leachate are shown in Figure 3. Generally, the variation trends of leachate pH for all these column reactors are similar during the bioleaching process (Figure 3(a)). The pH value of leachate slightly increased in the first two days, then started to decrease till the 12nd day. An increase of leachate pH was observed from the 12th day to the 18th day, and then the pH started to decrease again till the 24th day. The leachate pH of column reactor inoculated with consortium A was the lowest since the 6th day, and remained till the 24th day, while the pH of the leachate for the abiotic control was the highest. An increase of pH was observed after the replacement of leachate with fresh basic salt medium and penetration through ore column at the end of the 24th day. Then, the pH of all these column reactors started to decline till the 54th day. The leachate pH of column reactor inoculated with consortium C was much lower than that of others. The leachate pH values of consortium A and consortium B were comparable at this duration. A gradual decrease of pH value was also observed for abiotic control. The decrease of pH value of all these column reactors might be partially attributed to the formation of jarosite, an acid generation reaction, during the bioleaching. The much lower pH value of column reactors with microbial inoculum might be ascribed to the sulfur oxidation capabilities of the extreme thermoacidophiles.
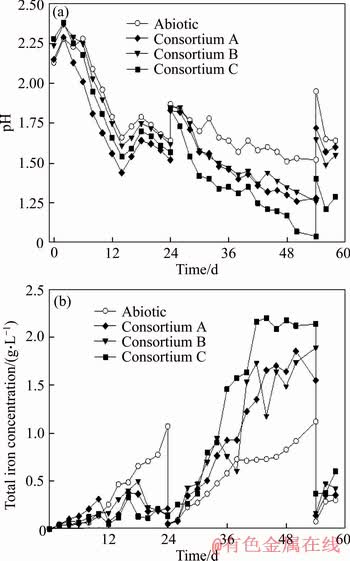
Figure 3 Changes of pH value (a) and total iron concentration (b) of leachate during bioleaching of low-grade copper ore
The iron in leachate of each column reactor remained almost as ferric iron throughout bioleaching process. The variation tendencies of total iron concentration in leachate of column reactors that inoculated with bioleaching consortium were similar (Figure 3(b)). The total iron concentration remained at low level (<0.5 g/L) in the first 24 d of bioleaching, and then it started to increase quickly to higher level (ranging from 1.6 to 2.2 g/L) till the 42nd day and maintained till the 54th day. The total iron concentrations in leachate of column reactors containing inoculum were lower than that of abiotic column reactor during the 12th day to 24th day. This is probably ascribed to the fact that the iron dissolved from ores was largely absorbed by the extracellular polymeric substances (EPS) of extreme thermoacidophiles attached on minerals surface. EPS is important for the function roles of single microorganism and consortium [24, 25]. Previous studies showed that the EPS of bioleaching acidophiles can absorb large amount of irons and play pivotal role in the bioleaching of copper sulfide minerals [26]. The increase of total iron concentration in leachate for these column reactors with inoculum between the 24th to 54th day was probably because the iron further dissolved from ores surpassed the iron-binding capacities of the EPS of extreme thermoacidophiles attached.
3.2 Verification of species-specific qPCR primer pairs
The designed species-specific primer pairs could only amplify a single target band from each genome respectively, and unspecific amplification from the genomic DNA of other species was not observed (Figure 4). Analyses of real-time PCR data show that only single melting peak is observed for each primer pair, which indicates that non- specific amplification from the genomic samples did not occur (data not shown).
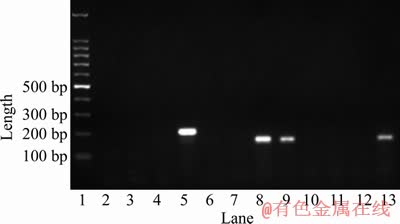
Figure 4 Verification analysis of strain specific primer pairs for extreme thermoacidophiles (Lane 1: 100 bp DNA marker; Lane 2: A. brierleyi DSM1651; Lane 3: M. sedula copA mutant; Lane 4: M. sedula ARS50-2; Lane 5: M. hakonensisi HO1-1; Lanes 2 to 5 amplified with primer pair Mha-F & Mha-R; Lane 6: A. brierleyi DSM1651; Lane 7: M. hakonensisi HO1-1; Lane 8: M. sedula copA mutant; Lane 9: M. sedula ARS50-2; Lanes 6 to 10 amplified with primer pair Mse-F & Mse-R; Lane 10: M. hakonensisi HO1-1; Lane 11: M. sedula copA mutant; Lane 12: M. sedula ARS50-2; Lane 13: A. brierleyi DSM1651; Lanes 10 to 13 amplified with primer pair A.b-F & A.b-R)
The species-specific qPCR primer pairs of typical moderate thermoacidophiles with bioleaching capabilities, such as Acidithiobacillus caldus, Leptospirillum ferriphilum, Sulfobacillus spp. and Ferroplasma spp., were designed based on their divergent 16S rRNA genes, and extensively applied to study the successions of microbial community consisting of these acidophiles during various bioleaching processes [27]. While, studies focusing on the succession of microbial community of extremely thermoacidophilic consortium during bioleaching of sulfide minerals are few, which is ascribed to the lack of species-specific primer pair [14]. The 16S rRNA genes of these biomining extreme thermoacidophiles in the genera Acidianus, Sulfolobus and Metallosphaera, are too similar to design their species-specific primer pairs for differentially quantitative analysis of each species from others when these strains coexisted in bioleaching reactor. It is easy to design the species-specific primer pairs for these bioleaching extreme thermoacidophiles with genome sequences available. While, it is quite challenging to design the primer pairs for these bioleaching extreme thermoacidophiles with only limited gene sequences available in the context of microbial consortium existing in the bioleaching niches. Adequate divergent regions were observed in the partial copA gene of these extreme thermoacidophiles employed in this study, which indicates that copA gene can also be informative for designing of species-specific qPCR primer pair for other bioleaching extreme thermoacidophiles without genome sequence available.
3.3 Microbial community analyses
The successions of microbial community of each consortium attached on minerals surface or planktonic in leachate on the 12th, 24th, 34th and 50th day during bioleaching process were quantitatively analyzed by qPCR.
M. hakonensis HO1-1 was the most abundant species inhabiting mineral surface in each consortium throughout the bioleaching process (Figure 5). Compared with consortium A, an increase of the viabilities of attached A. brierleyi DSM1651 and M. hakonensis HO1-1 was observed on the 24th day, while comparable viabilities maintained at other time points in consortium B (Figures 5(a) and (b)). Although an increase of viability of M. hakonensis HO1-1 was also observed at the 24th day in consortium C and a decrease of its copy number was found at other time points. The maximum number of sessile M. sedula copA mutant in consortium B was observed on the 12th day (3.22×107 cell/g ore), and remained as a major species till the 24th day. Then, it declined drastically to 1.74×106 cell/g ore at the 34th day. As low as 9.00×103 cell/g ore was observed for M. sedula copA mutant on the 50th day (Figure 5(b)). M. sedula ARS50-2 in consortium C was a major species (3.40×107 cell/g ore) attached on minerals surface since the 12th day, and its viability increased slightly to 3.91×107 cell/g ore at the 24th day, then decreased to 2.68×107 cell/g ore and evolved as the predominant species in consortium C on the 34th day (Figure 5(c)). The difference on viabilities between M. sedula copA mutant and M. sedula ARS50-2 attached on mineral surface since the 24th day would be related to their different Cu2+ resistance.
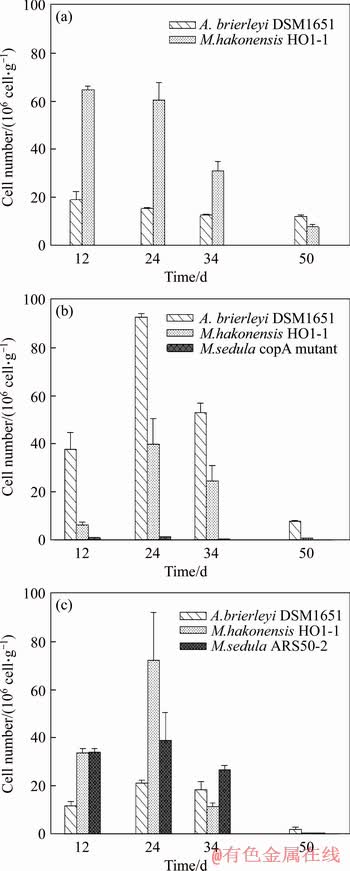
Figure 5 Microbial community of consortia attached on mineral surface for consortium A (a), consortium B (b) and consortium C (c) during bioleaching of low-grade copper ore
A. brierleyi DSM1651 was the predominant species that is planktonic in leachate of all these column reactors inoculated with extremely thermoacidophilic consortium throughout the bioleaching processes (Figure 6). The viability of A. brierleyi DSM1651 increased to the maximum on the 24th day in each consortium, then started to decrease. M. hakonensis HO1-1 was a minor species on the 12th day in each consortium, and then evolved as one of the major species on the 24th day. Both M. sedula copA mutant and M. sedula ARS50-2 existed in leachate as a minor species with low abundance throughout the bioleaching process (Figure 6(b) and (c)), in spite of great abundance was detected on the minerals surface for these two extreme thermoacidophiles. The discrepancies in relative abundances between the sessile and the planktonic of each species implied that different species might have different preferences to attach to the minerals surface under the physicochemical parameters employed in this study.
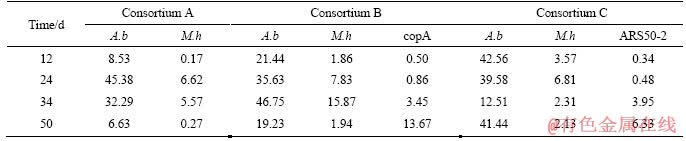
The proportion of planktonic cells versus the total cell number of each species (both planktonic and attached cells) was calculated for each consortium (Table 3). These data reveal that the M. hakonensis HO1-1, M. sedula copA mutant and M. sedula ARS50-2 were largely attached to the surface of minerals throughout the bioleaching process. On the contrary, considerable proportion of A. brierleyi DSM1651 existed in leachate in all these consortia at some time points during the bioleaching. The proportion of planktonic A. brierleyi DSM1651 varied in a similar tendency to consortium A and consortium B. Only low proportion (8.53% and 21.44% for consortium A and consortium B respectively) of A. brierleyi DSM1651 was planktonic in leachate on the 12nd day. Then, considerable proportion (percentage ranging from 32.29% to 46.75%) of A. brierleyi DSM1651 existed in leachate on the 24th and 34th day. Finally, the proportion of the planktonic A. brierleyi DSM1651 decreased to 6.63% and 19.23% for consortium A and consortium B respectively on the 50th day. The proportion of planktonic A. brierleyi DSM1651 varied in a different pattern from consortium C. Large amounts (percentage ranging from 39.58% to 42.56%) of planktonic A. brierleyi DSM1651 were observed for the 12th, 24th and 50th day, except for the 34th day.
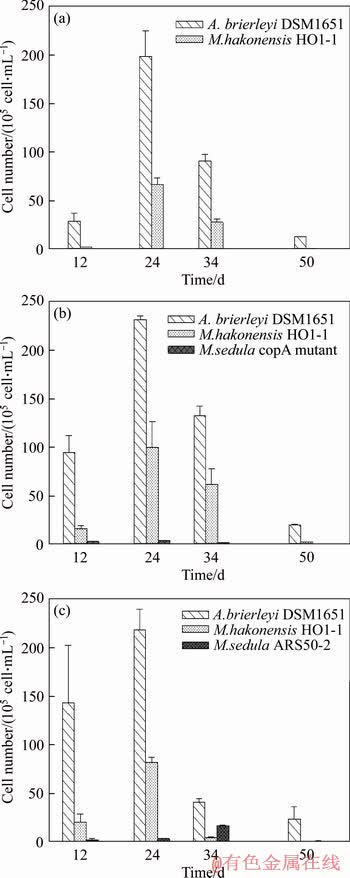
Figure 6 Microbial community of consortia in leachate for consortium A (a), consortium B (b) and consortium C (c) during bioleaching of low-grade copper ore
Table 3 Percentage of planktonic microbes versus total cell number of each species (both planktonic and attached cells) during bioleaching process in each consortium (A.b: A.brierleyi: M. h: M.hakonensis; copA: M. sedula copA mutant; ARS50-2: M. sedula ARS50-2)
The number of extreme thermoacidophiles attached on minerals surface outnumbers the corresponding planktonic cell population in each consortium during bioleaching process (Figure 7). No remarkable difference was observed regarding on the planktonic cell number in each consortium, while significant differences among these consortia were found for the numbers of extreme thermoacidophiles attached on minerals surface at several time points. The consortium C had the highest viability on the 24th and 34th day. Abundant extreme thermoacidophiles thriving on minerals surface on the 12th day might be attributed to their mixotrophic growth with tryptone, the reduced sulfur compounds and ferrous iron as substrates. The sessile extreme thermoacidophiles probably continued to proliferate, because the Cu2+ concentration in leachate of each column reactor was not inhibitive to the survival of each species at this time. The dissolved Cu2+ in leachate probably exerted inhibitory effects on these extreme thermoacidophiles species after the 16th day (Figure 2(a)).
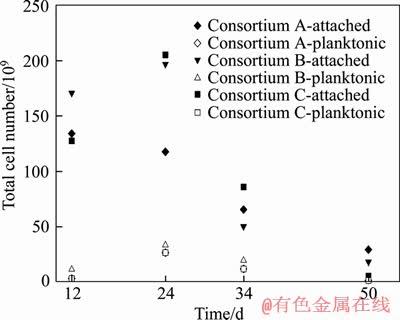
Figure 7 Biomass of sessile and planktonic extreme thermoacidophiles in each column bioreactor during bioleaching of low-grade copper ore
The concentration of Cu2+ in leachate of the column reactors inoculated with consortium A, consortium B and consortium C increased to 108, 97 and 133 mmol/L respectively on the 24th day, which was much higher than the MIC of each strain except for M. sedula ARS50-2. Although the increase of viabilities was observed for the consortium B and consortium C on the 24th day, their sulfur oxidation capacities could be severely inhibited. Despite the leachate was replaced with basic salt medium and supplemented with 0.05% (W/V) tryptone at the end of 24th day, a decrease in viabilities of attached extreme thermoacidophiles was observed on the 34th day in each consortium. This also indicates that the high Cu2+ concentration presented in leachate from the 18th day to 24th day would negatively affect the viabilities of these extreme thermoacidophiles. Higher concentration of copper was found in leachate of the column reactor inoculated with consortium C. The much lower pH value since the replacement of leachate on the 24th day indicates that the consortium C has higher sulfur oxidation capacity. The significant decrease of the viabilities in each column reactor inoculated with extremely thermoacidophilic consortium on the 50th day probably resulted from the shortage of energy substrates and the extreme low pH of leachate.
The temperature of leachate of each column reactor was around 52 °C throughout the bioleaching process, which was close to the limitation for these extreme thermoacidophiles employed in this study. Previous study shows that the temperature strongly influences the iron and sulfur oxidation rates of these bioleaching acidophiles [28]. Therefore, the iron and sulfur oxidation activity of the planktonic extreme thermoacidophile, if had any, was very low. So, it was the attached extreme thermoacidophiles that played pivotal role in the bioleaching of copper sulfide.
4 Conclusions
1) Introducing M. sedula copA mutant only had negligible effect on bioleaching; while introducing the M. sedula ARS50-2 substantially consolidated the bioleaching process, with an increase of copper recovery by 27.77% in 58 d.
2) M. hakonensis HO1-1 was the dominant species attached on minerals surface, meanwhile A. brierleyi DSM1651 was the most abundant species in leachate of these column reactors inoculated with extreme thermoacidophiles.
3) Although M. sedula copA mutant maintained as a major species in consortium till the 24th day, and as minority thereafter, the high concentration of Cu2+ in leachate probably inhibited its sulfur oxidation capacity. While, the Cu2+ in leachate probably exerted only negligible effects on the M. sedula ARS50-2 ascribing to its supranormal high Cu2+ resistance.
4) High Cu2+ resistance is a beneficial trait for extreme thermoacidophiles to extract copper sulfide ore under the stress of high concentration of Cu2+.
References
[1] NORTHEY S, MOHR S, MUDD G M, WENG Z, GIURCO D. Modelling future copper ore grade decline based on a detailed assessment of copper resources and mining [J]. Resources, Conservation and Recycling, 2014, 83: 190-201. DOI: 10.1016/j.resconrec.2013.10.005.
[2] WATLING H R. Review of biohydrometallurgical metals extraction from polymetallic mineral resources [J]. Minerals, 2015, 5: 1-60. DOI:10.3390/min5010001.
[3] ZHAO Hong-bo, ZHANG Yi-sheng, ZHANG Xian, QIAN Lu, SUN Meng-lin, YANG Yu, ZHANG Yan-sheng, WANG Jun, KIM H, QIU Guan-zhou. The dissolution and passivation mechanism of chalcopyrite in bioleaching: An overview. [J]. Minerals Engineering, 2019, 136: 140-154. DOI: 10.1016/j.mineng.2019.03.014.
[4] LIANG Yu-ting, HAN Jun-wei, AI Chen-bing, QIN Wen-qing. Adsorption and leaching behaviors of chalcopyrite by two extreme thermophilic archaea [J]. Transactions of Nonferrous Metals Society of China, 2018, 28(12): 2538-2544. DOI: 10.1016/S1003-6326(18)64900-3.
[5] PETERSEN J, DIXON D G. Thermophilic heap leaching of a chalcopyrite concentrate [J]. Minerals Engineering, 2002, 15: 777-785. DOI: 10.1016/S0892-6875(02)00092-4.
[6] PRADHAN N, NATHSARMA K C, SRINIVASA RAO K, SUKLA L B, MISHRA B K. Heap bioleaching of chalcopyrite: A review [J]. Minerals Engineering, 2008, 21(5): 355-365. DOI: 10.1016/j.mineng.2007.10.018.
[7] VALD S J, C RDENAS J P, QUATRINI R, ESPARZA M, OSORIO H, DUARTE F, LEFIMIL C, SEPULVEDA R, JEDLICKI E, HOLMES D S. Comparative genomics begins to unravel the ecophysiology of bioleaching [J]. Hydrometallurgy, 2010, 104(3, 4): 471-476. DOI: 10.1016/ j.hydromet.2010.03.028.
[8] ACOSTA M, GALLEGUILLOS P, GHORBANI Y, TAPIA P, CONTADOR Y, VEL SQUEZ A, ESPOZ C, PINILLA C, DEMERGASSO C. Variation in microbial community from predominantly mesophilic to thermotolerant and moderately thermophilic species in an industrial copper heap bioleaching operation [J]. Hydrometallurgy, 2014, 150: 281-289. DOI: 10.1016/j.hydromet.2014.09.010.
[9] BECK J. The role of bacteria in copper mining operations [J]. Biotechnology and Bioengineering, 1967, 9: 487-497. DOI: 10.1002/bit.260090405.
[10] PANDA S, AKCIL A, PRADHAN N, DEVECI H. Current scenario of chalcopyrite bioleaching: A review on the recent advances to its heap-leach technology [J]. Bioresource Technology, 2015, 196: 694-706. DOI: 10.1016/j.biortech. 2015.08.064.
[11] AI Chen-bing, YAN Zhang, CHAI Hong-sheng, GU Tian-yuan, WANG Jun-jun, CHAI Li-yuan, QIU Guan-zhou, ZENG Wei-min. Increased chalcopyrite bioleaching capabilities of extremely thermoacidophilic Metallosphaera sedula inocula by mixotrophic propagation [J]. Journal of Industrial Microbiology & Biotechnology, 2019, 46(8): 1113-1127. DOI: 10.1007/s10295-019-02193-3.
[12] AI Chen-bing, MCCARTHY S, ECKRICH V, RUDRAPPA D, QIU Guan-zhou, BLUM P. Increased acid resistance of the archaeon, Metallosphaera sedula by adaptive laboratory evolution [J]. Journal of Industrial Microbiology & Biotechnology, 2016, 43(10): 1455-1465. DOI: 10.1007/ s10295-016-1812-0.
[13] AI Chen-bing, LIANG Yu-ting, MIAO Bo, CHEN Miao, ZENG Wei-min, QIU Guan-zhou. Identification and analysis of a novel gene cluster involves in Fe2+ oxidation in Acidithiobacillus ferrooxidans ATCC 23270, a typical biomining acidophile [J]. Current Microbiology, 2018, 75(7): 818-826. DOI: 10.1007/s00284-018-1453-9.
[14] AI Chen-bing, MCCARTHY S, LIANG Yu-ting, RUDRAPPA D, QIU Guan-zhou, BLUM P. Evolution of copper arsenate resistance for enhanced enargite bioleaching using the extreme thermoacidophile Metallosphaera sedula [J]. Journal of Industrial Microbiology & Biotechnology, 2017, 44(12): 1613-1625. DOI: 10.1007/s10295-017- 1973-5.
[15] WATLING H R, WATKIN E L J, RALPH D E. The resilience and versatility of acidophiles that contribute to the bio-assisted extraction of metals from mineral sulphides [J]. Environmental Technology, 2010, 31(8, 9): 915-933. DOI: 10.1080/09593331003646646.
[16] WATLING H, SHIERS D, COLLINSON D. Extremophiles in mineral sulphide heaps: Some bacterial responses to variable temperature, acidity and solution composition [J]. Microorganisms, 2015, 3(3): 364-390. DOI: 10.3390/ microorganisms3030364.
[17] MCCARTHY S, AI Chen-bing, BLUM P. Enhancement of Metallosphaera sedula bioleaching by targeted recombination and adaptive laboratory evolution. [J]. Advances in Applied Microbiology, 2018, 104: 135-165. DOI: 10.1016/bs.aambs.2018.03.002.
[18] AI Chen-bing, MCCARTHY S, SCHACKWITZ W, MARTIN J, LIPZEN A, BLUM P. Complete genome sequences of evolved arsenate-resistant Metallosphaera sedula strains [J]. Genome Announcement, 2015, 3(5): e01142-15. DOI: 10.1128/genomeA.01142-15.
[19] MAEZATO Y, JOHNSON T, MCCARTHY S, DANA K, BLUM P. Metal resistance and lithoautotrophy in the extreme thermoacidophile Metallosphaera sedula [J]. Journal of Bacteriology, 2012, 194(24): 6856-6863. DOI: 10.1128/JB.01413-12.
[20] TAKAYANAGI S, KAWASAKI H, SUGIMORI K, YAMADA T, SUGAI A, ITO T, YAMASATO K, SHIODA M. Sulfolobus hakonensis sp. nov., a novel species of acidothermophilic archaeon [J]. International Journal of Systematic Bacteriology, 1996, 46(2): 377-382. DOI: 10.1099/00207713-46-2-377.
[21] LIANG Yu-ting, ZHU Shan, WANG Jun, AI Chen-bing, QIN Wen-qing. Adsorption and leaching of chalcopyrite by Sulfolobus metallicus YN24 cultured in the distinct energy sources [J]. International Journal of Minerals, Metallurgy, and Materials, 2015, 22(6): 549-552. DOI: 10.1007/s12613- 015-1106-y.
[22] MUIR M K, ANDERSEN T N. Determination of ferrous iron in copper-process metallurgical solutions by the o-phenanthroline colorimetric method [J]. Metallurgical and Materials Transactions B, 1977, 8(2): 517-518. DOI: 10.1007/bf02696942.
[23] CHIMPALEE N, CHIMPALEE D, SRITHAWEPOON S, PATJARUT T, BURNS D T. Flow-injection spectrophotometric determination of copper using bis(cyclohexanone) oxalyldihydrazone [J]. Analytica Chimica Acta, 1995, 304(1): 97-100. DOI: 10.1016/0003- 2670(94)00564-3.
[24] AI Chen-bing, YAN Zhang, ZHOU Han, HOU Shan-shan, CHAI Li-yuan, QIU Guan-zhou, ZENG Wei-min. Metagenomic insights into the effects of seasonal temperature variation on the activities of activated sludge [J]. Microorganisms, 2019, 7(12): E713. DOI: 10.3390/ microorganisms7120713.
[25] STOCKL M, TEUBNER N C, HOLTMANN D, MANGOLD K M, SAND W. Extracellular polymeric substances from Geobacter sulfurreducens biofilms in microbial fuel cells [J]. ACS Appl Mater Interfaces, 2019, 11(9): 8961-8968. DOI: 10.1021/acsami.8b14340.
[26] PENG Tang-jian, ZHOU Dan, LIU Xue-duan, YU Run-lan, JIANG Tao, GU Guo-hua, CHEN Miao, QIU Guan-zhou, ZENG Wei-min. Enrichment of ferric iron on mineral surface during bioleaching of chalcopyrite [J]. Transactions of Nonferrous Metals Society of China, 2016, 26(2): 544-550. DOI: 10.1016/S1003-6326(16)64143-2.
[27] PENG Tang-jian, ZHOU Dan, LIU Yuan-dong, YU Run-lan, QIU Guan-zhou, ZENG Wei-min. Effects of pH value on the expression of key iron/sulfur oxidation genes during bioleaching of chalcopyrite on thermophilic condition [J]. Annals of Microbiology, 2019, 69(6): 627-635. DOI: 10.1007/s13213-019-01453-y.
[28] FRANZMANN P D, HADDAD C M, HAWKES R B, ROBERTSON W J, PLUMB J J. Effects of temperature on the rates of iron and sulfur oxidation by selected bioleaching bacteria and archaea: Application of the Ratkowsky equation [J]. Minerals Engineering, 2005, 18: 1304-1314. DOI: 10.1016/j.mineng.2005.04.006.
(Edited by YANG Hua)
中文导读
极端嗜热菌群在70 °C柱浸反应器中浸出低品位硫化铜矿
摘要:本文研究了分别引入具有不同Cu2+抗性的M. sedula 菌株对极端嗜热菌群(由A. brierleyi DSM1651 和 M. hakonensis HO1-1组成)在70 °C柱浸反应器中浸出硫化铜矿的影响。引入Cu2+敏感的M. sedula copA 突变菌株对硫化铜矿的浸出几乎没有影响,铜浸出率没有提高。然而,引入Cu2+抗性M. sedula ARS50-2 能显著地强化浸出过程,铜浸出率在第58天提高了27.77%。第24 天后浸出液的pH表明M. sedula ARS50-2 的加入能提高极端嗜热菌群的硫氧化活性。定量PCR数据表明在浸出过程中,各反应器中矿物表面与浸出液中的极端嗜热菌群群落结构不同。极端嗜热菌在浸出过程中主要吸附在矿物表面。M. hakonensis HO 1-1是浸出过程中各个反应器矿物表面的主要菌株。M. sedula ARS50-2 在浸出的第34天仍然是矿物表面的主要的菌株。A. brierleyi DSM1651是浸出过程中各柱浸反应器浸出液中的主要菌株。相关数据表明加入高Cu2+抗性菌株有利于极端嗜热菌在高浓度Cu2+胁迫条件下对硫化铜矿的浸出。
关键词:极端嗜热菌;生物冶金;含铜硫化矿;铜离子抗性;微生物群落
Foundation item: Project(207154) supported by the Postdoctoral Research Funding of Central South University, China; Projects(31470230, 51320105006, 51604308) supported by the National Natural Science Foundation of China; Project (2017RS3003) supported by the Youth Talent Foundation of Hunan Province, China; Project(2018JJ2486) supported by the Natural Science Foundation of Hunan Province, China; Project(2018WK2012) supported by the Key Research and Development Projects in Hunan Province, China
Received date: 2019-06-28; Accepted date: 2020-03-13
Corresponding author: ZENG Wei-min, PhD, Professor; Tel: +86-13787288594; E-mail: zengweimin1024@126.com; ORCID: 0000- 0002-7155-6386

