�Ӻ�Cu, Ni, Fe��Zn���ӵ�Sarcheshmehͭ��ϳɼ���ҵ��Һ����ȡ�ͷ���Cu��Ni
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2020���2��
�������ߣ�Amirhossein SOEEZI Hadi ABDOLLAHI Sied Ziaedin SHAFAEI ESMAEIL RAHIMI
����ҳ�룺518 - 534
�ؼ��ʣ��ܼ���ȡ������ͭ��D2EHPA��LIX 984��Cyanex 302
Key words��solvent extraction; nickel; copper; D2EHPA; LIX 984; Cyanex 302
ժ Ҫ����D2EHPA��LIX 984��Cyanex 302��Chemorex CP 150��Acorga 5774Ϊ��ȡ����ú��Ϊϡ�ͼ�����ȡ��Sarcheshmehͭ��Cu(770 mg/L)��Ni(3200 mg/L)��Fe(800 mg/L)��Zn(200 mg/L)�ĺϳɼ���ҵ��Һ��Cu��Ni����ȡ�뷴�ͽ���ȫ���о�����ȡ�ο��ǵ�Ӱ�������л���ٶȺ�ʱ�䡢��ȡ��Ũ�ȡ�pH���¶ȡ����⣬���ò�ͬŨ�ȵĸ��������л���(���ᡢ���ᡢ�����������)��Cu��Ni���з��͡������������ʱ��Ϊ3 min������ٶ�Ϊ500 r/min���¶�Ϊ28 ��C�Լ�A/O��Ϊ1:1�������£�Cu��Ni����ȡ����ߡ����⣬��10%(v/v) LIX 984����Ч��ȡCu������30% (v/v) Cyanex 302����Ч��ȡNi����ȡCu��Ni�����pH�ֱ�Ϊ2~3��4~5���Ӻ�Ni��Cu��Zn��Fe���ӵĺϳɼ���ҵ��Һ�п�ѡ������ȡCu��Ni������ȡ�ʷֱ�Ϊ85%��93%��77%��82%���������ʵ�����������ϳɼ���ҵ��Һ��Ni�ķ����ʴ�96%��Cu�ķ�����Ϊ93%����ֱ���D2EHPA��LIX 984��Cyanex 302 3����ȡ����3������ȡZn��Fe��Cu��Ni 4��Ԫ�أ���������ȡ����͡�
Abstract: The extraction and stripping of Cu and Ni ions from synthetic and industrial solutions of Sarcheshmeh Copper Mine containing Cu (770 mg/L), Ni (3200 mg/L), Fe (800 mg/L) and Zn (200 mg/L) were comprehensively investigated using D2EHPA, LIX 984, Cyanex 302, Chemorex CP 150 and Acorga 5774 diluted in kerosene. The influential parameters such as mixing speed and time, concentration of extractant, pH and temperature were considered in extraction stage. Furthermore, stripping of Cu and Ni was examined using different inorganic and organic acids (sulfuric, hydrochloric, nitric and citric acids) with different concentrations. The results indicated that the highest extractions of Cu and Ni occurred within 3 min, with the mixing speed of 500 r/min, 28 ��C and A/O ratio of 1:1. Moreover, Cu was extracted by LIX 984 at the concentration of 10% (v/v), whereas Ni was extracted by Cyanex 302 at the concentration of 30% (v/v), efficiently. The optimal pH for Cu and Ni extraction is in ranges of 2-3 and 4-5, respectively. Cu and Ni were selectively extracted with the recoveries of 85%, 93% and 77%, 82% from synthetic and industrial solutions containing Ni, Cu, Zn and Fe ions, respectively. The results of optimal condition showed that 96% of Ni and 93% of Cu were stripped from the synthetic and industrial solutions. Finally, four elements Zn, Fe, Cu and Ni were extracted in three stages with D2EHPA, LIX 984 and Cyanex 302 extractants respectively with the least impurities.
Trans. Nonferrous Met. Soc. China 30(2020) 518-534
Amirhossein SOEEZI1, Hadi ABDOLLAHI1, Sied Ziaedin SHAFAEI1, Esmaeil RAHIMI2
1. School of Mining engineering, College of Engineering, University of Tehran, Tehran, Iran;
2. Department of Mining Engineering, Islamic Azad University, South Tehran Branch, Tehran, Iran
Received 16 April 2019; accepted 12 November 2019
Abstract: The extraction and stripping of Cu and Ni ions from synthetic and industrial solutions of Sarcheshmeh Copper Mine containing Cu (770 mg/L), Ni (3200 mg/L), Fe (800 mg/L) and Zn (200 mg/L) were comprehensively investigated using D2EHPA, LIX 984, Cyanex 302, Chemorex CP 150 and Acorga 5774 diluted in kerosene. The influential parameters such as mixing speed and time, concentration of extractant, pH and temperature were considered in extraction stage. Furthermore, stripping of Cu and Ni was examined using different inorganic and organic acids (sulfuric, hydrochloric, nitric and citric acids) with different concentrations. The results indicated that the highest extractions of Cu and Ni occurred within 3 min, with the mixing speed of 500 r/min, 28 ��C and A/O ratio of 1:1. Moreover, Cu was extracted by LIX 984 at the concentration of 10% (v/v), whereas Ni was extracted by Cyanex 302 at the concentration of 30% (v/v), efficiently. The optimal pH for Cu and Ni extraction is in ranges of 2-3 and 4-5, respectively. Cu and Ni were selectively extracted with the recoveries of 85%, 93% and 77%, 82% from synthetic and industrial solutions containing Ni, Cu, Zn and Fe ions, respectively. The results of optimal condition showed that 96% of Ni and 93% of Cu were stripped from the synthetic and industrial solutions. Finally, four elements Zn, Fe, Cu and Ni were extracted in three stages with D2EHPA, LIX 984 and Cyanex 302 extractants respectively with the least impurities.
Key words: solvent extraction; nickel; copper; D2EHPA; LIX 984; Cyanex 302
1 Introduction
Nowadays, some metals such as copper and nickel are extracted from their oxide and carbonate sources by the hydrometallurgical method [1-3]. Solvent extraction is based on a variety of organic solvents. The extractants with different formulae have been produced and utilized in order to extract elements such as copper, nickel and zinc from aqueous solution. The purpose of using solvent extraction method is to enrich the impregnated solution and reduce the impurities associated with valuable elements [4-6]. In general, the DSX (direct solvent extraction) method has been used for the selective extraction of different elements from the impregnated aqueous solution from leaching process (PLS) [7-10]. The DSX is associated with removing different elements from aqueous solution in different stages by different extractants (in single or combined forms of the extractants and directing them to the sedimentation stage such as electro-winning (EW) [11].
According to the previous studies, extractants like D2EHPA and Cyanex 272 have been used to separate cobalt, manganese and zinc ions from aqueous solution [12]. On the other hand, the extractants LIX [13] and Cyanex group [14,15] like Cyanex 301 and 302 have been utilized to extract copper and nickel ions, respectively. To date, several researches have been published for the separation of nickel and cobalt, but few of them have considered the extraction of nickel, copper, iron, zinc, and cobalt elements in one complex. REDDY et al [16] studied the extraction of copper, nickel, iron and cobalt elements using LIX 973 and LIX 984. They could extract copper using the extractant LIX group. However, after copper was extracted, the raffinate solution contained about 17 g/L of nickel. Furthermore, the concentration of sulfuric acid was determined as much as 157 g/L in copper stripping stage. KURSUNOGLU et al [10] investigated the nickel and cobalt extraction using the extractants Versatic 10 and TBP. The results of the experiments demonstrated that the best concentrations of Versatic 10 and TBP for nickel extraction were 15% and 5%, respectively. They could extract almost 100% of nickel in this concentration. But in this condition, other elements like cobalt, manganese and magnesium were also extracted as about 90%. They also used Cyanex 272 and TBP to extract cobalt from aqueous solution containing nickel, cobalt, manganese and calcium ions. Like the nickel extraction, this element was not separated selectively. MUBAROK and HANIF [17] studied the extraction of nickel and cobalt in a nitric environment using Cyanex 272 and Versatic 10. They could extract almost 99% of cobalt using Cyanex 272 in the concentration of 20%, whereas almost 80% of magnesium was also extracted. In other research, TANAKA et al [18] used LIX 84 to extract nickel from the electrolyte solution. According to studies conducted by SOEEZI et al [19], the extractant Chemorex CP 150 could have a significant effect on copper extraction from the pregnant aqueous solution obtained from leaching process (PLS). Moreover, the results also indicated that disturbing ions such as manganese can be very harmful for the process even in the mg/L (ppm)-scale, because this ion is able to oxidize the extractant and diminish the amount of recovery. According to the results of researches by MILLER et al [20] manganese is identified as the disturbing element in the electrolysis process which can be separated by the solvent extraction method. In addition, they reported the detrimental effects of the presence of manganese in the solvent extraction and electrowinning processes of copper which are listed as follows: (1) decreasing the chemical reaction rate; (2) decreasing the solvent capacity for the formation of copper chelate.
The main objective of the present study is selective solvent extraction and stripping of Cu and Ni from synthetic and industrial solutions containing Ni, Cu, Fe and Zn ions. A variety of solvent extractants including D2EHPA, LIX 984, Cyanex 302, Chemorex CP150, and Acorga 5774 were considered and the influential variables such as concentration and type of extractant, temperature, pH, mixing speed and time were also evaluated. It should be noted that DSX method was applied to extracting Cu, Ni, Zn and Fe in different stages. Firstly, Cu and Ni were selectively separated from each other in the synthetic solution containing two ions of Cu and Ni in binary system. The next stage was dealt with selective extraction of Cu and Ni from the synthetic aqueous solution containing four ions of Cu, Ni, Fe and Zn with similar concentrations of the solution obtained from the Sarcheshmeh Copper Refinery. For the next stage, the optimal condition was tested on the industrial sample of Sarcheshmeh Copper Mine and the selective extraction and stripping of the four abundant elements in the industrial solution (Zn, Fe, Ni and Cu) were evaluated. Finally, a new flowsheet was presented to selectively separate Cu and Ni from each other and from other disturbing elements such as Zn and Fe from industrial solution.
2 Experimental
2.1 Chemicals and sample
The organic solutions of D2EHPA (di-2- ethylhexylphosphoricacid), LIX 984, Acorga and Chemorex CP 150 (5-nonylsalicylaldloxime and 2-hydroxy-5-nonylacetophenone), Cyanex 302 (di-2,4,4-trimethylpentylmonothiophosphinic acid) with the purity of 97% were prepared from Cytec Co., Canada. All the extractants with different concentrations were diluted by kerosene. The CuSO4��5H2O, NiSO4��6H2O, FeSO4��7H2O and ZnSO4��7H2O were used to prepare the synthetic solutions containing Cu and Ni, and Cu, Ni, Fe and Zn ions, respectively. Also, industrial solution containing a variety of elements with different concentrations was prepared from the refinery solution of Sarcheshmeh Copper Mine. The concentrations of elements in the industrial solution are illustrated in Table 1.
Table 1 Concentrations of elements in refinery solution of Sarcheshmeh Copper Mine (mg/L)
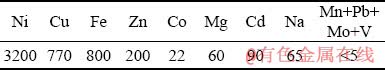
As indicated in Table 1, the industrial solution was prepared from the refinery of Sarcheshmeh Copper Mine which contains four dominant elements (Ni, Fe, Cu and Zn with concentration not less than 200 mg/L) and the other elements have lower concentration. Hence, these four elements were considered in this research. In the stripping stage, pure sulfuric (H2SO4), hydrochloric (HCl), nitric (HNO3) and citric (C6H8O7) acids were used. Analytical-grade reagents were used in these experiments without any purification. All the synthetic solutions were prepared with distilled and deionized water.
2.2 Method, instrument and analysis
Initially, synthetic Cu and Ni bearing solution (according to the solution concentration of Sarcheshmeh Copper Refinery) was prepared for evaluating the extraction of Cu and Ni from each other and analyzing the effect of various influential variables on the process. Cu and Ni were extracted using the solvent extraction method by different extractants D2EHPA, LIX 984, Cyanex 302, Acorga 5774 and Chemorex CP 150 with concentrations of 5%, 10%, 15%, 20%, 25% and 30%, respectively. To complete the process and enhance the extraction percentage, effective parameters including pH (2-6), mixing speed (100-1100) r/min), mixing time (1-15 min), and temperature (25-45 ��C) were considered. In the stripping stage, the type of acids (sulfuric, hydrochloric, nitric and citric acids) in different concentrations of 20-250 g/L was also taken into consideration. The next stage was to extract the Cu and Ni ions from the synthetic and industrial solutions containing four dominant elements of Ni (3200 mg/L), Cu (770 mg/L), Fe (800 mg/L) and Zn (200 mg/L). Three series stages of extraction and stripping were made to extract Cu and Ni ions from the synthetic and industrial solutions. It is noteworthy to mention that LIX 984 was stripped in two steps with different concentrations and Cyanex 302 was stripped in one step by sulfuric acid. To provide high precision results, the experiments were repeated two times and randomly three times and their results have been recorded in average.
The AAS analysis (Varian AA240FS) was applied to measuring the concentration of the elements in the aqueous solution and the extraction of the elements in the organic phase was calculated. All the experiments were carried out by the stirrer (Heidolph ST-1). The Memmert water bath (Water bath WNE 29) was used to make the temperature stable within the process [9,10].
The extractions of Cu and Ni were calculated by Eq. (1):
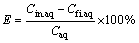 (1)
(1)
where Cin.aq represents the concentration of the elements in the primary impregnated aqueous solution (before the extraction stage) and Cfi.aq indicates the final concentration of the elements in the aqueous solution after the extraction stage. Equation (2) was used to calculate the distribution coefficient. The distribution coefficient is a factor by which the transition tendency of ions from aqueous to organic phase is measured. The distribution coefficient is calculated by Eq. (2), in which D is the distribution coefficient, Corg indicates the metal concentration in the organic phase and Caq represents the metal concentration in the aqueous phase [9,10].
 (2)
(2)
Also, Eq. (3) was used to calculate the separation factor. As this factor is far from 1, it indicates that a better extractant is selected, while the value of 1 implies that the extractant is indifferent to both types of the ions. In this equation, ��(Ni/Cu), D(Ni) and D(Cu) represent the separation factor of Ni from Cu ions and the distribution coefficients of the Ni and Cu ions, respectively [9,10].
��(Ni/Cu)= (3)
(3)
It is noteworthy to mention that the stripping rate of ions was calculated using the formula which is the opposite of Eq. (1).
3 Results and discussion
3.1 D2EHPA
This extractant is categorized in the group of cationic organophosphorus extractant. According to the literature reviews [21-24], this extractant has a high ability to extract Zn and Fe ions from solution. Since Ni is extracted by organophosphorus extractants like Cyanex 301, Cyanex 302 and Versatic 10 [25,26], it is predicted that D2EHPA plays an effective role in extracting Ni ion from solution. Therefore, the extractant D2EHPA was used to investigate the extraction of Cu and Ni ions from aqueous solution containing Cu and Ni ions in binary system. Figures 1(a) and (b) show the extractions of Cu and Ni ions using the D2EHPA in pH range of 2-6 with different concentrations of the extractant, respectively.

Fig. 1 Extractions of Cu (a) and Ni (b) from synthetic solution containing Cu and Ni elements by D2EHPA in pH range of 2-6 with different concentrations of extractant (28 ��C; 500 r/min; 3 min; O/A ratio 1:1)
As indicated in Fig. 1(a), D2EHPA did not have significant effect on Cu extraction. It is only capable of extracting the 45% of Cu from the aqueous solution containing Cu and Ni ions in the optimal condition. The results display that the highest extraction of Cu was achieved at pH=5 in the organic concentration of 15%. It is essential to mention that pH=6 reflects better results than other pH values in Cu extraction even though it is not suggested because of being close to copper sedimentation pH (according to the ��-pH diagram). However, almost 45% of Cu was extracted from the impregnated aqueous solution in this pH. The minimum of Cu extraction (20%) was recorded at pH=2 in the optimal condition. With pH increasing up to 5, it is observed that the extraction is enhanced. As the pH exceeds more than 6, the extraction is also reduced. Therefore, pH=5 was considered as the optimal pH for Cu extraction. The results were obtained at the D2EHPA concentration of 15%.
Figure 1(b) shows that D2EHPA is able to extract Ni from the aqueous solution containing Cu and Ni ions as about 50%. It should be pointed out that this amount of the extraction occurred at 28 ��C, mixing speed of 500 r/min, time of 3 min and organic concentration of 15%. The results specified that pH=5 provides the optimal condition for Ni extraction. The extractant D2EHPA is able to extract 47% of Ni under such conditions. Like Cu, the increment of pH has raised its extraction. The amount of Ni extraction at pH=6 has not been significantly increased in comparison with its previous pH. It is determined that the optimal pH for Ni extraction is 5. As the extractant D2EHPA did not properly extract Cu and Ni ions, other influential parameters such as temperature, mixing speed and time were not considered and optimized. This extractant is not suitable for the simultaneous and selective separation of Cu and Ni ions from aqueous solution. However, the literature reviews indicated that it is very efficient to remove Zn and Fe ions from aqueous solution [24,27].
3.2 LIX 984
3.2.1 Extraction stage
This extractant is a sub-division of the cationic chelating extractant. According to several researches, this extractant has a high ability to extract Cu and Fe ions at different levels of acidity (pH) [5,28]. In this part of research, the influence of this organic reagent on the extractions of Cu and Ni ions from the aqueous solution containing Cu and Ni ions was investigated. As mentioned before, all the tests were initially performed in the aqueous solution containing Cu and Ni ions in binary system in order to evaluate the effect of parameters on the extraction and stripping efficiency. Next, the influential parameters were optimized to extract Cu and Ni ions from the aqueous solution containing four elements of Cu, Zn, Ni and Fe. According to last research in Sungun Copper Mine it was found that Cu can be extracted by LIX 984 and Chemorex CP 150 at 3 min from its liquid solution [19]. So, the mixing time was fixed to be 3 min to optimize other parameters such as pH, mixing speed, temperature, and extractant concentration. It should be noted that Sungun Copper Mine and Sarcheshmeh Copper Mine are similar in genes. Figures 2(a) and (b) illustrate the results related to the extraction of Cu and Ni at different pH values and extractant concentrations. Figure 2(a) features out that the extractant LIX 984 is able to extract 100% of Cu in optimal condition. This amount of extraction occurred at pH=3. In contrast, the minimum Cu extraction is observed at pH=5-6. It is estimated that it is capable of extracting 97%-98% of Cu from aqueous solution. When the concentration of the extractant is moved up from 10% to 20% at pH=3, the Cu extraction is diminished to about 98%. As displayed in Fig. 2(b), the extractant LIX 984 is able to extract 68% of Ni in optimal condition. This extraction occurs at pH=5. When the pH increases to 6, the Ni extraction gets reduced and reaches 63%. The results of the experiment indicate that as the pH is decreased to 2, the Ni extraction reaches its minimum level which is almost 50%. As the pH increases from 2 to 3 and 4, the extraction is increased to 55% and 60%, respectively. It is noteworthy to mention that all the reported results were obtained at the extractant concentration of 10%. The results of these tests demonstrate that the amount of Ni extraction will be 40% when the concentration of the extractant is increased to 20% at pH=5. As a result, the Ni extraction gets decreased through enhancing or diminishing the pH in this concentration. As aforementioned, the pH values of 3 and 5 are identified as the best values to extract Cu and Ni ions from the aqueous solution containing Cu and Ni ions, respectively. According to the fact that Ni has a higher concentration in the solution, the rest of the tests were carried out at pH=5.
The mixing speed has been identified as another important parameter affecting the solvent extraction process. Therefore, the effects of mixing speed on Cu and Ni extractions were studied, and their results are shown in Figs. 2(c) and (d), respectively. The results of the experiments demonstrate that higher Cu extraction is observed at the mixing speed of 700 r/min than other mixing speed values. The Cu extraction is almost 99.8% at pH=5 and this mixing speed. As illustrated in Fig. 2(a), pH=3 is identified as the best pH for Cu extraction. However, with increasing the mixing speed at pH=5, the Cu extraction reaches nearly 100%. The Cu extraction is reduced to 97% through increasing the mixing speed up to 1100 r/min. On the other hand, the decrease of mixing speed to 100 r/min has reduced the Cu extraction to 90%. It should be pointed out that all the aforementioned results were obtained at the concentration of 10% of extractant. As the concentration of the extractant is increased at mixing speeds over 700 r/min, the Cu extraction has not been increased and it has passed a straight direction. However, the increment of the extractant concentration has increased the Cu extraction up to only 1%-2% at lower mixing speeds like 100 and 300 r/min. The highest Cu extraction happened at 700 r/min, but it is ignored because it is close to the mixing speed of 900 r/min at which it makes the solution become turbulent. So, the mixing speed of 500 r/min was selected as the optimal mixing speed. Another reason is that of Cu extraction at 500 r/min is close to that at 700 r/min. For this purpose, the mixing speed was set to be 500 r/min for the rest of experiments. In addition, the reduction of energy consumption is also considered as another reason of choosing the mixing speed of 500 r/min. Today, most of the processing plants are searching for methods to minimize the energy consumption. Thus, it is suggested that the mixing speed sets in the minimum value.

Fig. 2 Extractions of Cu and Ni from synthetic solution containing two elements of Cu and Ni by LIX 984 in different conditions
Figure 2(d) indicates that the extractant LIX 984 has ability to extract almost 70% of Ni ions in suitable condition. This extraction occurred at pH=5. Figure 2(d) illustrated the effect of mixing speed on the extraction of Ni ions from Cu-and Ni-bearing solution. As indicated, the best extraction of Ni (71%) was achieved at 700 r/min. Also, the extraction of Ni was 70% at 500 r/min. This figure clearly illustrated that the best extraction was at 700 r/min which was 71%. Whereas, the studies specified that the extraction of Ni was decreased to 58% at 1100 r/min. On the other hand, the Ni extraction was decreased to 45% and 55% at 100 and 300 r/min, respectively. All the aforementioned tests were carried out at the concentration of 10% of LIX 984. The results of the experiments figure out that the decrease and increase of the concentration of LIX 984 have reduced the Ni extraction. For example, the extraction was about 53% at the extractant concentration of 5% and 700 r/min. But, the extraction reached 71% at this mixing speed under the extractant concentration of 10%. The Ni extraction was diminished to 50% when the concentration of the extractant was increased to 20% at the similar mixing speed. As pointed out, the highest Ni extraction was obtained at 700 r/min. However, it is ignored because it is close to the mixing speed of 900 r/min at which the solution gets turbulent. Also, the Ni extraction at the mixing speed of 500 r/min is close to that at 700 r/min. Hence, the speed of 500 r/min was selected as the optimal mixing speed for the rest of the experiments.
Furthermore, mixing time was also considered as another significant parameter affecting the solvent extraction process. Figures 2(e) and (f) reveal the extraction results of elements Cu and Ni at different time and concentrations of the extractant LIX 984. As shown in Figs. 2(e) and (f), the Cu and Ni extractions were increased with increasing the mixing time by this extractant. In other words, the extractions of Cu and Ni ions were enhanced to 99.99% and 71.00% by increasing the mixing time to 7 min. In contrast, the extractions of both elements were decreased by increasing the time to 15 min. It is better to choose the lowest time which has the best percentage of extraction. For example, LIX 984 is able to extract 99.50% of Cu and 70.00% of Ni at 3 min. So, the duration of 3 min was specified as the optimal mixing time. Another reason of choosing the mixing time of 3 min is associated with the fact that Ni can be extracted more selectively from the aqueous solution containing Cu and Ni ions. More clearly, by increasing the time to 7 min, the Cu extraction was increased compared to Ni, but Ni extraction was not increased significantly and was about 0.25%. Therefore, the mixing time was selected to be 3 min in the rest of experiments. In other words, this time can be the optimal time to extract Cu and Ni ions. It is noteworthy to mention that all the results were at a concentration of 10% of extractant.
Also, the results of extraction, distribution coefficient and separation factor for Cu and Ni elements are presented in Table 2. This table postulates that the extractant LIX 984 is able to extract more than 99% of Cu at pH=2-3 and near 68% of Ni at pH=5. Larger distribution coefficient represents greater desire to transfer ions into the organic phase. According to the results, the best separation between the two elements of Cu and Ni in binary system occurs at pH=3 at which the separation factor is 8182.48. At pH=5-6, the minimum separation of two elements from each other was observed. For the purpose of simultaneous extraction of Cu and Ni elements, pH=5 was selected as the optimal pH for further investigation.
Another important parameter affecting the process efficiency is temperature. The results of extractions of Cu and Ni ions versus different temperatures are shown in Figs. 3(a) and (b), respectively. As shown in Fig. 3(a), Cu extraction decreases with increasing the temperature. The best temperature for extraction of this element is in the range of 25-30 ��C. When the temperature increases to 45 ��C, the extraction of Cu ions is decreased to 96.5%. Higher temperatures disrupt the chemical characteristic of LIX 984 and consequently reduce its performance. Therefore, the best temperature for this extractant is determined in the range of 25-30 ��C. Also, with increasing the temperature, the Ni extraction decreases which is similar to the Cu extraction behaviour (Fig. 3(b)). The optimal temperature for extraction of Ni is also in the range of 25-30 ��C. As the temperature rises to 45 ��C, Ni extraction is reduced to 68%.
Table 2 Extraction, distribution coefficient and separation factor of Cu and Ni ions from solution using LIX 984 (10% v/v, 28 ��C, 500 r/min, 3 min)
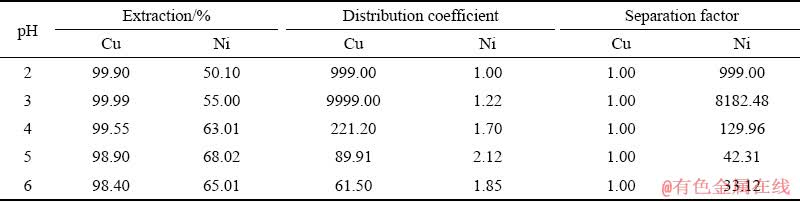
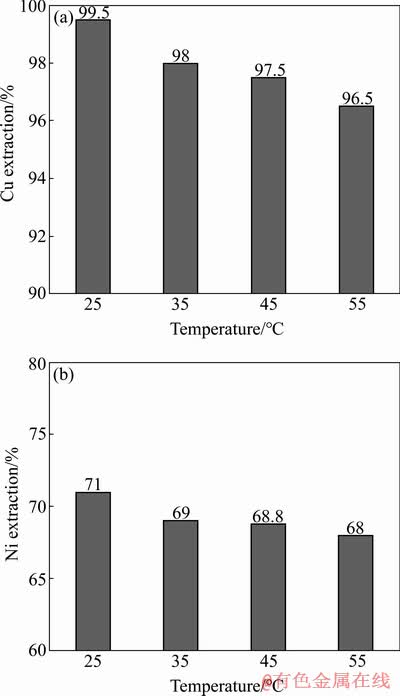
Fig. 3 Extractions of Cu (a) and Ni (b) from synthetic solution containing two elements of Cu and Ni by LIX 984 at different temperatures (pH=5, 500 r/min, 3 min, O/A ratio 1:1)
3.2.2 Stripping stage
As mentioned earlier, after the extraction stage and optimizing the effective parameters, the stripping of the elements from the extractants was investigated. Different acids such as sulfuric acid, hydrochloric acid, nitric acid, and citric acid were used at various concentrations and the effects of each acid on the stripping of Cu and Ni were evaluated. The results are shown in Fig. 4.

Fig. 4 Stripping rates of Cu and Ni with different acids and concentrations (28 ��C, mixing speed 500 r/min, 3 min, concentration of extractant 10%, O/A ratio 1:1)
As illustrated in Fig. 4(a), the Cu stripping rate can be increased or decreased as the result of changing the type of acid in the stripping stage. It is evidently concluded that the enhancement of concentration of hydrochloric acid from 130 to 180 g/L is able to reach Cu stripping rate from 82% to 90%. The results also showed that the stripping rate of Cu with nitric acid is lower than that with hydrochloric and sulfuric acids. Thus, 92%-94% of Cu was stripped at the concentration of 210 g/L sulfuric and hydrochloric acids, respectively. But using nitric acid only 78% of Cu was stripped with the same concentration. Besides, increasing the concentration of hydrochloric acid from 210 to 250 g/L has increased stripping rate from 93% to 94% approximately, respectively. Subsequently, the organic acid (citric acid) was not effective in stripping stage and it was not able to compete with other inorganic acids. In the best condition, its Cu stripping rate reached 61%. Due to the fact that sulfuric and hydrochloric acids affect the Cu stripping process efficiently, these two acids were utilized to strip Ni from organic phase. Figure 4(b) demonstrates that changing the type of acid did not remarkably influence the Ni stripping rate. There is no any meaningful difference between hydrochloric acid and sulfuric acid in the stripping rate of Ni. The results of the experiment reveal that almost 84% of Ni can be separated from the organic phase at the concentration of 20 g/L sulfuric acid. As the acid concentration increases to 40 g/L, more Ni is separated to the aqueous solution and the stripping rate reaches 91%. When the concentration of hydrochloric acid reaches 40 g/L, it is able to strip Ni up to 92%. According to Fig. 4(b), it can be seen that low concentrations of hydrochloric or sulfuric acids can be used to remove Ni from the organic phase and to separate it from Cu. It is observed that at concentrations of 20-40 g/L, more than 80% of Ni is removed while the amount of Cu introduced into the aqueous phase is less than 15%. It is noteworthy to mention that the stripping rates with hydrochloric acid at the concentrations of 20 and 30 g/L are almost similar to those with sulfuric acid. So, sulfuric acid was selected for stripping of Ni in further experiments.
Sulfuric acid is cheaper and more accessible than hydrochloric acid, and most of copper melting plants release plenty of SO2 into the environment because of roasting and smelting of sulfide ores. Plants producing sulfuric acid are usually established adjacent to the plants of roasting and smelting of sulfide ores in order to decrease the emission of SO2 through the environment [28-30]. Hence, sulfuric acid was selected as a suitable acid for the stripping stage in this research. Figure 5 represents the extraction data and their conditions and the stripping of Cu and Ni elements from the organic phase. As observed in Fig. 5, solution containing Cu and Ni ions with respective concentrations of 770 mg/L and 3200 mg/L is entered into the mixer and is separated by LIX 984 with the concentration of 10% at pH=2.5. In this stage, about 99.5% of Cu and 71% of Ni are extracted. After that, the organic phase is entered into stripping stage. Two stripping steps at different concentrations of sulfuric acid are applied for the selective extraction of these elements. Initially, the organic phase is washed by sulfuric acid with the concentration of 40 g/L. In this step, approximately 90% of Ni and 7% of Cu are separated from the organic phase. Once more, the organic phase is washed by sulfuric acid with the concentration of 210 g/L. In this step, about 97% Cu and 9.9% Ni are stripped from extractant, respectively.
3.3 Cyanex 302
3.3.1 Extraction stage
This extractant is categorized in the group of cationic organophosphorus extractant. According to the literature reviews, this extractant has a high ability to extract Ni, Mg and Co ions from aqueous solution. Therefore, this extractant was used to extract Ni ions from the solution [10]. The extraction results of Cu and Ni ions by this extractant at different concentrations and pH values are shown in Figs. 6(a) and (b), respectively.

Fig. 5 Selective extraction and stripping of Ni and Cu using LIX 984 from aqueous solution containing Cu and Ni ions (28 ��C, pH=2.5, 500 r/min, 3 min, concentration of extractant 10%, O/A ratio 1:1)
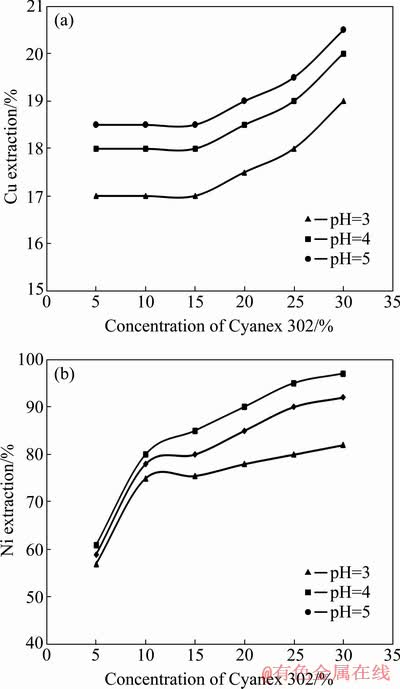
Fig. 6 Extractions of Cu (a) and Ni (b) from synthetic solution containing Cu and Ni elements by Cyanex 302 extractant at different concentrations and pH (28 ��C, 500 r/min, 3 min, O/A ratio 1:1)
As observed in Fig. 6(b), the extractant Cyanex 302 is capable of extracting the 97% of Ni ions from the aqueous solution. This extraction was observed at pH=4 and the extractant concentration of 30%. Also, this extractant is able to extract 95% of Ni ions at pH=4 with the concentration of 25%. The results show that decreasing the concentration of the extractant to 5% is directed into reducing the Ni extraction in all pH values. Remarkably, the extractant Cyanex 302 is able to extract only 20.5% of Cu ions from the solution (Fig. 6(a)).
The effect of mixing speed on the extractions of Cu and Ni ions is presented in Fig. 7. It is postulated that with increasing the mixing speed from 500 to 700 r/min, the extraction of Ni ions is enhanced. Likewise, the mixing speed increment from 500 to 700 r/min at the extractant concentration of 30% leads to the increase Ni extraction from 97% to 97.5%. The results indicate that the increase of mixing speed at any concentration from the organic phase is directed into enhancing the Ni extraction. The minimum Cu extraction is achieved by the extractant Cyanex 302. At any mixing speed, the Cu extraction from the aqueous solution is less than 20%. The mixing speed enhancement increases the extractions of Ni and Cu ions. Evidently, with increasing the mixing speed from 500 to 700 r/min, the Cu and Ni extractions increase. The mixing speed of 500 r/min with the minimum Cu extraction was considered for the selective extraction in order to prevent more Cu extraction from the aqueous solution.
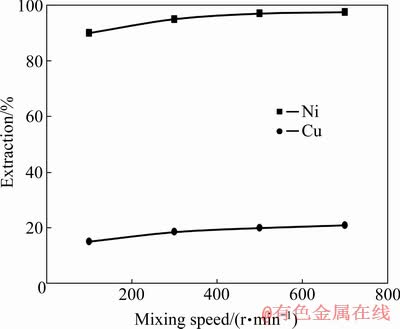
Fig. 7 Extractions of Cu and Ni by extractant Cyanex 302 at different mixing speeds (pH=4, 3 min, O/A ratio 1:1, concentration of extractant 30%)
The effect of mixing time on the extractions of Cu and Ni ions from the solution containing Cu and Ni ions is presented in Fig. 8. As observed, increasing the mixing time is directed into increasing the extractions of Cu and Ni ions by Cyanex 302. Clearly, as the mixing time is increased from 3 to 7 min, the extraction of Ni ions is increased by about 0.5%. However, this increment leads to higher Cu extraction of 23%. As a result, it was determined that the duration of 3 min is the best choice for the extraction of Ni ions from the aqueous solution containing Cu and Ni ions. In this condition, the extraction of Cu is lower than the others.
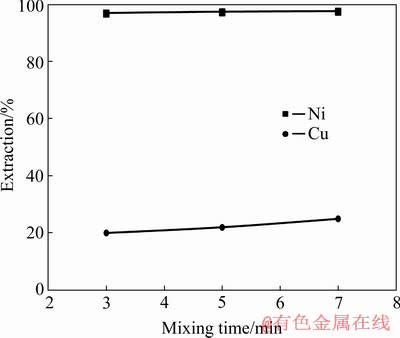
Fig. 8 Extractions of Cu and Ni by extractant Cyanex 302 at different mixing time (pH=4, 500 r/min, O/A ratio 1:1, concentration of extractant 30%)
Also, the extraction, distribution coefficient and separation factor for Cu and Ni ions are presented in Table 3. This table figures out that the extractant Cyanex 302 is able to extract about 19.50% of Cu and 97.00% of Ni at pH=4. Larger distribution coefficient represents greater desire to transfer ion into the organic phase. According to the results of its separation factor, it can be seen that the highest Ni separation from Cu occurs at pH=4, which is 134.71. According to the results, the best separation between the Ni and Cu ions occurs at pH=4, in which the separation factor is 134.71. At pH=3, the minimum separation of two elements from each other was observed.
The increase of the temperature increased the extractions of Cu and Ni ions from the solution (Fig. 9). The extraction of Ni ions was increased by 0.7% through raising the temperature from 25 to 45 ��C. However, this increase enhanced the Cu extraction to 20.6%. Regarding to the Cu extraction, it was specified to set the temperature at 25-30 ��C for the selective extraction of Ni ions from the aqueous solution containing Cu and Ni ions. According to the results, this extractant is considered as a suitable organic reagent to extract Ni ions selectively from the aqueous solution containing Cu and Ni ions. As displayed in Fig. 8, Cyanex 302 extracted almost 97% of Ni ions from the aqueous solution containing these two ions in optimal condition (environment temperature, mixing speed of 500 r/min, and extractant concentration of 30%). In such condition, about 19.5% of Cu was extracted.
Table 3 Extraction, distribution coefficient and separation factor of Cu and Ni ions from solution by Cyanex 302 (extractant concentration 30%, 28 ��C, 500 r/min, 3 min)
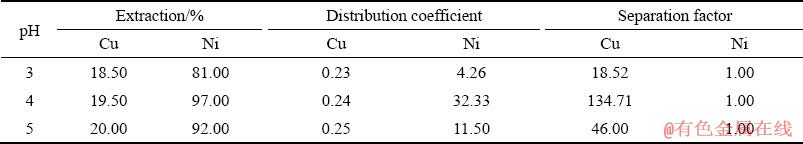

Fig. 9 Extractions of Cu (a) and Ni (b) ions using Cyanex 302 extractant at different temperatures (pH=4, 500 r/min, O/A ratio 1:1, concentration of extractant 30%)
3.3.2 Stripping stage
The current research considered the stripping of Cyanex 302 by sulfuric and hydrochloric acids in different concentrations of 40 to 210 g/L (extracted Ni and Cu ions: 83% and 16%, respectively). The stripping results of Ni and Cu ions using sulfuric and hydrochloric acids in different concentrations are presented in Fig. 10. As illustrated, changing the type of acid and concentration did not significantly affect the Ni stripping. However, increasing the concentration of sulfuric and hydrochloric acids increased the Cu stripping rate. As Ni stripping is not changed remarkably, the Ni stripping rate was raised by 1% approximately by increasing the sulfuric acid concentration from 130 to 210 g/L. Almost 99% of Ni and 18% of Cu were stripped at the sulfuric acid concentration of 80 g/L. Evidently, the Cu stripping rate was decreased through reducing the acid concentration from 130 g/L even though the Ni stripping rate is not changed. As the concentration of sulfuric acid is reduced from 80 to 40 g/L, the Ni stripping starts decreased from 99% to 98%. For this purpose, the 80 g/L of sulfuric acid was selected as the optimal concentration of stripping reagent. On the other hand, the stripping rate of Cu was 38% by hydrochloric acid in the concentration of 80 g/L, which was two times more than that of sulfuric acid. Eventually, sulfuric acid was selected as the best stripping reagent in the stripping stage.

Fig. 10 Stripping rates of Cu and Ni from Cyanex 302 extractant in different concentrations of sulfuric and hydrochloric acids (500 r/min, 3 min, O/A ratio 1:1, extractant concentration 30%)
3.4 Acorga 5774 and Chemorex CP 150
The extractants Acorga 5774 and Chemorex CP 150 are categorized as cationic chelating extractants. The natures of these two extractants are very close to those of the LIX group. Therefore, the optimal condition obtained from the experiments by LIX 984 is considered for these extractants. All parameters such as temperature, mixing speed and time and O/A ratio were constant except pH and extractant concentration which had intense effect on the extraction rate. So, the parameters pH and concentration of extractant were considered as variables for these two extractants to compare their extractions with that of LIX 984. The extraction of Cu and Ni ions by Acorga 5774 and Chemorex CP 150 versus different extractant concentrations and pH values are presented in Figs. 11(a) and (b), respectively.
As observed, the highest extraction of Cu ions was achieved by the extractant Acorga 5774 at the concentration of 15% and pH=3. In such conditions, the Cu extraction is about 95% which is 4% less than that of the extractant LIX 984. It should be pointed out that by increasing the pH, the extraction is reduced to 90%. According to the results, LIX 984 extracted the highest amount of Cu ions compared with other extractants. The reason of this increase can be related to the variety of the nature or purity of the LIX 984. Considerably, Acorga 5774 was able to extract 52% of Ni ions at pH=5 with concentration of 15%. A simple comparison between LIX 984 and this extractant reveals that Acorga 5774 has a less extraction capacity than LIX 984 for both Ni and Cu elements.

Fig. 11 Extractions of Cu and Ni from synthetic solution by Acorga 5774 (a) and Chemorex CP 150 (b) extractants at different concentrations and pH values (25 ��C, 500 r/min, 3 min, O/A ratio 1:1)
Figure 11(b) shows that the maximum Cu extraction happens at the concentration of 15% and pH=3 by the extractant CP 150. The Cu extraction is about 95% in such conditions which is the same as that of the extractant Acorga 5774. Therefore, it can be concluded that CP 150 acts just like the extractant Acorga 5774 to extract Cu ions from aqueous solution. However, the extractant CP 150 is not suitable organic reagent to extract Ni ions even in the best condition (only 35%). The extractant CP 150 acts weakly to extract Ni ions compared with LIX 984. The extractants Chemorex CP 150 and Acorga 5774 and LIX 984 are chemically so similar to each other. Their only difference is the company in which they are produced and their purity. According to the results, it was observed that the extractant LIX 984 is able to extract Cu and Ni ions better than Chemorex CP 150 and Acorga 5774 and therefore, the LIX 984 was selected as the best extractant between these three similar organic reagents.
3.5 Selective separation of Cu and Ni from synthetic solution containing Cu, Ni, Fe and Zn ions
This part of research highlights the selective separation of Cu and Ni ions from the synthetic solution containing ions of Cu, Ni, Fe and Zn (at concentrations similar to those from the refinery of Sarcheshmeh Copper Mine, Ni: 3200 mg/L, Cu: 770 mg/L, Fe: 800 mg/L, and Zn: 200 mg/L) using extractants D2EHPA, LIX 984, and Cyanex 302 in the three series steps.
As shown in Fig. 12, Cu and Ni ions are selectively extracted by the extractants D2EHPA, LIX 984, and Cyanex 302 in different three steps. The results of the experiments figure out that the extraction of Fe and Zn ions is performed by D2EHPA at pH=2-3. Initially, the impregnated aqueous solution containing Cu, Ni, Fe and Zn is mixed with the extractant D2EHPA (concentration of 15%). After mixing and the first extraction step, 96% of Zn, 75% of Fe, 8% of Cu and 8% of Ni ions are extracted. In the second step, the aqueous solution is mixed with the LIX 984 (concentration of 10%) at pH=2.5. In this step, 91% of Cu, 12.5% of Fe, 16% of Ni and 0.1% of Zn ions are extracted. In the last step of extraction stage, the solution is mixed with Cyanex 302. In this case, 95% of Ni, 5% of Fe, 0.9% of Cu and 0.03% of Zn are extracted. In the stripping stage, the LIX 984 is washed in two steps by sulfuric acid at two concentrations of 40 and 210 g/L. So, 14% of Ni and 90% of Cu are separated in two different stripping steps. Finally, 96% of Ni present in Cyanex 302 is stripped by sulfuric acid in the concentration of 80 g/L in the third steps. Totally, 96% of Ni and 90% of Cu are separated successfully from each other and can be transferred to the EW stages. It is noteworthy to mention that the extractant D2EHPA is just used to extract Fe and Zn ions from the synthetic solution it for the next steps of extraction by extractants LIX984 and then Cyanex 302.
3.6 Selective separation of Cu and Ni ions from industrial refinery solution
According to the results obtained from the previous experiments on the synthetic solution, the selective separation of Cu and Ni ions from the industrial solution (elements and their concentrations presented in Table 1) was investigated using D2EHPA, LIX 984, and Cyanex 302 with the same process pattern. The results are show in Fig. 13. The extraction and stripping rate are decreased because of the variety of elements presented in the solution. Also, different cations, rare earth elements, anions and complex components are presented in the industrial solution which are not listed in Table 1. So, these elements, ions or components, can affect the test results and decrease the extraction efficiency. For example, the extraction of Cu ions was 85% using LIX 984, while 89% of Ni ions was extracted by Cyanex 302. In comparison with the synthetic solution, the extractions of Cu and Ni ions were reduced by about 6% and 6%, respectively. It is noteworthy to mention that the stripping rates of Cu and Ni were also reduced in comparison with those of the synthetic solution. Indeed, stripping rates of Cu and Ni were decreased from 90% to 88% and from 96% to 90%, respectively. Totally, 82% of Ni and 77% of Cu are separated successfully from each other and can be transferred to the EW stages. Also, the extractant D2EHPA was used to extract Fe and Zn elements as impurities from the industrial solution for the next steps of extraction by extractants LIX984 and then Cyanex 302.

Fig. 12 Extraction and stripping of Cu and Ni from synthetic solution containing Cu, Ni, Fe and Zn ions (Extraction parameters: 28 ��C, 500 r/min, 3 min, O/A ratio 1:1; Stripping parameters: 28 ��C, 500 r/min, 3 min, A/O ratio 1:1)
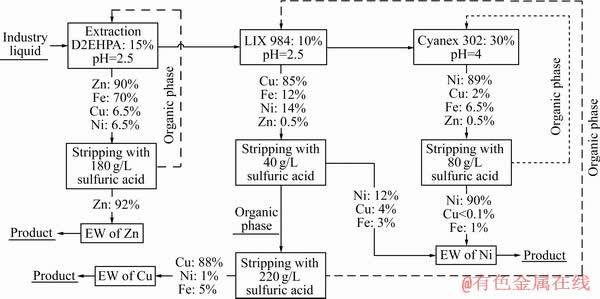
Fig. 13 Selective extraction and stripping of Cu and Ni from industrial solution of Sarcheshmeh Copper Mine (Extraction parameters: 28 ��C, 500 r/min, 3 min, O/A ratio 1:1; Stripping parameters: 28 ��C, 500 r/min, 3 min, A/O ratio 1:1)
Table 4 Some previous studies related to present work

According to the results of the experiments, it can be concluded that using the three steps of the solvent extraction process with D2EHPA, LIX 984, and Cyanex 302 and the two steps of the stripping process with sulfuric acids with different concentrations, four dominant ions of the industrial solution containing Cu, Ni, Fe and Zn ions can be separated effectively. The results of some studies related to the present work are listed in Table 4. As shown, this is for the first time that selective solvent extraction and stripping of four ions Cu, Ni, Fe and Zn from two kinds of synthetic and industrial solutions have been comprehensively investigated.
4 Conclusions
(1) Selective solvent extraction and stripping of Cu and Ni elements from synthetic and industrial solutions containing Ni, Cu ions and Ni, Cu, Fe and Zn ions were comprehensively investigated. Also, the effects of different extractants including D2EHPA, LIX 984, Cyanex 302, Chemorex CP 150 and Acorga 5774 were studied and the effective parameters were optimized.
(2) The results show that using the three steps of the extraction process with D2EHPA, LIX 984, and Cyanex 302 in series pattern and the two steps of the stripping process with sulfuric acid at two different concentrations, Cu and Ni ions can be separated effectively from synthetic and industrial solutions containing four main elements of Cu, Ni, Zn and Fe.
(3) The extractant D2EHPA recovered about 75% of Fe and 96% of Zn ions at pH=2.5 from synthetic solution and also 70% of Fe and 90% of Zn ions from industrial solution. LIX 984 can perfectly extract Cu ions as much as 99.9% at pH=2-3. As the pH is increased to 5, this extractant is able to extract 70% of Ni ions from aqueous solution. Cyanex 302 can extract 97% of Ni ions at the concentration of 30% of the organic phase and pH=4 with minimum amount of Cu extraction.
(4) In the synthetic solution, for the first step of extraction, D2EHPA extracted almost 96% of Zn, 75% of Fe, 8% of Cu and 8% of Ni ions. In the second step, 91% of Cu, 12.5% of Fe, 16% of Ni and 0.1% of Zn ions were extracted with the LIX 984. In the third step, about 95% of Ni, 0.9% of Cu, 5% of Fe and 0.03% of Zn ions were extracted by Cyanex 302. In the industrial solution, for the first step of extraction, D2EHPA extracted almost 90% of Zn, 70% of Fe, 6.5% of Cu and 6.5% of Ni ions. In the second step, 85% of Cu, 12% of Fe, 14% of Ni and 0.5% of Zn ions were extracted with the LIX 984. In the third step, about 89% of Ni, 2% of Cu, 6.5% of Fe and 0.5% of Zn ions were extracted by Cyanex 302.
Acknowledgments
The authors would like to appreciate all the ones who contributed to prepare this work specifically Mr. Mojtaba KARAMI and Mr. Hossein LOTFI for preparing the chemicals.
References
[1] DEVI N. Solvent extraction and separation of copper from base metals using bifunctional ionic liquid from sulfate medium [J]. Transactions of Nonferrous Metals Society of China, 2016, 26: 874-881. https://doi.org/10.1016/S1003- 6326(16)64179-1.
[2] BALAZ P, ALACOVA A, ACHIMOVICOVA M, FICERIOVA J, GODOCIKOVA E. Mechanochemistry in hydrometallurgy of sulphide minerals [J]. Hydrometallurgy, 2005, 77: 9-17. https://doi.org/10.1016/j.hydromet.2004.09. 009.
[3] SHEN Yong-feng, XUE Wen-ying, LI Wei, LI Shan-de, LIU Xiang-hua. Recovery of Mn2+, Co2+ and Ni2+ from manganese nodules by redox leaching and solvent extraction [J]. Transactions of Nonferrous Metals Society of China, 2007, 17: 1105-1111. https://doi.org/10.1016/S1003-6326 (07) 60233-7.
[4] LI Li-qing, ZHONG Hong, CAO Zhan-fang, YUAN Lu. Recovery of copper (II) and nickel (II) from plating wastewater by solvent extraction [J]. Chinese Journal of Chemical Engineering, 2011, 19: 926-930. https://doi.org/ 10.1016/S1004-9541(11)60073-6.
[5] LIU Qing-ming, YU Run-lan, QIU Guan-zhou, FANG Zheng, CHEN Ai-liang, ZHAO Zhong-wei. Optimization of separation processing of copper and iron of dump bioleaching solution by LIX 984N in Dexing Copper Mine [J]. Transactions of Nonferrous Metals Society of China, 2008, 18: 1258-1261. https://doi.org/10.1016/S1003-6326 (08)60213-7.
[6] GUAN Qing-jun, WEI Sun, ZHOU Gui-ying, LIU Jia-peng, YIN Zhi-gang. Recovery of cobalt and nickel in the presence of magnesium and calcium from sulfate solutions by Versatic 10 and mixtures of Versatic 10 and Cyanex 301 [J]. Transactions of Nonferrous Metals Society of China, 2016, 26: 865-873. https://doi.org/10.1016/S1003-6326(16) 64178-X.
[7] QIU Yin-xuan, YANG Li-mei, HUANG Song-tao, JI Zhong-guang, LI Yan. The separation and recovery of copper (II), nickel (II), cobalt (II), zinc (II), and cadmium (II) in a sulfate-based solution using a mixture of Versatic 10 acid and Mextral 984H [J]. Chinese Journal of Chemical Engineering, 2017, 25: 760-767. https://doi.org/10.1016/j. cjche.2016.10.013.
[8] PANDA S, PARHI P K, PRADHAN N, MOHAPATRA U B, SUKLA L B, PARK K H. Extraction of copper from bacterial leach liquor of a low grade chalcopyrite test heap using LIX 984N-C [J]. Hydrometallurgy, 2012, 121-124: 116-119. https://doi.org/10.1016/j.hydromet.2012.03.008.
[9] ASHKENNY M, IBANA D, BARNARD K. Reagent selection for recovery of nickel and cobalt from nitric acid nickel laterite leach solutions by solvent extraction [J]. Minerals Engineering, 2015, 77: 42-51. https://doi.org/10. 1016/j.mineng.2015.02.010.
[10] KURSUNOGLU S, ICHLAS Z T, KAYA M. Solvent extraction process for the recovery of nickel and cobalt from Caldag laterite leach solution: The first bench scale study [J]. Hydrometallurgy, 2017, 169: 135-141. https://doi.org/10. 1016/j.hydromet.2017.01.001.
[11] JHA M K, GUPTA D, CHOUBEY P, KUMAR V, JEONG J, LEE J C. Solvent extraction of copper, zinc, cadmium and nickel from sulfate solution in mixer settler unit (MSU) [J]. Separation and Purification Technology, 2014, 122: 119-127. https://doi.org/10.1016/j.seppur.2013.10.045.
[12] MISHRA R K, ROUT P C, SARANGI K, NATHSARMA K C. Solvent extraction of zinc, manganese, cobalt and nickel from nickel laterite bacterial leach liquor using sodium salts of TOPS-99 and Cyanex 272 [J]. Transactions of Nonferrous Metals Society of China, 2016, 26: 301-309. https://doi.org/ 10.1016/S1003-6326(16)64119-5.
[13] EL-NAGGAR M, EL-SHERIEF E, MEKHEMAR H. Performance of geopolymers for direct immobilization of solvent extraction liquids: Metakaolin/LIX-84 formulations [J]. Journal of Hazardous Materials, 2018, 360: 670-680. https://doi.org/10.1016/j.jhazmat.2018.08.057.
[14] DESSOUKY S I, EL-NADI Y A, AHMEDA I M, SAAD E A, DAOUDA J A. Solvent extraction separation of Zn(II), Fe(II), Fe(III) and Cd(II) using tributylphosphate and Cyanex 921 in kerosene from chloride medium [J]. Chemical Engineering and Processing, 2008, 47: 177-183. https://doi.org/10.1016/ j.cep.2007.03.002.
[15] WANG L Y, LEE M S. Separation of Co(II) and Ni(II) from chloride leach solution of nickel laterite ore by solvent extraction with Cyanex 301 [J]. International Journal of Mineral Processing, 2017, 166: 45-52. https://doi.org/10. 1016/j.minpro.2017.07.004.
[16] REDDY R, PARK K H, MOHAPATRA D. Process development for the separation and recovery of copper from sulphate leach liquors of synthetic Cu�CNi�CCo�CFe matte using LIX 84 and LIX 973N [J]. Hydrometallurgy, 2007, 87: 51-57. https://doi.org/10.1016/j.hydromet.2007.01.004.
[17] MUBAROK M Z, HANIF L I. Cobalt and nickel separation in nitric acid solution by solvent extraction using Cyanex 272 and Versatic 10 [J]. Procedia Chemistry, 2016, 19: 743-750. https://doi.org/10.1016/j.proche.2016.03.079.
[18] TANAKA M, HUANG Y, YAHAGI T, HOSSAIN M K, SATO Y, NARITA H. Solvent extraction recovery of nickel from spent electroless nickel plating baths by a mixer-settler extractor [J]. Separation and Purification Technology, 2008, 62: 97-102. https://doi.org/10.1016/j.seppur.2007.12.022.
[19] SOEEZI A, RAHIMI E, MOHAGHEGH N. Investigating the effect of concentration and stirring time on copper extraction process from pregnant liquid solution (pls), case study: Sungun copper mine [J]. Chem Metall, 2015, 2: 33-35. http://dx.doi.org/10.15242/IJRCMCE.E0915008.
[20] MILLER G M, READETT D J, HUTCHINSON P. Experience in operating the girilambone copper SX-EW plant in changing chemical environments [J]. Minerals Engineering, 1997, 10: 467-481. https://doi.org/10.1016/ S0892-6875(97)00026-5.
[21] JAFARI H, ABDOLLAHI H, GHARABAGHI M, BALESINI A. Solvent extraction of zinc from synthetic Zn-Cd-Mn chloride solution using D2EHPA: Optimization and thermodynamic studies [J]. Separation and Purification Technology, 2018, 197: 210-219. https://doi.org/10.1016/ j.seppur.2018.01.020.
[22] VAHIDI E, RASHCHI F, MORADKHANI D. Recovery of zinc from an industrial zinc leach residue by solvent extraction using D2EHPA [J]. Minerals Engineering, 2009, 22: 204-206. https://doi.org/10.1016/j.mineng.2008.05.002.
[23] CHENG C Y, BARNARD K, ZHANG W S, ZHU Z W, PRANOLO Y. Recovery of nickel, cobalt, copper and zinc in sulphate and chloride solutions using synergistic solvent extraction [J]. Chinese Journal of Chemical Engineering, 2016, 24: 237-248. https://doi.org/10.1016/j.cjche.2015. 06.002.
[24] MELLAH A, BENACHOUR D. The solvent extraction of zinc and cadmium from phosphoric acid solution by di-2-ethyl hexyl phosphoric acid in kerosene diluent [J]. Chemical Engineering and Processing, 2006, 45: 684-690. https://doi.org/10.1016/j.cep.2006.02.004.
[25] SULAIMAN R, OTHMAN N. Synergistic green extraction of nickel ions from electroplating waste via mixtures of chelating and organophosphorus carrier [J]. Hazardous Materials, 2017, 340: 77-84. https://doi.org/10.1016/j. jhazmat. 2017.06.060.
[26] TSAKIRIDIS P E, AGATZINI S L. Simultaneous solvent extraction of cobalt and nickel in the presence of manganese and magnesium from sulfate solutions by Cyanex 301 [J]. Hydrometallurgy, 2004, 72: 269-278. https://doi.org/10. 1016/S0304-386X(03)00180-4.
[27] MANSUR M, ROCHA S, SILVA M, BENEDETTO D S. Selective extraction of zinc(II) over iron(II) from spent hydrochloric acid pickling effluents by liquid�Cliquid extraction [J]. Journal of Hazardous Materials, 2008, 150: 669-678. https://doi.org/10.1016/j.jhazmat.2007.05.019.
[28] ONI A O, FADARE D A, SHARMA S, RANGAIAH G P. Multi-objective optimisation of a double contact double absorption sulphuric acid plant for cleaner operation [J]. Journal of Cleaner Production, 2018, 181: 652-662. https:// doi.org/10.1016/j.jclepro.2018.01.239.
[29] ZHOU E, HE Y F, MA X X, LIU G Q, HUANG Y, CHEN C, WANG W. Study of the combination of sulfuric acid treatment and thermal regeneration of spent powdered activated carbons from decolourization [J]. Chemical Engineering & Processing: Process Intensification, 2017, 121: 224�C231. https://doi.org/10.1016/j.cep.2017.09.008.
[30] TAIEB D, BRAHIM A B. Electrochemical method for sulphur dioxide removal from flue gases: Application on sulphuric acid plant in Tunisia [J]. Comptes Rendus Chimie, 2013, 16: 39-50. https://doi.org/10.1016/j.crci.2012.08.009.
[31] SINHA M K, SAHU S K, PRAMANIK S, PRASAD L, PANDEY B D. Recovery of high value copper and zinc oxide powder from waste brass pickle liquor by solvent extraction [J]. Hydrometallurgy, 2016, 165: 182-190. https:// doi.org/10.1016/j.hydromet.2015.09.012.
[32] HACHEMAOUI A, BELHAMEL K. Simultaneous extraction and separation of cobalt and nickel from chloride solution through emulsion liquid membrane using Cyanex 301 as extractant [J]. International Journal of Mineral Processing, 2017, 161: 7-12. https://doi.org/10.1016/j. minpro.2017.02. 002.
[33] SINHA M, SAHU S, MESHRAM P, PANDEY B. Solvent extraction and separation of zinc and iron from spent pickle liquor [J]. Hydrometallurgy, 2014, 147-148: 103-111. https:// doi.org/10.1016/j.hydromet.2014.05.006.
[34] YANG Rui-si, WANG Shi-xiong, DUAN Heng-pan, YUAN Xiao-hong, HUANG Zhang-jie, GUO Hong, YANG Xiang-jun. Efficient separation of copper and nickel from ammonium chloride solutions through the antagonistic effect of TRPO on Acorga M5640 [J]. Hydrometallurgy, 2016, 163: 18-23. https://doi.org/10.1016/j.hydromet.2016.03.006.
[35] SATPATHY S, MISHRA S. Kinetics and mechanisms of solvent extraction and separation of La(III) and Ni(II) with DEHPA in petrofin [J]. Transactions of Nonferrous Metals Society of China, 2019, 29: 1538-1548. https://doi.org/10. 1016/S1003-6326(19)65061-2.
Amirhossein SOEEZI1, Hadi ABDOLLAHI1, Sied Ziaedin SHAFAEI1, Esmaeil RAHIMI2
1. School of Mining engineering, College of Engineering, University of Tehran, Tehran, Iran;
2. Department of Mining Engineering, Islamic Azad University, South Tehran Branch, Tehran, Iran
ժ Ҫ����D2EHPA��LIX 984��Cyanex 302��Chemorex CP 150��Acorga 5774Ϊ��ȡ����ú��Ϊϡ�ͼ�����ȡ��Sarcheshmehͭ��Cu(770 mg/L)��Ni(3200 mg/L)��Fe(800 mg/L)��Zn(200 mg/L)�ĺϳɼ���ҵ��Һ��Cu��Ni����ȡ�뷴�ͽ���ȫ���о�����ȡ�ο��ǵ�Ӱ�������л���ٶȺ�ʱ�䡢��ȡ��Ũ�ȡ�pH���¶ȡ����⣬���ò�ͬŨ�ȵĸ��������л���(���ᡢ���ᡢ�����������)��Cu��Ni���з��͡������������ʱ��Ϊ3 min������ٶ�Ϊ500 r/min���¶�Ϊ28 ��C�Լ�A/O��Ϊ1:1�������£�Cu��Ni����ȡ����ߡ����⣬��10%(v/v) LIX 984����Ч��ȡCu������30% (v/v) Cyanex 302����Ч��ȡNi����ȡCu��Ni�����pH�ֱ�Ϊ2~3��4~5���Ӻ�Ni��Cu��Zn��Fe���ӵĺϳɼ���ҵ��Һ�п�ѡ������ȡCu��Ni������ȡ�ʷֱ�Ϊ85%��93%��77%��82%���������ʵ�����������ϳɼ���ҵ��Һ��Ni�ķ����ʴ�96%��Cu�ķ�����Ϊ93%����ֱ���D2EHPA��LIX 984��Cyanex 302 3����ȡ����3������ȡZn��Fe��Cu��Ni 4��Ԫ�أ���������ȡ����͡�
�ؼ��ʣ��ܼ���ȡ������ͭ��D2EHPA��LIX 984��Cyanex 302
(Edited by Wei-ping CHEN)
Corresponding author: Hadi ABDOLLAHI; Tel/Fax: +98-21-88008838, +98-21-82084567, +98-912-5032335; E-mail: H_abdollahi@ut.ac.ir
DOI: 10.1016/S1003-6326(20)65232-3

