
Formation of unidirectional nanoporous structures in thickly anodized aluminum oxide layer
Hyun-Chae NA1, Taek-Jin SUNG1, Seok-Heon YOON1, Seung-Kyoun HYUN1, Mok-Soon KIM1,
Young-Gi LEE2, Sang-Hyun SHIN2, Seok-Moon CHOI2, Sung YI2
1. Division of Materials Science and Engineering, Inha University, 253 Yonghyun-dong, Nam-gu,
Incheon, 402-751, Korea;
2. Samsung Electro-Mechanics Co., Ltd., 314, Maetan3-dong, Yeongtong-gu,
Suwon, Gyunggi-do, 443-743, Korea
Received 18 June 2008; accepted 10 March 2009
Abstract: A series of anodic aluminum oxide(AAO) was grown on the commercially pure 1050 aluminum sheet by controlling electrolyte temperature (2-15 ℃) and anodizing time (0.5-6 h), using a fixed applied current density of 3 A/dm2 in diluted sulfuric acid electrolyte. A crack-free thick AAO with the thickness of 105-120 μm and containing unidirectional nano sized pores (average pore diameter of 5-7 nm) is successfully achieved in the specimens anodized for 2 h, irrespective of electrolyte temperature. When anodizing time reaches 6 h, very thick AAO with the thickness of 230-284 μm is grown, and average diameter of unidirectional pores is in the range of 6-24 nm. The higher values in both the AAO thickness and pore diameter are attained for the specimens anodized at higher temperatures of 10-15 ℃. A crack is observed to exist in the AAO after anodizing up to 4 h and more. A higher fraction (more than 9%) of the crack is shown in the specimens anodized at higher temperatures of 10-15 ℃ for 6 h and a considerable amount of giant cracks are contained.
Key words: aluminum; anodizing; unidirectional nanopore; anodic aluminum oxide
1 Introduction
Anodizied aluminum oxide(AAO) is well known to be formed by the anodizing (electrochemical oxidation) of aluminum in an electrolytic cell in which the aluminum acts as the anode. If the anodizing is performed in an acid electrolyte such as sulfuric acid, oxalic acid or phosphoric acid, the AAO contains a periodic array of nanosized pores aligned in one direction within the rigid alumina[1-4]. Recently, the nanoporous AAO has attracted considerable interest because the unidirectional nanoporous alumina is applicable as nanotemplates in fabricating one- dimensional nanoscale materials including nanowires and nanotubes[5-8], which can be used in the electronic, photonic and biological devices[9-13]. Since the length of one-dimensional nanoscale materials is governed by the thickness of nanotemplates, the thicker AAO is essential to obtain the longer nanowires and nanotubes. Especially, the crack-free (sound) thick AAO should be developed to make the nanoporous alumina fit for practical usage. In this study, we attempted to fabricate a crack-free thick (larger than 100 μm) AAO containing the unidirectional nanoporous structures on the commercially pure 1050 aluminum sheet.
2 Experimental
The commercially pure 1050 aluminum sheet (99.5% in purity, 40 mm×40 mm×0.5 mm in size) was used in this study. In order to clean the surface of aluminum before anodizing, a pretreatment was carried out as follows: ultrasonic cleaning in acetone for 30 s, followed by acid etching using 15% (volume fraction) sulfuric acid solution at 50 ℃ for 180 s, and then rinsed thoroughly in deionized (DI) water. The pretreated aluminum sample was masked by an acid-resistant tape in order to expose the surface area of 20 mm×100 mm during anodization. The prepared aluminum sample was anodized in diluted sulfuric acid electrolyte using a mesh shaped Pt cathode and a fixed current density of 3 A/dm2. Anodization was carried out at different electrolyte temperatures of 2, 10 and 15 ℃ for various anodizing time from 0.5 to 6 h. After anodizing, the acid-resistant tape was removed by ultrasonic cleaning in acetone, and immediately the microstructure of the anodized aluminum surface was observed by a scanning electron microscope(SEM). The thickness of AAO was measured from an optical microscope(OM) observation of the polished cross section. The fracture section of the AAO was also observed by SEM after fracturing in liquid nitrogen.
3 Results and discussion
The average thickness of AAO formed on the pure 1050 aluminum specimen is presented in Fig.1 as a function of the anodizing time at different electrolyte temperatures. For the specimens anodized at 10 ℃ and 15 ℃, the thickness of AAO increases almost linearly with increase in the anodizing time. In contrast, a parabolic increment in the thickness of AAO is found to occur with anodizing time for the specimens anodized at 2 ℃. When the anodizing time reaches 2 h, a thick AAO larger than 100 μm is formed at all the electrolyte temperatures. Namely, the thicknesses of AAO are measured to be 120, 105 and 114 μm for the specimens anodized at 2, 10 and 15 ℃, respectively. When the anodizing time increases up to 6 h, very thick AAO layers with the thickness of 230, 279 and 284 μm are grown on the specimen anodized at 2, 10 and 15 ℃, respectively.
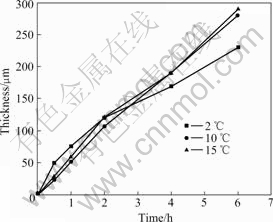
Fig.1 Variation of AAO thickness with anodizing time at different electrolyte temperatures
Interestingly, for the specimens anodized up to 4 h and more, a crack appears in the AAO layer, and the fraction of the crack depends upon the anodizing time and electrolyte temperature. The three OM images taken from the cross-section of AAO containing cracks are exhibited in Fig.2. In this figure, the cracks are indicated by arrows. A smaller (<100 μm) crack (Fig.2(a), anodized at 2 ℃ for 4 h), larger (>300 μm) crack (Fig.2(c), anodized at 15 ℃ for 6 h), and intermediate sized (100-300 μm) crack (Fig.2(b), anodized at 10 ℃ for 4 h) are observed in the AAO layer.
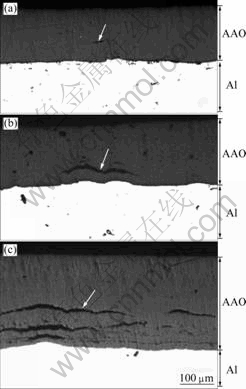
Fig.2 OM images of cross section for AAO containing cracks anodized at 2 ℃ for 4 h (a), at 10 ℃ for 4 h (b) and at 15 ℃ for 6 h (c)
The fraction of crack existing in the AAO is summarized in Table 1. As shown from this table, when the anodizing time changes from 4 to 6 h, the fraction of crack increases rapidly for the specimens anodized at 10 and 15 ℃, compared with the specimen anodized at 2 ℃. As a result, for the specimens anodized up to 6 h, a lower fraction (<2%) of crack is obtained for the specimen anodized at 2 ℃, although noticeably higher fraction (>9%) of crack is attained for the specimens anodized at higher temperatures of 10 and 15 ℃.
Table 1 Summary of areal fraction (%) of crack existing in cross-section of AAO

In order to investigate the relationship between the crack size and the fraction of crack, relative frequency of crack length is measured by the OM micrographs taken from the whole cross-section area of AAO. Fig.3 shows the plot of relative frequency of crack length for the specimens containing cracks in AAO. For the specimens with crack fraction of 0.2% (Fig.3(a), anodized at 2 ℃ for 4 h) and 0.3% (Fig.3(c), anodized at 15 ℃ for 4 h), only small sized cracks (crack length<100 μm ) exist in AAO. An example of such a crack can be seen from Fig.2(a). In the case of the specimen with the crack fraction of 1.5% (Fig.3(b), anodized at 10 ℃ for 4 h), the intermediate sized cracks (crack length of 100-300 μm, as shown in Fig.2(b)) are also observed in addition to the small sized cracks. For the specimen having the crack fraction of 1.8% (Fig.3(d), anodized at 2 ℃ for 6 h), a considerably small amount (relative frequency of 4.3%) of the large sized crack (crack length>300 μm) exists in addition to both the small and intermediate sized cracks. In contrast, in the case of the specimens showing the higher fraction of crack, such as 13.8% (Fig.3(f), anodized at 15℃ for 6 h) and 9.4% (Fig.3(e), anodized at 10 ℃ for 6 h), relative frequencies of the large sized cracks reach around 27% and 12%, respectively. It is concluded that the higher fraction of the crack (9.4% and more) exhibited in the specimens anodized at higher temperatures (10-15 ℃) for longer time (6 h) is closely related to the development of a giant (large sized) crack during anodizing process. Also, although a detailed mechanism concerning the formation of the giant crack is not understood yet, it is clear that decrease in the electrolyte temperature during anodizing is effective to retard the formation of the giant crack.
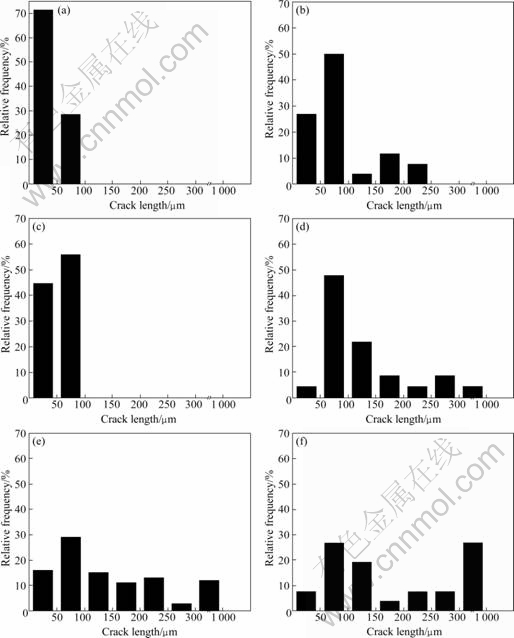
Fig.3 Relative frequency of crack length for cracks existing in AAO anodized at 2 ℃ for 4 h (a), at 10 ℃ for 4 h (b), at 15 ℃ for 4 h (c), at 2 ℃ for 6 h (d), at 10 ℃ for 6 h (e) and at 15 ℃ for 6 h (f)
Fig.4 shows SEM images of fracture section of AAO grown at 10 ℃ for 2 h. In the surface region of AAO (Fig.4(a)), nano-sized (about 7 nm) porous structure is well developed and a uniform distribution of the nanopores is observed. In the cross-section region of AAO (Figs.4(b) and (c)), the pores and cell walls are clearly seen. Fig.4(d) shows a cross-section morphology in the vicinity of the interface between porous layer and barrier layer.

Fig.4 SEM images of surface (a) and cross section (b-d) of specimen anodized at 10 ℃ for 2 h
The average pore diameter measured from the SEM observation on the surface region of AAO is summarized in Table 2. According to this table, at a given electrolyte temperature, the pore diameter tends to increase with increasing anodizing time except the case of 6 h at the electrolyte temperature of 2 ℃.
Table 2 Average pore diameter (nm) measured from surface of AAO

4 Conclusions
A series of anodic aluminum oxide (AAO) was grown on the commercially pure 1 050 aluminum sheets by controlling the electrolyte temperature from 2 to 15 ℃, and changing the anodizing time from 0.5 to 6 h, using a fixed applied current density of 3 A/dm2 in diluted sulfuric acid electrolyte.
1) A crack-free thick AAO with the thickness of 105-120 μm and containing unidirectional nano-sized pores (average pore diameter of 5-7 nm) is successfully achieved at the anodizing time of 2 h, irrespective of electrolyte temperature.
2) When anodizing time reaches 6 h, very thick AAO with the thickness of 230-284 μm is formed. The average pore diameter of unidirectional pores is in the range of 6-24 nm. The higher values in both the AAO thickness and pore diameter are attained for the specimens anodized at higher temperatures of 10-15 ℃.
3) A crack is found to exist in the AAO after anodizing up to 4 h and more. A higher fraction (more than 9%) of the crack is shown in the specimens anodized at higher temperatures of 10-15 ℃ for 6 h and a considerable amount of giant cracks are contained.
References
[1] WERNICK S, PINNER R, SHEASBY P G. The surface treatment and finishing of aluminum and its alloys [M]. 5th ed. Ohio, USA: ASM International, Metals Park, 1987.
[2] SCHWIRN K, LEE W, HILLEBRAND R, STEINHART M, NIELSCH K, GO¨SELE U. Self-ordered anodic aluminum oxide formed by H2SO4 hard anodization [J]. ACS NANO, 2008, 2(2): 302-310.
[3] LEE W, SCHWIRN K, STEINHART M, PIPPEL E, SCHOLZ R, GO¨SELE U. Structural engineering of nanoporous anodic aluminium oxide by pulse anodization of aluminium [J]. Nature Nanotechnology, 2008, 3: 234-239.
[4] LEE W, JI R, GO¨SELE U, NIELSCH K. Fast fabrication of long- range ordered porous alumina membranes by hard anodization [J]. Nature Materials, 2006, 5: 741-747.
[5] MARTIN C R. Nanomaterials: A membrane-based synthetic approach [J]. Science, 1994, 266: 1961-1966.
[6] MASUDA H, FUKUDA K. Ordered metal nanohole arrays made by a two-step replication of honeycomb structures of anodic alumina [J]. Science, 1995, 268: 1466-1468.
[7] WANG Z, BRUST M. Fabrication of nanostructure via self-assembly of nanowires within the AAO template [J]. Nanoscale Res Lett, 2007, 2: 34-39.
[8] MONTERO-MORENO J, SARRET M, MULLER C. Influence of the aluminum surface on the final results of a two step anodizing [J]. Surface & Coatings Technology, 2007, 201: 6352-6357.
[9] NALWA H S. Handbook of nanostructured materials and nanotechnology [M]. San Diego, USA: Academy Press, 2000.
[10] NIELSCH K, WEHRSPOHN R, BARTHEL J, KIRSCHNER J, GO¨SELE U. Hexagonally ordered 100 nm period nickel nanowire arrays [J]. Applied Physics Letters, 2001, 79(9): 1360-1362.
[11] LEE S. B, MITCHELL D, TROPIN L, NEVANEN T, SODERLUND H, MARTIN C. Antibody-based bio-nanotube membranes enantiomeric drug separations [J]. Science, 2002, 296: 2198-2200.
[12] LEE W, SCHOLZ R, NEILSCH K, GO¨SELE U. A template-based electrochemical method for the synthesis of multisegmented metallic nanotubes [J]. Angew Chem Int Ed, 2005, 44: 6050-6054.
[13] TABERNA P, MITRA S, POIZOT P, SIMON P, TARASCON J. High rate capabilities Fe3O4-based Cu nano-architectured electrodes for lithium-ion battery applications [J]. Nature Materials, 2006, 5: 567-573.
Corresponding author: Mok-Soon KIM; Tel: +82-32-8607541; E-mail: mskim@inha.ac.kr
DOI: 10.1016/S1003-6326(08)60398-2
(Edited by YANG Bing)