
J. Cent. South Univ. (2018) 25: 1573-1581
DOI: https://doi.org/10.1007/s11771-018-3849-x
Derivation and application of concentration formula of alloy genes of DO3-type in BCC alloy systems
PENG Hong-jian(彭红建)1, 2, LI Ling(李玲)1, LI Xiao-bo(李小波)2, XIE You-qing(谢佑卿)2
1. School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China;
2. School of Materials Science and Engineering, Central South University, Changsha 410083, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2018
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2018
Abstract: Taking Ta–W alloy system as an example, the concentration formulae of alloy genes of DO3-type alloys in BCC structures were derived on the basis of the theory of alloy genes and the number of coordination atoms. The concentrations of alloy genes of DO3-TaW3 and DO3-Ta3W ordered alloys were calculated as functions of composition xW and ordering degree (s). When s=smax, the concentrations of alloy genes of stoichiometric DO3-TaW3 compound are equal to those of alloys, that is,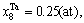

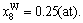 The concentrations of alloy genes of stoichiometric DO3-Ta3W compound are also equal to those of alloys, that is,
The concentrations of alloy genes of stoichiometric DO3-Ta3W compound are also equal to those of alloys, that is,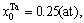

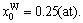 As ordering degree decreases, alloy genes of DO3-TaW3 and DO3-Ta3W ordered alloys will split. And the degree of splitting of alloy genes increases with the ordering degree decreasing. The atomic states and properties of DO3-TaW3 and DO3-Ta3W ordered alloys were calculated as a function of composition xW. The reason was pointed out that preparation of DO3-TaW3 and DO3-Ta3W intermetallic compounds is difficult due to small differences in their cohesive energies. It will provide theoretical guidance for the scientific designation to new candidate for ultra- high-temperature materials in areo-engine applications.
As ordering degree decreases, alloy genes of DO3-TaW3 and DO3-Ta3W ordered alloys will split. And the degree of splitting of alloy genes increases with the ordering degree decreasing. The atomic states and properties of DO3-TaW3 and DO3-Ta3W ordered alloys were calculated as a function of composition xW. The reason was pointed out that preparation of DO3-TaW3 and DO3-Ta3W intermetallic compounds is difficult due to small differences in their cohesive energies. It will provide theoretical guidance for the scientific designation to new candidate for ultra- high-temperature materials in areo-engine applications.
Key words: Ta–W alloy system; BCC structure; alloy gene
Cite this article as: PENG Hong-jian, LI Ling, LI Xiao-bo, XIE You-qing. Derivation and application of concentration formula of alloy genes of DO3-type in BCC alloy systems [J]. Journal of Central South University, 2018, 25(7): 1573–1581. DOI: https://doi.org/10.1007/s11771-018-3849-x.
1 Introduction
The target of material science development is to continuously design and develop new materials to meet modernization and future needs. To develop material design by multi-structure levels and various properties, materials science should be systematic. After nearly 30 years of efforts by XIE et al, the framework of systematic science of alloys been had established on the basis of energy band theory, valence bond theory and statistical thermodynamics. The core is the theory of the characteristic crystals and points out direction of alloy design. The theory has been successfully applied to Ag–Cu alloys [1, 2] and Au–Cu alloys [3–6] in FCC structure, Ti–Al alloys with coexistence of FCC and HCP structures [7, 8].
Ta–W alloys show a series of desirable properties, such as high density, high melting point, good heat conductivity, good fracture toughness, excellent corrosion resistance and weldability. So, they are excellent candidates for ultra-high temperature structural materials in areo-engine applications [9–12]. The manufacturing technology for refractory metals is difficult. However, there is little research on alloys in BCC structure by the theory [13, 14]. The purpose of this paper is to choose Ta–W alloys in BCC structure as research subject. The concentration formulae of alloy genes of DO3-type alloys in BCC structures were derived. The concentrations of alloy genes of DO3-TaW3 and DO3-Ta3W ordered alloys were calculated as functions of composition xW and ordering degree (s). The atomic states and properties of DO3-TaW3 and DO3-Ta3W ordered alloys are calculated as a function of composition xW. The reason that the preparation of DO3-TaW3 and DO3-Ta3W intermetallic compounds is difficult according to their cohesive energies analyzed. It will provide theoretical basis for the scientific designation to new candidate for refractory metals in areo-engine applications.
2 Crystal structure of DO3-TaW3 alloy
Figure 1 shows the crystal structure of DO3-TaW3 alloy. Its space group is Fm3m. Each Ta atom has 8 W2 atoms and each W1 atom also has 8 W2 atoms as the nearest neighbors. But each W2 atom has 4 Ta atoms and 4 W1 atoms.
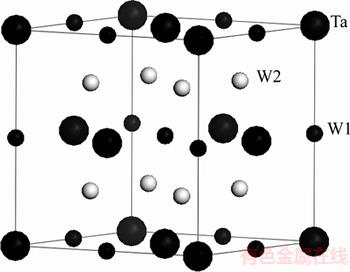
Figure 1 Crystal structure of DO3-TaW3 alloy
The crystalline of DO3-TaW3 compound contains sublattices I and II. The numbers of sublattices I and II are represented by N1 and N2 , respectively, then N (=N1+N2) denotes the total number of sublattices in the unit cell. v1 and v2 respectively represent the position fractions of sublattices I and II.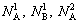 and
and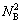 represent the numbers of sublattices I and II by A and B atoms. A and B atoms (A, B are Ta, W, respectively) have priority to occupy sublattices I and II.
represent the numbers of sublattices I and II by A and B atoms. A and B atoms (A, B are Ta, W, respectively) have priority to occupy sublattices I and II. 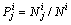 represents the probability with component j (j=A or B) in the sublattice point i (i= I, II). All the parameters should satisfy Eq. (1).
represents the probability with component j (j=A or B) in the sublattice point i (i= I, II). All the parameters should satisfy Eq. (1).
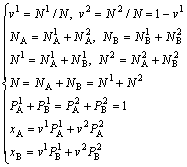 (1)
(1)
The arrangement of atoms in strict stoichiometric alloys will be destroyed due to elevated temperature or deviation of compositions from stoichiometric ratio. Alloys that deviate from stoichiometric ratio maybe form ordered alloys again. Therefore, we must consider influence of ordering degree and establish the formula between the concentration of alloy genes and composition and ordering degree (s). Only considering the influence of the nearest neighbor atoms, the ordering degree is defined as
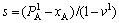 (2)
(2)
According to Eq. (2), we can know as follows: ①When xA=v1 and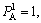 the alloys are completely stoichiometric and sublattice I is occupied by A atom and ordering degree (s) is equal to 1.② When the compositions deviate from stoichiometric, the maximal ordering degree is
the alloys are completely stoichiometric and sublattice I is occupied by A atom and ordering degree (s) is equal to 1.② When the compositions deviate from stoichiometric, the maximal ordering degree is
a) smax=xA/v1 for the alloy xA1;
b) smax=(1–xA)/(1–v1) for the alloy xA>v1, that is, xA1 or xA>v2, the ordering degree is unequal to 1.
The function can be derived as follows according to ordering degree and the definition of atomic occupancy probability:
 (3)
(3)
According to the sublattice point model, various of sublattice occupancy fractions of several types of intermetallic compounds are determined. For AB type, v1=1/2; for A3B type, v1=3/4; for AB3 type, v1=1/4.
3 Concentrations  and
and  of DO3- type ordered alloys
of DO3- type ordered alloys
3.1 Derivation of concentrations formulae and
and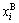
According to the sublattice model, the concentration  of the ith alloy gene A is equal to the summation of concentrations in sublattices I and II, that is,
of the ith alloy gene A is equal to the summation of concentrations in sublattices I and II, that is,
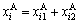 (4)
(4)
where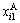 and
and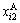 respectively represent concentrations of alloy genes A in sublattices I and II which has i B atoms in its neighbor configuration.
respectively represent concentrations of alloy genes A in sublattices I and II which has i B atoms in its neighbor configuration. 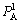 and
and represent the occupancy probabilities of alloy genes A in sublattices I and II.
represent the occupancy probabilities of alloy genes A in sublattices I and II. 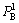 and
and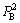 represent the occupancy probabilities of alloy genes B in sublattices I and II. The concentration
represent the occupancy probabilities of alloy genes B in sublattices I and II. The concentration of alloy genes A is
of alloy genes A is

 (5)
(5)
According to the same method of concentration of alloy genes A, the concentration 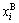 of alloy genes B is
of alloy genes B is

 (6)
(6)
According to Eqs. (5) and (6), the concentrations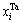 and
and of Ta and W alloy genes of DO3-TaW3 ordered alloys as a function of composition xW are shown in Figure 2. When s=smax, the distribution characteristics are as follows.
of Ta and W alloy genes of DO3-TaW3 ordered alloys as a function of composition xW are shown in Figure 2. When s=smax, the distribution characteristics are as follows.
1) When xW=0.75, alloy is stoichiometric DO3-TaW3 compound. There are only three types of alloy genes, that is, 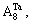
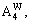
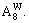 The concentrations of alloy genes are equal to the compositions of alloys, that is,
The concentrations of alloy genes are equal to the compositions of alloys, that is,
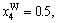
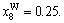
2) When xW>0.75, 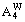 and
and in DO3-TaW3 ordered alloys will split into
in DO3-TaW3 ordered alloys will split into 
 and the concentration of W alloy genes will be
and the concentration of W alloy genes will be  while Ta alloy gene will be still
while Ta alloy gene will be still 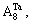 the concentration of Ta alloy genes will be
the concentration of Ta alloy genes will be 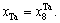
3) When xW<0.75, in DO3-TaW3 ordered alloys will split into
in DO3-TaW3 ordered alloys will split into  the concentration of Ta alloy genes will be
the concentration of Ta alloy genes will be 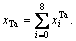
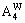 and
and 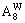 will also split into
will also split into 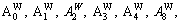 the concentration of W alloy genes will be
the concentration of W alloy genes will be 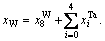
From the above, the alloy genes of splitting are different in rich Ta and rich W DO3-TaW3 ordered alloys, which leads to great influence on atomic states, crystal structure parameters and physical properties.
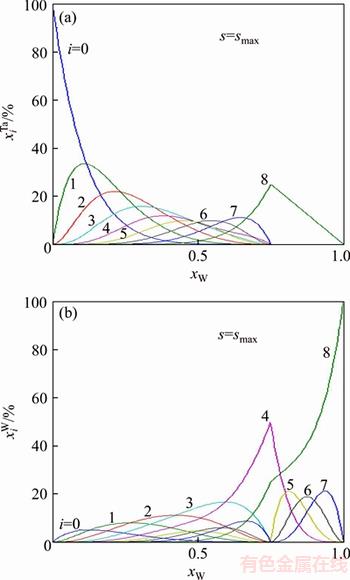
Figure 2 Concentrations 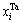 (a) and
(a) and  (b) of alloy genes of DO3-TaW3 ordered alloys as a function of composition xW with maximal ordering degree
(b) of alloy genes of DO3-TaW3 ordered alloys as a function of composition xW with maximal ordering degree
3.2 Concentrations 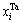 and
and  of alloy genes of DO3-TaW3 ordered alloys as functions of composition xW and ordering degree (s)
of alloy genes of DO3-TaW3 ordered alloys as functions of composition xW and ordering degree (s)
The concentrations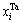 and
and of alloy genes as functions of composition xW and ordering degree (s=0.8smax, s=0.6smax and s=0) of DO3-TaW3 ordered alloy are shown in Figure 3. From Figure 3, when ordering degree decreases, alloy genes
of alloy genes as functions of composition xW and ordering degree (s=0.8smax, s=0.6smax and s=0) of DO3-TaW3 ordered alloy are shown in Figure 3. From Figure 3, when ordering degree decreases, alloy genes 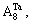
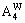 and
and 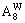 in DO3-TaW3 ordered alloys will split into
in DO3-TaW3 ordered alloys will split into

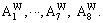 The splitting degree of alloy genes will be larger with the ordering degree decreasing. As ordering degree decreases, the concentrations of various alloy genes of ordered alloys are approaching to those of disordered alloys.
The splitting degree of alloy genes will be larger with the ordering degree decreasing. As ordering degree decreases, the concentrations of various alloy genes of ordered alloys are approaching to those of disordered alloys.
4 Concentrations and
and of alloy genes of DO3-Ta3W ordered alloys as functions of composition xW and ordering degree (s)
of alloy genes of DO3-Ta3W ordered alloys as functions of composition xW and ordering degree (s)
On the basis of Eqs. (5) and (6), the concentrations 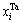 and
and  of Ta and W alloy genes of DO3-Ta3W ordered alloys as a function of composition xW are shown in Figure 4. When s=smax, the distribution characteristics are as follows.
of Ta and W alloy genes of DO3-Ta3W ordered alloys as a function of composition xW are shown in Figure 4. When s=smax, the distribution characteristics are as follows.
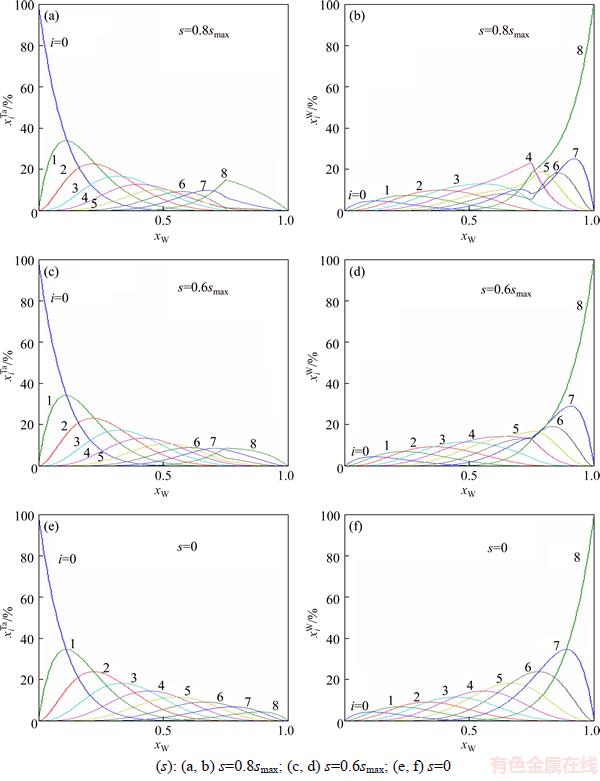
Figure 3 Concentrations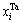 (a, c, e) and
(a, c, e) and (b, d, f)of alloy genes of DO3-TaW3 type ordered alloys as functions of composition xW and ordering degree (s):
(b, d, f)of alloy genes of DO3-TaW3 type ordered alloys as functions of composition xW and ordering degree (s):
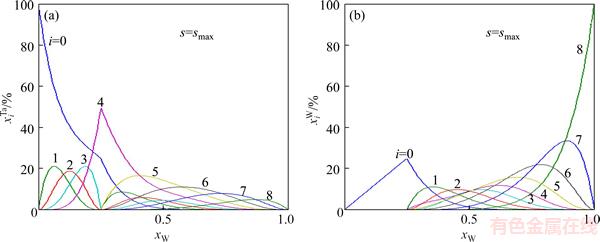
Figure 4 Concentrations 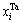 (a) and
(a) and  (b) of alloy genes of DO3-Ta3W ordered alloys as a function of composition xW with maximal ordering degree
(b) of alloy genes of DO3-Ta3W ordered alloys as a function of composition xW with maximal ordering degree
1) When xW=0.25, alloy is stoichiometric DO3-Ta3W compound. There are only three types of alloy genes, that is
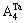 and
and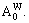 The concentrations of alloy genes are equal to the compositions of alloy, that is,
The concentrations of alloy genes are equal to the compositions of alloy, that is,
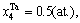

2) When xW<0.25,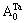 and
and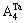 of DO3-TaW3 ordered alloys with a maximal ordering degree split into
of DO3-TaW3 ordered alloys with a maximal ordering degree split into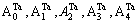 and the concentration of Ta alloy genes is
and the concentration of Ta alloy genes is 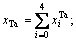 while
while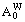 does not split and the concentration of W alloy genes is
does not split and the concentration of W alloy genes is 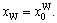
3)When xW>0.25,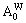 atoms of DO3- TaW3 ordered alloys with a maximal ordering degree split into
atoms of DO3- TaW3 ordered alloys with a maximal ordering degree split into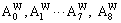 and the concentration of W alloy genes is
and the concentration of W alloy genes is 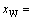
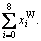
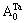 and
and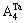 are also split into
are also split into 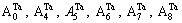 and the concentration of Ta alloy genes is
and the concentration of Ta alloy genes is 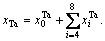
From the above, the alloy genes of constitute are different between rich Ta and rich W DO3-TaW3 ordered alloys, which leads to great influence on atomic state, crystal structure parameters and physical properties.
The concentrations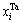 and
and of alloy genes as functions of composition xW and ordering degree (s=0.8smax, s=0.6smax and s=0) of DO3-Ta3W ordered alloys are shown in Figures 5. From Figure 5, when ordering degree decreases, alloy genes
of alloy genes as functions of composition xW and ordering degree (s=0.8smax, s=0.6smax and s=0) of DO3-Ta3W ordered alloys are shown in Figures 5. From Figure 5, when ordering degree decreases, alloy genes 
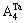 and
and  in DO3-Ta3W ordered alloys will split into
in DO3-Ta3W ordered alloys will split into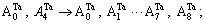
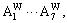
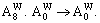 The splitting degree of alloy genes will be larger with the ordering degree decreasing. As ordering degree decreases, the concentrations of various alloy genes of ordered alloys are approaching to those of disordered alloys.
The splitting degree of alloy genes will be larger with the ordering degree decreasing. As ordering degree decreases, the concentrations of various alloy genes of ordered alloys are approaching to those of disordered alloys.
5 Atomic states and properties of DO3- TaW3 and DO3-Ta3W ordered alloys as a function of composition xW
The properties of disordered and ordered alloys are described by average properties of components. The properties of disordered alloys are obtained by the additive law of alloy genes. Therefore, the atomic states and properties of DO3-TaW3 and Ta3W ordered alloys as a function of composition xW are listed in Tables 1 and 2. The variation trend of properties of DO3-TaW3 and Ta3W ordered alloys is almost the same as that of atomic states. Cohesive energies of disordered Ta1–xWx alloys DO3-Ta3W and DO3-TaW3 ordered alloys as functions of composition xW are shown in Figure 6. From Figure 6, we can know that the differences of cohesive energies among disordered Ta1–xWx alloys, DO3-Ta3W and DO3-TaW3 ordered alloys are very small. Therefore, it is difficult to prepare DO3-Ta3W and DO3-TaW3 intermetallic compounds. The results are excellent agreement with experimental results. That will provide theoretical guidance for preparing Ta–W alloys materials.
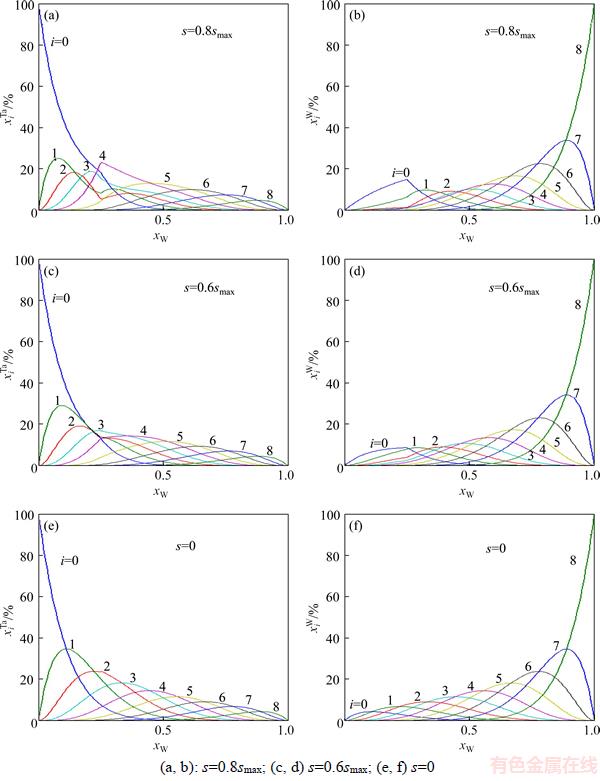
Figure 5 Concentrations (a, c, e) and
(a, c, e) and  (b, d, f) of alloy genes of DO3-Ta3W ordered alloys as functions of composition xW and ordering degree (s):
(b, d, f) of alloy genes of DO3-Ta3W ordered alloys as functions of composition xW and ordering degree (s):
6 Conclusions
1) Taking Ta–W alloy system as an example, the concentration formulae of alloy genes of DO3-type alloys in BCC structures were derived on the basis of the theory of alloy genes and the number of coordination atoms. The concentrations of alloy genes of DO3-TaW3 and DO3-Ta3W ordered alloys were calculated as functions of composition xW and ordering degree (s).
2) When s=smax, the concentrations of alloy genes of stoichiometric DO3-TaW3 compound are equal to those of alloys, that is,
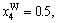
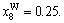 The concentrations of alloy genes of stoichiometric DO3-Ta3W compound are also equal to those of alloys, that is,
The concentrations of alloy genes of stoichiometric DO3-Ta3W compound are also equal to those of alloys, that is, 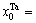 0.25,
0.25,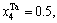
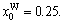 As ordering degree decreases, alloy genes of DO3-TaW3 and DO3-Ta3W ordered alloys will split. And the degree of splitting of alloy genes increases with the ordering degree decreasing.
As ordering degree decreases, alloy genes of DO3-TaW3 and DO3-Ta3W ordered alloys will split. And the degree of splitting of alloy genes increases with the ordering degree decreasing.
Table 1 Atomic states and properties of DO3-TaW3 ordered alloys as a function of composition xW
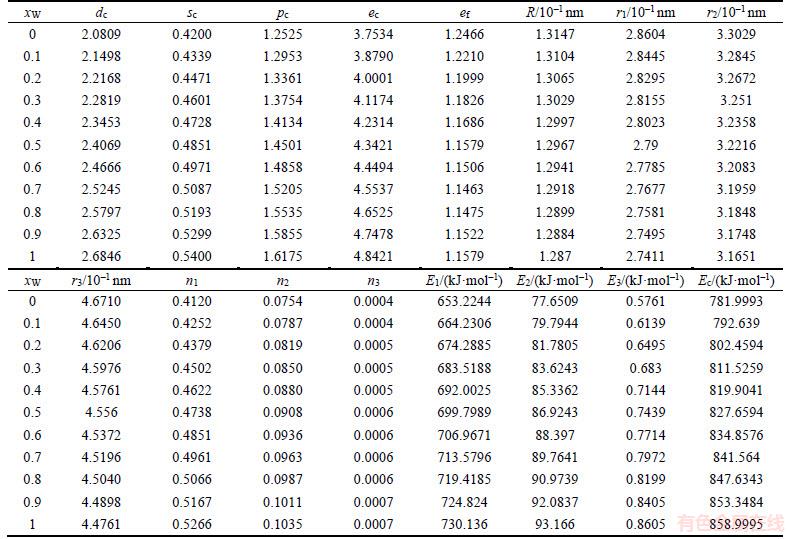
Table 2 Atomic states and properties of DO3-Ta3W ordered alloys as a function of composition xW
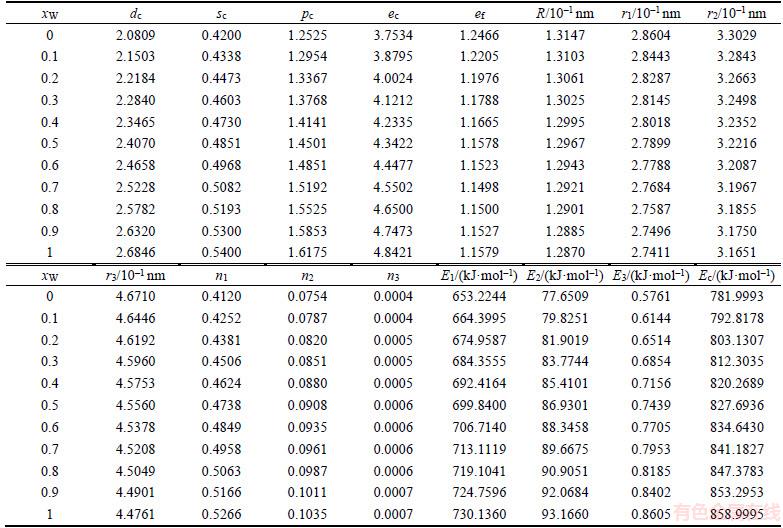
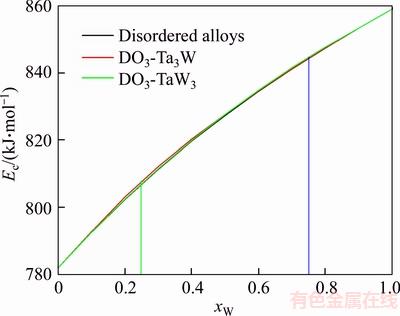
Figure 6 Cohesive energies of disordered Ta1–xWx alloys, DO3-Ta3W and DO3-TaW3 ordered alloys as functions of composition xW
3) The atomic states and properties of DO3-TaW3 and DO3-Ta3W ordered alloys were calculated as a function of composition xW. The main reason is that it is difficult to prepare DO3-Ta3W and DO3-TaW3 intermetallic compounds due to their small differences in cohesive energies. It will provide theoretical guidance for the scientific designation to new candidate for ultra-high- temperature materials in areo-engine applications.
References
[1] XIE Y Q. Atomic energies and Gibbs energy functions of Ag-Cu alloys. [J]. Science in China: Technological Sciences, 1998, 41(2): 146–156. DOI: 10.1007/BF02919677.
[2] XIE Y Q, ZHANG X D. Electronic structure of Ag-Cu alloys [J]. Science in China: Technological Sciences, 1998, 41(3): 225–236. DOI: 10.1007/BF02919432.
[3] XIE You-qing, LIU Xin-bi, LI Xiao-bo, PENG Hong-jian, NIE Yao-zhuang. Holographic alloy positioning design system and holographic network phase diagrams of Au-Cu system [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(3): 885–906. DOI: 10.1016/S1003- 6326(15)63678-0.
[4] XIE You-qing, NIE Yao-zhuang, LI Xiao-bo, PENG Hong-jian, LIU Xin-bi. Alloy gene Gibbs energy partition function and equilibrium holographic network phase diagrams of Au3Cu-type sublattice system [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(1): 211–240. DOI: 10.1016/S1003-6326(15)63598-1.
[5] XIE You-qing, LI Xiao-bo, LIU Xin-bi, NIE Yao-zhuang, PENG Hong-jian. Alloy gene Gibbs energy partition function and equilibrium holographic network phase diagrams of AuCu3-type sublattice system [J]. Transactions of Nonferrous Metals Society of China, 2014, 24(11): 3585–3610. DOI: 10.1016/S1003-6326(15)63598-1.
[6] XIE You-qing, PENG Hong-jian, LIU Xin-bi, LI Xiao-bo, NIE Yao-zhuang. New atom movement mechanism for tracking path on disordering AuCuI  compound [J]. Transactions of Nonferrous Metals Society of China, 2014, 24(10): 3221–3256. DOI: 10.1016/S1003- 6326(14)63464-6.
compound [J]. Transactions of Nonferrous Metals Society of China, 2014, 24(10): 3221–3256. DOI: 10.1016/S1003- 6326(14)63464-6.
[7] XIE Y Q, TAO H J, PENG H J, LIU X B. Atomic states, potential energies, volumes, stability and brittleness of ordered FCC TiAl2 type alloys [J]. Physica B, 2005, 366: 17–37. DOI: 10.1016/j.physb.2005.04.041.
[8] XIE Y Q, PENG H J, LIU X B, PENG K. Atomic states, potential energies, volumes, stability and brittleness of ordered FCC Ti3Al-type alloys [J]. Physica B, 2005, 362: 1–17. DOI: 10.1016/j.physb.2005.01.197.
[9] YU Quan-zhi, LU You-lian, HU Zhi-liang, ZHOU Bin, YIN Wen, LIANG Tian-jiao. Decay heat calculations for a 500 kW W–Ta spallation target [J]. Nuclear Instruments and Methods in Physics Research B, 2015, 351: 41–45. DOI: 10.1016/j.nimb.2015.03.087.
[10] WANG S, CHEN C, JIA Y L, XIAO Z, WANG M P, LI Z, WU Y C. Effects of grain size on the microstructure and texture of cold-rolled Ta-2.5W alloy [J]. Int Journal of Refractory Metals and Hard Materials, 2016, 58: 125–136. DOI: 10.1016/j.ijrmhm.2016.04.018.
[11] WANG Shan, WANG Ming-pu, CHEN Chang, JIA Yan-lin, XIA Fu-zhong, ZUO Bo. Microstructures and properties of annealed Ta-7.5%W alloy foils [J]. Journal of Central South University: Science and Technology, 2012, 43(8): 2951–2957. (in Chinese)
[12] HU Kong-sheng, XIAO Xuan, DONG Xian-juan, LEI Xue-hai, ZHOU Tian-ning. Research progress on strengthening and toughening of refractory tantalum based alloys for ultra-high-temperature applications [J]. Hot Working Technology, 2014, 43(24): 19–22.
[13] PENG Hong-jian, ZHOU Jiao-lian, LI Xiao-bo, XIE You-qing. Research on properties of DO3- and B2-type ordered alloys and disordered alloys in Nb-Mo alloy system in BCC structure [J]. Science in China: Technological Sciences, 2011, 41(12): 1684–1690. DOI: 10.1007/s11431- 011-4473-2.
[14] PENG Hong-jian, WU Qing, LI Xiao-bo, XIE You-qing. Computation of concentrations of characteristic atoms of alloys in BCC structure [J]. Science in China: Technological Sciences, 2011, 41(9): 1167–1171. DOI: 10.1007/s11431- 011-4432-y.
(Edited by YANG Hua)
中文导读
BCC 合金系中DO3-型合金基因浓度公式的推导和应用
摘要:本文以Ta–W合金系为例, 根据合金基因理论和配位原子数,推导出了BCC结构中DO3-型合金基因浓度的公式。计算了DO3-TaW3和DO3-Ta3W有序合金的基因浓度随成分xW和有序度(s)的变化。当s=smax时,化学计量比的DO3-TaW3化合物的合金基因浓度等于合金的浓度,即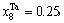
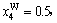
 化学计量比的DO3-Ta3W化合物的合金基因浓度也等于合金的浓度,即
化学计量比的DO3-Ta3W化合物的合金基因浓度也等于合金的浓度,即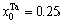
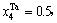
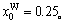 随着有序度的降低,DO3-TaW3和DO3-Ta3W有序合金将会发生分裂,且随着有序度的降低,合金基因分裂程度增加。计算了DO3-TaW3和DO3-Ta3W有序合金的原子状态和性质随成分xW的变化,指出了DO3-TaW3和DO3-Ta3W 金属间化合物的制备非常困难的原因主要是由于它们的结合能差异小,为新一代超高温材料在航空发动机上的应用设计提供科学理论指导。
随着有序度的降低,DO3-TaW3和DO3-Ta3W有序合金将会发生分裂,且随着有序度的降低,合金基因分裂程度增加。计算了DO3-TaW3和DO3-Ta3W有序合金的原子状态和性质随成分xW的变化,指出了DO3-TaW3和DO3-Ta3W 金属间化合物的制备非常困难的原因主要是由于它们的结合能差异小,为新一代超高温材料在航空发动机上的应用设计提供科学理论指导。
关键词:Ta–W合金系;BCC结构;合金基因
Foundation item: Project(50954006) supported by the National Natural Science Foundation of China; Project (2014-12) supported by the Environmental Protection Department of Hunan, China; Project(2016TP1007) supported by the Hunan Provincial Science and Technology Plan Project, China; Project(2016-01) supported by the Development and Reform Commission of Hunan Province, China
Received date: 2016-11-02; Accepted date: 2017-02-23
Corresponding author: PENG Hong-jian, PhD, Associate Professor; Tel: +86–731–88879287; E-mail: hongjianpeng@126.com