Trans. Nonferrous Met. Soc. China 20(2010) s837-s841

Influence of surface liquid segregation on corrosion behavior of semi?solid metal high pressure die cast aluminium alloys
E. P. MASUKU 1, H. M?LLER1, U. A. CURLE1, P. C. PISTORIUS2, W. LI2
1.Materials Science and Manufacturing, Council for Scientific Industrial Research, Pretoria, South Africa;
2.Department of Materials Science and Engineering, Carnegie Mellon University, Pittsburgh, USA
Received 13 May 2010; accepted 25 June 2010
Abstract: The corrosion behavior (in chloride medium) of the surface layer of SSM-HPDC plates of alloys 7075-T6 and 2024-T6 was compared with that of the wrought alloys 7075-T6 and 2024-T6. Potentiodynamic testing was performed in deaerated 3.5%NaCl solution. In separate tests, the open-circuit potential was monitored in aerated 3.5% NaCl for 30 min after immersion. The electrochemical tests show that there is no significant difference in the pitting potential of the wrought alloys and that of the Cu-enriched surfaces of the SSM-HPDC alloys.
Key words: semi-solid metal (SSM) forming; wrought aluminium alloys; alloy 7075; alloy 2024; corrosion; surface liquid segregation
1 Introduction
The semi-solid metal (SSM) process inherently offers the opportunity to produce high integrity near net shape components. Since its discovery in the early 1970s, a wide spectrum of routes and processes has been developed to produce semi-solid metal slurries[1]. The aim is to obtain a semi-solid structure (which is free of dendrites) with the solid present in a near spherical form. This semi-solid mixture flows homogeneously, behaving as a thixotropic fluid with viscosity depending on the shear rate and fraction of solid[2].
The nature of the SSM slurry with solid spherical grains suspended in liquid makes it susceptible to liquid segregation during forming processes. The modeling and quantifying of typical segregation phenomena in SSM forming have been studied[3-7]. It has also been shown by the authors that a surface liquid segregation (SLS) phenomenon existed in SSM-HPDC (high pressure die cast) plates of A356/7[8] and A201 casting alloys[9] and 2024, 6082 and 7075 wrought alloys[9]. This SLS layer (or eutectic layer) in the A356/7 was found to vary in thickness of between 0.7 and 1.1 mm (using the Si upper limit specification of 7.5% as the criterion for determining the thickness based on OES analysis[8]). It was proposed that the surface segregation phenomenon that was observed is a characteristic of SSM forming and that the degree of segregation will be dependent on alloy composition, die design and processing parameters [8].This SLS layer differs in chemical composition from the centre (or bulk) of the castings. Therefore, it has different properties such as hardness, wear resistance and corrosion.
High strength Al alloys such as 7075 and 2024 are commonly used in aerospace and other industrial applications. The addition of Cu as one of the alloying elements for both alloys greatly improves the mechanical strength of the 7xxx and 2xxx series alloys by precipitation hardening. However, these Cu containing alloys in the T6 temper condition are susceptible to various forms of localized corrosion in chloride environments[10]. One of the main advantages of SSM processing is that it is possible to produce near net-shape components from alloys that are conventionally wrought (such as the 2000, 6000 and 7000 series)[9,11] and from difficult-to-conventionally-cast high strength casting alloys such as the 200 series[9,12]. A study by the authors on the pitting corrosion behavior of the SLS layer of SSM-HPDCA356/7 showed that the heterogeneous structures produced in SSM HPDC plates made it more susceptible to pitting corrosion in media containing chloride ions[13]. A number of studies have been conducted on comparing the tensile properties of these high performance SSM processed alloys with their wrought counterparts[11,14]. However, the presence of a SLS layer in these alloys and its influence on properties (such as fatigue, wear and corrosion) have not been studied. In this study, the corrosion behavior of the surface layer of SSM-HPDC plates was compared with the wrought material of high performance alloys of 7075 and 2024.
2 Experimental
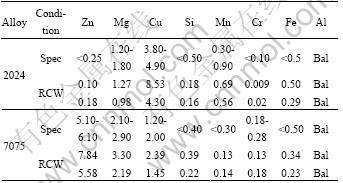
The wrought alloys used in this study were supplied in form of 270 mm diameter billets for the 7075 and rolled plate for the 2024 (The upper and lower limit specifications for the different elements are given in Table 1). Semi-solid metal slurries of alloys 2024 and 7075 were prepared using an electric resistance melting furnace and the CSIR (Council for Scientific and Industrial Research) rheocasting process (single coil version)[15]. Plates (6 mm �� 55 mm �� 100 mm) were cast in steel moulds with a 130 t HPDC machine. The plates were sectioned in order to perform the microstructural evaluation, chemical composition and hardness test for both wrought and cast alloys in T6 temper condition. The surface chemical composition was measured using a Thermo Quantris optical emission spectrometer (OES).
Table 1 Chemical composition limits for Al alloys 7075 and 2024, as well as surface compositions of alloys used in this study in rheocast (RC) and wrought (W) condition[9] (mass percentage, %)
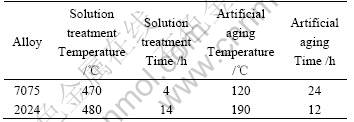
The heat treatment parameters used for the T6 temper condition can be found in Table 2 for both SSM-HPDC plates and wrought samples. More information regarding the actual SSM-HPDC and surface segregation characterization can be found in Ref.[9]. The solution treatment temperatures were kept relatively low to prevent incipient melting of low-melting phases, especially at the surface of the castings[9,11]. Vickers hardness (19.6 N) measurements (average of at least five measurements per sample) on the surface of the plates were made in the T6 temper conditions. Metallographic samples of cross-sections of the castings were also prepared, etched using 0.5%HF and evaluated using optical microscopy. Corrosion tests were performed in 3.5% NaCl solution at 22��. The alloy surfaces were wet ground on silicon carbide paper (finishing with 600 grit paper). Potentiodynamic testing was performed in deaerated 3.5%NaCl solution, using a scan rate of 0.5 mV/s. In separate tests, the open-circuit potential was monitored in aerated 3.5% NaCl for 30 min after immersion. Corrosion tests were performed in an Avesta-type corrosion cell, to avoid crevice corrosion at the seal between the corrosion sample and the electrochemical cell[16]. A three-electrode configuration was employed: using the alloy in this study as working electrode, with a surface area of 1.23 cm2, graphite rod counter electrodes (diameter of 8 mm and height of 60 mm) and a silver-silver chloride (SSC) reference electrode (with saturated KCl filling solution).
Table 2 T6 heat treatment cycles applied to different SSM-HPDC plates and wrought samples[9,11]
3 Results and discussion
Fig. 1 shows the typical light micrograph images of SSM-HPDC alloys in the T6 temper condition. Microstructural analyses of cross-sections of the castings reveal the presence of the SLS layer on SSM-HPDC samples. Closer to the centre of the casting, microconstituents are mainly globular a primary grains and smaller amounts of eutectic. The purpose of the T6 solution heat treatment is to dissolve solutes which lead to the formation of a large number of strengthening precipitates during subsequent artificial aging. It also reduces micro-segregation of elements in the aluminium matrix. However, the SLS layer in all the alloys is still clearly visible. The SLS layer has a higher concentration of alloying elements than the bulk. Specifically, the Cu and Fe contents are greater than the corresponding wrought alloys (Table 1)[9]. The possible significance of this is that these elements form intermetallic compounds, which affect corrosion. In general, intermetallic compounds affect the pitting corrosion resistance of aluminium alloys because the intermetallic compounds are more efficient cathodic sites than the aluminium surface, and hence give a higher corrosion potential. This galvanic effect overrides the slight increase in pitting potential with the increase of copper content[10].

Fig.1 Optical micrographs of SSM-HPDC alloys in T6 condition: (a) alloy 2024; (b) alloy 7075
Fig.2 displays a typical rolled grain structure with elongated grains for alloy 2024 and equiaxed microstructure for alloy 7075. Both wrought alloys show a finely homogeneous dispersion of phases throughout the structure.

Fig.2 Optical micrographs of wrought alloys in T6 condition: (a) alloy 2024; (b) alloy 7075
The hardness for the surface of SSM-HPDC plates in T6 temper condition is much higher than that for the wrought alloy in T6 condition (Fig. 3)[9]. This is due to the high concentration of the alloying elements on the surface of the SSM-HPDC plates (Table 1). This implies that the solution treatment is not successful in eliminating the concentration gradients throughout the plate[9]. This is as expected: for the higher of the two solution temperatures (480 ��) the diffusion coefficient of Cu in aluminium is approximately 2��10?14 m2/s[17], which means that the diffusion distance during the 14 h heat treatment is approximately (Dt)0.5=0.03 mm, where D is the diffusivity and t time.
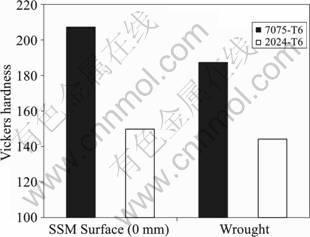
Fig.3 Vickers hardness for SSM-HPDC and wrought samples in T6 temper condition
The results of potentiodynamic tests for 7075 alloy are shown in Fig.4, where noisy current is characteristic of pitting corrosion. The noise from pit precursors does not allow a definite pitting potential to be identified, but both the wrought and SSM-HPDC (surface) alloys show strong increases in current�Cfrom stably growing pits�Cabove approximately ?0.7 V(vs Ag/AgCl), which is similar to the pitting potentials reported by MENG and FRANKEL[10] for 7xxx series aluminium alloys containing 1.4%-2% Cu in acidified 0.5 mol/L NaCl. Similarly, the pitting potential of both SSM-HPDC and wrought 2024 is found to be approximately ?0.78 V (vs Ag/AgCl) (Fig.5). From these results there does not appear to be a significant difference in pitting potential for wrought and SSM-HPDC alloys. MENG and FRANKEL [10] found a linear relationship between the pitting potential and the logarithm of copper concentration, with a 0.11 V increase in pitting potential for every decade of copper content. This means that the difference in pitting potential between the 7075 wrought alloy (1.45% Cu) and the surface of the 7075 SSM-HPDC alloy (2.4%Cu) is expected to be only some 0.11 lg (2.4/1.45)=0.02 V. The difference for 2024 wrought alloy (4.3% Cu) and the surface of the 2024 SSM-HPDC alloy (8.5%Cu) is expected to be 0.11 lg (8.5/4.3)=0.03 V.
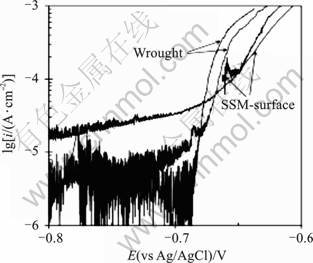
Fig.4 Potentiodynamic polarisation results for wrought and SSM-HPDC (surface) 7075 alloys in deaerated 3.5% NaCl at 22?C and scan rate of 0.5 mV/s
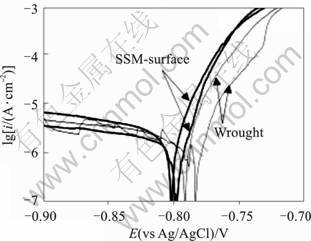
Fig.5 Potentiodynamic polarisation results for wrought and SSM-HPDC (surface) 2024 alloys in deaerated 3.5% NaCl at 22��, and scan rate of 0.5 mV/s
The absence of a significant difference in pitting potential is confirmed by the corrosion potential measurements (Fig.6 and Fig.7) for both alloys. For alloys containing a significant copper content (0.7% Cu or more in the work of MENG and FRANKEL[10]), the corrosion potential in aerated solutions is found to be equal to the pitting potential: the cathodic reactions on the intermetallic compounds support pitting of the aluminium matrix. As Figs.6 and 7 show, after an initial difference (which may have been caused by slight differences in the surface preparation of the samples) the corrosion potentials of the wrought and SSM-HPDC alloys are essentially identical, around ?0.73 V (vs Ag/AgCl) for alloy 7075 and ?0.8 V (vs Ag/AgCl) for alloy 2024.
4 Conclusions
(1) The electrochemical tests show no significant difference in the pitting potential of the wrought alloy and that of the Cu-enriched surface of the SSM alloy for both alloys 2024 and 7075.

Fig.6 Corrosion potentials of wrought and SSM-HPDC (surface) 7075 alloys in aerated 3.5% NaCl
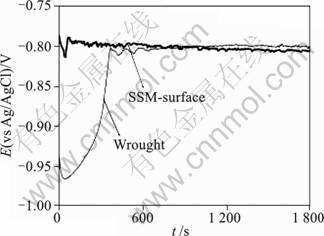
Fig.7 Corrosion potentials of wrought and SSM HPDC(surface) 2024 alloys in aerated 3.5%NaCl
(2) Longer-term exposure testing would be required to determine whether there are any differences in the morphology of pitting attack between these alloys; pit morphology may be affected by the spatial distribution of intermetallic compounds in the alloy microstructure, which is likely to differ between the SSM-HPDC and wrought alloys.
References
[1] ATKINSON H V. Modelling the semisolid processing of metallic alloys[J]. Progress in Materials Science, 2005, 50 (3): 341-412.
[2] FLEMINGS M C, MARTINEZ R A. Principles of microstructural formation in semi-solid metal processing[J]. Solid State Phenomena, 2006, 116/117: 1-8.
[3] NOLL T, FRIEDRICH B, HUFSCHMIDT M, MODIGELL M, NOHN B, HARTMANN D. Evaluation and modelling of chemical segregation effects for thixoforming processing[J]. Advanced Engineering Materials, 2003, 5 (3): 156-160.
[4] KANG C G, JUNGIALS H K. A study on solutions for avoiding liquid segregation phenomena in thixoforming process: Part I. Constitutive modelling and finite element method simulations for die design[J]. Metallurgical and Materials Transactions B: Process Metallurgy and Meterials Processing Science, 2001, 32(1):119-127.
[5] KANG C G, JUNG H K. A study on solutions for avoiding liquid segregation phenomena in thixoforming process: Part II. Net shape manufacturing of automotive scroll component[J]. Metallurgical and Materials Transactions B: Process Metallurgy and Meterials Processing Science, 2001, 32 (1): 129-136.
[6] HUFSCHMIDT M, MODIGELL M, PETERA J. Modelling and simulation of forming processes of metallic suspensions under non-isothermal conditions[J]. Journal of Non Newtonian Fluid Mechanics, 2006, 134 (1/3): 16-26.
[7] SEO P K, KIM D U, KANG C G. The effect of the gate shape on the microstructural characteristic of the grain size of Al-Si alloy in the semi-solid die casting process[J]. Materials Science and Engineering A, 2007, 445/446: 20-30.
[8] GOVENDER G, M?LLER H. Evaluation of surface chemical segregation of semi-solid metal cast aluminium alloy A356[J]. Solid State Phenomena,2008, 141/143: 433-438.
[9] M?LLER H, CURLE U A, MASUKU E P. Characterization of surface liquid segregation in SSM-HPDC aluminium alloys 7075, 2024, 6082 and A201[C]//Submitted for the 11th International Conference on Semi-solid Processing of Alloys and Composites. Beijing, 2010.
[10] MENG Q, FRANKEL G S. Effect of Cu content on the corrosion behavior of 7xxx series aluminum alloys[J] Journal of the Electrochemical Society, 2004,151: B271-B283.
[11] CURLE U A. Semi-solid near-net shape rheocasting of heat treatable wrought aluminum alloys[C]//Submitted for the 11th International Conference on Semi-solid Processing of Alloys and Composites. Beijing, 2010.
[12] MASUKU E P, GOVENDER G, IVANCHEV L, M?LLER H. Rheocasting of Al-Cu alloy A201 with different silver contents[J]. Solid State Phenomena,2008, 141/143: 151-156.
[13] M?LLER H, MASUKU E P. The influence of liquid surface segregation on the pitting corrosion behaviour of semi-solid metal high pressure die cast alloy F357[J]. Open Corrosion Journal,2009, 2: 216-220.
[14] LIU D, ATKINSON H V, KAPRANOS P, JIRATTITICHAROEAN W, JONES H. Microstructural evolution and tensile mechanical properties of thixoformed high performance aluminium alloys[J]. Materials Science and Engineering A,2003, 361(1/2): 213-224.
[15] BRUWER R, WILKINS J D, IVANCHEV L H, ROSSOUW P, DAMM O F R A. Method of and apparatus for processing of semi-solid metal alloys: US7368690[P]. 2008.
[16] QVARFORT R. New electrochemical cell for pitting corrosion testing[J]. Corrosion Science, 1988, 28 (2): 135-140.
[17] DU H, CHANG Y A, HUANG B, GONG W, JIN Z, XU H, YUAN Z, LIU Y, HE Y, XIE F-Y. Diffusion coefficients of some solutes in fcc and liquid Al: critical evaluation and correlation[J]. Materials Science and Engineering A, 2003, 363 (1/2): 140-151.
(Edited by ZHAO Jun)
Corresponding Author: E. P. Masuku; Tel: +27-12-8413052; Fax: +27-12-8413378; E-mail: emasuku@csir.co.za