
Corrosion resistance, composition and structure of
RE chemical conversion coating on magnesium alloy
YU Sheng-xue(于升学)1, 2, CAO Jing-yu(曹靖瑜)2, CHEN Ling(陈 玲)2,
HAN Jing(韩 婧)2, ZHANG Rui-jun(张瑞军)1
1. State Key Laboratory of Metastable Materials Science and Technology, Yanshan University,
Qinhuangdao 066004, China;
2. Department of Applied Chemistry, Yanshan University, Qinhuangdao 066004, China
Received 12 June 2008; accepted 5 September 2008
Abstract: Golden yellow rare earths chemical conversion coating was obtained on the surface of magnesium alloy by immersing in cerium sulfate solution. The corrosion resistance of RE conversion coating was evaluated using immersion test and potentiodynamic polarization measurements in 3.5% NaCl solution. The morphologies of samples before corrosion and after corrosion were observed by SEM. The structures and compositions of the RE conversion coating were studied by means of XPS, XRD and IR. The results show that, the conversion coating consists of mainly two kinds of element Ce and O, the valences of cerium are +3 and +4, and OH- exists in the coating. The anti-corrosion property of magnesium alloy is increased obviously by rare earths conversion coating, Its self-corrosion current density decreases and the coating has self-repairing capability in the corrosion process in 3.5% NaCl solution.
Key words: magnesium alloy; chemical conversion coating; corrosion resistance; rare earths
1 Introduction
Magnesium alloy possesses the lowest density among all the metal materials. It is about 33% lighter than that of aluminum, about 73% than that of zinc alloy and about 77% than that of steel. Magnesium alloy has many other special characteristics, so it is widely applied in fields such as automotive industry, aerospace, communication, optical instrument, and computer[1-4]. But the application of magnesium alloy is restricted seriously by its poor anti-corrosion property[5-7]. Therefore, magnesium alloy must be treated before preparing anti-corrosive and decorative coatings[8-12]. Because of easy manipulation and low cost, chemical conversion coating on magnesium alloy has attracted more and more attention. At present, chromate is industrially used as chemical conversion coating on magnesium alloy, while Cr6+ is so toxic that its application is restricted, so it is very important to develop a new chemical conversion coating technology without Cr6+ for magnesium alloy[13-14].
Although rare earths solution can also be used to prepare chemical conversion coating on magnesium alloy, this technology has not been applied widely so far[15-16]. In the work, RE cerium conversion coating was prepared on magnesium alloy, its microstructure and chemical composition were studied and its anti-corrosion property was evaluated and discussed.
2 Experimental
Samples were magnesium alloy AZ81E (Al 8.4, Mn 0.4, and Zn 0.6) with the size of 25 mm×2.0 mm×25 mm. The samples surfaces were firstly polished by the abrasive papers (No.600, 800, and 1000) and the buffing machine and then cleaned in the distilled water, acetone and absolute alcohol. Parameters of RE conversion treatment is listed in Table 1.
The polarization curves of rare earths conversion coating in 3.5% NaCl solution were tested to evaluate the anti-corrosion property. The saturated calomel electrode was used as reference electrode, and the large-area Pt electrode as the assistant electrode. The area of sample exposed in solution was 1 cm2. The scanning speed was 10 mV/s.Morphologies of the conversion coating were observed by SEM(KYKY2800-B). Structure analysis was carried out by means of Dmax-rB X-ray diffractometer with the accelerating voltage of 50 kV, current density of 50 mA and Cu Kα radiation.
Table 1 Treatment process and suitable technological process of rare earths conversion coating

Composition of RE conversion coating was analyzed by XPS(ESCALABMKⅡ) with Al Kα standard source radial ray. The voltage, current and pressure were 12.5 kV, 20 mA and 6 mPa, respectively.
Functional groups of RE conversion coating were obtained by IR (EQUINOX55).
3 Results and discussion
3.1 Anti-corrosion properties of conversion coatings
The polarization curves of the magnesium alloy samples with and without RE conversion coating in 3.5% NaCl solution are shown in Fig.1. It is obvious that the self-corrosion current of the sample with rare earths conversion coating is much smaller than that of the sample without rare earths conversion coating, and its corrosion potential is more positive. In the anodic part of the curve, the corrosion current of the bare sample increases gradually and smoothly with potential, while that of the coated sample shows zigzag in the anodic part of the curve. This kind of change in the anodic part of polarization curve is probably due to the self-repairing of the RE conversion coating during corrosion process.
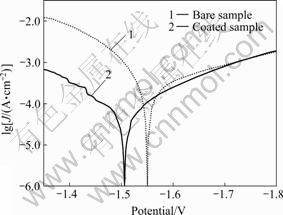
Fig.1 Tafel curves of samples in 3.5% NaCl solution
The surface morphologies of the rare earths conversion coatings are shown in Fig.2(a). The surface morphologies of magnesium alloy samples with and without RE conversion coating that has been corroded in 3.5% NaCl for 30 min are shown in Figs.2(b) and 2(c). It can be seen that the substrate surface is covered evenly by RE conversion coating (Fig.2(a)). The bare sample without conversion coating is corroded seriously. Its surface is uneven and there are some white corrosion product and black corrosion holes on the surface (Fig.2(b)). While no obvious corrosion happens on the surface of coated sample except that the top layer drops. Thus, it can be deduced that the adhesion between the coating and the substrate is tight and the RE conversion coating protects the substrate from corrosion.

Fig.2 Surface morphologies of magnesium alloy samples: (a) With conversion coating; (b) Bare sample after immersion in NaCl solution; (c) With conversion coating after immersion in NaCl solution
The open circuit potential―time curve of the coated sample was recorded in 3.5% NaCl solution on CHI660A type electrochemical station at room temperature. The saturated calomel electrode was used as reference electrode and Pt electrode as the assistant electrode with the sample interval of 1 mV(shown in Fig.3). It can be seen that the potential declines first, increases to a plateau near -1.315 V, and then declines again slowly. The change of the potential can be related to the change of coating structure. At the beginning of corrosion, corrosive medium is easy to enter the coating through the holes on top coating to result in the decline of the open circuit potential. Ce in the coating with passivation capability can repair the destroyed coating, and the inner layer may be compact enough to resist corrosion, therefore the open circuit potential increases to a plateau. With the time going along, outside layer is destroyed, Cl- in NaCl solution begins to corrode the inner layer, the integrality of inner is destroyed and part of substrate is exposed to be corroded, which results in the declined potential.

Fig.3 Curves of potential―time of sample with coating in 3.5% NaCl solution
3.2 Phase analysis of RE conversion coating
The XRD spectrum of rare earths conversion coating on magnesium alloy is shown in Fig.4. Compared with standard pattern, it can be found that Peak 1, 11, 12, 17 and 21 are from peaks of Ce2O3 and Peak 8, 9, 15 and 23 from the magnesium matrix, Peak 4, 6, 16 and 18 are diffraction peaks of CeO2. Because X-ray can penetrate tens of micron, the peaks of the substrate appear in the pattern from the surface to substrate. Besides the mentioned peaks, other peaks can not be identified. Therefore, XPS analysis of conversion coating was carried out in order to identify the composition further.
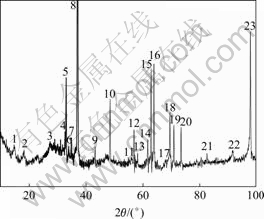
Fig.4 XRD spectrum of rare earths coating
3.3 Composition analysis of RE conversion coating
In the survey spectrum of XPS analysis for rare earths conversion coating (Fig.5), it can be seen that peaks of O 1s, Ce 3d, and Ce 4d are strong, which indicates that the main components of conversion coating are Ce and O. In order to affirm the exiting form of Ce and O in the coating, the peaks of Ce and O were analyzed separately.
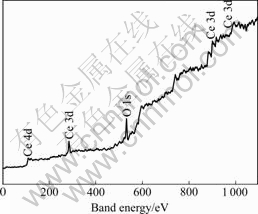
Fig.5 Survey spectrum of XPS analysis for rare earths conversion coating
XPS spectrum of Ce 3d region for Ce conversion coating is shown in Fig.6. The binding energy data according to the peaks in Fig.6 are: 880.4 eV (Peak 1), 882.7 eV (Peak 2), 884.3 eV (Peak 3), 885.6 eV (Peak 4), 889.4 eV (Peak 5), 898.9 eV (Peak 6), 900.3 eV (Peak 7), 901.7 eV (Peak 8), 902.9 eV (Peak 9), 908.2 eV (Peak 10) and 917.5 eV (Peak 11). Peak 1 to Peak 6 attribute to Ce 3d5/2; Peak 7 to Peak 9 attribute to Ce 3d3/2. Because Peak 6 and Peak 11 attribute to Ce4+ 3d94f0, Peak 2 and Peak 5 are symbols of compounds of Ce4+. It can be affirmed that there is compound of Ce4+ in the coating. Peak 1, 4 and 10 are symbols of compounds of Ce3+ (for example Ce2O3, Ce(OH)3, etc). From the analysis of XPS spectrum above, it can be concluded that the valences of cerium in magnesium alloy conversion coating are +3 and +4.

Fig.6 XPS of Ce 3d region for Ce conversion coating
XPS spectrum of O 1s region for Ce conversion coating is shown in Fig.7. It can be seen that the binding energy value corresponding to the peak is about 531.98 eV, and the spike is wide. After fitting, the peak can be divided into two different peaks, which means the oxygen in conversion coating has more than two valences. One peak, 530.35 eV stands for O 1s in metal oxide (binding energy of O 1s in Ce2O3 is 530.3 eV, binding energy of O 1s in CeO2 is 529.43 eV). And for other peaks, 531.98 eV stands for O 1s in M―OH. IR spectrum of conversion coating is shown in Fig.8. It can be seen that there are strong absorption peak between 2 800 cm-1 and 3 700 cm-1. The peaks between 3 200 cm-1 and 3 700 cm-1 are the characteristic peaks of O―H. Combined with XPS spectrum of O 1s, it can be concluded that there is hydroxyl in Ce conversion coating.
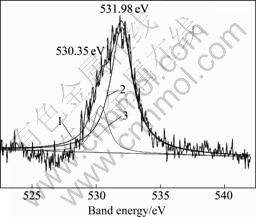
Fig.7 XPS of O 1s region for Ce conversion coating: 1―O 1s before fitting; 2―O 1s in M―OH; 3―O 1s in metal oxide
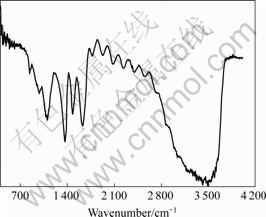
Fig.8 IR region for Ce conversion coating
From the analysis above, it can be realized that the main components of magnesium alloy RE conversion coating are cerium oxide and cerium hydrate, and valences of cerium are +3 and +4. On one hand, magnesium alloy RE conversion coating separates the substrate from corrosive medium; on the other hand, Ce+4 in the coating has passivation capability and it can repair the destroyed coating. This repairing process can be confirmed by the zigzag tooth shape in anodic part of polarization curve.
3.4 Analysis and discussion
In cerium sulfate solution, many micro-batteries form on the surface of magnesium alloy sample because of its electrochemical unevenness. The anode reaction is the dissolution of magnesium, and the cathode reaction is the deoxidizing of H+. The electrode reactions are as follows:
Anode reaction: Mg-2e= Mg2+ (1)
Cathode reaction: 2H+ +2e = H2↑ (pH=2.0) (2)
At the same time, because the electrode potential of Ce(SO4)2+e=Ce3++SO42- is 1.44 V and that of Mg2++2e=Mg is -2.372 V, another reaction will occur as the following:
Mg+Ce4+=Mg2++Ce3+ (3)
Besides, part of Ce4+ turns to Ce3+ because of the high oxidation ability of Ce4+:
4Ce4+ + 2H2O2 → 4Ce3+ + 2O2↑+ 4H+ (4)
Therefore, there are Ce4+, Ce3+ and Mg2+ in the liquid layer of sample surface. When pH value rises to a certain number, hydrates coating will deposit on sample surface. Because Ksp[Ce(OH)3]=1.0×10-20, Ksp[Ce(OH)4]=4.1× 10-51, and Ksp[Mg(OH)2]=5.61×10-12, the concentration of Ce4+ and Ce3+ reach a balance in solution, and there are both Ce(OH)3 and Ce (OH)4 in conversion coating. The forming reactions of the coating are as follows:
Ce4+ + 4OH-=Ce(OH)4↓ (5)
Ce3+ + 3OH-=Ce(OH)3↓ (6)
After some time, parts of hydrates of Ce3+ and the hydrate of Ce4+ are dehydrated to Ce2O3 and CeO2. So it can be concluded that rare earths conversion coating on magnesium alloy consists of cerium oxide and cerium hydrate, and the valences of cerium are +3 and +4. This conclusion can be confirmed by the XRD, XPS and IR spectra shown above.
4 Conclusions
1) The anti-corrosion properties of the magnesium alloy is enhanced obviously by rare earths conversion coating. The self-corrosion current density decreases and the self-repairing characteristics can be found in the anodic part of the polarization curve.
2) The rare earths conversion coating on the magnesium alloy consists of cerium oxide and cerium hydrate, and the valences of cerium are +3 and +4.
References
[1] LIU Zheng, WANG Yue, WANG Zhong-guang, LI Feng, SHEN Zhi-yong. Developing trends of research and application of magnesium alloys[J]. Chinese Journal of Materials Research, 2000, 14(6): 449-456. (in Chinese)
[2] ZENG Rong-cang, KE Wei, XU Yong-bao. Recent development and application of magnesium alloys[J]. Actametallurgica Sinica, 2001, 37(7): 673-685. (in Chinese)
[3] ZHANG Chun-xiang, CHEN Pei-lei, CHEN Jun. Application and research progress of magnesium alloys in automobile industry[J]. Foundry Technology, 2008, 29(4): 531-535. (in Chinese)
[4] XU Hong-xia, ZHANG Xiu-li. Green enviroment―friendly materials for 21 century―magnesium alloys[J]. Journal of Shanghai University of Engineering Science, 2007, 21(4): 322-325. (in Chinese)
[5] ZHAO Ming, WU Shu-sen, AN Ping, FUKUDA Y, NAKAE H. Growth of multi-elements complex coating on AZ91D magnesium alloy through conversion treatment[J]. Journal of Alloys and Compounds, 2007, 427(1/2): 310-315.
[6] CHEN Fei, ZHOU Hai, YAO Bin, QIN Zhen, ZHANG Qing-feng. Corrosion resistance property of the ceramic coating obtained through microarc oxidation on the AZ31 magnesium alloy surfaces[J]. Surface and Coatings Technology, 2007, 201(9/11): 4905-4908.
[7] ANIK M, K?RPE E. Effect of alloy microstructure on electroless NiP deposition behavior on Alloy AZ91[J]. Surface and Coatings Technology, 2007, 201(8): 4702-4710
[8] ZHU Li-qun, LI Wei-ping, SHAN Dan-dan. Effects of low temperature thermal treatment on zinc and/or tin plated coatings of AZ91D magnesium alloy[J]. Surface and Coatings Technology, 2006, 201(6): 2768-2775.
[9] JIN Fan-ya, CHUB P K, XUA Gui-dong, ZHAO Jun, TANG De-li, TONG Hong-hui. Structure and mechanical properties of magnesium alloy treated by micro-arc discharge oxidation using direct current and high-frequency bipolar pulsing modes[J]. Materials Science and Engineering A, 2006, 436/446(5): 123-126.
[10] SHI Hui-ying, YANG Wei, JIANG Bai-ling. Composite technology and coatings obtained by micro-arc oxidation and electrophoresis of AZ31 Mg-based[J]. Journal of Chinese Society for Corrosion and Protection, 2008, 28(3): 155-160. (in Chinese)
[11] HSIAO H Y, TSUNG H C. TSAI W T. Anodization of AZ91D magnesium alloy in silicate-containing electrolytes[J]. Surface & Coatings Technology, 2005, 199(2/3): 127-134.
[12] HOCHE H, SCHEERER H, PROBST D, BROSZEIT E, BERGER C. Development of a plasma surface treatment for magnesium alloys to ensure sufficient wear and corrosion resistance[J]. Surface and coatings Technology, 2003, 174/175: 1018-1023.
[13] UMEHARA H, TAKAYA M, TERAUCHI S. Chrome-free surface treatments for magnesium alloy[J]. Surface and Coatings Technology, 2003, 169/170: 666-669.
[14] AMY L R, BRESLIN C B, MANSFELD F. The Corrosion protection afforded by rare earth conversion coatings applied to magnesium[J]. Corrosion Science, 2000, 42(2): 275-288
[15] ZHANG Qing, LI Quan-gan, WEN Jiu-ba, ZHANG Xing-yuan. Application of rare earth in corrosion prevention of magnesium alloy[J]. Corrosion Science and Protection Technology, 2007, 19(2): 119-121. (in Chinese)
[16] LIU Jian-rui, GUO Yi-na, HUANG Wei-dong. Study on the corrosion resistance of phytic acid conversion coating for magnesium alloys[J]. Surface and Coatings Technology, 2006, 201: 1536-1541.
(Edited by CHEN Can-hua)
Corresponding author: YU Sheng-xue; Tel: +86-335-8061569; E-mail: yshxue@ysu.edu.cn