
Preparation and thermal decomposition mechanism of Mg,Al-hydrotalcite nano-crystals with titania doping
REN Qing-li(������)1, LUO Qiang(�ޡ�ǿ)2
1.School of Technical Physics, Xidian University, Xi��an 710071, China;
2. The Second Artillery Engineering College, Xi��an 710025, China
Received 10 April 2006; accepted 25 April 2006
Abstract: The highly pure nano-crystal Mg,Al-hydrotalcite with titania doping was synthesized by one-step liquid reaction method at atmospheric pressure. The preparation of the Mg,Al-hydrotalcite nano-crystal after doping titania was investigated according to the results of XRD, TEM, IR and DSC. Moreover, based on the DSC test results, the thermal mechanism functions of the Mg,Al-hydrotalcite with titania doping were studied after calculation and comparison.
Key words: Mg, Al-hydrotalcite; nano-crystal; kinetics; titania doping
1 Introduction
Flame-retardant materials are extensively used for safety in cable industry. This kind of material is required to have the following properties: avoiding short-circuits between neighboring wires, producing a minimum of toxic gases and dark smoke during pyrolysis, and keeping good mechanical properties. Such fine behaviors can be achieved by developing polymer compounds whose formulations contain a variable loading of inorganic fillers. Hydrotalcite is a new kind of such inorganic fillers. However, previous attempts to synthesize hydrotalcite are not practical for industrial scale production owing to high pressure and low purity. Moreover, the system theory study about Mg,Al-hydrotalcite is very limited. Besides, it is clear that thermal property is one of the most important properties of the materials [1-13], especially for the Mg, Al-hydrotalcite used as the flame-retardant agent.
The purpose of this study is to investigate the preparation and the thermal decomposition mechanism of Mg, Al-hydrotalcite nano-crystal with titania doping.
2 Experimental
The reagents MgCl2, NaOH, Al2(CO3)3 and TiO2 were weighed in a proper molar ratio according to the nominal composition Mg6-xTixAl2(OH)16CO3?4H2O and mixed in water by stirring for 16 h and about 90 �� and atmospheric pressure. The slurry was then dried at 90 ��.
X-ray diffraction (XRD) data were obtained with a Rigaku D/MAX-2400 X-ray diffractometer. Differential scanning calorimeter (DSC) experiments were performed with America SDT2960 type thermal analyzer at different heating rates: 2.5, 5.0, 10.0 and 20.0 �桤min-1. The microstructures of the samples were observed by JEM-200CX transmission electron microscope (TEM). The infrared (IR) analyses were conducted with German Bruker infrared analyzer. Mg2+ concentration was analyzed by the rules of the EDTA complex titration determination. And Al3+ concentration was analyzed by the ultraviolet UV-1100 spectrophotometer.
3 Results and discussion
3.1 Preparation of Mg,Al-hydrotalcite with titania doping
Fig.1 shows the XRD patterns for samples with titania doping. The highly pure nanometer Mg, Al-hydrotalcite (JCPDS22-700) with titania doping can be obtained when the molar fraction of titania added is 1%, 2%, 4%, respectively. However, the sample with the composition of Mg,Al-hydrotalcite, Mg(OH)2, and TiO2 can be obtained when the molar fraction of titania added is larger than 10%. The crystal grain size is calculated according to the following Equation:
 (1)
(1)
where B is the diffraction line width; l is the wavelength of the diffraction line; �� is the BRAGGER angle.
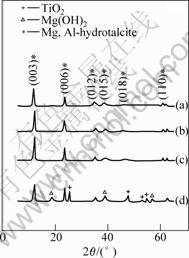
Fig.1 XRD patterns of Mg, Al-hydrotalcite with different molar fractions of titania doping: (a) 4%; (b) 2%; (c) 1%; (d) 10%
Fig.2 shows the TEM photos of Mg, Al-hydrotalcite with 4%(molar fraction) titania before the technological process of drying. The samples are nanosize crystal grains. And the diffraction shows that they belong to the hexagonal crystal lattice hydrotalcite.
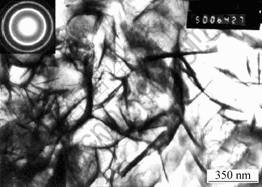
Fig.2 TEM photo and electronic diffraction of Mg, Al-hydrotalcite with 4%(malar fraction) titania doping
Fig.3 shows the IR patterns for samples with/without titania doping. IR analyses results show that there are two characteristic absorption peaks from 550 cm-1 to 750 cm-1 for the samples with titania doping (see Fig.3(b)), which are the feature peaks of Ti��O.
Fig.4 shows the patterns of DSC of Mg, Al- hydrotalcite samples with 4% titania doping. There are

Fig.3 IR patterns of the Mg,Al-hydrotalcite: (a) Original; (b) with 4%(molar fraction) titania doping

Fig.4 DSC curves of Mg,Al-hydrotalcite with 4%(molar fraction) titania doping tested at different heating rates: (a) 2.5 ��?min-1; (b) 5 ��?min-1; (c) 10 ��?min-1; (d) 20 �桤min-1
two endothermic peaks during the thermal decomposition of the Mg,Al-hydrotalcite with titania doping.
It is well known that hydrotalcite belongs to the hexagonal crystal lattice whose space lattice radius is 3 m. Hydroxyl (OH-) piles up and constructs the concentrated double-deck where Mg2+ and Al2+ are distributed in the octahedral structure space randomly. Therefore, the positively charged basic layer [Mg6Al2(OH)16]2+ is formed. And the negatively charged middle layer [CO32-+4H2O]2- is sandwiched between the two positively charged basic layers.
The test results of the reaction time depending on Mg2+ and Al3+ concentration show that the concentrations of both Mg2+ and Al3+ are quickly reduced when the reaction just begin for the samples with less than 10%(molar fraction) titania doping. However, when the titania doping amount is larger than 20%, the concentrations of both Mg2+ and Al3+ are almost not reduced; and the TiO2 crystal sample is obtained. So, it is reasonable to consider that Mg2+ and Al3+ which are
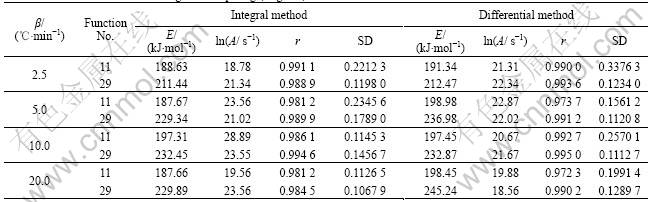
Table 1 Choice data after calculating and comparing (stage ��)
consumed are used to produce the Mg, Al-hydrotalcite crystal embryo and Ti4+ ions (r=0.068 nm) to enter the octahedral structure space of the hydroxyl (OH-) and replace Mg2+ ions (r=0.072 nm) after taking into account the results of XRD and IR furthermore (see Figs.1 and 3). Moreover, the number that Ti4+ enters the octahedral structure space of the hydroxyl (OH-) and replace Mg2+ is very limited.
3.2 Thermal decomposition mechanism
There are two endothermic stages during the Mg, Al-hydrotalcite thermal decomposition. In the first decomposition stage, the crystal water is released. And in the second decomposition stage, the hydroxyl (OH-) octahedral structure is destroyed, the hydroxyl (OH-) and the gas of H2O and CO32- anion release, and CO2, MgO, Al2O3 and TiO2 are formed. And the mechanism function of the second thermal decomposition stage is studied so as to promote the study of the electrical cable made by compounding Mg, Al-hydrotalcite flame-retardant agent with other organic materials.
The Sativa-Sestak integral method, Achar differential method and Kissinger method are chosen here. And their mechanism equations are as follows[14]:
 (2)
(2)
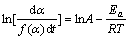 (3)
(3)
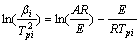 (4)
(4)
where���� is the decomposition rate; T is the absolute temperature; A is the former factor; G(��) and f(��) are the mechanism functions; �� is the heating rate.
Based on the test results of DSC, lgG(��) and 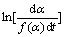 of the forty-three recognized mechanism functions [16] are calculated according to the basic data of T, �� and ��. And all of these data are compared. Two of them for the Mg, Al-hydrotalcite with titania doping are chosen (shown in Table 1).
of the forty-three recognized mechanism functions [16] are calculated according to the basic data of T, �� and ��. And all of these data are compared. Two of them for the Mg, Al-hydrotalcite with titania doping are chosen (shown in Table 1).
The optimum mechanism function should satisfy the following rules [14]:
1) Its linear correlation coefficient (r) is greater than 0.98;
2) Its standard error (SD) is smaller than 0.3;
3) Its kinetic parameter must be in concordance with the calculated results by using other differential or integral methods.
So, according to the calculated results of Kissinger method, whose calculated apparent activation energy (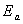 ) is 235.67 kJ/mol, r is 0.993 2 and SD is 0.1016, the rational integral mechanism function should be (1-��)-1-1 (function No.29).
) is 235.67 kJ/mol, r is 0.993 2 and SD is 0.1016, the rational integral mechanism function should be (1-��)-1-1 (function No.29).
4 Conclusions
The highly pure flake nanocrystalline Mg,Al-hydrotalcite with titania doping is synthesized. And its integral mechanism function is confirmed as the functional form of (1-��)-1-1.
References
[1] TOMASZEWICZ E, KOTFICA M. Mechanism and kinetics of thermal decomposition of nickel(II) sulfate(VI) hexahydrate[J]. J Thermal Analysis and Calorimetry, 2004, 77(1): 25-31.
[2] STEPANOV R S, ASTAKHOV A M, KRUGLYAKOVA L A, PEKHOTIN K V. Kinetics and mechanism of liquid-phase thermal decomposition of cyanoethyl-n-nitramines[J]. Russian Journal of General Chemistry, 2002, 72(8): 1256-1258.
[3] IU B, THOMAS P S, RAY A S, WILLIAMS R P. The effect of sampling conditions on the thermal decomposition of electrolytic manganese dioxide [J]. Journal of Thermal Analysis and Calorimetry, 2004, 76(1): 115-122.
[4] ZELI? J, RU?I? D, KRSTULOVI? R. Kinetic analysis of thermal decomposition of Ca(OH)2 formed during hydration of commercial portland cement by DSC[J]. Journal of Thermal Analysis and Calorimetry, 2002, 67(3): 613-622.
[5] ZHORIL R, MASHLAM M, PAPAEFTHYMIOU V, HADJIPANAYIS G. Thermal decomposition of Fe2(SO4)3: Demonstration of Fe2O3 polymorphism[J]. Journal of Radioanalytical and Nuclear Chemistry, 2004, 255(3): 413-417.
[6] ISAKOV G N. Mechanism and microkinetics of soot formation upon thermal decomposition of carbon and fiberglass plastics under one-sided radiant-convective heating[J]. Combustion, Explosion, and Shock Waves, 2005, 38(2): 169-175.
[7] KHAIROU K S. Kinetics and mechanism of the non-isothermal decomposition[J]. Journal of Thermal Analysis and Calorimetry, 2004, 69(2): 583-588.
[8] POPOV E A, AHOKAS J, ELORANTA J, KUNTTU H. Thermal decomposition mechanism of N2-impurity helium solids[J]. J Low Temperature Physics, 2004, 139(5-6):557-562.
[9] GOTOR F J, MACIAS M, ORTEGA A, CRIADO J M. Comparative study of the kinetics of the thermal decomposition of synthetic and natural siderite samples[J]. J Thermal Analysis and Calorimetry, 2002, 67(3): 613-622.
[10] LIKACHEVA Y A, VENIAMINOV S A and PAUKSHTIS E A. Thermal decomposition of NH4-analcime[J]. Physics and Chemistry of Minerals, 2004, 31(5): 306-312.
[11] DEUKKL. Kinetic evaluation of mechanistic models for O2 release from ZSM-5-supported [Cu2+ �CO�CCu2+] ions by thermal reduction or chemical interaction with impinging N2O molecules[J]. J Thermal Analysis and Calorimetry, 2003��68(3): 331-339.
[12] GALWEY A K, BROWN B E. Thermal Decomposition of Ionic Solid[M]. Amsterdam: Elsevier Science, 1999.
[13] BANO A C, BRITO Z, PAREDES H, DOMINGUEZ N and SANCHEZ Y. Application of romero Garc��a method to the degradation process of DGEBA/ETDA filled with ultra fine metallic copper[J]. Polymer Bulletin, 2005, 54(6): 399-407.
[14] SHI Z, HU R Z and GAO S L. Thermal Analysis Kinetics[M]. Beijing: Science Press, 2001. (in Chinese)
(Edited by CHEN Can-hua)
Foundation item: Project (2004E113) Supported by Natural Science Basic Research Plan of Shaanxi Province of China
Corresponding author: REN Qing-li; Tel: +86-29-82551108; E-mail: qlren@mail.xidian.edu.cn, abcdrenqingli@263.sina.com