Trans. Nonferrous Met. Soc. China 23(2013) 1520-1524
Preparation of nanostructure silver powders by mechanical decomposing and mechanochemical reduction of silver oxide
Gholam Reza KHAYATI1, Kamal JANGHORBAN2
1. Department of Material Science and Engineering, Shahid Bahonar University of Kerman, P. O. Box No. 76135-133, Kerman, Iran;
2. Materials Science and Engineering Department, School of Engineering, Shiraz University, Zand Avenue, Shiraz, Iran
Received 2 July 2012; accepted 16 January 2013
Abstract: The mechanical decomposing and mechanochemical reductions of silver oxide for preparation of nanocrystalline silver powders by high planetary ball mill was investigated. XRD and HRSEM techniques were used to characterize the structural evolution and morphological changes of products. The results show that the nanostructured silver with an average crystallite size of 14 nm and internal strain of 0.75% is synthesized by mechanical decomposing of Ag2O after 95 h milling. While, the product of mechanochemical reduction of silver oxide using graphite after 22 h milling is nanostructured silver with an average crystallite size of 28 nm and internal strain of 0.44%.
Key words: nanocrystalline silver; silver oxide; mechanical decomposing; mechanochemical reduction
1 Introduction
Mechanochemistry is a branch of science referring to the chemical and physicochemical reactions of substances due to the influence of mechanical force [1]. Recently, mechanochemistry has been widely applied in many fields, such as preparation of intermetallic compounds, extractive metallurgy, crystal engineering, coal industry, composites and complex oxides, nanocrystalline substances, building industry, agriculture, pharmacy and waste treatment [2-6]. According to the literature, a broad variety of Ag nanoparticle applications were reported ranging from sterilization of medical devices and purifying appliances to water treatment. Furthermore, due to the unique characteristics, Ag nanoparticles have also been used in a broad variety of application such as magnetic, biomedical, optoelectronic, dielectric, hygienic and healing purposes [7,8].
In our previous work, mechanochemical reduction of Ag2O was carried out in a planetary ball mill [9]. However, no information is available on the structural evolution of Ag2O mechanical decomposing as well as the effect of graphite on reaction mechanism and product properties. In this study, nanostructured silver has been synthesized using mechanical decomposing of Ag2O by high energy ball milling without any reducing agent, and the results are compared with the results of mechanochemical reduction of Ag2O using graphite.
2 Experimental
Raw materials were commercially pure Ag2O powder (99%, 5-40 ��m) and graphite (99.9%, 10-50 ��m). The powder samples were milled in a high energy planetary ball mill. Details of ball milling process for both mechanochemical reduction and mechanical decomposing are given in Table 1. The only difference between two types of reduction process was the composition of initial powder. Ag2O powder in mechanical decomposing and a mixture of graphite- Ag2O in mechanochemical reduction are used as initial materials.
The samples were characterized by high-resolution transmission electron microscopy (HRSEM, Hitachi S-4160) and X-ray diffraction (XRD, Philips PW-1730) using Cu K��. The broadening owing to the instrument was computed from Warren��s method [10]. Peak positions (2��) and the full-width at half maximum (FWHM) were obtained from the XRD spectra by OriginPro 8 from the Ag2O planes selected for the profile analysis. The line broadening due to the instrument was calculated from Warren��s method [11]. The average crystallite size and internal strain were estimated using Williamson-Hall plot [12].
Table 1 Details of planetary ball milling process
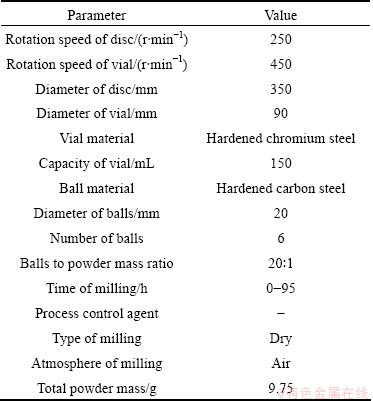
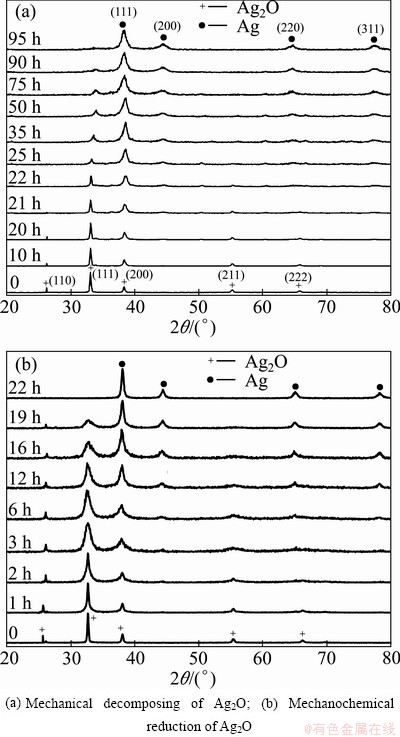
Fig. 1 XRD patterns as function of milling times
3 Results and discussion
Figure 1(a) shows the XRD spectra of mechanical decomposing of Ag2O milled for various times of 0, 10, 20, 21, 22, 25, 35, 50, 75, 90 and 95 h. Analysis of the XRD patterns shows that, up to 20 h milling, gradual refinement is the only considerable occurrence in Ag2O powders, and no detectable transformation is happens to introduce any new phase. The traces of silver could be produced after 21 h milling. The XRD results also show that the Ag2O peaks progressively disappear with increasing the milling time. In fact, they are well below the resolution limit of XRD after 95 h of milling, which suggests the completion of dissociation reaction as follows [13]:
Ag2O(s)=2Ag(s)+1/2O2(g), ��H=61080 J (1)
Figure 1(b) shows the XRD patterns of the mechanochemical reduction of Ag2O together with 40% (molar fraction) of extra graphite as reducing agent according to Eq. (2) [13]:
2Ag2O(s)+C(s)=4Ag(s)+CO2(g), ��H=-363560 J (2)
The samples were milled for various times up to 22 h. It could be seen that Ag peaks appear after a relatively short milling time of 3 h in comparison with that of mechanical decomposing of Ag2O, where Ag peaks are first observed after 21 h milling. The XRD results also show that the Ag2O peaks progressively disappear with increasing milling time. The fraction of Ag2O reduced during milling, ��, is estimated by considering the intensity of Ag2O (111) peak as follows [5]:
��=1-(It/I0) (3)
where It is the Ag2O (111) peak intensity of sample milled for a certain time and I0 is the intensity of Ag2O (111) peak of unmilled sample. Figure 2 shows the calculated values of �� as a function of milling time for mechanical decomposing and mechanochemical reduction of Ag2O. An incubation time of approximately 22 h in mechanical decomposing and 3 h in mechanochemical reduction of Ag2O are observed, then the rate of reduction reaction is increased. The presence of graphite in mechanochemical reduction performed as the reducing agent enhances the reducing potential of Ag2O in comparison to the mechanical decomposing and consequently the time dependence of �� is more drastic in mechanochemical reduction than in mechanical decomposing. It seems that the reduction energy barrier diminishes by increasing milling time due to the increasing of imperfection sites in both cases. Meanwhile, as a general trend, the rate of Ag2O reduction decreases slowly at the final stages of reduction process in both cases.
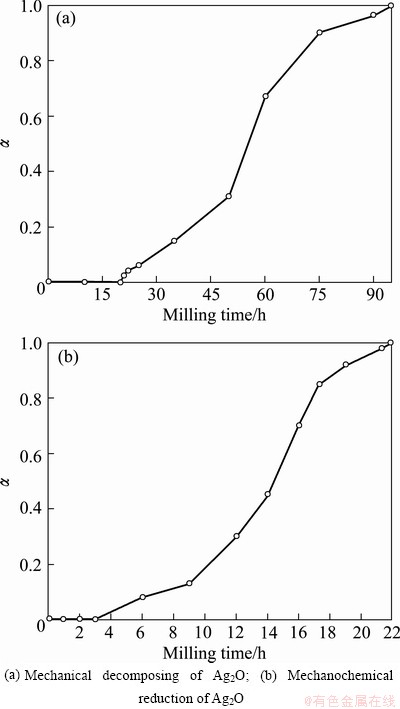
Fig. 2 Calculated values of �� as function of ball milling time
The changes in crystallite size and lattice strain of mechanical decomposing and mechanochemical reduction of Ag2O are shown in Fig. 3. The average crystallite size and internal strain of Ag particles prepared from mechanical decomposing of Ag2O (called S1 sample) show irregulartrend in comparison of Ag particles prepared from mechanochemical reduction of Ag2O (called S2 sample). As a general trend in Fig. 3, the milling process had two stages.
1) In the former, the Ag crystallite size decreases with milling time whereas the internal strain increases in Ag prepared from S1 sample. While, the changes of Ag crystallite size and strain prepared from S2 sample are exactly in opposite direction. This trend can be interpreted by the following reasons:
First, due to the lower reduction potential of Ag2O in S1 sample, the process prolongs in a longer time (up to 50 h). The longer time of milling enhances the dislocation density due to the plastic deformation and refining the structure by the formation of dislocation cells. As illustrated in Fig. 3(b), the major fraction of reaction (2) in S2 sample is completed up to 19 h, consequently a large fraction of strain-free silver particles are prepared in this period. These particles are deformed by further milling, but high preparation rate of strain-free silver could balance it. Second, the mechanical decomposing of Ag2O is an endothermic transformation (Eq. (1)) and consequently the operational temperature of vial is lower. However, the mechano- chemical reduction of Ag2O with graphite is an exothermic transformation (Eq. (2)) and the reaction in sample S2 is done in an enhanced temperature condition. Thus, dynamic recovery and recrystallization occur in the silver particles with high internal strains [13].
2) In the later, according to Fig. 3(a), the internal strain decreases and crystallite size increases in S1 sample. It can be seen that under specific conditions of mechanical decomposing of Ag2O for S1 sample, enhancing the temperature relatively reduces the rate of changes of crystallite size and internal strain with milling time. The trends of changes are reversed by increasing the internal strain and decreasing the crystallite size in S2 sample. In this case, the effect of severe plastic deformation on Ag particles prepared from S2 sample dominates the generation of latest strain free particles as well as the recrystallization and dynamic recovery.
Fig. 3 Evolution of crystallite size and internal strain of Ag particles as function of milling time
The strain and crystallite size of Ag powder prepared by mechanical decomposing and mechanochemical reduction of Ag2O are summarized in Table 2.
Table 2 Strain and crystallite size of Ag powder prepared from samples S1 and S2
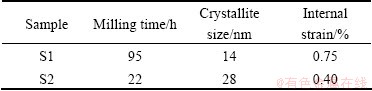
The crystallite size of Ag in S1 sample reaches to a stable size of about 14 nm in 95 h, and in S2 sample it reaches to 28 nm in 22 h (Table 2). The lattice strains of Ag in S1 and S2 samples are 0.75% and 0.40%, respectively. Thus, the silver powders with the smaller crystallite size contain larger lattice residual strains. This is related to longer milling time of silver powders produced by mechanical decomposing.
Moreover, from the start point of Ag preparation in Figs. 1(a) and 1(b), the smaller crystallite size of S1 sample after 95 h milling probably refers to the size of Ag2O particles. The size of Ag2O particles formed in S1 sample after 21 h is smaller than that of the Ag2O particles in S2 sample after 3 h, and consequently due to prolonged mechanical decomposing duration in S1 sample, the high density of defects is generated in fine Ag2O particles. Therefore, the number of Ag nucleation sites in the S1 sample is increased. So, the kinetics of nucleation of Ag from Ag2O increases in S1 sample due to the increase in nucleation sites as well as the reduction of the activation energy for non-homogeneous nucleation. As the ratio of the nucleation to growth rates is high, many nuclei will create before the reduction is completed, and fine crystallite sizes will be obtained.
Figure 4 shows the HRSEM images of Ag powders prepared from S1 and S2 samples. The Ag particles after 95 h (Fig. 4(a)) are in irregular shape and have a wide distribution in particle size ranging from about 2 to 30 ��m. Moreover, the agglomeration of particles of silver in large aggregates and plate-like nature can be observed. As silver is ductile, extending milling flattens the surface of Ag particles by ball�Cpowder�Cball collisions.
Figure 4(b) shows the HRSEM of S2 sample. Although the silver particles are ductile and deformed by increasing milling time, the high lubricating effect of graphite could compensate it. Consequently, the tendency to fracture dominates over cold welding. The shape of Ag particles after 22 h is regular with relatively spherical geometry in the nanoscale regime.

Fig. 4 HRSEM images of Ag prepared from sample S1 (a) and sample S2 (b)
4 Conclusions
1) The preparation of Ag powder by mechanical decomposing and mechanochemical reduction of Ag2O under the given experimental conditions takes place after 95 h and 22 h milling respectively.
2) The crystallite size and internal strain of the silver powders for reduction of Ag2O with milling energy reach to 14 nm and 0. 75%, respectively, after 95 h of ball milling.
3) The crystallite size and internal strain for silver produced through reduction of Ag2O with graphite are 28 nm and 0.40%, respectively, after 22 h of ball milling.
4) Smaller crystallite size and larger lattice residual strains in Ag produced through mechanical decomposing of Ag2O may result in better mechanical properties.
References
[1] BALAZ P. Mechanochemistry in nanoscience and minerals [M]. Heidelberg, Berlin: Springer-Verlag, 2008: 18.
[2] WIECZOREK-CIUROWA K, GAMRAT K. Some aspects of mechanochemical reactions [J]. Materials Science-Poland, 2007, 25: 219-232.
[3] KHAYATI G R, JANGHORBAN K. A thermodynamic approach to synthesis of silver nanocrystalline by mechanical milling of silver oxide [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(2): 543-547.
[4] KHAYATI G R, JANGHORBAN K. An investigation on the application of process control agents in the preparation and consolidation behavior of nanocrystalline silver by mechanochemical method [J]. Adv Powder Technol, 2012, 23: 808-813.
[5] KHAYATI G R, JANGHORBAN K, SHARIAT M H. Isothermal kinetic study of mechanochemically and thermally synthesized Ag from Ag2O [J]. Transactions of Nonferrous Metals Society of China, 2012, 22: 935-942.
[6] RAZAVI TOUSI S S, YAZDANI RAD R, SALAHI E, RAHIMIPOUR M R, KAZEMZADE A, RAZAVI M. Structural Evolution of Al-20%(Wt) Al2O3 system, during ball milling stages [J]. International Journal of Engineering, 2009, 22(2): 169-178.
[7] AHAMED M, ALSALHI M S, SIDDIQUI M K. Silver nanoparticle applications and human health [J]. Clinica Chimica Acta, 2010, 411(23-24): 1841-1849.
[8] TOLAYMAT T M, EL BADAWY A M, GENAIDY A, SCHECKEL K G, LUXTON T P, SUIDAN M. An evidence-based environmental perspective of manufactured silver nanoparticle in syntheses and applications: A systematic review and critical appraisal of peer-reviewed scientific papers [J]. Science of the Total Environment, 2010, 408(5): 999-1006.
[9] KHAYATI G R, JANGHORBAN K. The nanostructure evolution of Ag powder synthesized by high energy ball milling [J]. J Advanced Powder Technology, 2011, 23: 393-397.
[10] CULLITY B D, STOCK S R. Elements of X-ray diffraction [M]. Prentice Hall, NJ: Upper Saddle River, 2001: 399.
[11] BALZAR D. X-ray diffraction line broadening: Modeling and application to high-Tc superconductors [J]. Research of the National Institute of Standards and Technology, 1993, 98: 321-353.
[12] WILLIAMSON G. K, HALL W H. X-Ray line broadening from filed aluminum and wolfram [J]. Acta Materialia, 1953, 1: 22-31.
[13] GASKELL D R. Introduction to metallurgical thermodynamics [M]. Fourth Ed. Taylor & Francis Books, Inc, 2003.
��е�ֽ��Լ���е��ѧ��ԭ�������Ʊ�������ĩ
Gholam Reza KHAYATI1, Kamal JANGHORBAN2
1. Department of Material Science and Engineering, Shahid Bahonar University of Kerman, P. O. Box No. 76135-133, Kerman, Iran;
2. Materials Science and Engineering Department, School of Engineering, Shiraz University, Zand Avenue, Shiraz, Iran
ժ Ҫ�����ø���������ĥ�о���е�ֽ��Լ���е��ѧ��ԭ���������Ʊ�������ĩ���ա�����XRD��HRSEM�Բ���Ľṹ�Լ���ò�ı仯���б�����������������û�е�ֽ�������������ĥ95 h��õ���������ĩ��ƽ������ߴ�Ϊ14 nm����Ӧ��Ϊ0.75%����ͨ����е��ѧ��ԭ��������������ʯī����ĥ22 h��õ���������ĩ��ƽ������ߴ�Ϊ28 nm����Ӧ��Ϊ0.44%��
�ؼ��ʣ�������������������е�ֽ⣻��е��ѧ��ԭ
(Edited by Chao WANG)
Corresponding author: Gholam Reza KHAYATI; Tel: +98-915-1903477; Fax: +98-341-2111860; E-mail: khayati@uk.ac.ir
DOI: 10.1016/S1003-6326(13)62625-4