�����Ʊ����ṹ����п�Ĵ�ѧ��ѧ����
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2015���12��
�������ߣ�Zahra MOUSAVI Mahdiyeh ESMAEILI-ZARE Masoud SALAVATI-NIASARI
����ҳ�룺3980 - 3986
�ؼ��ʣ�����п�����ṹ������ˮ�ԣ���ѧ���ܣ���ѧ����
Key words��zinc chromite; nanostructures; superhydrophilicty; magnetic property; optical property
ժ Ҫ�����������Ʊ����ṹ����п(ZnCr2O4)�������������700 ��C�˻��ZnCr2O4Ϊ�������õļ⾧ʯ�ṹ���о���Ӧʱ��������书�ʶ�ZnCr2O4�����ߴ����ò��Ӱ�졣�ֱ����X��������(XRD)��ɨ��羵(SEM)��������ɢX����(EDX)����羵(TEM)�����������(DRS)�����·���(PL)���ס�����Ҷ�������(FTIR)�ȼ���������Ʒ��ǿ�ƶ����ϳɵ�ZnCr2O4��Ʒ���б����������������(DRS)����õ�ZnCr2O4���ṹ�Ĺ����϶Ϊ3.50 eV�����·��������Ҳ֤ʵ�˸ý����
Abstract: The nanostructures of zinc chromite (ZnCr2O4) were fabricated by the microwave method. It was shown that the well-crystallized spinel structure is formed after annealing at 700 ��C. The influence of reaction time and irradiation power of oven on the size and shape of the as-prepared ZnCr2O4 samples was studied. The synthesized samples were characterized by X-ray diffraction (XRD), scanning electron microscopy (SEM), energy dispersive X-ray (EDX), transmission electron microscopy (TEM), diffuse reflectance spectroscopy (DRS), photoluminescence (PL) spectroscopy, Fourier transform infrared (FTIR) spectra and vibrating sample magnetometry (VSM), respectively. The optical band gap calculated using DRS was found to be 3.50 eV for ZnCr2O4 nanostructures. Photoluminescence measurements also confirmed this result.
Trans. Nonferrous Met. Soc. China 25(2015) 3980-3986
Zahra MOUSAVI, Mahdiyeh ESMAEILI-ZARE, Masoud SALAVATI-NIASARI
Institute of Nano Science and Nano Technology, University of Kashan, Kashan 87317-51167, Iran
Received 23 January 2015; accepted 7 May 2015
Abstract: The nanostructures of zinc chromite (ZnCr2O4) were fabricated by the microwave method. It was shown that the well-crystallized spinel structure is formed after annealing at 700 ��C. The influence of reaction time and irradiation power of oven on the size and shape of the as-prepared ZnCr2O4 samples was studied. The synthesized samples were characterized by X-ray diffraction (XRD), scanning electron microscopy (SEM), energy dispersive X-ray (EDX), transmission electron microscopy (TEM), diffuse reflectance spectroscopy (DRS), photoluminescence (PL) spectroscopy, Fourier transform infrared (FTIR) spectra and vibrating sample magnetometry (VSM), respectively. The optical band gap calculated using DRS was found to be 3.50 eV for ZnCr2O4 nanostructures. Photoluminescence measurements also confirmed this result.
Key words: zinc chromite; nanostructures; superhydrophilicty; magnetic property; optical property
1 Introduction
Zinc chromite (ZnCr2O4) is a mixed oxide which crystallizes in the cubic system and has a normal spinel structure. Non-magnetic Zn2+ and magnetic Cr3+ ions have strong preference for the tetrahedral A- and the octahedral B-sites, respectively. ZnCr2O4 is very attractive as air depollution catalytic material, for a variety of reactions like oxidative dehydrogenation of hydrocarbons, oxidation of hydrocarbons, synthesis of methanol [1], gas sensing [2], as photocatalyst [3,4] and humidity sensing [5]. Photocatalytic degradation processes have been widely applied as techniques of destruction of organic pollutants in wastewater and effluents [6]. ZnCr2O4 was synthesized previously by multifarious methods including mechanical activation [7], high-temperature solid-state reaction [8], micro-emulsion method [2], solution method [9] and spray pyrolysis [10]. The most general method for preparing spinels involves solid state reaction of the parent metal oxides that are mechanically mixed in the form of finely divided powders [7]. However, for completion of the reaction, a temperature of about 1100 ��C for several days is needed [5]. The damages of solid-state routes such as inhomogeneity, larger particle size and lack of stoichiometry control are avoided when the material is synthesized using a solution-based method. It is very important to recognize that proper choice of the synthesis route and precursor and the knowledge of its composition and structure are crucial to tailor-make a pure product. For this reason, in order to obtain spinel zinc chromite nanoparticles, we selected a synthesis method belonging to wet chemistry. Using microwave energy to synthesize the materials in a convenient and simple way has been recognized, since the method is faster, more economical and cleaner [11]. A variety of inorganic materials such as chalcogenides [12], nitrides [13], complex oxides [14], silicides [15] and zeolites [16], were synthesized using the microwave approach. Because microwave irradiation is easily generated, various Zn-related nanostructures were synthesized using this method [17-23].
In this work, we reported a novel and rapid microwave method for the preparation of ZnCr2O4 nanostructures. The nanostructures were synthesized from reaction between Zn(NO3)2��6H2O and CrCl3��6H2O in the presence of sodium dodecyl-benzene-sulfonate (SDBS) as surfactant. The effects of different parameters such as power of oven and time of irradiation were also studied.
2 Experimental
2.1 Materials and physical measurements
All the chemical reagents used in our experiments were of analytical grade, were purchased from Merck and used as received without further purification. The XRD patterns of the products were recorded by a Rigaku D-max C III XRD using Ni-filtered Cu K�� radiation. SEM images were obtained on Philips XL-30 ESEM equipped with an energy dispersive X-ray spectroscopy. The EDX analysis with an accelerating voltage of 20 kV was done. Room temperature photoluminescence (PL) was studied on a Perkin Elmer (LS 55) fluorescence spectrophotometer. Fourier transform infrared (FTIR) spectra were recorded on a Shimadzu Varian 4300 spectrophotometer in KBr pellets. UV�CVis absorption spectra of the samples were obtained with a UV�CVis DRS spectrophotometer (Shimadzu, model UV-3101) with a scan rate of 5 nm/s. TEM image was obtained on a Philips EM208 transmission electron microscope with an accelerating voltage of 200 kV.
2.2 Synthesis of ZnCr2O4 nanostructures
In a general procedure, 0.2 g Zn(NO3)2��6H2O precursor and 0.1 g SDBS as surfactant were dissolved in 20 mL of distilled water under vigorous stirring. By adding 0.35 g CrCl3��6H2O and raising the pH to 11 with NaOH, a compound was precipitated. Afterward, the solution was exposed to microwave irradiation with different powers and time. The microwave oven followed a working cycle of 30 s on and 60 s off (30% power). After heating under microwave irradiation, the nanoparticles were cooled to room temperature naturally. Precipitates were flittered, washed with deionized water and ethanol, and then dried at room temperature. Finally, dried precipitates were annealed in air at 700 ��C for 3 h. Table 1 shows the conditions of reactions in detail.
Table 1 Reaction conditions for preparation of ZnCr2O4 nanostructures

3 Results and discussion
3.1 XRD analysis
Figure 1(a) shows XRD pattern of the as-prepared ZnCr2O4, which indicates that this sample is a pure-phase compound. The product has peaks corresponding to the cubic ZnCr2O4 (space group: Fd3m) phase with cell constants a=b=c=8.3275 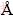 , which are in agreement with JCPDS No. 22-1107. The intense and sharp diffraction peaks suggest that the obtained product is well crystallized. Figure 1(b) shows the XRD pattern of ZnCr2O4 nanostructure before calcination. According to this figure, with calcination of the ZnCr2O4 samples at 700 ��C for 3 h, the crystallinity of the products is increased.
, which are in agreement with JCPDS No. 22-1107. The intense and sharp diffraction peaks suggest that the obtained product is well crystallized. Figure 1(b) shows the XRD pattern of ZnCr2O4 nanostructure before calcination. According to this figure, with calcination of the ZnCr2O4 samples at 700 ��C for 3 h, the crystallinity of the products is increased.
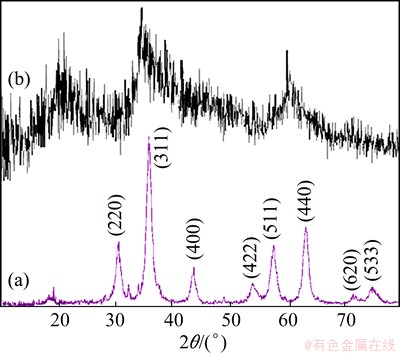
Fig. 1 XRD pattern of Sample 2 (a) and Sample 8 (b)
3.2 Morphology and microstructure analysis
To investigate the effect of irradiation time on the morphology of products, the reaction was performed in 4, 6 and 8 min at 600 W, as shown in Figs. 2(a)-(c), respectively. At first, when the reaction took place for 4 min, which was not enough for the separation of the particles, the agglomerated particles were obtained (Fig. 2(a)). By increasing the time to 6 min, the particles were separated. In addition, uniform particles were formed due to sufficient time for the reaction (Fig. 2(b)). When the reaction occurred for 8 min, the particles were agglomerated. This was due to the fact that after optimum amount of reaction time, further irradiation would increase as the prepared particles�� kinetic energy and collision of these particles would increase, which led to the agglomeration of particles (Fig. 2(c)). For investigating the effect of surfactant on the morphology and particle size of the products, one test was carried out in the absence of SDBS for 6 min at 600 W (Fig. 2(d)). This figure shows that the particles coalesced and turned into bulk structures.

Fig. 2 SEM images of different samples

Fig. 3 SEM images of as-prepared nanostructures
SEM images of the as-synthesized products at 6 min and different powers of 750 and 900 W are exhibited in Figs. 3(a) and (b), respectively. When the irradiation power was 600 W, small particles were obtained (Fig. 2(b)). Based on Figs. 3(a) and (b), it can be confirmed that by increasing the irradiation power to 750 and then 900 W, the agglomerated particles were produced. This is due to the fact that the particles have higher kinetic energy when the irradiation power is increased, and particles can fuse together again. By increasing the reaction time to 8 min at 900 W (Fig. 3(c)), the chances of collision between nanoparticles increase and the average diameter of nanoparticles decreases.
The morphological and structural features of ZnCr2O4 products, shown in Fig. 4, were characterized with transmission electron microscope (TEM), which show the shape of semi-sphere. The diameter of these nanoparticles is in the range of 10-15 nm.
3.3 FTIR analysis
FTIR spectroscopy has been widely applied in solid state chemistry because it can provide information on structural characteristics of inorganic solids in crystalline and amorphous states. Figure 5 demonstrates the FTIR spectrum of Sample 2 obtained after annealing up to 700 ��C. Two high frequency modes corresponding to peaks at 515.13 and 627.44 cm-1 mainly involve the displacement of oxide anions relative to the chromium cations along the direction of the octahedral chains, and tentatively assigned to Cr��O stretching [7].
3.4 EDS and PL analyses
X-ray energy dispersive spectroscopy (EDS) analysis measurements were used to characterize the chemical composition of the products (Fig. 6(a)). The results for ZnCr2O4 in the presence of SDBS at 600 W for 6 min (Sample 2) show that only elements Zn, Cr and O exist. In addition, neither N nor C signals were detected in the EDS spectrum, which means that no solvent or capping agent exists in the sample. Figure 6(b) shows the PL spectrum of ZnCr2O4 prepared by the microwave method. A sharp peak at 365 nm was observed and band gap of sample was calculated about 3.39 eV.

Fig. 4 TEM images of Sample 2

Fig. 5 FTIR spectrum of as-synthesized ZnCr2O4 nanostructures (Sample 2)
3.5 Contact angle analysis
The contact angle (CA) of the ZnCr2O4 nanoparticles (Sample 2) is shown in Fig. 7. Contact angle is a quantitative measure of the wettability of a surface. Hydrophilicity basically refers to hydrogen bonding with water molecules [24]. The ZnCr2O4 showed a hydrophilic surface at a contact angle of 5.3��.

Fig. 6 EDS pattern (a) and PL spectrum (b) of Sample 2

Fig. 7 Photograph of measured contact angle on rough surface of ZnCr2O4 materials (Sample 2)
3.6 DRS and VSM analyses
The optical properties of ZnCr2O4 nanostructures were studied by UV�CVis DRS spectra. The spectra were recorded in wavelength region between 200 and 800 nm at room temperature. The estimated values of the band gap energy (Eg) of ZnCr2O4 nanoparticles are shown in Fig. 8(a). The optical gap was calculated using Tauc relation by plotting (��h��)2 against h��, where �� and h�� denote the absorption coefficient and photon energy, respectively. The direct band gap energy of ZnCr2O4 is found to be 3.50 eV, which is in a good agreement with the value reported in Ref. [25].

Fig. 8 Diffuse reflectance spectrum (a) and M-H curve of ZnCr2O4 nanostructures (Sample 2) (b)
Figure 8(b) shows the dependence of magnetization with the applied magnetic field (M-H curve). ZnCr2O4 is a cubic spinel at room temperature with magnetic ions (Cr3+, s=3/2) at the B sites and is a typical example of the geometrically frustrated system. Temperature variation of magnetic susceptibility measurement confirms that the ceramic material is paramagnetic at above -173 ��C. Below this temperature, anti-ferromagnetic interaction develops. However, the geometry of the cubic lattice results in a frustrated spin system [16].
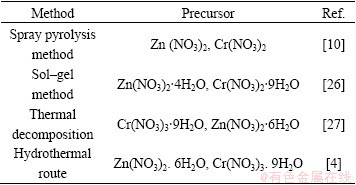
In comparison with other similar works illustrated in Table 2, our method is simpler and more commodious. We used microwave method for the preparation of products. Microwave synthesis of inorganic materials gains importance due to less complex synthesis procedure and shorter reaction time.
Table 2 Characterization comparison of ZnCr2O4 nanostructures with other similar works
Oxides with spinel structure are some of the most studied compounds in solid-state sciences due to interesting physico-chemical properties and are found to be useful in many technological applications such as magnetic materials [28], super hard materials [29], and high-temperature ceramics [30]. In particular, zinc chromite (ZnCr2O4) ceramic spinels are commonly used as humidity sensors, magnetic material and catalytic materials in lots of heterogeneous chemical processes such as CO oxidation, catalytic combustion of hydrocarbons, reduction of several organic molecules and sensing properties [1-5].
4 Conclusions
The present study demonstrates successful synthesis of zinc chromite nanostructures via microwave method. The XRD patterns reveal that the ZnCr2O4 nanostructures prepared by annealing the precursor at 700 ��C for 3 h have good crystallinity with fine cubic structure. The nanostructures exhibit a regular morphology with homogeneous particle size distribution. The FTIR spectrum also confirms nanostructure of prepared products. According to the magnetic measurement, ZnCr2O4 nanostructure exhibits the room temperature paramagnetic behaviour. In addition, it is believed that this simple process can also be applied to synthesizing other functional materials.
References
[1] GABR R M, GIRGIS M M, El-AWAD A M. Formation conductivity and activity of zinc chromite catalyst [J]. Journal of Materials Chemistry and Physics, 1992, 30: 169-177.
[2] NIU Xin-shu, DU Wei-ping, DU Wei-min. Preperation and gas sensing properties of ZnM2O4 (M=Fe, Co, Cr) [J]. Journal of Sensors and Actuators B: Chemical, 2004, 99: 405-409.
[3] BOUMAZA S, BOUGUELIA A, BOUARAB R, TRARI M. Physical and photoelectrochemical studies for hydrogen photo-evolution over the spinel ZnCr2O4 [J]. International Journal of Hydrogen Energy, 2009, 34: 4963-4967.
[4] PENG Cheng, GAO Lian. Optical and photocatalytic properties of spinel ZnCr2O4 nanoparticles synthesized by a hydrothermal route [J]. Journal of the American Ceramic Society, 2008, 91: 2388-2390.
[5] POKHREL S. Humidity-sensing properties of ZnCr2O4�CZnO composites [J]. Journal of Materials Letters, 2003, 57: 3543-3548.
[6] OPPENNLANDER T. Photochemical purification of water and air [M]. Weinheim: Wiley-VCH, 2003.
[7] MARINKOVIC STANOJEVIC Z V, MANCIC L, MARCIC L, MILOSEVICL O. Microstructural characterization of mechanically activated ZnO-Cr2O3 system [J]. Journal of the European Ceramic Society, 2005, 25: 2081-2084.
[8] MARINKOVIC STANOJEVIC Z V, ROMCEVIC N, STOGANOVIC B. Spectroscopic study of spinel ZnCr2O4 obtained from mechanically activated ZnO�CCr2O3 mixtures [J]. Journal of the European Ceramic Society, 2007, 27: 903-907.
[9] ADAK A K, PATHAK A, PRAMANIK P. Polyvinyl alcohol evaporation method for preparation of submicron chromite powders [J]. Journal of British Ceramic Transactions, 1999, 98: 200-203.
[10] MARINKOVIC STANOJEVIC Z V, MANCIC L, MARCIC L, MILOSEVICL O. Preparation of nanostructured Zn-Cr-O spinel powders by ultrasonic spray pyrolysis [J]. Journal of the European Ceramic Society, 2001, 21: 2051-2055.
[11] MINGOS M. Microwave syntheses of inorganic materials [J]. Journal of Advanced Materials, 1993, 5: 857-859.
[12] VAIDHYANATHAN B, GANGULI M, RAO K J. Fast solid-state synthesis of metal vanadates and chalcogenides using microwave irradiation [J]. Journal of Materials Research Bulletin, 1995, 30: 1173-1177.
[13] VAIDHYANATHAN B, RAO K J. Synthesis of Ti, Ga, and V nitrides: Microwave assisted carbothermal reduction and nitridation [J]. Chemistry of Materials, 1997, 9: 1196-1200.
[14] VAIDHYANATHAN B, RAIZADA P, RAO K J. Microwave assisted fast solid-state synthesis of niobates and titanates [J]. Journal of Materials Scicence Letters, 1997, 16: 2022-2025.
[15] VAIDHYANATHAN B, RAO K J. Microwave assisted synthesis of technologically important transition metal silicides [J]. Journal of Materials Research, 1997, 12: 3225-3229.
[16] PURNENDU P, VARADHARAJAN M. Microwave metathesis approach for the synthesis and characterization of ZnCr2O4 [J]. Journal of European Ceramic Society, 2008, 28: 1665-1670.
[17] AKULA V, SARKARI R, CHATLA A, VANKUDOTH K, KUMAR MANDARI K. Synthesize 2-methylpyrazine using aqueous glycerol and ethylenediamine over zinc oxide�Czinc chromite catalysts: Structure�Cactivity relationship [J]. Applied Catalysis A: General, 2012, 441: 108-118.
[18] GINGASU D, MINDRU L, CULITA D C, PATRON L, CALDERON-MORENO J M, PREDA S, OPREA O, OSICEANU P, PINEDA E M. Investigation of nanocrystalline zinc chromit obtained by two soft chemical routes [J]. Materials Research Bulletin, 2014, 49: 151-159.
[19] KAJBAFVALA A, QHORBANI H, PARAVAR A, SAMBERG J P, KAJBAFVALA E, SADNEZHAAD S K. Effects of morphology on photocatalytic performance of zinc oxide nanostructures synthesized by rapid microwave irradiation methods [J]. Superlattices and Microstructures, 2012, 51: 512-522.
[20] BARUAH S, DUTTA J. Hydrothermal growth of ZnO nanostructures [J]. Science and Technology of Advanced Materials, 2009, 10: 013001.
[21] ZHUO R F, FENG H T, CHEN J T, YAN D, FENG J J, LI H J, GENG B S, CHENG S, XU Z Y, YAN P X. Multistep synthesis, growth mechanism, optical, and microwave absorption properties of ZnO dendritic nanostructures [J]. The Journal of Physical Chemistry, 2008, 112: 11676-11775.
[22] KAJBAFVALA A, SHAYEGH M R, MAZLOUMI M, ZANGANEH S, LAK A, MOHAJERANI M S. Nanostructure sword-like ZnO wires: Rapid synthesis and characterization through a microwave- assisted route [J]. Journal of Alloys and Compounds, 2009, 469: 293-297.
[23] KRIISHNAKUMAR T, JAYAPRAKASH R, PINNA N, SINGH V N, MEHTA B R, PHANI A R. Microwave-assisted synthesis and characterization of flower shaped zinc oxide nanostructures [J] Materials Letters, 2009, 63: 242-245.
[24] ZOU L D, VIDALIS I, STEELE D, MICHELMORE A, LOW S P, VEBERK J. Surface hydrophilic modification of RO membranes by plasma polymerization for low organic fouling [J]. Membrane Science, 2011, 369: 420-428.
[25] GINGAUS D, MINDRU I, PATRON L, CULITA D C, CALDERON-MORENO J M, DIAMANDESCULI L, FEDER M, OPREA O. Precursor method��A nonconventional route for the synthesis of ZnCr2O4 spinel [J]. Journal of Physics Chemistry Solids, 2013, 74: 1295-1302.
[26] YAZDANBAKHSH M, KHOSRAVI I, GOHARSHADI E K, YOUSSEFI A. Fabrication of nanospinel ZnCr2O4 using sol�Cgel method and its application on removal of azo dye from aqueous solution [J]. Journal of Hazardous Materials, 2010, 184: 684-689.
[27] STEFANESCU M, BARBU M, VLASE T, BARVINSCHI P, BARBU-TUDORAN L, STOIA M. Novel low temperature synthesis method for nanocrystalline zinc and magnesium chromites [J]. Thermochimica Acta, 2011, 526: 130-136.
[28] MARTINHO H D S, MORENO N O, SANJURJO J A, RETTORI C, GARCIA-ADEVA A J, LAWERNCE HUBRE D. Magnetic properties of the frustrated anti-ferromagnetic spinel ZnCr2O4 and the spin-glass Zn1-xCdxCr2O4 (x=0.05, 0.10) [J]. Phys Rev B, 2001, 64: 1-6.
[29] ZERR A, MIEHE G, SERGHIOU G, SCHWARZ M, KROKE E, RIEDEL R. Synthesis of cubic silicon nitride [J]. Nature, 1999, 400: 340-342.
[30] KIM B N, HIRAGA K, MORITA K, SAKKA Y A. High-strain-rate superplastic ceramic [J]. Nature, 2001, 413: 288-291.
Zahra MOUSAVI, Mahdiyeh ESMAEILI-ZARE, Masoud SALAVATI-NIASARI
Institute of Nano Science and Nano Technology, University of Kashan, Kashan 87317-51167, Iran
ժ Ҫ�����������Ʊ����ṹ����п(ZnCr2O4)�������������700 ��C�˻��ZnCr2O4Ϊ�������õļ⾧ʯ�ṹ���о���Ӧʱ��������书�ʶ�ZnCr2O4�����ߴ����ò��Ӱ�졣�ֱ����X��������(XRD)��ɨ��羵(SEM)��������ɢX����(EDX)����羵(TEM)�����������(DRS)�����·���(PL)���ס�����Ҷ�������(FTIR)�ȼ���������Ʒ��ǿ�ƶ����ϳɵ�ZnCr2O4��Ʒ���б����������������(DRS)����õ�ZnCr2O4���ṹ�Ĺ����϶Ϊ3.50 eV�����·��������Ҳ֤ʵ�˸ý����
�ؼ��ʣ�����п�����ṹ������ˮ�ԣ���ѧ���ܣ���ѧ����
(Edited by Wei-ping CHEN)
Corresponding author: Masoud SALAVATI-NIASARI; Tel: +98-31-55912383; Fax: +98-31-55913201; E-mail: salavati@kashanu.ac.ir
DOI: 10.1016/S1003-6326(15)64007-9

