Neutral pyrimidine C-H donor as anion receptor
来源期刊:中南大学学报(英文版)2016年第5期
论文作者:王洪波 袁迎雪 吴娜娜 韩逸飞 宋相志
文章页码:1023 - 1031
Key words:pyrimidine; pyrrole; C―H bond; anion receptor
Abstract: Anion receptors including pyrimidine subunit were designed and synthesized and their binding abilities with various anions were investigated by fluorescence and 1H NMR titration experiments. DFT calculations provided some information for anion recognition. It is confirmed that both of two new pyrimidine anion receptors have the selectivity for Cl-.
J. Cent. South Univ. (2016) 23: 1023-1031
DOI: 10.1007/s11771-016-0351-1

YUAN Ying-xue(袁迎雪)1, WU Na-na(吴娜娜)1, HAN Yi-fei(韩逸飞)1,
SONG Xiang-zhi(宋相志)1, WANG Hong-bo(王洪波)1, 2
1. College of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China;
2. Key Laboratory of Optoelectronic Chemical Materials and Devices of Ministry of Education
(Jianghan University), Wuhan 430056, China
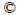 Central South University Press and Springer-Verlag Berlin Heidelberg 2016
Central South University Press and Springer-Verlag Berlin Heidelberg 2016
Abstract: Anion receptors including pyrimidine subunit were designed and synthesized and their binding abilities with various anions were investigated by fluorescence and 1H NMR titration experiments. DFT calculations provided some information for anion recognition. It is confirmed that both of two new pyrimidine anion receptors have the selectivity for Cl-.
Key words: pyrimidine; pyrrole; C―H bond; anion receptor
1 Introduction
Anion recognition has been commonly referred to as “anion coordination chemistry”, which was first proposed by LEHN [1]. The development of anion coordination chemistry, however, is relatively slow for a number of reasons [2]. Compared with cations, anions are larger than the equivalent isoelectronic cations and therefore have a lower charge to radius ratio. They also have a wide range of geometries, such as spherical, linear, trigonal planar, tetrahedral, and octahedral. In addition, anions’ pH sensitivity plays a role in the formation of these species. These constraints result in a challenge for the design and synthesis of effective recognition motifs.
As structural knowledge of supramolecular complexes involving anions has been accumulated, a number of factors (such as hydrogen bonding, topology and charge) which influence the strength, selectivity, and structures have been observed in anion recognition [3]. Binding between anions and their hosts is mainly attributed to hydrogen bonding and/or electrostatic interactions, because hydrogen bonding is more influential in promoting selective binding through topological complementarity. In these cases, the lone pair of electrons is donated from the anion to a hydrogen atom on the host, resulting in hydrogen-bond formation (Scheme 1). Many reported receptors are based on quaternary ammonium, guanidinium, amidinium, and thiouronium motifs. Others such as indole or pyrrole systems and linear analogues have also been extensively studied. Most of these known synthetic receptors include N-H groups or N+-H motifs as hydrogen bond donors [4].
As demonstrated, the N-H motifs are strong hydrogen bond donors, and this property has been widely applied in the development of anion receptors. However, the importance of C-H as hydrogen bonding donor in molecular assembly processes [5], protein-protein interactions [6], extended crystal lattices [7] and anion recognition [8], recently has come to be appreciated. CAI and SESSLER [9] has summarized the use of neutral and cationic C-H hydrogen-bonded donors including phenyl C-H, triazole C-H, imidazolium (C-H)+ and triazolium (C-H)+ for anion recognition (Scheme 2) [9]. Plorized C-H bonds conjugated with an alternating arrangement of several components can form a binding pocket to selectively encapsulate chloride anions [10]. Herein, a novel type of anion receptors based on pyrimidine was synthesized and characterized. Their binding properties with different anions were investigated via 1H NMR titrations and fluorescence titrations.
2 Experimental
2.1 General information
All the receptors and anions (as their tetrabutylammonium salts) were dried under vacuum. Dichloromethane was redistilled and stored in the presence of anhydrous sodium sulfate. CDCl3 and DMSO-d6 were purchased from Cambridge Isotope Inc. Other reagents were obtained from commercial sources without further purification. Fluorescence titration experiments were carried out on HITACHI F7000 fluorescence emission spectrophotometer. 1H NMR, 13CNMR spectra and titrition experiments were carried out on Ascend TM 400 MHz or 500 MHz spectrometers.

Scheme 1 N―H hydrogen-bond binding mode of anion and some hydrogen bond donor motifs

Scheme 2 C―H hydrogen-bond binding mode of anion and some hydrogen bond donor motifs
2.2 Synthesis of 1H-pyrrole-2-carbaldehyde [11]
A 100 mL round bottom flask was charged with anhydrous DMF (4.0 g, 53 mmol). Then, the flask was immersed in ice-bath, followed by dropwise addition of phosphorus oxychloride (7.7 g, 50 mmol) over 10 min. Pyrrole (3.3 g, 50 mmol) in acetonitrile (50 mL) was added into DMF solution and the mixture was stirred at room temperature for 12 h. After completion of reaction, saturated sodium bicarbonate solution was added into the reaction mixture until pH≈7. The aqueous mixture was extracted with ethyl acetate (3×100 mL), washed with brine (3×50 mL) and dried over Na2SO4. The organic solvent was evaporated under reduced pressure to yield 1H-pyrrole-2-carbaldehyde (4.024 g, 84.2%) as colorless liquid which was used without further purification. 1H NMR (500 MHz, CDCl3) △ (10-6): 10.18 (s, 1H), 9.50 (br s, 1H ), 7.16-7.14 (m, 1H), 7.00-6.98 (m, 1H), 6.36-6.33 (m, 1H).
2.3 Synthesis of 1-(1H-pyrrole-2-yl) ethanone [11]
A 100 mL round bottom flask was charged with anhydrous dimethyl acetamide (4.62 g, 53 mmol). The flask was then immersed in ice-bath, followed by dropwise addition of phosphorus oxychloride (7.7 g, 50 mmol) over 10 min. Pyrrole (3.3 g, 50 mmol) in acetonitrile (50 mL) was added into the reaction solution and then the mixture was stirred at room temperature for 12 h. After completion of the reaction, saturated aqueous sodium bicarbonate solution was added into the reaction mixture until pH≈7. The aqueous mixture was extracted with ethyl acetate (3×100 mL), washed with brine (3×50 mL) and dried over Na2SO4. The organic solvent was removed under reduced pressure to yield 1-(1H-pyrrole- 2-yl) ethanone (4.132 g, 75.8%) as colorless solid which was used without further purification in the next step. 1H NMR (500 MHz, CDCl3) △ (10-6): 9.96 (br s, 1H),7.43 (t, 1H), 6.77 (t, 1H), 6.63-6.62 (m, 1H), 2.44 (s, 3H).
2.4 Synthesis of 1,3-di(1H-pyrrole-2-yl)prop-2-ene- 3-one 3
A mixture of 1H-pyrrole-2-carbaldehyde (4.655 g, 49 mmol), 1-(1H-pyrrole-2-yl) ethanone (5.341 g, 49 mmol) and NaOH (2.91 g, 7.35 mmol) was dissolved in methanol (30 mL) and then refluxed for 12 h. After the reaction solution was cooled down to room temperature, the precipitate was filtered and washed with water. The crude product was further purified via being recrystallized in ether to yield pure yellow crystal (2.6g, 28.5%). 1H NMR (500 MHz, CDCl3) △ (10-6): 11.93 (s, 1H), 11.69 (s, 1H), 7.48 (d, 1H), 7.35 (d, 1H), 7.07 (s, 3H), 6.62 (s, 1H), 6.25 (s, 1H), 6.19 (s, 1H).
2.5 Synthesis of 4-bromobenzamidine hydrochloride
4-bromobenzonitrile (7.28 g, 40 mmol) was dissolved in anhydrous methanol (30 mL), followed by the slow addition of 1 mL sodium methoxide in methanol (0.96 mol/L) under nitrogen atmosphere. After the reaction mixture was stirred at room temperature for 36 h, ammonium chloride (2.12 g, 40 mmol) was added in portion and refluxed for another 36 h. When the mixture was cooled to room temperature, the product was obtained by filtration and washed by anhydrous methanol to afford pure product as white solid (5.697 g, 60.3%).
2.6 Synthesis of 1
4-bromobenzamidine hydrochloride (590 mg,2.5 mmol), 1,3-di(1H-pyrrole-2-yl)prop-2-ene-3-one (930 mg, 5 mmol) and KOH (280 mg, 5 mmol) were dissolved in anhydrous ethanol (20 mL). The mixture was monitored via TLC and refluxed for about 24 h. Then, the reaction mixture was cooled to room temperature and then water was added into the mixture. The aqueous layer was extracted by dichloromethane (100 mL) and dried over Na2SO4. The organic layer was removed under reduced pressure to obtain the residue as crude product. The residue was purified by column chromatography (silica gel, petroleum ether/ dichloromethane=20:1) to give pure product as white solid (126 mg, 40.9%). M.p.248-249 °C. 1H NMR (500 MHz, CDCl3) △ (10-6):9.78 (s, 2H), 8.43-8.41 (d, 2H), 7.67-7.65 (d, 2H),7.51 (s, 1H), 7.06 (d, 2H), 6.99 (d, 2H), 6.42 (s, 2H). 13C NMR (100 MHz, CDCl3) △ (10-6): 162.1, 157.2, 137.5, 131.6, 130.9, 129.9, 124.7, 122.9, 111.6, 110.5, 105.4.
2.7 Synthesis of 2-phenyl-1H-pyrimidine-4,6-dione [12]
Benzamidine hydrochloride hydrate (3.92 g,25 mmol) and diethyl malonate (4.28 g, 27 mmol) were dissolved in anhydrous ethanol (40 mL). A solution of CH3ONa (5.16 g, 91 mmol) in CH3OH (24 mL) was added dropwise into the ethanol solution and then it was refluxed for 3 h. When the reaction was completed, the pH of ethanol solution was adjusted to 3-4 via the concentrated hydrochloric acid successively. The precipitate in ethanol solution was filtered and then washed with water to afford pure pale yellow solid (2.45 g, 52.1%), which was used without further purification.
2.8 Synthesis of 2-phenyl-4,6-dichloro pyrimidine [13]
In a 100 mL round bottom flask, 2-phenyl-1H- pyrimidine-4,6-dione(1.75 g, 9.30 mmol) and POCl3 (3.57 g, 23.23 mmol) was added and then heated at 110°C. The reaction was monitored via TLC. After about 3 h, the reaction mixture was poured into ice water slowly to afford precipitate as crude product. The crude product was further purified via recrystallization in ethanol to obtain pale yellow solid yield (1.79 g, 86.1%). 1HNMR (400 MHz, CDCl3) △ (10-6): 8.40 (m, 2H), 7.54 (m, 3H), 7.30 (s, 1H).
2.9 Synthesis of 1-dodecylimidazole [14]
A mixture of imidazole (0.68 g, 10 mmol), 1-bromododecane (2.50 g, 10 mmol) and anhydrous K2CO3 (2.76 g, 20 mmol) was dissolved in acetone (20 mL) and refluxed for 7 h. The solvent was removed under reduced pressure and the residue was subjected to silica gel chromatography with ethyl acetate as eluent to give pure product (2.12 g, 90%).1H NMR (400 MHz, CDCl3)△ (10-6):0.81 (t,J=7, 3H), 1.22 (br s, 18H), 1.70 (m, 2H), 3.85 (t,J=7, 2H), 6.86 (s, 1H), 6.99 (s, 1H), 7.42 (s, 1H).
2.10 Synthesis of 2-phenyl-4,6-bis(3- dodecylimidazolium)pyrimidine dichloride
A 100 mL three necked round bottom flask was charged with 40 mL toluene, followed by adding 2-phenyl-4,6-dichloro pyrimidine (1.13 g, 5 mmol) and 1-dodecylimidazole (2.60 g, 11 mmol) under nitrogen atmosphere. After being refluxed for 24 h, the reaction solution was cooled to obtain crude product as precipitate, which was further purified via recrystallization in CH2Cl2 to afford pale waxy solid (1.98 g, 56.9%). M.p. 212-213 °C. 1H NMR (400 MHz, CDCl3): △ (10-6) 11.65 (s, 2H), 9.66 (s, 1H), 9.37 (s, 1H), 8.55 (s, 2H), 7.64 (s, 2H),7.45(s, 2H) 4.48 (t, J=7.2 Hz, 4H), 2.11 -2.04 (m, 4H), 1.46 -1.20 (m, 36H), 0.87 (t, J=7.2 Hz, 6H).
2.11 Synthesis of 2
2-phenyl-4,6-bis(3-dodecylimidazolium) pyrimidine dichloride (0.70 g, 1 mmol) in CH3OH (20 mL) was added dropwise to LiBF4 (0.21 g, 2.21 mmol) aqueous solution (10 mL). The reaction mixture was stirred for about 30 min. The precipitate in solution was filtered and washed with ethyl acetate to obtain product (0.74 g, 92.5%). M.p.223-225 °C. 1H NMR (400 MHz, DMSO-d6): △ (10-6) 10.52 (s, 2H), 8.80 (d, 2H), 8.71 (d, 2H), 8.63 (s, 1H), 8.22 (d, 2H), 7.70 (m, 3H), 4.40 (t, 4H), 1.95 (s, 4H), 1.30 (d, 36H), 0.84 (d, 6H). 13CNMR (100 MHz, DMSO-d6): △ (10-6) 164.60, 156.58, 136.81, 134.47, 133.77, 129.47, 124.99, 119.75, 97.78, 50.53, 31.77, 29.68, 29.52, 29.49, 29.44, 29.33, 29.19, 28.92, 26.03, 22.56, 14.40.
2.12 Fluorescence titration experiments
The binding stoichiometry is obtained from Job plot. Compound YW-1 was excited at 316 nm. Excited voltage was set as 600 V and slit was 5 nm/5 nm. Compound YW-2 was excited at 280 nm. Excited voltage was set as 700 V and slit was 5 nm/5 nm.
Take the molar ratio of anion for X-axis and concentration of the stable complex for Y-axis. Job plot was drawn by ORIGIN 8.5. Concentration of complex was obtained from
[HG]=(I0-I)/I0×[H] (1)
where [HG] is the concentration of complex, [H] is the concentration of receptor, I0 stands for the fluorescence intensity of the receptor only, and I stands for the fluorescence intensity of the series.
During fluorescence titration, we kept the concentration of receptor at the fixed concentration (5×10-6 mol/L). Fluorescence intensity decreased with the equivalent addition of anion solution. Taking the concentration of anion as X-axis and fluorescence intensity at maximum emission wavelength as Y-axis resulted in Scatter diagram.
2.13 DFT calculations
All geometry structures and frequency calculations in this work were carried out under DFT/B3LYP/ 6-311G(d) calculation level. Furthermore, BSSE calculations of corresponding structures were performed at DFT/B3LYP/6-311++G(d, p) level.
3 Results and discussion
3.1 Design, synthesis and characterization of anion receptors
A number of cyclic and linear anion receptors containing two or more pyrrole N-H groups, have shown strong binding ability and selectivity. As shown in Scheme 3, SESSLER et al [15] reported 1,3-bis (pyrrol-2-yl)benzene I as an alternative anion receptor and revealed both of two pyrrole N-H groups pointing toward the center of the resulting cleft [16]. And a series of dipyrrolyldiketone difluoroboron complexes II and dipyrrolylpyrazoles III were synthesized, started from 1,3-dipyrrolyl-1,3-propanedione [17-18]. For compound IV calix[4]pyrrole, four pyrroles concurrently donate their hydrogen bonds to the anionic gust molecule and this binding conformation provides the system typical bowl shape similar with other calixarene systems [19-20].
On the other hand, pyrimidine can interact with anionic species through hydrogen bonds, and thus the combination of pyrrole and pyrimidine groups within one molecule may realize functionality to bind various anionic guests. Reports on neutral C―H bond as the binding site for anions were few, not to mention the combination of neural C―H bond with other interactions. As shown in Fig. 1, four types of anion acceptors were computed to figure out the best choice for anion binding. Frequency calculation toward four structures revealed that complex A and D are unstable even if their optimized structures satisfy tight convergence standards apparently. The reported binding constants of complex A and D are lower than 104 L/mol, which are not sufficiently strong. Those experimental results are consistent with our computational results. Although the optimized structures of complex B and C are obtained without imaginary frequency, we can make choice from the relative value of △G. The binding free energies of B and C are -76.59 kJ/mol and -89.82 kJ/mol, respectively; what’s more, the chloride can only effectively bind with two sites of dipyrrolypyrazole scaffold. DFT calculations further demonstrate the superiority of pyrrole and pyrimidine-based anion receptor in the field of anion supramolecular chemistry.

Scheme 3 Some known anion receptors based on C―H bond

Fig. 1 Optimized structures of complex A, B, C and D
Scheme 4 Synthesis of new anion receptors:
For the synthesis of new anion receptors (Scheme 4), we first tried the approach of 1,3-di-2-pyrrolyl-1,3- propanedione and 4-bromobenzamidine hydrochloride. However, we barely got YW-1. The reason for this situation probably was the relative low activity of 1,3-di-2-pyrrolyl-1, 3-propanedione. Furthermore, during the synthesis of pyrimidine ring, KOH is better than K2CO3 or KHCO3 because of its strong alkalinity. In order to improve anion receptor in less polar solvents, we used two long alkyl chains in YW-2.
3.2 Anion binding properties
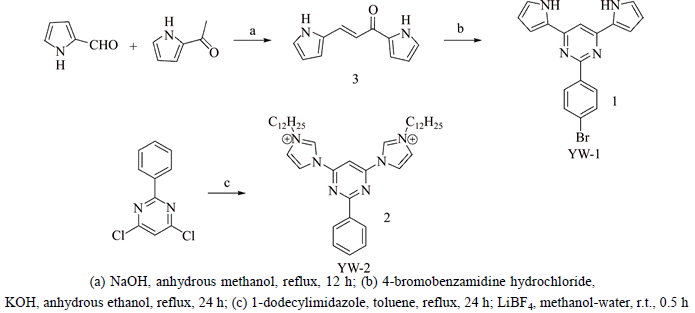
1) Fluorescence titration experiments. The binding property between receptors and anions (F-, Cl-, Br-, I-, HSO4-, AcO-, H2PO4- as their tetrabutylammonium salts) was analyzed by means of fluorescence titration experiments. From the Job plot and fluorescence titration experiments, we can conclude that the binding stoichiometry of host-guest is 1:1 and both the receptors exhibit a better alternative to Cl- than other anions (Br-, I-, HSO4-, AcO-, H2PO4-).
For YW-1, the addition of various anions exhibits no emission wavelength change at 391.8 nm. However, with continuous titration of anion, fluorescence intensity is decreased gradually. Compared to the more basic anion H2PO4- (Ka=3.2×104 L/mol), YW-1 prevents a stronger hydrogen-bond interaction for Cl- (Table 1), which is rarely reported in host-guest chemistry. This indicates that compatibility of space between host’s cavity and guest’s size and structure has a tremendous impact on the binding interaction (Fig. 1).
Table 1 Calculated geometry information and △Gbinding during formation of complex A, B, C and D

Fluorescence titration experiment for YW-2 and anions system was carried out in the similar way. After equivalent addition of anions, the compound shows decreased emission intensity and at the same time, the peaks induce a significant hypochromic shift from 361.6 nm to around 350 nm (Fig. 2). Meanwhile, the binding ability of YW-2 with different anions is stronger than that of YW-1, which is shown in Table 2. This probably results from the assistance of static electricity interaction apart from C-H hydrogen bond.
From Job plots, we conclude that the ratio of host-to-guest is 1:1, so the curve fitting is figured out by ORIGIN 8.5 based on the Eq. (2) and association constant is obtained by non-linear least-squares:

Fig. 2 Fluorescence titration of YW-1 (5×10-6 mol/L in CH2Cl2) with response to anions:(Insert: variation of fluorescence intensity at λ= 391.8 nm and fitted curve upon addition of equivalent anion and Job plot)
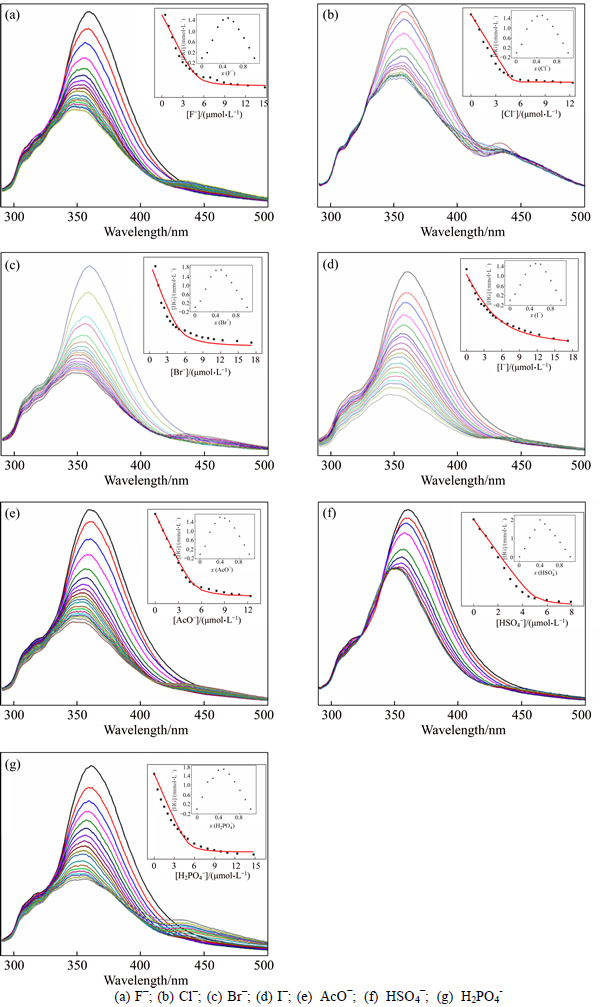
Fig. 3 Fluorescence titration of YW-2 (5×10-6 mol/L in CH2Cl2) with response to 0-3 equiv anion:(Insert: variation of fluorescence intensity at λ=361.6 nm and fitted curve upon addition of equiv anion and Job plot)
Table 2 Association constants Ka (L・mol-1) determined by fluorescence titrations with tetra-n-butylammonium salts of anions in chloroform

I=I0+(Ilim-I0)/2CH{CH+CG+1/K-[(CH+CG+1/K)2-4CHCG]1/2} (2)
where I0 and I stand for the fluorescence intensity of system before and after addition of anion, respectively; CH is the concentration of receptor (5×10-6 mol/L for both); CG is the concentration of anion and K stands for the association constant of the receptor and anion.
2) 1H NMR titration experiments. In order to obtain a deeper understanding of binding mechanism, we performed the 1H NMR titration experiments of YW-1 in CDCl3 (0.01 mmol, 298 K). Slowly addition of Cl- results in the concurrent downfield movements of pyrrole N―H protons (from 9.78 to 12.0) and pyrimidine C―H proton (from 7.51 to 8.02) (Fig. 4). It is predicted that the binding sites of the Pyrimidine A is the N―H and C―H hydrogen-bond.
3.3 Anion recognition analysis
Compared with known compounds I-IV, a new type of neutral C―H bond recognizer consisting of pyrimidine has more stable conformation and stronger binding ability, which is supported by the DFT and titration experiments. YW-1 has the selectivity for Cl- and its association constant is 4300 L/mol, which is less than YW-2 for about 30 times. This is probably because of the notable polarization of C―H bond in YW-1. For compound Ⅱ including BF2, the binding efficiency for anions is too low, due to the requirement for reversion of pyrrole groups to interact with anions. While pyrazole- N-blocked dipyrrolylpyrazole derivative compound Ⅲ is reported with 1600 L/mol association constant for AcO-. In addition, our anion receptors YW-1 and YW-2 have better solubility in organic solvent and superior pyrimidine skeleton.
The affinity sites for YW-1 are pyrrole N-H bond and pyrimidine C―H bond, while the joint property of hydrogen-bond and electrostatic interactions for YW-2 is predicted to be dominant. The stronger binding ability of neutral C-H donor in contrasts to the reported ones is due to the stable and highly polarized pyrimidine C―H bond and combination with other secondary interactions.
Fig. 4 1H NMR titration of YW-1 (0.01 mmol) with increasing equivalents of tetrabutylammonium chloride in CDCl3
4 Conclusions
We have designed and synthesized two kinds of anion receptors consisting of neutral pyrimidine backbone and two pyrrole subunits, which exhibit the N―H…X- and C―H…X- binding sites. The other one includes imidazole framework recognize anions through (N―H)+…X- and C―H…X- pattern. Both two anion receptors produce higher affinity for Cl- over other anions. Furthermore, the association ability of YW-2 binding to anions is notably stronger than that of YW-1, which is demonstrated by fluorescence and 1H NMR titration experiments. The detailed research on the pyrimidine systems is in progress.
References
[1] LEHN J M. Cryptates: The chemistry of macropolycyclic inclusion complexes [J]. Accounts of Chemical Research, 1978, 11: 49-57.
[2] SESSLER J L, CAMIOLO S, GALE P A. Pyrrolic and polypyrrolic anion on binding agents [J]. Coordination Chemistry Reviews, 2003, 240(1): 17-55.
[3] BOWMAN-JAMES K. Alfred Werner revisited: The coordination chemistry of anions [J]. Accounts of Chemical Research, 2005, 38(8): 671-678.
[4] AMENDOLA V,  D, FABBRIZZI L, LICCHELLI M. What anions do to N-H-containing receptors [J]. Accounts of Chemical Research, 2006, 39(5): 343-353.
D, FABBRIZZI L, LICCHELLI M. What anions do to N-H-containing receptors [J]. Accounts of Chemical Research, 2006, 39(5): 343-353.
[5] DESIRAJU G R. C―H…O and other weak hydrogen bonds from crystal engineering to virtual screening [J]. Chemical Communications, 2005, 24: 2995-3001.
[6] JIANG Lin, LAI Lu-hua. CH…O hydrogen bonds at protein-protein interfaces [J]. The Journal of Biological Chemistry, 2002, 277: 37732-37740.
[7] BIRADHA K. Crystal engineering: From weak hydrogen bonds to co-ordination bonds [J]. Cryst Eng Comm, 2003, 66(5): 374-384.
[8] CAI Jia-jia, SESSLER J L. Neutral CH and cationic CH donor groups as anion receptors [J]. Chemical Society Reviews, 2014, 43: 6198-6213.
[9] HUA Yu-ran, FLOOD A H. Click chemistry generates privileged CH hydrogen-bonding triazoles: The latest addition to anion supramolecular chemistry [J]. Chemical Society Reviews, 2010, 39: 1262-1271.
[10] ROSSOM W V, HILL J P, TERENTYECA T G, SODEYAMA K, MATSUSHITA Y, TATEYAMA Y, ARIGA K. Arylpyrrole oligomers as tunable anion receptors [J]. Organic & Biomolecular Chemistry, 2014, 12: 5492-5499.
[11] MENGES N, SARI O, ABDULLAYEV Y, ERDEM S S, BALCI M. Design and synthesis of pyrrolotriazepine derivatives: An experimental and computational study [J]. The Journal of Organic Chemistry, 2013, 78: 5184-5195.
[12] ZHANG Xiang, GAO Yong-xin, LIU Hui-jun, GUO Bao-yuan, WANG Hui-li. Design, synthesis and antifungal activities of novel strobilurin derivatives containing pyrimidine moieties [J]. Bulletin of the Korean Chemistry Society, 2012, 8(33): 2627-2634.
[13] TANG Cai-fei, LI Zhi-ming, WANG Quan-rui. IBD-mediated oxidative cyclization of pyrimidinylhydrazones and concurrent Dimroth rearrangement: Synthesis of [1, 2, 4] triazolo [1, 5-c] pyrimidine derivatives [J]. Beilstein Journal of Organic Chemistry, 2013, 9: 2629-2634.
[14] ZHANG Yong-hui, ZHU Wei, LIU Yi-liang, WANG Hong, WANG Ke, LI Kai, NO J H, AYONG L, GULATI A, PANG Ran, FREITAS- JUNIOR L, MORITA C T, OLDFIELD E. Chem-Immunotherapeutic Antimalarials targeting isoprenoid biosynthesis [J]. ACS Medicinal Chemistry, 2013, 4(4): 423-427.
[15] SESSLER J L, AN D, CHO W S, LYNCH V, MARQUEZ M. Calix[n]bispyrrolybenzenes: Synthesis, characterization, and preliminary anion binding studies [J]. Chemistry-A European Journal, 2005, 11(7): 2001-2011.
[16] HIROMITSU M, YOSHIHIRO I, YUKIO K, TAKASHI N. Dipyrrolypyrazoles: Anion receptors in protonated form and efficient building blocks for organized structures [J]. Chemical Communications, 2007, 11: 1136-1138.
[17] HIROMITSU M. Supramolecular chemistry of acyclic oligopyrroles [J]. European Journal of Organic Chemistry, 2007, 2007, 32: 5313-5325.
[18] MAEDA H, KUSUNOSE Y. Dipyrrolydiketone difluoroboron complexes: Novel anion sensors with C―H…X- interactions [J]. Chemistry, 2005, 19: 5661-5666.
[19] SESSLER J L, ANZENBACHER P, JURSIKOVA K, MIYAJI H, GENGE J W, TVERMOES N A, ALLEN W E, SHRIVER J A, GALE P A, KRAL V. Functionalized calix[4]pyrroles [J]. Pure and Applied Chemistry, 1998, 70(12): 2401-2408.
[20] SAHA I, LEE J T. Recent advancements in calix[4]pyrrole-based anion-receptor chemistry [J]. European Journal of Organic Chemistry, 2015, 2015(18): 3859-3885.
(Edited by YANG Bing)
Foundation item: Project(21302232) supported by the National Natural Science Foundation of China
Received date: 2015-03-18; Accepted date: 2015-09-27
Corresponding author: WANG Hong-bo, Professor; Fax: +86-731-88879616; E-mail: hongbo.wang@jhun.edu.cn

