J. Cent. South Univ. (2015) 22: 1066-1070
DOI: 10.1007/s11771-016-0356-9
Increased photo-catalytic removal of sulfur using titania/MWCNT composite
Molood Barmala, Abdolsamad Zarringhalam Moghadam, Mohammad Reza Omidkhah
Chemical Engineering Department, Tarbiat Modares University, Jalal Ale Ahmad Highway,
P. O. Box: 14155-143, Tehran, Iran
 Central South University Press and Springer-Verlag Berlin Heidelberg 2015
Central South University Press and Springer-Verlag Berlin Heidelberg 2015
Abstract: Titania coating of multi wall carbon nano tube (MWCNT) was carried out by sol-gel method in order to improve its photo catalytic properties. The effect of MWCNT/TiO2 mass to volume ratio on adsorption ability, reaction rate and photo-catalytic removal efficiency of dibenzothiophene (DBT) from n-hexane solution was investigated using a 9 W UV lamp. The results show that the addition of nanotubes improves the photo-catalytic properties of TiO2 by two factors; however, the DBT removal rate versus MWCNT content is found to follow a bimodal pattern. Two factors are observed to affect the removal rate of DBT and produce two optimum values for MWCNT content. First, large quantities of MWCNTs prevent light absorption by the solution and decrease removal efficiency. By contrast, a low dosage of MWCNT causes recombination of the electron holes, which also decreases the DBT removal rate. The optimum MWCNT contents in the composite are found to be 0.25 g and 0.75 g MWCNT per 80 mL of sol.
Key words: photo oxidation; desulfurization; sol-gel; titania; multi wall carbon nano tube (MWCNT)
1 Introduction
Sulfur removal from liquid fuels has been increased in importance in recent years. In addition to environmental concerns, it is also a factor in combustion systems. Although hydrodesulfurization is the usual method for removing sulfur, it requires high energy, high temperatures, high pressure and high consumption of hydrogen in the presence of a metal catalyst. Yet the elimination of dibenzothiophene (DBT) compounds using this process is difficult [1-4]. Alternative processes such as alkylation, extraction, sedimentation, oxidation and adsorption have been investigated to decrease energy consumption during sulfur removal [1]. Among these, adsorption and oxidation processes are favoured because they can be used at ambient temperatures and pressure [1, 5-6]. Oxidation converts thiophene into easily separable compounds like sulfone and sulfoxide [1, 3, 5].
DBT is a compound that cannot be removed by photolysis [1, 6]. TAO et al [1] showed that there is sulfide compounds in Kerosene whose photo oxidation removal rates are up to a hundred times greater than those of Dibenzo thiophene, and even with the use of low-pressure lamps, they can be completely removed within the first an half hour of photo oxidation. DBT is more highly resistant and there is no noticeable change in concentration after 5 h [1].
When a catalyst is employed (photo-catalytic oxidation), the removal rate of sulfur compounds increases or oxidation occurs in less time. Also oxidants could be added to increase the hydroxyl radical concentration [2].
Among semiconductors, titania (TiO2) shows good potential as a photo-catalyst for the removal of dyes and organic materials to produce hydrogen by water splitting. It is highly stable, resistant to corrosion, relatively non-toxic and chemically inert [7-10]. Despite these good properties, its low photo-catalytic efficiency resulting from the quick recombination of electron holes hinders its commercial use [3, 7, 10-11].
Photo-catalytic reaction rate can be controlled by light absorption, the transmission of electron holes to the photo-catalytic surface, the recombination of electron holes, the electron-hole reaction and mass transfer rate of the reactant on the surface of the photo-catalyst. Efficient photo-catalysts must have high photon conversion efficiency and a large specific surface area [9].
Photo-catalytic efficiency can be improved by decreasing the possibility of recombination of electron holes, preventing agglomeration of particles, increasing adsorption of the catalyst and decreasing the band gap [3, 7]. One way of achieving these is using TiO2 combined with a suitable substrate [7-8, 12].
Carbon nanotubes (CNTs) have chemical stability, high specific surface areas and high electrical conductivity and they are materials considered applicable as substrates for catalysts [3, 7, 11, 13-14]. CNTs show
electrical conductivity similar to that of copper, which makes them suitable electron acceptors when combined with semiconductors [13]. They also show photo- catalytic activity in the range of visible light [12-13].
Introduction of multi wall carbon nano tubes MWCNTs decreases the crystallite size of composite recombination chance [10-11].
SALEH [12] stated that, since CNTs are black, large amount in a CNT/semiconductor composite disperses light and prevents it from reaching the bulk solution. This decreases the efficiency of the process and indicates that there must be an optimal amount of CNTs in a structure [12]. It has also been shown that a large amount of catalyst in a solution decreases the photo-catalytic efficiency for the same reason [6-8, 14].
In the present work, the removal of sulfur by photo- catalytic oxidation was investigated. MWCNT/TiO2 composites with different MWCNT fractions were synthesized and characterized to study the effect of MWCNTs on the photo-catalytic activity of the composite. As one of the most refractory sulfide compounds present in liquid fuels [2, 13], DBT was used as a sample component and its removal rate from n-hexane solution was investigated.
2 Experimental
2.1 Preparation and characterization of photo catalysts
MWCNT/TiO2 composites were synthesized using sol-gel method. Sol was made according to the molar ratio used by JITIANU et al [15-16]. Tetra ethyl orthotitanate (98%, Merck) and isopropanol (99%, Dr. Mojallali Company) were mixed for 30 min. A suitable amount of nitric acid (65%, Merck) diluted with distilled water (solution, pH=3) was added to the solution and stirred to form a hydrolyzed sol. After 1 h, MWCNT (10-20 nm in diameter and 10 ��m in length; RIPI, Iran) of 2, 1, 0.75, 0.5, 0.35, 0.25, 0.15, 0 g per 80 mL of sol (samples 1-8 abbreviated as S1-S8, respectively) was added to the sol and magnetically stirred for 3 h.
This solution was kept at room temperature for at least 48 h to complete the gelation process. After solvent evaporation, the samples were calcinated at 450 ��C. The heating and cooling rates were 5 ��C/min and the soaking time was 1 h.
2.2 Photo-catalytic oxidation testing
To evaluate the photo-catalytic properties of the composites and the oxidation of DBT, a solution of DBT in n-hexane was used at a mass fraction of 2��10-4; 0.5 g catalyst was added to 100 mL solution. The sample was initially stirred for 2 h in the dark to reach adsorption equilibrium and the final concentration was measured. This value was considered to be the initial concentration for photo-catalytic activity. At appropriate time intervals, the solution was sampled to determine the time required to reach adsorption equilibrium. Later, samples were irradiated for 2 h using a low pressure mercury lamp (254 nm, Philips, 9 W) in a mirror box. The DBT removal rates during this period were recorded.
The DBT concentration was measured using a UV spectrophotometer (325 nm in wavelength). Changes in concentration over time during irradiation were recorded to determine the DBT reaction rate constant. Composite structural characterization was carried out using FTIR, SEM and BET.
3 Results and discussion
3.1 Catalyst structure characterization
3.1.1 FTIR
Figure 1(a) shows the FTIR spectrum of raw MWCNT. Peak at 1580 nm-1 signifies the C=C linking, which is characteristic of MWCNT. The band at 1160 nm-1 signifies the C��C bond [5, 8, 12, 14, 17]. The peak in the region of 2800-3000 nm-1 signifies the C��H bond [8, 18]. There is a peak at 2300 nm-1 that signifies CO2 produced during heating and trapped in the CNT structure [19]. The peak in district 600 cm-1 signifies the Ti��O��Ti bond [5, 14, 20-21].
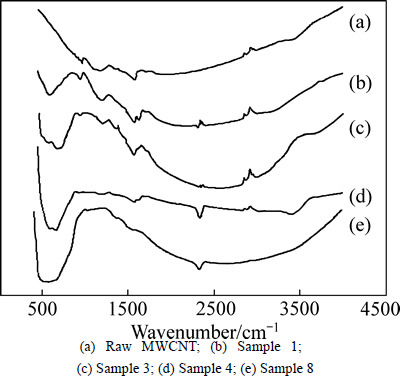
Fig. 1 FTIR spectra:
3.1.2 SEM
The SEM images of several samples are shown in Fig. 2. Figure 2(a) shows the SEM image of raw MWCNT and the rest are images of composites. In all composite images, the TiO2 coverage of the MWCNT is evident. This increases as the ratio of MWCNT to sol decreases and agglomerates of TiO2 are created.
3.1.3 BET
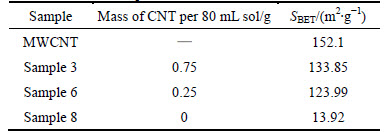
The specific surface areas of some samples are listed in Table 1. The lowest specific surface area is 13.92 m2/g for sample 8 (TiO2 without MWCNT). The
highest specific surface area is 152.1 m2/g for raw MWCNT. The specific surface areas of the composites fall between these values; the greater the MWCNT content is, the higher is the specific surface area is [3, 10, 12-13, 15, 20].

Fig. 2 SEM images of MWCNT (a), sample 1 (b), sample 3 (c), sample 4 (d), sample 6 (e) and sample 6 with higher magnification (f)
Table 1 SBET of sample
3.2 Adsorption ability of composites
The equilibrium content of the solution after 2 h of stirring in the dark for an average of at least 3 replications is presented in Fig. 3. It can be seen that by decreasing the MWCNT content in composite, the ability of composite to adsorb dibenzo thiophene decreases due to the reduction of specific surface area.
Figure 4 shows the change in content versus time for the composites and pure samples. It can be seen that increasing the MWCNT content decreases the time required to reach equilibrium. This again is due to the increased surface area and adsorption ability due to the addition of MWCNT to composite.
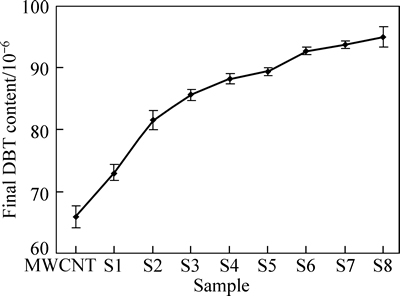
Fig. 3 Equilibrium content after adsorption (in dark)
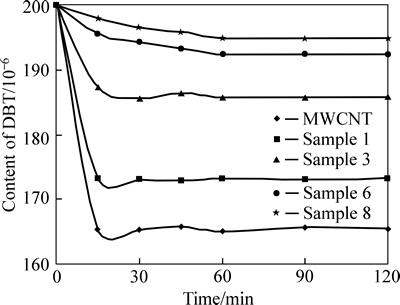
Fig. 4 Content change with time
3.3 Photo-catalytic properties
It is found that no change in DBT content has occurred without addition of a catalyst (photolysis). DBT is resistant to photolysis, which is consistent with the results of previous studies [1-2, 6]. Figure 5 shows the DBT content change versus MWCNT content for the different composites.
It is apparent from Fig. 5 that pure MWCNT has low photo-catalytic properties [12, 22]. In principle, ambient oxygen acts as an oxidizing agent and decreases the DBT content [1].
Samples 1-7 (MWCNT/TiO2 composites) show higher photo-catalytic activity than samples with pure TiO2 or MWCNT. TiO2 without MWCNT (sample 8) shows low removal efficiency resulting from electron hole recombination. Since nanotubes can act as electron acceptors, the addition of MWCNT to the composite increases adsorption and decreases recombination of electrons holes, increasing the removal efficiency. Similar results have been reported from previous studies, but these studies often used single composites and compared the efficiency of the semiconductor and raw CNT [3, 12-13, 17, 20].
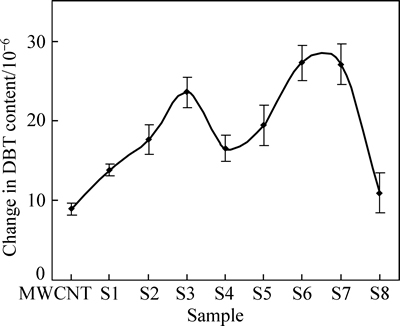
Fig. 5 Content change after 2 h irradiation
The present study synthesized and studied several composites. Figure 5 shows that the change in photo-catalytic activity by MWCNT content is a bimodal trend showing two maxima. The two maximum values are related to the simultaneous influence of DBT adsorption on the catalyst surface and oxidation by the electron hole.
Figure 5 shows the maximum DBT content change in sample 7 due to good photo-catalytic activity of the composite resulting from the high TiO2 content. Although adsorption of this composite is low, the DBT molecules (adsorbed by the composite) are efficiently oxidized by the photo-catalyst.
The other peak in Fig. 5 (sample 3) is the result of good adsorption by the composite. The DBT molecules are easily adsorbed onto the composite, come into contact with the TiO2 molecules and are oxidized. As the MWCNT content increases, molecules adsorbed onto the surface are no longer in good contact with TiO2, and photo-catalytic ability decreases. Also large quantities of MWCNTs prevent light absorption by the solution and decrease removal efficiency.
SALEH et al [18] examined the removal efficiency of acetaldehyde from water at ZnO/MWCNT mass ratios of 0.5:1, 1:1, 10:1, 20:1 and 50:1 and found that increased amounts of ZnO and MWCNT both decreased photo- catalytic activity.
HINTSHO et al [22] used a combination of titanium oxyacetylacetonate and MWCNT as a photo-catalyst taking into account the TiO2 relative to MWCNT of 10%, 20%, 30%, and 40%. The removal rate of methylene blue and the photo-catalytic properties were investigated and the trends were found to be similar to those of SALEH et al [18], with the best ratio of 20%.
3.4 Reaction rate constant
The elimination reaction of DBT is a first order reaction [2, 4, 6, 10]:
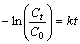 (1)
(1)
where Ct is the concentration at a given moment, C0 is the initial concentration and t is time.
Figure 6 shows the -ln(Ct/C0) plotted versus time and the rate constants calculated based on the slope of the resulting line. The reaction rate is constant after 10 h and its variation versus MWCNT content is shown.
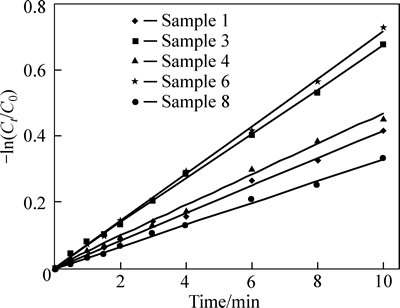
Fig. 6 Reaction rate constant diagram
As shown, it follows the same trend for photo- catalytic properties. Sample 6 shows the greatest decrease in concentration after 10 h and the maximal slope, indicating the highest reaction rate constant at 2��10-5 s. This value is more than twice the elimination rate constant without MWCNT (sample 8) and represents a large increase in the photocatalytic activity of TiO2 with the addition of MWCNT.
4 Conclusions
1) Adding MWCNT to TiO2 improves the photo- catalytic activity as a result of increased surface area and adsorption ability of composite.
2) Two factors, adsorption and oxidation, influence the photo catalytic process, so two optimal MWCNT contents (0.75 g and 0.25 g per 80 mL of sol) are recommended.
3) The DBT elimination reaction is a first order equation and the rate constants at two optimal points are calculated to be 2��10-5 s and 1.8��10-5 s, which is almost twice the rate constant without MWCNT.
References
[1] TAO H, NAKAZATO T, SATO S. Energy-efficient ultra-deep desulfurization of kerosene based on selective photooxidation and adsorption [J]. Fuel, 2009, 88: 1961-1969.
[2] DEDUAL G, MACDONALD M J, ALSHAREEF A, WU Z , TSANG D C W , YIP A C K. Requirements for effective photocatalytic oxidative desulfurization of a thiophene- containing solution using TiO2 [J]. Journal of Environmental Chemical Engineering. 2014, 2: 1947-1955.
[3] VU T H T, NGUYEN T T T, NGUYEN P H T, DO M H, AU H T, NGUYEN T B, NGUYEN D L, PARK J S. Fabrication of photocatalytic composite of multi-walled carbon nanotubes/TiO2 and its application for desulfurization of diesel [J]. Materials Research Bulletin, 2012, 47: 308-314.
[4] ZHAO D, ZHANG J, WANG J, LIANG W, LI H. Photocatalytic oxidation desulfurization of diesel oil using Ti�Ccontaining zeolite [J]. Petroleum Science and Technology, 2009, 27: 1-11.
[5] HUANG D, WANG Y J, CUI Y C, LUO G S. Direct synthesis of mesoporous TiO2 and its catalytic performance in DBT oxidative desulfurization [J]. Microporous and Mesoporous Materials, 2008, 116: 378-385.
[6] ZHANG Juan, ZHAO Di-shun, YANG Li-yan, LI Yong-bo. Photocatalytic oxidation dibenzothiophene using TS-1 [J]. Chemical Engineering Journal, 2010, 156: 528-531.
[7] TAKENAKA S, ARIKE T, MATSUNE H, KISHIDA M. Coverage of carbon nanotubes with titania nanoparticles for the preparation of active titania-based photocatalysts [J]. Applied Catalysis B: Environtal, 2012, 125: 358-366.
[8] GEORGIOS P G. Titania carbon nanotube composites for enhanced photocatalysis [D]. Gainesville: University of Florida, 2006.
[9] KOCI K, OBALOVA L, MATEJOVA L, PLACHA D, LACNY Z, JIRKOVSKY J, SOLCOVA O. Effect of TiO2 particle size on the photocatalytic reduction of CO2 [J]. Applied Catalysis B: Environtal, 2009, 89: 494-502.
[10] LIU Hao, YU Xian-qun, YANG Hong-min. The integrated photocatalytic removal of SO2 and NO using Cu doped titanium dioxide supported by multi-Walled carbon nanotubes [J]. Chemical Engineering Journal, 2014, 243: 465-472.
[11] LIU Hao, ZHANG Hai-ru, YANG Hong-min. Photocatalytic removal of nitric oxide by multi-walled carbon nanotubes-supported TiO2 [J]. Chinese Journal of Catalysis, 2014, 35: 66-77.
[12] SALEH T A. The role of carbon nanotubes in enhancement of photocatalysis [M]// Synthesis and Applications of Carbon Nanotubes and their Composites, 2013: 479-493.
[13] AAZAM E S. Visible light photocatalytic degradation of thiophene using Ag-TiO2/multi-walled carbon nanotubes nanocomposite [J]. Ceramics International, 2014, 40: 6705-6711.
[14] ADAMS L A, OKI A, LUO Z P. Sol-gel and hydrothermal approach to single-walled carbon nanotubes TiO2 nanoparticles [J]. Journal of Science and Research Development, 2011, 13: 151-159.
[15] JITIANU A, CACCIAGUERRA T, BENOIT R, DELPEUX S, BEGUIN F, BONNAMY S. Synthesis and characterization of carbon nanotube-TiO2 nanocomposites [J]. Carbon, 2004, 42: 1147-1151.
[16] JITIANU A, CACCIAGUERRA T, BERGER M H, BENOIT R, BEGUIN F, BONNAMY S. New carbon multiwall nanotubes-TiO2 nanocomposites obtained by the sol-gel method [J]. Journal of Non-Crystalline Solids, 2004, 345-346: 596-600.
[17] ZHU Lu-ping, LIAO Gui-hong, HUANG Wen-ya, MAC Li-li, YANG Yang, YU Ying, FU Shao-yun. Preparation, characterization and photocatalytic properties of ZnO-coated multi-walled carbon nanotubes [J]. Materials Science and Engineering, 2009, B, 163: 194-198.
[18] SALEH T A, GONDAL M A, DRMOSH Q A, YAMANI Z H, AL-YAMANI A. Enhancement in photocatalytic activity for acetaldehyde removal by embedding ZnO nano particles on multiwall carbon nanotubes [J]. Chemical Engineering Journal, 2011, 166: 407-412.
[19] CHRISTOPHER M, CHEN L, MILTON S, EDWARD B, KARL J J, BRADLEY B. Trapped CO2 in carbon nanotube bundles [J]. The Journal of Physical Chemistry B, 2003, 107: 12930-12941.
[20] LI You-ji, LI Lei-yong, LI Chen-wan, CHEN Wei, ZENG Meng-xiong. Carbon nanotube/titania composites prepared by a micro-emulsion method exhibiting improved photocatalytic activity [J]. Applied Catalysis A: General, 2012, 427-428: 1-7.
[21] SAWANTA M S, CHIRAYATH B A, POPATRAO B N, PATIL P S. Synthesis, characterization of hydrothermally grown MWCNT-TiO2 photoelectrodes and their visible light adsorption properties [J]. Journal of Solid State Science and Technology, 2012, 1: M15-M23.
[22] HINTSHO N, NECHAEV E, TITINCHI S, NDUNGU P. Photo- catalytic activity of titanium dioxide carbon nanotube nano- composites modified with silver and palladium nanoparticles [J]. Applied Catalysis B: Environmental, 2014, 156-157: 273-283.
(Edited by FANG Jing-hua)
Received date: 2014-09-17; Accepted date: 2015-01-25
Corresponding author: Abdolsamad Zarringhalam Moghadam; Tel: +98-2182883337; E-mail: zarrin@modares.ac.ir