Effect and mechanism of dolomite with different size fractions onhematite flotation using sodium oleate as collector
来源期刊:中南大学学报(英文版)2016年第3期
论文作者:印万忠 罗溪梅 王云帆 孙传尧 马英强 刘建
文章页码:529 - 534
Key words:dolomite; hematite; sodium oleate; mineral dissolution; depressing effect
Abstract: The effect of dolomite with different particle size fractions on hematite flotation was studied using sodium oleate as collector at pH of about 9. The effect mechanism of dolomite on hematite flotation was investigated by means of solution chemistry, ultraviolet spectrophotometry (UV), inductively coupled plasma atomic emission spectrometry (ICP-AES) and X-ray photoelectron spectroscopy (XPS). It is observed that dolomite with different size fractions has depressing effect on hematite flotation using sodium oleate as collector, and dolomite could be the “mineral depressant” of hematite using sodium oleate as collector. The reasons for that are concerned with sodium oleate consumption and the adsorption onto hematite of dissolved species of dolomite.
J. Cent. South Univ. (2016) 23: 529-534
DOI: 10.1007/s11771-016-3099-8
LUO Xi-mei(罗溪梅)1, YIN Wan-zhong(印万忠)2, WANG Yun-fan(王云帆)3,
SUN Chuan-yao(孙传尧)4, MA Ying-qiang(马英强)2, LIU Jian(刘建)1
1. State Key Laboratory of Complex Nonferrous Metal Resources Clean Utilization (Faculty of Land and
Rescource Engineering, Kunming University of Science and Technology), Kunming 650093, China;
2. College of Zijin Mining, Fuzhou University, Fuzhou 350116, China;
3. Faculty of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China;
4. Beijing General Research Institute of Mining & Metallurgy, Beijing 100044, China
 Central South University Press and Springer-Verlag Berlin Heidelberg 2016
Central South University Press and Springer-Verlag Berlin Heidelberg 2016
Abstract: The effect of dolomite with different particle size fractions on hematite flotation was studied using sodium oleate as collector at pH of about 9. The effect mechanism of dolomite on hematite flotation was investigated by means of solution chemistry, ultraviolet spectrophotometry (UV), inductively coupled plasma atomic emission spectrometry (ICP-AES) and X-ray photoelectron spectroscopy (XPS). It is observed that dolomite with different size fractions has depressing effect on hematite flotation using sodium oleate as collector, and dolomite could be the “mineral depressant” of hematite using sodium oleate as collector. The reasons for that are concerned with sodium oleate consumption and the adsorption onto hematite of dissolved species of dolomite.
Key words: dolomite; hematite; sodium oleate; mineral dissolution; depressing effect
1 Introduction
Iron is one of the most common elements in the Earth’s crust. The primary industrial types of iron-bearing minerals include oxides and hydroxides and to a lesser extent including carbonate: magnetite (Fe3O4), hematite (Fe2O3), goethite (FeO(OH)), limonite (FeO(OH)・nH2O) and siderite (FeCO3).
Previous studies on the flotation of iron ores have shown that iron ores can be floated with both anionic and cationic collectors [1-4]. Although the reverse cationic flotation remains the most popular flotation route used in the iron ore industry, the reverse anionic flotation of iron ores in China is successful [5]. Reverse cationic flotation is more sensitive toward desliming the flotation feed, while the anionic route is more sensitive toward the ionic composition of the pulp [6]. The use of direct flotation of iron oxides seems still attractive in the case of low grade ores, marginal ores that may be floated to increase the stripping ratio, and also in the recovery of material stored in tailings pond.
Hematite is of significant interest due to its variability in chemical and phy-chemical properties, from one sample to another, which modifies, sometimes significantly, its floatability behavior [7]. The flotation of hematite using saturated fatty acid with 6-18 carbon atoms in the chain has been investigated [8].
In recent years, mineral processors dealing with hematite have faced a challenge that the flotation results of hematite are poor in the presence of carbonates, such as dolomite and siderite [9-11]. The Fe grade of iron concentrate decreases with the increase of carbonate content in iron ores. But so far researchers have not been able to find the exact reasons for the phenomenon.
Carbonates are salt-type minerals. Their dissolution characteristics play a major role in determining the nature of the interactions taking place in the bulk solution or on the mineral surfaces [12-13]. The dissolved mineral species can undergo the reactions such as hydrolysis, complexation, adsorption, and surface and bulk precipitations, which can inhibit the selective interaction between reagents and minerals [14-15]. HU [16] reported that the dissolution behavior had a crucial effect on collector and modifier, thus affecting the mineral separation. Dolomite, a salt-type mineral, in aqueous dispersions is dissolved and gives ions of Ca and Mg and certainly hydrolysis products [17-21]. The investigation of solution chemistry is particularly important for salt-type mineral flotation.
In this work, the pure mineral flotation tests were conducted using sodium oleate as collector to discuss the effect of dolomite on hematite flotation. The aim of the present study was to investigate the effect mechanism of dolomite on hematite flotation by means of solution chemistry, UV, ICP-AES and XPS.
2 Materials and methods
2.1 Materials and reagents
The dolomite sample was obtained from Kuandian prefecture, Liaoning province, China. The hematite sample was purchased from Anshan city, Liaoning province, China. The hematite sample was carefully ground in an iron ball mill, processed by gravity concentration and sieved to 45-106 μm size fractions. The dolomite sample was carefully ground in a porcelain ball milling with porcelain grinding balls, and sieved to 45-106 μm, 18-45 μm and <18 μm size fractions separately. X-ray diffraction analysis and chemical analysis of two samples showed that the dolomite and hematite samples were of high purity. The dolomite sample contained about 30.13% CaO and 21.33% MgO, the hematite sample had a purity of 67.45% Fe.
Sodium oleate with 98% purity was used as the anionic collector. Analytical grade NaOH was used for pH adjustment to keep the pH at about 9. Distilled water was used in all experiments.
2.2 Method
Flotation experiments were carried out in a 30 mL flotation cell with impeller speed of 1500 r/min. A 2.0 g sample composed of dolomite and hematite in different proportions by mass according to test design was mixed with 25 mL distilled water in flotation cell for 1 min. Then, NaOH was added to keep the pH at about 9 and the pulp was conditioned for 2 min. And then sodium oleate was added and the pulp was conditioned for 2 min. The flotation time was fixed for 3 min at room temperature (25℃). After flotation, the floated product and non-floated product were collected, filtered, dried, weighed and analyzed respectively.
The sodium oleate adsorption capacity adsorbed on minerals was measured by ultraviolet spectrophotometer with the type of Spectro Flex 6600. The steps taken were similar to Ref. [21].
The concentration of Mg2+ and Ca2+ in pulp was measured by inductively coupled plasma atomic emission spectrometry (ICP-AES) on inductively coupled plasma spectrum instrument with the type of Prodigy XP.
X-ray photoelectron spectroscopy (XPS) analysis of hematite before and after reaction with Ca2+ was carried out on photoelectron spectrometer with the type of VGESCALAB MK.
3 Results and discussion
3.1 Effect of dolomite with different particle size fractions on hematite flotation
Figure 1 shows the hematite recovery as a function of fraction of dolomite with different size fractions using sodium oleate as collector at pH of about 9. As can be seen, the hematite recovery decreases gradually when the fraction of dolomite with 45-106 μm size increases. The hematite recovery significantly decreases first and then increases when 18-45 μm or <18 μm size fraction increases. When the fraction of dolomite with 18-45μm or <18 μm size fractions is above 5%, the increase of hematite recovery may be due to the function of fine particle like frother, which can increase the bubbles quantities and enhance the froth stability, observed from experiment phenomenon. VIEIRA and PERES [22] also pointed out that the presence of fine particle (<74 μm size fractions) affected the aspect of the froth, decreasing the bubble size and improving the froth stability. Meanwhile, the results indicate that the hematite recovery in presence of dolomite is sharply declined compared with that in absence of dolomite. When the fraction of dolomite with size fraction of 18-45 μm or <18 μm is 5%, the hematite recovery decreases from 77.24% to 18.56% and 13.23%, respectively. The results also show the effect of dolomite with <18 μm size fraction on hematite recovery is most prominent, followed by 18-45 μm fraction and finally 45-106 μm fraction.
In conclusion, the results above indicate that dolomite with different size fractions has strong negative impact on hematite flotation using sodium oleate as collector at pH of about 9.
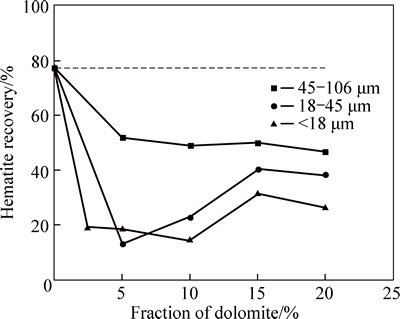
Fig. 1 Hematite recovery as function of fraction of dolomite with different size fractions (sodium oleate dosage: 120 mg/L; pH: 8.93-9.11; 2.0 g sample composed of dolomite and hematite in different proportions by mass)
3.2 Effect of sodium oleate dosage on hematite flotation from hematite-dolomite mixed minerals
Hematite recovery as a function of sodium oleate dosage in the presence of dolomite (fraction is 10%) using sodium oleate as collector at pH about 9 is shown in Table 1. The results indicate that the hematite recovery in presence of dolomite increases slightly with the sharp increase of sodium oleate dosage, but much less than that in the absence of dolomite.
Table 1 Hematite recovery as function of sodium oleate dosage in the presence and absence of dolomite
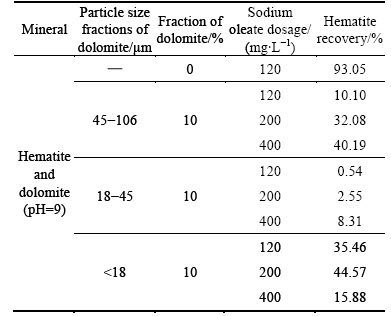
Table 2 indicates the sodium oleate adsorption capacity on different minerals in sodium oleate solution with a concentration of 120 mg/L at pH about 9. It can be seen that except dolomite with 45-106 μm size fraction, the sodium oleate adsorption capacity on dolomite with both 18-45 μm and <18 μm size fractions are higher than that on hematite. The results from both Table 1 and Table 2 indicate the increase of hematite recovery is not large with sharply increasing sodium oleate dosage, although there is difference in sodium oleate adsorption capacity between dolomite and hematite. Therefore, it can be speculated the main cause of the decrease of hematite recovery in presence of dolomite is not just sodium oleate consumption by dolomite.
The hematite recovery as a function of pH and Ca2+ concentration is shown in Fig. 2. The results show that Ca2+ has a strong depressing effect on hematite in neutral and alkali pulp environment. Therefore, it can be concluded from the results above that the main cause of the decrease of hematite recovery in the presence of dolomite may be the depressing effect of Ca2+ dissolved species of dolomite on hematite.
Table 2 Sodium oleate adsorption capacity on different minerals

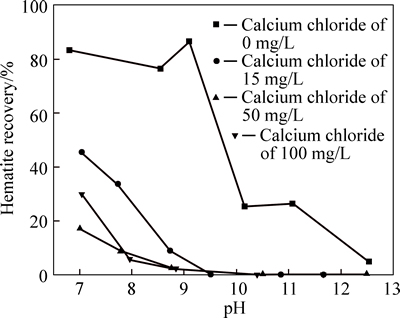
Fig. 2 Hematite recovery as a function of pH and calcium chloride dosage (sodium oleate dosage: 120 mg/L)
3.3 Effect of dolomite dissolution on hematite flotation
3.3.1 Dissolved components of dolomite in water
It is shown by the data [23-25], dolomite can be dissolved in water. The dissolution of dolomite is decided by the following formulas [13, 26]:
 =
= ,
,
 (1)
(1)
 =CaOH+, β1=101.4 (2)
=CaOH+, β1=101.4 (2)
Ca2++OH-=Ca(OH)2, β2=102.77 (3)
Mg2++OH-=MgOH+, β3=102.58 (4)
Mg2++2OH-=Mg(OH)2, β4=103.58 (5)
 =
=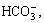
 (6)
(6)
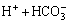 =H2CO3,
=H2CO3,  (7)
(7)
H2CO3=CO2+H2O, K0=101.47 (8)
If  Pa in atmospheres, then
Pa in atmospheres, then 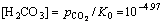 and lg[H2CO3]=-4.97.
and lg[H2CO3]=-4.97.
The relationship between the rest of component concentration and pH is as follows:
 (9)
(9)
Different composition concentration dissolved from dolomite as a function of pH is shown in Fig. 3 according to formulae (1) to (9). Figure 3 shows that HCO3- and CO32- in the solution are the main ions in alkaline condition, and Ca2+, Mg2+ and their hydroxide complexes are the principal ions in neutral and acidic condition.
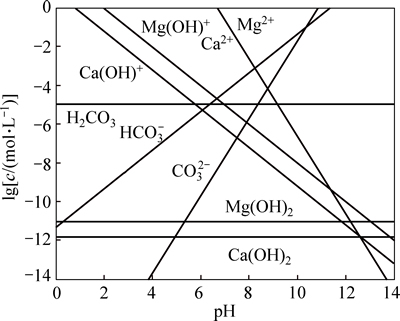
Fig. 3 Dissolved components logarithm diagram of dolomite
3.3.2 Concentration of Ca2+ and Mg2+ ion dissolved from dolomite with different size fractions
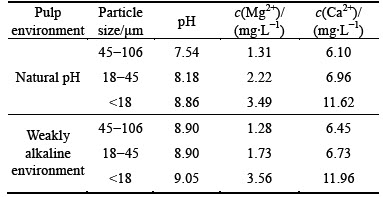
The concentration of Ca2+ and Mg2+ dissolved species of dolomite with different size fractions after stirring for 8 min in distilled water was measured by means of ICP-AES at natural pH and at pH of about 9 respectively. The results shown in Table 3 indicate that at natural pH, the concentration of Ca2+ and Mg2+ ion increases with decreasing the particle size of dolomite. Meanwhile, the same trend at pH of about 9 appears. That indicates that the smaller particle size of dolomite is, the more easily dolomite dissolves.
Table 3 Concentration of Ca2+ and Mg2+ ion dissolved from dolomite with different size fractions in distilled water
3.3.3 Adsorption of Ca2+ onto hematite
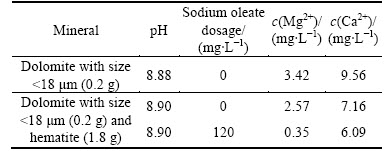
In order to confirm that the adsorption of ion dissolved from dolomite onto hematite occurs, take dolomite with <18 μm size for example, Ca2+ and Mg2+ ion concentration in dissolution of dolomite after stirring for 8 min at pH about 9 as a function of hematite content and sodium oleate dosage is measured. It can be seen from Table 4 that the concentration of Ca2+and Mg2+ in dolomite dissolution in presence of hematite decreases compared with that in absence of hematite. The results indicate that the adsorption of Ca2+and Mg2+ dissolved species of dolomite onto hematite occurs. Meanwhile, It can be also seen that the concentration of Ca2+and Mg2+ in dolomite and hematite dissolution decreases in presence of sodium oleate with a dosage of 120 mg/L in contrast to that in absence of sodium oleate. The results demonstrate that part of sodium oleate can be consumed by Ca2+and Mg2+ dissolved species of dolomite, which results in decreasing the adsorption of sodium oleate onto hematite.
Table 4 Ca2+ and Mg2+ concentration in <18 μm dolomite dissolution as a function of hematite content and sodium oleate dosage
3.3.4 Mechanism of depressing effect of Ca2+ on hematite flotation
From the results above, it can be estimated that the decrease of hematite recovery in presence of dolomite using sodium oleate as collector at pH about 9 is mainly due to two reasons. On the one hand, part of sodium oleate is consumed by dolomite for the adsorption capacity difference of sodium oleate between two minerals, which results in decreasing the adsorption of sodium oleate onto hematite. On the other hand, it is the most important reason that ions (HCO3-, CO32-, Ca2+ and Mg2+) dissolve from dolomite. The competitive adsorption onto hematite between ions (HCO3- and CO32-) dissolve species of dolomite and sodium oleate occurs. Ca2+and Mg2+ ions dissolved from dolomite have passive effect on hematite flotation in two ways. They are consumption of sodium oleate and ions adsorption onto hematite. Because the solubility product of ferric oleate (Sfo=34.2) is much less than that of calcium oleate (Sco=34.2) and magnesium oleate (Smo=13.8), the bond between oleate and Ca2+or Mg2+ is more difficult than that between oleate and Fe3+, which leads to depressing effect of Ca2+ on hematite flotation. Therefore, dolomite can be the “mineral depressant” of hematite using sodium oleate as collector.
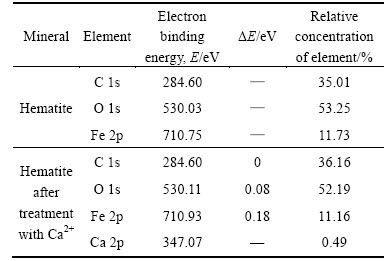
In order to further validate the depressing effect mechanism of Ca2+ on hematite, the XPS analysis of hematite before and after treatment with Ca2+ is carried out. It can be seen from Table 5 that Ca element appears on hematite and the relative concentration of C, O and Fe elements changes after treatment with calcium chloride. Meanwhile, the change in electron binding energy (ΔE) of C, O and Fe elements after treatment with Ca2+ is 0 eV, 0.08 eV and 0.18 eV, respectively, which is less than the error value of experiment equipment. That means that physical adsorption between hematite and Ca2+ occurs by the possible forms of dehydration or hydrogen bond. The bond between oleate and Ca2+ is harder than that between oleate and Fe3+, so Ca2+ from dolomite dissolution adsorbing on hematite has depressing effect on hematite flotation.
Table 5 XPS analysis results of hematite before and after treatment with Ca2+
To further verify that the other reason of depressing effect of Ca2+ on hematite is sodium oleate consumption by Ca2+, the experiment of sodium oleate dosage is carried out. The hematite recovery as a function of sodium oleate dosage in presence of calcium chloride with a dosage of 15 mg/L at pH 9 is shown in Fig. 4. Theresults indicate that the hematite recovery increases gradually with increasing the dosage of sodium oleate. These results above confirm the correctness of the above reasons.
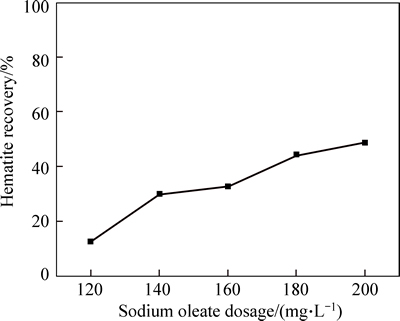
Fig. 4 Hematite recovery as a function of sodium oleate dosage in the presence of 15 mg/L calcium chloride at pH 9
4 Conclusions
1) Dolomite has negative effect on hematite flotation using sodium oleate as collector at pH about 9. The hematite recovery in presence of dolomite with different size fractions is sharply declined compared with that in absence of dolomite. The smaller particle size of dolomite is, the greater the effect is.
2) Dolomite can dissolve in water. There is a great deal of Ca2+ and Mg2+ ions dissolved from dolomite in solution at pH about 9.The smaller particle size of dolomite is, the easily dolomite dissolves.
3) The decrease of hematite recovery in presence of dolomite using sodium oleate as collector at pH about 9 is mainly due to sodium oleate consumption and dolomite dissolution by means of solution chemistry, UV, ICP-AES and XPS. On the one hand, part of sodium oleate is consumed by dolomite for the adsorption capacity difference of sodium oleate between two minerals. On the other hand, it is the most significant reason that ions (HCO3-, CO32-, Ca2+ and Mg2+) dissolve from dolomite. The competitive adsorption onto hematite between ions (HCO3- and CO32-) dissolved species of dolomite and sodium oleate occurs. Ca2+and Mg2+ dissolved species of dolomite have negative effect on hematite flotation in two ways. They are consumption of sodium oleate and ions adsorption onto hematite by physical action. The bond between oleate and Ca2+or Mg2+ is more difficult than that between oleate and Fe3+, which leads to depressing effect of Ca2+ on hematite flotation.
References
[1] ARAUJO A C, VIANA P R M, PERES A E C. Reagents in iron ores flotation [J]. Minerals Engineering, 2005, 18(2): 219-224.
[2] MOWLA D, KARIMI G, OSTADNEZHAD K. Removal of hematite from silica sand ore by reverse flotation technique [J]. Separation and Purification Technology, 2008, 58(3): 419-423.
[3] LIMA N P, VALAD O G E S, PERES A E C. Effect of amine and starch dosage on the reverse cationic flotation of an iron ore [J]. Minerals Engineering, 2013, 45: 180-184.
O G E S, PERES A E C. Effect of amine and starch dosage on the reverse cationic flotation of an iron ore [J]. Minerals Engineering, 2013, 45: 180-184.
[4] FILIPPOV L O, SEVEROV V V, FILIPPOVA I V. An overview of the beneficiation of iron ores via reverse cationic flotation [J]. International Journal of Mineral Processing, 2014, 127: 62-69.
[5] MA M. Froth flotation of iron ores [J]. Int J Min Eng Miner Process, 2012, 1(2): 56-61.
[6] MA X, MARQUES M, GONTIJO C. Comparative studies of reverse cationic/anionic flotation of Vale iron ore [J]. International Journal of Mineral Processing, 2011, 100(1/2): 179-183.
[7] MONTES-SOTOMAYOR S, HOVOT R, KONGOLO M. Technical notes. Flotation of silicate gangue iron ores: mechanism and effect of starch [J]. Minerals Engineering, 1998, 11: 71-76.
[8] QUAST K. Flotation of hematite using C6-C18 saturated fatty acid [J]. Minerals Engineering, 2006, 19(6/7/8): 582-597.
[9] ZHANG Zhao-yuan, LV Zheng-fu, YIN Wan-zhong, HAN Yue-xin. Influence of the siderite in donganshan iron ore on reverse flotation [J]. Metal Mine, 2008(10): 52-55. (in Chinese)
[10] YANG Bin. Study on separation technology and mechanism of siderite and hematite [D]. Changsha: Central South University, 2010. (in Chinese)
[11] LUO Xi-mei, YIN Wan-zhong, YAO Jin, SUN Chuan-yao, CAO Yang, MA Ying-qiang, HOU Ying. Flotation separation of magnetic separation concentrate of refractory hematite containing carbonate with enhanced dispersion [J]. The Chinese Journal of Nonferrous Metals, 2013, 23(1): 238-245. (in Chinese)
[12] van CAPPELLEN P, CHARLET L, STUMM W, WERSIN P. A surface complexation model of the carbonate mineral-aqueous solution interface [J]. Geochim Cosmochim Acta, 1993, 57(15): 3505-3518.
[13] CHEN Gong-lun, TAO D. Effect of solution chemistry on flotability of magnesite and dolomite [J]. International Journal of Mineral Processing, 2004, 74(1/2/3/4): 343-357.
[14] NUNES A P L, PERES A E C, de ARAUJO A C, VALAD O G E S. Electrokinetic properties of wavellite and its floatability with cationic and anionic collector [J]. Journal of Colloid and Interface Science, 2011, 361(2): 632-638.
O G E S. Electrokinetic properties of wavellite and its floatability with cationic and anionic collector [J]. Journal of Colloid and Interface Science, 2011, 361(2): 632-638.
[15]  S D. Electrokinetic properties of hydroxyapatite under flotation conditions [J]. Journal of Colloid and Interface Science, 2010, 343(1): 239-245.
S D. Electrokinetic properties of hydroxyapatite under flotation conditions [J]. Journal of Colloid and Interface Science, 2010, 343(1): 239-245.
[16] HU Yue-hua. Research on solution chemistry and floatability of salt-type minerals [D]. Changsha: Central South University, 1989. (in Chinese)
[17] HU Y, CHI R, XU Z. Solution chemistry study of salt-type mineral flotation systems: Role of inorganic dispersants [J]. Industrial and Engineering Chemistry Research, 2003, 42(8): 1641-1647.
[18] POKROVSKY O S, GOLUBEV S V, SCHOTT J, CASTILLO A. Calcite, dolomite and magnesite dissolution kinetics in aqueous solutions at acid to circumnetral pH, 25 to 150℃ and 55 atm pCO2 : New constrains on CO2 sequestration in sedimentary basins [J]. Chemical Geology, 2009, 260(3/4): 317-329.
[19] NERMIN G, NURGUL O. pH dependence of electrokinetic behavior of dolomite and magnesite in aqueous electrolyte solutions [J]. Applied Surface Science, 2005, 252(23): 8057-8061.
[20] POKROVSKY O S, GOLUBEV S V, SCHOTT J, CASTILLO A. Calcite, dolomite and magnesite dissolution kinetics in aqueous solutions at acid to circumnetral pH, 25 to 150 °C and 1 to 55 atm pCO2 : New constrains on CO2 sequestration in sedimentary basins [J]. Chemical Geology, 2009, 265(1/2): 20-32.
[21] JIANG Hao, LIU Guo-rong, HU Yue-hua, XU Long-hua, YU Ya-wen, XIE Zhen, CHEN Hao-chuan. Flotation and adsorption of quaternary ammonium salts collectors on kaolinite of different particle size [J]. International Journal of Mining Science and Technology, 2013, 23(2): 249-253.
[22] VIEIRA A M, PERES A E C. The effect of amine type, pH and size range in the flotation of quartz [J]. Minerals Engineering, 2007, 20(10): 1008-1013.
[23] NERMIN G, NURGUL O. pH dependence of electrokinetic behavior of dolomite and magnesite in aqueous electrolyte solutions [J]. Applied Surface Science, 2006, 252(23): 8057-8061.
[24] GAUTELIER M, SCHOTT J, OELKERS E H. An experimental study of dolomite dissolution rates at 80 °C as a function of chemical affinity and solution composition [J]. Chemical Geology, 2007, 242(3/4): 509-517.
[25] LIU A, NI W, WU W. Mechanism of separating pyrite and dolomite by flotation [J]. Journal of University of Science and Technology Beijing: Mineral, Metallurgy, Material, 2007, 14(4): 291-296.
[26] FENG Yin. Study on direct flotation separation between apatite and dolomite [D]. Changsha: Central South University, 2011. (in Chinese)
(Edited by FANG Jing-hua)
Foundation item: Project(51374079) supported by the National Natural Science Foundation of China; Project(KKSY201521031) supported by Talent Cultivation Foundation of Kunming University of Science and Technology, China; Project(2015Y067) supported by Foundation of Yunnan Educational Committee, China
Received date: 2014-09-02; Accepted date: 2015-01-31
Corresponding author: YIN Wan-zhong, Professor, PhD; Tel: +86-24-83673958; E-mail: yinwanzhong@mail.neu.edn.cn

