
Surface characteristics of chemical conversion coating for Mg-Al alloy
Masazumi OKIDO1, Ryoichi ICHINO1, Seong-Jong KIM2, Seok-Ki JANG2,
1. Nagoya University, Furo-cho, Chikusa-ku, Nagoya, 464-8603, Japan;
2. Division of Marine Engineering, Mokpo Maritime University, Mokpo City, Jeonnam, 530-729, Korea
Received 18 June 2008; accepted 10 March 2009
Abstract: The chemical conversion coating was formed on Mg alloy for low cost and harmlessness in environment by using the colloidal silica as the main component. The film formed at 298 K was thick, which was thought to be the combination of Si and O. In salt spray test, the ratio of black rust on the specimen that did not conducted chemical conversion treatment was five times or more than those of the chemical conversion treated specimen. The film of chemical conversion coating produced by alkali treatment process was thinner than the specimen produced in basic chemical conversion treatment solution.
Key words: chemical conversion coating; Mg alloy; salt spray test; alkali treatment
1 Introduction
Recently, the demand of light metals for lightweight of automobile, cellar phone etc was increased. Chromate chemical conversion coating was used in past 50 years or more as a surface treatment method of Mg-Al alloy because it has the superior corrosion resistance property. But, it was appointed to be the carcinogenesis substance of class 1 at the pollutant release and transfer register (the PRTR)[1]. In Europe, the use of lead, mercury, cadmium and Cr(Ⅵ) for the electric and electronic product was prohibited. A plan for collecting waste electric and electronic equipments(WEEE) was announced in June 2000. The use of Cr(Ⅵ) is gradually prohibited, and it will be banned totally by 2007[2]. The use of Cr(Ⅵ) will be prohibited in the automobile industry after July 2007[3], necessitating chrome-free conversion[4-7]. However, in the consi- deration of environment, the removal of carcinogenesis and the harmful matter becomes very important matter.
Therefore, in this work, the method to develop chemical conversion coating of the non-chrome type which does not use Cr(Ⅵ) was investigated with extremely simple operation.
2 Experimental
The specimens used in this experiment were AZ91D and AZ31B. The acid activation of specimen for chemical conversion treatment was executed in 2% HNO3 at 298 K for 5 s. The basic chemical conversion solution contained 200 mmol/L colloidal silica (Nissan Chemical Industries, Ltd., Snowed) with 10-20 nm SiO2 particles, 4.2 mmol/L Ti(SO4)2, 1.8 mmol/L CoSO4, and 4.2 mmol/L C2H4(COOH)2. The chemical conversion films formed on the specimens were dried at 353 K for 5 min. This solution was defined as basic chemical conversion treatment solution. The electrochemical system consisted of a Pt coil as counter electrode, and an Ag/AgCl saturated KCl as reference electrode. The anodic polarization curves was measured with a scanning rate of 1 mV/s in 0.1 mol/L NaCl and 0.1 mol/L Na2SO4 solution at 298 K. In addition, the salt spray test (SST) was carried out at a spray rate of (0.018 75±0.006 25 mL/(h?cm2) using (50±5) g/L NaCl solution at (308±1) K. The surface of the coated film was observed by SEM equipped with EDX.
3 Results and discussion
The surface morphology, elements distribution and EDX spectrum of the specimen after the chemical conversion treatment in basic condition are shown in Fig.1. The molar ratios of elements (mean values) in coating are O 78.64%, Si 2.56%, Mg 17.07%, Al 1.64%, and Zn 0.09%. The white part(A) on the coating at SEM photograph contains O 91.07%, Si 3.56%, Mg 4.82%, and Al 0.54%. Especially, the contents of Mg and Al are little, and those of O and Si are dominate. Mg 23.75%, Al 3.33%, O 70.79%, Si 1.70% are included in crack parts(B), and the Mg and Al are the main components. It is ascertained that the combination of Si and O contains mainly in the chemical conversion coating; also the Mg, Al and OH are included simultaneously.

Fig.1 SEM and EDX results after standard chemical conversion treatment: (a) Surface morphology; (b) Mg distribution; (c) Al distribution; (d) Si distribution; (e) O distribution; (f) Zn distribution; (g) EDX spectrum
Fig.2 shows the effect of immersion time on surface morphology of specimen formed in basic chemical conversion treatment solution. The surface film formed at 60 s is not complete since scratch can be seen on the surface. The film formed at immersion time of 90-180 s does not change largely. However, the combination part of Si and O (white color parts) in 90 s is wide, and the formed film is thick. On the other hand, the film formed after immersion for 300 s becomes rough. The reason is thought that the quantity of film formed with increasing immersion time is smaller than that of dissolution by activation reaction.
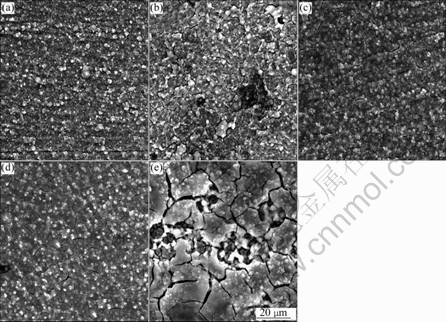
Fig.2 Effect of immersion time on morphology of specimen formed in standard chemical conversion solution: (a) 60 s; (b) 90 s; (c) 150 s; (d) 180 s; (e) 300 s
The pickling with nitric acid can remove the surface deterioration layer of magnesium alloy, the hydroxide and the oxide, so the surface of Mg alloy is activated. Then, the pretreatment for die-cast specimen concerns alkali treatment and sealing effect by hot water. KOH alkaline solution of pH 10 was prepared; and the specimen was produced with Process (1) (Alkali cleaning→hot water washing→nitric acid activation→distilled water washing→silica type chemical conversion treatment→hot water washing→warm air drying), Process (2) (Alkali cleaning→distilled water washing→nitric acid activation→distilled water washing→silica type chemical conversion treatment→distilled water washing→warm air drying), Process (3) (Nitric acid activation→ distilled water washing→alkali cleaning→distilled water washing→silica type chemical conversion treatment→distilled water washing→warm air drying), and Process (4) (Nitric acid activation→distilled water washing→silica type chemical conversion treatment→distilled water washing→hot water immersion for 10 min at 100 ℃→warm air drying).
SEM photographs of specimen surface produced from alkali treatment Process (1)-(3) are shown in Fig.3. The combination of Si and O (white parts) on the surface of chemical conversion coating (Fig.3(a)) which is produced by alkali treatment before the activation in the nitric acid is dispersed compared with chemical conversion coating produced in basic conditions (Fig.3(c)). On the surface of chemical conversion coating formed from Process (1), the microcracks are fine and uniform. The microcracks in specimen surface formed by Process (2) are larger compared with that by Process (1). It is thought that the magnesium alloy is unevenly dissolved since standard process (Process (3)) is activated, and the hydroxide film on the surface by alkali treatment is formed. Fig.4 shows the anodic polarization curves of the chemical conversion coating formed in basic condition, and Process (1), (2), (3). The potential with the increase of anodic current decreases in comparison with that in basic treatment condition. The Process (1) among various conditions is slightly high, but corrosion resistance does not improve. The microcracks in the film formed on the surface by anodizing of the magnesium alloy were sealed by hot water[8-10]. In the same way, sealing treatment was conducted after chemical conversion treatment to bury microcracks. It is thought that the hydration oxide is formed by the hydration reaction with the film, and the volume of the film expands[11]. Then, the Process (4) by hot water was carried out after chemical conversion treatment process in basic condition.

Fig.3 SEM photographs of specimens produced by different processes: (a) Process (1); (b) Process (2); (c) Process (3) (standard process)
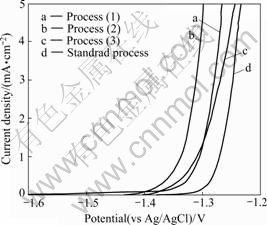
Fig.4 Anodic polarization curves after chemical conversion treatment
The SEM photograph, and EDX cross-section analysis of the specimen surface produced by Process (4), are shown in Fig.5. It is seen that the microcracks of the specimen surface after the hot water treatment became fine in comparison with the specimen produced in basic solution condition. From the result of EDX analysis, the chemical conversion coating formed by Process (4) became thinner than the chemical conversion coating (2-3 μm) produced under basic chemical conversion treatment solution condition. Furthermore, the distribution of chemical composition was very irregular. It is thought that the adhesion between the chemical conversion coating and the base metal is deteriorated because hot water makes microcracks dissolve in the silica type chemical conversion coating. In the anodic polarization experiment, the corrosion resistance in sealing by hot water after chemical conversion treatment in basic condition gets worse than that in basic condition.

Fig.5 SEM photographs (a, b) and EDX cross-sectional analysis (c, d) on chemical conversion coating in hot water for 10 min at 373 K
Fig.6 shows the morphologies of salt spray test of chemical conversion coating on AZ91D formed in various solution conditions. Each specimen after salt spray test for 10, 48, 96 h was washed, dried, and observed. The surface of specimen of only polishing (Fig.6(a)) formed the black rust and the white rust after the salt spray test for 10 h. The corrosion range expanded with time increasing. Simultaneously it cannot judge that the filamentous rust exists clearly until 10 h. The black filamentous rust appeared at 48 h, and the rust grew with time increasing. The area ratio of the black rust to whole specimen area appeared to be 27.95% after 10 h, 33.84% after 48 h, and 43.61% after 96 h. The specimen produced under basic chemical conversion treatment condition (Fig.6(b)) showed the rust on the surface after salt spray test for 10 h. The filamentous rust grew with salt spraying time increasing. The area ratios of the black rust after 10, 48, and 96 h were 1.57%, 6.24% and 9.62%, respectively. The white rust could not be verified almost. In alkali treatment process, the specimen of Process (1) had the similar appearance color to the specimen produced at standard condition. After the salt spraying test for 10 h, the black filamentous rust grew gradually and the area ratios were 1.03% for 48 h and 3.66% for 96 h, respectively. And also, the specimens of Process (2), (3) and hot water treatment had the similar appearance color to the specimen produced in standard condition. In Process (2), the black filamentous rust generated after 10 h was 1.69%, 2.88% after 48 h, and 3.90% after 96 h. In Process (3), the black filamentous rust produced after 10 h occupied 0.72% of the total area, 1.63% after 48 h, and 2.74% after 96 h. The area ratio of the filamentous black rust on film produced by alkali treatment was almost the same. It was decreased compared with that of chemical conversion coating produced in standard condition. In Process (4), the black filamentous rust after the salt spraying test for 10 h occupied 2.66% of the total area, 4.46% for 48 h, 8.92% for 96 h. The filamentous rust was almost the same as film produced in standard condition. The ratio of black rust on specimen which did not conduct chemical conversion treatment was five times or more than that of chemical conversion treated specimen. The specimens of the Process (1), (2) and (3) which executed alkaline treatment have almost similar corrosion-resistant quality, while the specimen of the Process (3) after salt spray test for 96 h shows the lowest ratio of black rust.
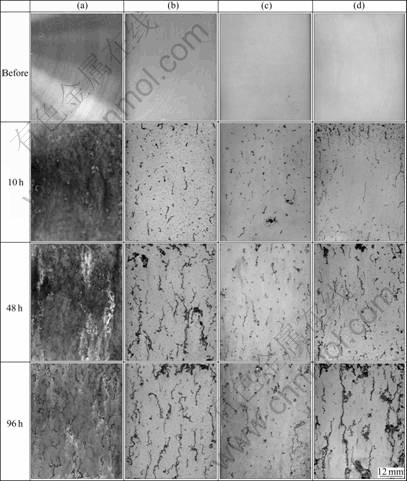
Fig.6 Morphologies of chemical conversion coating after salt spray test (white color on whole surface, black filamentous): (a) After polishing only; (b) Under standard condition; (c) By coating in alkali treatment; (d) In hot water
In salt spray test of AZ31B specimen, for only polishing specimen, the surface was corroded extensively after salt spraying test for 24 h, and the area of rust occupied 74% of the whole surface. While, the filamentous rust on the specimen chemical conversion treated in standard conditions occupied 34% of the whole surface. For only polishing specimen, the area of corrosion after the salt spraying test of 48 h was 72%, and the appearance of rust changed a little. The corrosion product during salt spray test for only polishing specimen was generated in large quantities. It is thought that the corrosion product during salt spraying test was already flow because the adhesion between the corrosion product and the base metal becomes worse. The film of chemical conversion after 48 h occurred exfoliation, and the area ratio of rust decreased to 27%. The corrosion resistance of AZ91D alloy is better than that of AZ31B alloy, in general. The filamentous rust area ratio of the AZ91D specimen of chemical conversion treatment after salt spray test for 48 h decreased from 33.84% to 6.24%, but that of the AZ31B decreased from 72% to 27%. The corrosion resistance by chemical conversion treatment for AZ31B was improved. However, it must be considered that the adhesion between the chemical conversion film and the AZ31B base metal is poor; and the possibility of being let flow with the corrosion product is high.
4 Conclusions
From the anodic polarization experiment result, the corrosion resistance of Mg alloy by sealing in hot water after chemical conversion treatment in standard condition get worse than that in standard condition only. The area ratio of black rust of specimen, which did not conduct chemical conversion treatment in salt spray test, was five times or more than that of chemical conversion treated specimen. The film of chemical conversion coating produced by alkali treatment process is thinner than that of the specimen produced in standard chemical conversion treatment solution.
References
[1] SUZUKI K. On the PRTR as a synthesis management for chemical matter [J]. Alutopia, 2000, 5: 40-43.
[2] The European Parliament and Council. Draft proposal for a direct on the restriction of the use of certain hazardous substances in electrical and electronic equipment, 2000.
[3] KUNIED N. Trend of tecnology for Cr+ coating in automible industry [C]// 107th Meeting of the Surface Finishing Society of Japan. 2003: 296.
[4] MIZUTANI Y, KIM S J, ICHINO R, OKIDO M. Anodizing of Mg alloys on alkaline solutions [J]. Surface and Coatings Technology, 2003, 169/179: 143-146.
[5] KIM S J, OKIDO M, MIZUTANI Y, ICHINO R, TANIKAWA S, HASEGAWA S. Formation of anodic films on Mg-Al alloys in NaOH solutions at constant potentials [J].Materials Transactions, 2003, 44(5): 1036-1041.
[6] KIM S J, ZHOU Y, ICHINO R, OKIDO M, TANIKAWA S. Characterization of the chemical conversion films that form on Mg-Al alloy in colloidal silica solution [J]. Metals and Materials International, 2003, 9(2): 207-213.
[7] KIM S J, HARA M, ICHINO R, OKIDO M, WADA N. Characterization of silica conversion film formed on Zinc-electroplated steel [J]. Materials Transactions, 2003, 44(4): 782-786.
[8] KIM S J, MIZUTANI Y, ICHINO R, OKIDO M. Improvement of film property by sealing after environmental conscious anodizing of Mg-Al alloys [C]// Third International Symposium on Biomimetic Materials Processing (BMMP-3). Nagoya, Japan, 2003: 544.
[9] KIM S J, KIM J I, OKIDO M. Sealing effects of anodic oxide films formed in Mg-Al alloys [J]. The Korean Journal of Chemical Engineering, 2004, 21(4): 915-920.
[10] KIM S J, MIZUTANI Y, ICHINO R, OKIDO M. Surface morphology and electrochemical property of Mg-Al alloys anodized in alkaline solutions and sealed [C]// 203rd Meeting of the Electrochemical Society. Paris, France, 2003: 244.
[11] SHIMAJIRI Y, NAKATA S, AONO T. Chemical conversion treatment of magnesium alloys in non chromium solution [J]. The Surface Finishing Society of Japan, 2001, 52(6): 469-470.
Corresponding author: Seong-Jong KIM; Tel: +82-61-2407226; Fax: +82-61-2407201; E-mail: ksj@mmu.ac.kr
DOI: 10.1016/S1003-6326(08)60372-6
(Edited by YUAN Sai-qian)