Syntheses, characteristics and fluorescence properties of complexes of terbium with benzoic acid and its derivatives
ZHOU Zhong-cheng(���ҳ�)1, SHU Wan-gen(������)2, RUAN Jian-ming(���)1,HUANG Bai-yun(�Ʋ���)1, LIU You-nian(������)2
(1. State Key Laboratory of Power Metallurgy, Central South University,Changsha 410083, China;
2. School of Chemistry and Chemical Engineering, Central South University,Changsha 410083, China)
Abstract: The binary complexes of terbium with benzoic acid and its derivatives (phthalic acid, iso-phthalic acid, o-aminobenzoic acid, salicylic acid, sulfosalicylic acid) were synthesized and their compositions were identified by elemental analyses. UV, IR of the complexes were investigated. The UV spectra indicate that the complexes�� ultraviolet absorption is mainly the ligands�� absorption, but the location of peak drifts. The IR spectra show that the IR spectra of complexes are different from those of free ligands, and the band at 400-500cm-1, due to the stretching vibration of Tb��O, is absent for free ligands. The fluorescence properties were investigated by using luminescence spectroscope, the results show that all the six complexes of terbium exhibit excellent luminescence, due to the transition from the lowest excited state 5D4 to 7F ground state manifold, the complexes of terbium with sulfosalicylic acid have the strongest fluorescence intensity, and is stronger than o-aminobenzoic acid-terbium, whose fluorescence intensity is regarded as the strongest one in the literature, and even stronger than some phosphor of terbium.
Key words: rare earth; terbium; benzoic acid; fluorescence; complexes CLC number: O614.33
Document code: A
1 INTRODUCTION
Rare earth complexes with aromatic carboxylic acids are a kind of luminescent materials with good properties[1, 2]. Much attention has been paid to the fluorescence properties of these complexes. There are some reports about fluorescence properties of the complexes of a part of rare earth with phthalic acid and terbium with benzoic acid and salicylic acid[3-9]. However, no report on systemic study of the fluorescence properties concerning rare earth complexes with different aromatic carboxylic acids has been published. For this purpose, we have synthesized fourteen complexes of Sm, Eu, Tb, Dy with benzoic acid derivatives[10-14], such as RC6H4COOH (R=o-NH2, H, p-NH2, o-OH, m-Cl, o-COOH, m-COOH, p-COOH, p-OH, m-NO2, 3,5-Dinitro, p-Br, o-I, 2-OH-5-SO3H) and it is found that the complexes of Sm and Dy with these aromatic acids show little fluorescence , and Eu and Tb with some of these aromatic acids show strong fluorescence in the excitation of UV, and that the complexes of terbium with sulfosalicylic acid had the strongest fluorescence intensity, and was stronger than that of o-aminobenzoic acid-terbium, whose fluorescence intensity was regarded as the strongest one in the literature, and even stronger than some phosphor of terbium. In this paper, terbium complexes with aromatic acids are synthesized and their fluorescence properties were investigated.
2 EXPERIMENTAL
2.1 Materials
The purity of terbium oxides was 99.99%, p-phthalic acid was chemical pure and iso-phthalic acid, o-aminobenzoic acid, salicylic acid, sulfosalicylic acid, sodium benzoate were analytical reagents.
2.2 Synthesis
Terbium chloride stock solution was obtained by dissolving terbium oxides in hydrochloric acid, evaporating excess acid and then diluting to predetermined concentration. The stock solution of sodium benzoate was prepared by dissolving the sodium benzoate to water, and the stock solution of phthalic acid, iso-phthalic acid, o-aminobenzoic acid, salicylic acid, sulfosalicylic acid was prepared by dissolving them respectively to 95% ethanol.
The aqueous TbCl3 solution was slowly added to ligand solution according to the molar ratio of Tb3+ to ligand 1��3 or 2��3(phthalic acid, isophthalic acid) at 80�� under stirring. The pH of the mixture was adjusted to 6.0-6.5 by addition of sodium hydroxide solution. Thus, white precipitates of the complexes came into being. After another 2h, stop stirring, the mixture was cooled to room temperature. The white precipitates of the complexes were filtered, washed with deionized water and ethanol till no chloridion could be found, then dried at 85�� under vacuum for 4h.
2.3 Measurements
Infrared spectra were recorded using KBr pellets on NEXUS-470 Fourier Transform infrared spectrophotometer (Nicolet Co. USA). Elemetary analyses were carried out on Perkin Elemer-240 elemtary analysis insrument. UV-visible absorption spectra were recorded on UV-754 spectrophotometers. The luminescence spectra were measured on Hitachi F-2500 fluorospectrophotometers. The method was as follows: finely powered sample was added to a cylindrical plastic sample cell, debulked and leveled the surface of the sample with a glass panel, then the fluorescence spectra was measured in a solid sample bracket.
3 RESULTS AND DISCUSSION
3.1 Composition of complexes
Using xvlenol orange as denote agent, the terbium contents in complexes were determined by EDTA capacity methods. The element analysis of C, H, N was performed according to the ordinary methods. The results are shown in Table 1.
Table 1 Elementary analysis of complexes

From Table 1, it can be seen that the results of element analysis are almost the same as the results of calculation according to the formula.
3.2 IR spectra
The infrared spectra of the samples in KBr pellets were recorded in the region of 4000-400cm-1. Special attention was paid to their IR around 1700-1200cm-1. The IR spectra of complexes have changed much in comparison with the uncomplexed ligands. Compared with sodium carboxylate, the symmetric stretching vibration of carboxyl(��s, COO-) is shifted from 1260-1283cm-1 to 1384-1430cm-1 and the asymmetric stretching vibration of carboxyl(��as, COO-) is shifted from 1558-1582cm-1 to 1529-1600cm-1 and the absorption becomes weak. The changes of stretching vibration of carboxyl suggests that carboxyl are indeed bound to the Tb(��) ion. Moreover, the band at 400-500cm-1 , due to the stretching vibration of Tb��O, is absent for uncomplexed ligands.
The IR spectra of Tb(o-amino)3 and Tb2(Ssal)3 are shown in Figs.1 and 2. It is clearly seen from Fig.1 that, in comparison with o-aminobenzoic acid, the symmetric stretching vibration of amino-group(��s, NH2) is shifted from 3239.26 to 3311.64cm-1 and the asymmetric stretching vibration of amino-group(��as, NH2) is shifted from 3322.19 to 3367.22cm-1; and that the symmetric stretching vibration of carboxyl(��s, COO-) is shifted from 1484.10 to 1454.55cm-1 and the asymmetric stretching vibration of carboxyl(��as, COO-) is shifted from 1666.40 to 1617.69cm-1; and that the wide band at 2564cm-1 in the free ligand disappear after forming complexes. All these changes suggest that complexes have been synthesized.

Fig.1 IR spectrum of Tb(o-amino)3
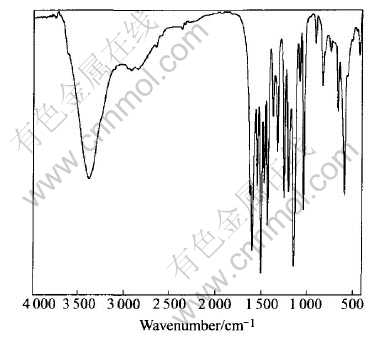
Fig.2 IR spectrum of Tb2(Ssal)3
In the IR spectrum of Tb2(Ssal)3 (Fig.2), the IR spectrum of the complexes is apparently different from the ligand. The stretching vibration of carboxyl of ligand at 1686cm-1 disappears and the symmetric stretching vibration and asymmetric stretching vibration of carboxyl at 1601 and 1489cm-1 appears. The stretching vibration of Ar��OH is shifted from 1282 to 1225cm-1, which overlay the characteristic peak of sulfonic acid group at 1230cm-1 forming an wide band. Additionally, the characteristic peak of the stretching vibration of Tb��O is at 448cm-1. From the analyses we can conclude that the carboxyl and phenol take part in coordination.
3.3 UV-visible absorption spectra
Table 2 illustrates the UV absorption of ligands and their complexes in ethanol solution. It is clearly seen that the absorption of complexes are characterized by the high intensity bands of the corresponding ligands, which have intense absorption than rare earth ion in the region of 200-400nm, and whose absorption energy can transfer to terbium ion after forming complexes.
Table 2 UV absorption peaks of ligands and their complexes

3.4 Fluorescence properties
3.4.1 Excitation spectra
The excitation spectra were measured when the emission wavelength was fixed at 545nm and the excitation spectra of complexes are shown in Fig.3.

Fig.3 Fluorescence excitation spectra of Tb2(Ssal)3(a), Tb(o-amino)3(b),Tb(Sal)3(c), Tb(BA)3(d), Tb2(Phth)��4H2O(e) and Tb2(iso-Phth)��4H2O(f)
Fig.3 shows that all complexes give a wide band from 250 to 400nm but the peak differ greatly. The excitation intensity of complexes decreases in the following order: Tb2(Ssal)3>Tb(o-amino)3>Tb(Sal)3>Tb(BA)3>Tb2(Phth)��4H2O>Tb2(iso-Phth)��4H2O and the strongest excitation peak in turn is 353, 376, 340, 292, 290, 295nm. Regarding the UV absorption of free ligands which have a strong absorption in the region of 250-380nm, we know that the excitation spectra of complexes are due to the ����* transition of ligands.
3.4.2 Emission spectra
Under the excitation of UV , the six complexes are strongly luminescent, due to the transition from the lowest excited state 5D4 to 7F ground state manifold, that is 5D4��7F6 (491nm), 5D4��7F5(545nm), 5D4��7F4(586nm), 5D4��7F3(622nm) etc. This suggests that the lowest triplet state energy level of these ligands and Tb(��)matches very much[15]. Take Tb(o-amino)3 as example, the relative intensity of the emission peak is 2411��5326��384.6��173.7.
Table 3 shows the assignment and relative intensity of fluorescence emission spectra of complexes. Fig.4 shows the fluorescence emission spectra of complexes.
From Table 3 and Fig.4, it is obvious that, according to the changes of substituting group of benzene ring, the fluorescence intensity of complexes decreases in the following order as: Tb2(Ssal)3>Tb(o-amino)3>Tb(Sal)3>Tb(BA)3>Tb2(iso-Phth)��4H2O>Tb2(Phth)��4H2O, and of which Tb2(Ssal)3 has the strongest fluorescence
Table 3 Fluorescence emission peak positions and their relative intensities and assignments

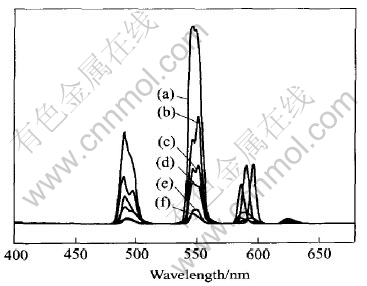
Fig.4 Fluorescence emission spectra of Tb2(Ssal)3(a), Tb(o-amino)3(b), Tb(Sal)3(c),Tb(BA)3(d), Tb2(iso-Phth)��4H2O(e)and Tb2(Phth)��4H2O(f)
intensity, and is stronger than o-aminobenzoic acid-terbium, whose fluorescence intensity is regarded as the strongest one in the literature[9], and even stronger than some phosphor of terbium.
4 CONCLUSIONS
1) The binary complexes of terbium with benzoic acid and its derivatives (phthalic acid, isophthalic acid, o-aminobenzoic acid, salicylic acid, sulfosalicylic acid) are synthesized.
2) The UV spectra indicated that the complexes�� ultraviolet absorption is mainly the ligands�� ultraviolet absorption, but the location of peak drifts. The IR spectra of complexes are different from those of free ligands and suggests that coordination bonds form between ligands and terbium ion.
3) All the six complexes of terbium exhibit excellent luminescence, according to the changes of substituting group of benzene ring, the fluorescence intensity of complexes decreases in the following order as: Tb2(Ssal)3>Tb(o-amino)3>Tb(Sal)3>Tb(BA)3>Tb2(iso-Phth)��4H2O>Tb2(Phth)��4H2O.
4) Tb2(Ssal)3 is firstly synthesized which has the strongest fluorescence intensity, and is stronger than o-aminobenzoic acid-terbium, whose fluorescence intensity is regarded as the strongest one in the literature[9], and even stronger than some phosphor of terbium.
REFERENCES
[1]Weissan S I. Intramolecular energy transfer-the fluorescence of complexes of europium[J]. J Chem Phys, 1942,10: 214-217.
[2]LI Wen-lian. Rescent advance in luminescence study of rare earth organic complexes[J]. Chemistry, 1991, 7(1): 1-8. (in Chinese)
[3]SHOU Han-sen, YE Jian-pin, YU Qun. Luminescence properties of benzoic acid-terbium complexes[J]. J Lumin, 1988, 42: 29-34.
[4]LI Wen-lian, LI Wei-li, YU Gui, et al. Luminescence enhancement of Eu(��) or Tb(��) complexes with organic ligands by Ln(��)(Ln=Y, La, Gd, Lu)[J]. Journal of Alloys and Compounds, 1993, 192: 34-36.
[5]LI Wen-lian, YU Gui, HUANG Shi-hua. Enhancement effects of Y(��) ions on Tb(��) fluorescence in Tb(��)-Y(��)-benzoic acid complexes[J]. Journal of Alloys and Compounds, 1993, 194: 19-22.
[6]LI Wen-lian, YU Gui, ZHAO Xu. The preparation and luminescence of Eu(��) activated o-phenanthroline containing Gd(��) succinate phosphors[J]. Journal of Alloys and Compounds, 1994, 206: 195-199.
[7]SHOU Han-sen, YE Jian-ping, YU Qun. Fluorescent property of benzoic acid terbium complexes[J]. Chinese J Appl Chem, 1988, 5(3): 9-14. ( in Chinese)
[8]YANG Yu-hua, CAI Qiang, MENG Ji-wu, et al. High efficient luminescence of Salcylic acid-Tb3+(Gd3+)complexes[J]. Chinese J lumin, 1991, 12(2): 151-154. ( in Chinese)
[9]ZHANG Gan-bin, CHENG Zhong-lu, LI Zhi-long, et al. Fluorescence properties of several binary complexes of terbium substituted benzoic acid[J]. J Hubei University (Natural Edition), 1998, 20(2): 162-164. ( in Chinese)
[10]SHU Wan-yin, ZHOU Zhong-cheng, NIU Chong-wei. Fluorescence of complexes of Eu(��)with aromatic carboxylic acid-1,10-phenanthroline[J]. Trans Nonferrous Met Soc China, 2001, 11(5): 106-109.
[11]ZHOU Zhong-cheng, SHU Wan-gen, WAGN Zheng-xiang. Spectra analysis and fluorescence sensation of solid complexes Tb1-xGdxA(A=o-Aminobenzoic acid, x=0.0-0.9) [J]. Chinese J Spec Lab, 2002, 19(5): 569-572. ( in Chinese)
[12]ZHOU Zhong-cheng, SHU Wan-gen, RUAN Jian-ming, et al. Fluorescence properties of Tb1-xLaxA(A=o-aminobenzoic acid, x=0.0-0.9) [J]. Rare Metal, 2003, 3: 154-157. (in Chinese)
[13]ZHOU Zhong-cheng, SHU Wan-gen, RUAN Jian-ming, et al. Fluorescence properties of the complexes of terbium with o-aminobenzoic acid[J]. Rare Earth, 2003, 4: 22-25. (in Chinese)
[14]WAGN Zheng-xiang, SHU Wan-gen, ZHOU Zhong-cheng, et al. Fluorescence properties and application of doping complexes Eu1-xLxTTA3Phen as light conversion agents[J]. J Cent South Univ Technol, 2003, 10(4): 342-346.
[15]Latva M, Takalo H, Mukkala V M. Correlation between the lowest triplet state energy level of the ligand and lanthanide(��)luminescence quantum yield[J]. J Lumin, 1997, 75(2): 149-169.
(Edited by LONG Huai-zhong)
Foundation item: Project (02JJY2021) supported by the Natural Science Foundation of Hunan Province
Received date: 2004-06-15; Accepted date: 2004-10-06
Correspondence: ZHOU Zhong-cheng, PhD; Tel: +86-731-8830614; E-mail: zhouzc@mail.csu.edu.cn