���������Ȼ��ˮ�Ե��ܶȷ��������о�
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2014���2��
�������ߣ��Դ仪 �½��� �Ⲯ�� �����
����ҳ�룺491 - 498
�ؼ��ʣ���ˮ��������Ȼ�ɸ��ԣ��ܶȷ�������
Key words��sulfide minerals; water adsorption; natural floatability; density functional theory
ժ Ҫ�������ܶȷ��������о���ˮ���������������Լ��������Ȼ�ɸ��ԡ�Ϊ���ų��������������ص�Ӱ�죬���еļ���ģ�Ͷ�������ջ����½����ġ�ˮ�������ڻ���������п��ı��棬��������ˮ����ڷ�Ǧ��ͭ�����ͻ����ˮ��������ˮ�����������Щ��ı��档��һ���棬�������������������ˮ������п����治������ˮ�������������Ǧ�����ͭ���������ˮ�ģ�����п������ˮ�ġ����������һ������ˮ�ԣ���������������������������Ϊ��������ˮ������Ҫ���뵪��������������ˣ�������Ǧ��ͭ������������Ȼ�ɸ��ԡ�
Abstract: Adsorption of water on sulfide surfaces and natural floatability of sulfide minerals were studied using density functional theory (DFT) method. All computational models were built in a vacuum environment to eliminate the effects of oxygen and other factors. H2O molecule prefers to stay with pyrite and sphalerite surfaces rather than water, whereas for galena, chalcocite, stibnite, and molybdenite, H2O molecule prefers to stay with water rather than the mineral surfaces. On the other hand, pyrite surface favors N2 more than water, while sphalerite surface cannot adsorb N2. These results show that galena, stibnite, chalcocite, and molybdenite are hydrophobic, while sphalerite is hydrophilic. Although pyrite has certain hydrophilicity, it tends to be aerophilic because the reaction of pyrite with H2O is weaker than pyrite with N2. Thus, pyrite, galena, chalcocite, stibnite and molybdenite all have natural floatability.
Trans. Nonferrous Met. Soc. China 24(2014) 491-498
Cui-hua ZHAO1,2,3, Jian-hua CHEN1,4, Bo-zeng WU2, Xian-hao LONG1
1. School of Chemistry and Chemical Engineering, Guangxi University, Nanning 530004, China;
2. Guangxi China Tin Group Co., Ltd, Liuzhou 545006, China;
3. College of Materials Science and Engineering, Guangxi University, Nanning 530004, China;
4. College of Resources and Metallurgy, Guangxi University, Nanning 530004, China
Received 23 February 2013; accepted 28 July 2013
Abstract: Adsorption of water on sulfide surfaces and natural floatability of sulfide minerals were studied using density functional theory (DFT) method. All computational models were built in a vacuum environment to eliminate the effects of oxygen and other factors. H2O molecule prefers to stay with pyrite and sphalerite surfaces rather than water, whereas for galena, chalcocite, stibnite, and molybdenite, H2O molecule prefers to stay with water rather than the mineral surfaces. On the other hand, pyrite surface favors N2 more than water, while sphalerite surface cannot adsorb N2. These results show that galena, stibnite, chalcocite, and molybdenite are hydrophobic, while sphalerite is hydrophilic. Although pyrite has certain hydrophilicity, it tends to be aerophilic because the reaction of pyrite with H2O is weaker than pyrite with N2. Thus, pyrite, galena, chalcocite, stibnite and molybdenite all have natural floatability.
Key words: sulfide minerals; water adsorption; natural floatability; density functional theory
1 Introduction
Flotation is a surface chemistry based process for the separation of fine solids, taking advantage of the difference in wettability of the solid particle surfaces. Hydrophobic surfaces solids are often naturally non-wettable by water. Such surfaces are also typically air attracting, known as aerophilic surface. They are strongly attracted to an air interface, readily displacing water on the solid surface. The floatability of minerals depends on the wettability degree of the surfaces with water. Over the last few decades, many studies have been performed on the natural floatability of certain sulfide minerals, but various researchers reached different conclusions [1-4]. For example, as early as 1940, RAVITZ [1] suggested that galena is naturally floatable; however, this premise has been object by other investigators [2-4]. Whether sulfides have natural floatability has been a controversial issue over the years.
FINKELSTEIN et al [4] and LEPETIC [5] observed natural floatability under certain conditions for chalcopyrite. HEYES and TRAHAR [6] showed that floatability in the absence of collectors occurs under oxidizing conditions and found that flotation could not be achieved under reducing conditions. GARDNER and WOODS [7] confirmed these observations with potentiostatic experiments. The presence of sulfur was suggested by these authors to be critical for chalcopyrite flotation in the presence of collectors. However, YOON [8] showed that chalcopyrite responds well to collectorless flotation process after sodium sulfide is added to the system. Sulfur was not detected on the chalcopyrite surface under these reducing conditions; thus, the natural floatability of sulfides depends on adsorption environment.
On the other hand, FUERSTENAU and SABACKY [9] studied the natural floatability of sulfide minerals (galena, chalcopyrite, chalcocite, pyrite and sphalerite) from various sources. The system used in this research was in an atmosphere containing less than 10�C6 oxygen in mole fraction and water containing less than 5��10-6 oxygen without addition of any collector or frother. The results showed that chalcocite, chalcopyrite, galena, and pyrite are naturally floatable under specific conditions (low oxygen content, pH 6.8, no collector, no frother), which disagrees with the results reported [2-4]. According to results of FUERSTENAU and SABACKY [9], oxygen is a critical factor affecting the natural floatability of sulfide minerals. In the presence of oxygen, oxidation of the surfaces of these sulfides to sulfur-oxy species occurs. Under these conditions, water molecules are hydrogen bonded to the surface and the sulfide minerals lose their natural floatability.
Influence of oxygen on the process cannot be completely eliminated in any flotation system, and absolutely clean surface of minerals is hardly obtained in certain conditions and environments, which may result in differences in natural floatability of sulfide minerals observed by several investigators.
In recent years, a great deal of research for H2O adsorption on sulfide surfaces has been carried out. Our research group [10] studied the adsorption of water on sulfide surfaces (pyrite, sphalerite, galena and molybdenite) by microcalorimetry technique. The results showed that galena and molybdenite are hydrophobic, while pyrite and sphalerite are hydrophilic. The heat of adsorption is in decreasing order of pyrite, sphalerite, galena and molybdenite. The adsorption kinetics parameters of hydrophobic galena and molybdenite surfaces are close, while those of hydrophilic pyrite and sphalerite surfaces are very different. The adsorption rate of water on the sphalerite surface is larger than that of water on the pyrite surface. STIRLING [11] studied water interaction with the (100) surface of pyrite by means of ab initio molecular dynamics simulations. The results showed that a very strong preference for molecular adsorption on the surface iron sites. Hydrogen bonding plays an important role in the stabilization of the adsorbed water. Water forms a coordinative covalent bond with the surface iron. GUEVREMONT et al [12] investigated the interaction of water with atomically clean FeS2 (100). The results showed that the binding sites on clean FeS2 (100) can be broadly classified as being associated with stoichiometric FeS2 (100) and a sulfur-deficient surface. These latter sites bind H2O more strongly than the former. WRIGHT et al [13] studied the reaction of water on the surface of PbS (galena). The results from both semi-empirical and ab initio levels of theory suggested that on a perfect (001), water is a stable species and dissociation does not occur. However, at a small step-like feature the reaction PbS+ H2O��Pb(OH)++HS- is exothermic with a sufficiently low barrier that a facile reaction occurs at ambient temperature. ROSSO et al [14] studied the interaction of gaseous O2, H2O and their mixtures with clean (100) surfaces of pyrite in ultra-high vacuum by scanning tunneling microscopy and spectroscopy (STM-STS), ultraviolet photoelectron spectroscopy (UPS) and ab initio calculations. The results indicated oxidative consumption of low binding-energy electrons occupying dangling bond surface states localized on surface Fe atoms, and the formation of Fe��O bonds. No such changes in the valence band spectra are observed for pyrite surfaces exposed to H2O. The combined gases more aggressively oxidize the surface compared with equivalent exposures of pure O2. Ab initio cluster calculations of adsorption energies and the interaction of O2 and water species with the surface indicated that H2O dissociatively sorbs when O2 is present on the surface. The study on ROSSO suggests that O2 can be influential on H2O adsorption on sulfide surfaces.
In this work, H2O adsorption on sulfide surfaces and the natural hydrophobicity of sulfide minerals were studied by density functional theory (DFT) method. The sulfide minerals studied included pyrite, sphalerite, galena, chalcocite, stibnite and molybdenite. All calculations were carried out in the vacuum environment to completely eliminate the effects of oxygen and other similar factors.
2 Computational and experimental methods and models
2.1 Computational method
Based on the DFT method, all calculations were performed by CASTEP (Cambridge serial total energy package) program module developed by PAYNE et al [15], which is a first-principle pseudopotential method based on DFT. The DFT calculations have been performed using plane wave (PW) basis sets and ultrasoft pseudopotentials [16,17]. The exchange correlation functional used was the generalized gradient approximation (GGA), developed by PERDEW and WANG (PW91) [16]. The interactions between valence electrons and ionic core were represented with ultrasoft pseudopotentials. Valence electrons configuration considered in this study included Fe 3d64s2, S 3s23p4, Cu 3d104s1, Pb 5d106s26p2, Sb 5s25p3, Zn 3d104s2 and Mo 4s24p64d55s1 states. Based on the test results, plane wave cut-off energies of pyrite and galena are 270 eV and 280 eV, respectively, and others (sphalerite, chalcocite, stibnite, molybdenite) are all 300 eV. The thicknesses of vacuum layer for all six sulfides are 15  , which is the most stable. The convergence tolerances for geometry optimization calculations were set to be the maximum displacement of 0.002
, which is the most stable. The convergence tolerances for geometry optimization calculations were set to be the maximum displacement of 0.002  , the maximum force of 0.08 eV/
, the maximum force of 0.08 eV/  , the maximum energy change of 2.0��10�C5 eV/atom and the maximum stress of 0.1 GPa, and the self-consistent field (SCF) convergence tolerance was set to be 2.0��10�C6 eV/atom.
, the maximum energy change of 2.0��10�C5 eV/atom and the maximum stress of 0.1 GPa, and the self-consistent field (SCF) convergence tolerance was set to be 2.0��10�C6 eV/atom.
2.2 Computational model
Common pyrite (FeS2) possesses a cubic crystal structure and has a space group of The common cleavage plane is face (100) along bond Fe��S. Each Fe atom on the surface coordinates with adjacent five S atoms, while each S atom coordinates with adjacent two Fe atoms and one S atom (Fig. 1(a)). Sphalerite has cubic crystal structure with space group of
The common cleavage plane is face (100) along bond Fe��S. Each Fe atom on the surface coordinates with adjacent five S atoms, while each S atom coordinates with adjacent two Fe atoms and one S atom (Fig. 1(a)). Sphalerite has cubic crystal structure with space group of  with surface (110). Each Zn atom of the surface coordinates with three S atoms, while each S atom coordinates with two Zn atoms and one S atom (Fig. 1(b)). Galena (PbS) also belongs to cubic crystal structure with a space group of
with surface (110). Each Zn atom of the surface coordinates with three S atoms, while each S atom coordinates with two Zn atoms and one S atom (Fig. 1(b)). Galena (PbS) also belongs to cubic crystal structure with a space group of  Common cleavage plane is face (100) along bond Pb��S. Each Pb atom of the surface coordinates with adjacent five S atoms, and each S atom coordinates with five Pb atoms (Fig. 1(c)). Low chalcocite has an orthorhombic unit cell with a space group of P2(1)/c, whose slab models surface (100) is shown in Fig. 1(d). S atom of the surface coordinates with four Cu atoms or three Cu atoms, and Cu atom coordinates with three S atoms and one Cu atom, or two S atoms and two Cu atoms, or three S atoms and two Cu atoms. Stibnite crystallizes in an orthorhombic space group (Pnma). Common cleavage plane is face (010), whose slab model is shown in Fig. 1(e). S atom of the surface coordinates with two Sb or three Sb atoms, and each Sb atom coordinates with three S atoms. Molybdenite is a hexagonal crystal with a space group of P63/mmc, whose cleavage plane is (001). Slab model of molybdenite is shown in Fig. 1(f).
Common cleavage plane is face (100) along bond Pb��S. Each Pb atom of the surface coordinates with adjacent five S atoms, and each S atom coordinates with five Pb atoms (Fig. 1(c)). Low chalcocite has an orthorhombic unit cell with a space group of P2(1)/c, whose slab models surface (100) is shown in Fig. 1(d). S atom of the surface coordinates with four Cu atoms or three Cu atoms, and Cu atom coordinates with three S atoms and one Cu atom, or two S atoms and two Cu atoms, or three S atoms and two Cu atoms. Stibnite crystallizes in an orthorhombic space group (Pnma). Common cleavage plane is face (010), whose slab model is shown in Fig. 1(e). S atom of the surface coordinates with two Sb or three Sb atoms, and each Sb atom coordinates with three S atoms. Molybdenite is a hexagonal crystal with a space group of P63/mmc, whose cleavage plane is (001). Slab model of molybdenite is shown in Fig. 1(f).
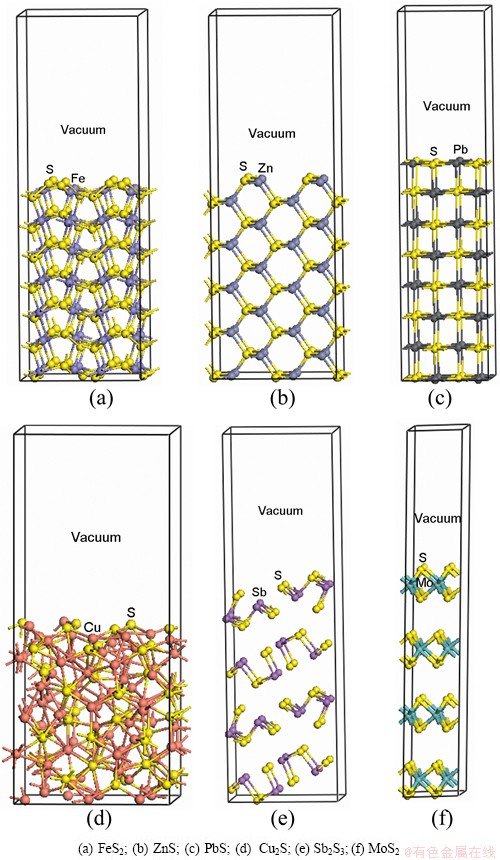
Fig. 1 Slab models of sulfides
All surfaces were obtained from the bulk sulfides with the optimum unit cell volume and were modeled by a supercell approach (2��2��1) except for chalcocite (1��1��1), where the central cell, periodic in 3D, contains a slab with two surfaces and a vacuum gap above and below the surfaces separating adjacent mirror images of the slab. The surface energies of a range of surfaces with varying slab thicknesses were calculated to determine the slab size. Figure 1 shows the most stable slab models resulted from DFT calculations. During all geometry optimization calculations, the central atomic layer of the slab is kept fixed to prevent the slab from drifting vertically along the supercell.
2.3 Calculation of adsorption energy and heat
The adsorption energies of H2O and N2 on sulfide surfaces are calculated as
 (1)
(1)
where  is the adsorption energy;
is the adsorption energy;  is the energy of the H2O or N2 molecules calculated in a cubic cell (x= H2O or N2);
is the energy of the H2O or N2 molecules calculated in a cubic cell (x= H2O or N2);  is the energy of the pyrite, sphalerite, galena, chalcocite, stibnite, or molybdenite slab;
is the energy of the pyrite, sphalerite, galena, chalcocite, stibnite, or molybdenite slab;  is the energy of the pyrite, galena, sphalerite, chalcocite, stibnite, or molybdenite slab with adsorbed H2O and N2.
is the energy of the pyrite, galena, sphalerite, chalcocite, stibnite, or molybdenite slab with adsorbed H2O and N2.
3 Results
3.1 Interaction between water and surface of sulfide minerals surfaces
There are many adsorption sites for H2O interaction with sulfide surface. Each adsorption site would provide a lot of important information. To determinate the optimal adsorption site of H2O on sulfide surfaces which is the most stable structure, adsorption site and adsorption configuration were examined. The calculation results show that the interaction between oxygen of H2O molecules and metal atom of mineral surfaces is the strongest. Figure 2 shows the adsorption models of H2O molecules on the surface of pyrite, sphalerite, galena, chalcocite, stibnite and molybdenite, which are the most stable adsorption configuration through optimization test of various adsorption sites.

Fig. 2 Adsorption models of H2O molecule on various sulfides surfaces (Numbers indicate the distance in  )
)
It is obviously observed from the models in Fig. 2 that H2O molecules show significantly different adsorption modes on various minerals surfaces. Table 1 presents the distance changes between O atoms of H2O molecules and adsorption atom (metal) of minerals surfaces (dads) compared with the sum of the atomic radius of O atom and adsorption atom (d0). After adsorption, the distance between O of H2O molecules and Fe atom of pyrite surface obviously decreases (-0.186  ), indicating that the strong interaction takes place between H2O and pyrite surface. The distance between O atoms of H2O molecules and Zn atom of sphalerite surface slightly changes (-0.022
), indicating that the strong interaction takes place between H2O and pyrite surface. The distance between O atoms of H2O molecules and Zn atom of sphalerite surface slightly changes (-0.022  ), suggesting weak interactions between H2O molecules and sphalerite surface. Whereas for the other four sulfides (galena, chalcocite, stibnite, and molybdenite), the distances between O atoms of H2O molecules and adsorption atoms of all sulfide surfaces after adsorption increase. Among them, the distance change between O atoms of H2O molecules and Mo atom of molybdenite surfaces is the largest (1.512
), suggesting weak interactions between H2O molecules and sphalerite surface. Whereas for the other four sulfides (galena, chalcocite, stibnite, and molybdenite), the distances between O atoms of H2O molecules and adsorption atoms of all sulfide surfaces after adsorption increase. Among them, the distance change between O atoms of H2O molecules and Mo atom of molybdenite surfaces is the largest (1.512  ), then stibnite (0.797
), then stibnite (0.797  ), chalcocite (0.679
), chalcocite (0.679  ), and galena (0.529
), and galena (0.529  ) in order, suggesting that H2O molecule is repelled by these mineral surfaces. The above results indicate that pyrite and sphalerite are hydrophilic, but galena, chalcocite, stibnite, and molybdenite are hydrophobic.
) in order, suggesting that H2O molecule is repelled by these mineral surfaces. The above results indicate that pyrite and sphalerite are hydrophilic, but galena, chalcocite, stibnite, and molybdenite are hydrophobic.
Table 1 Variations of distance between O atoms of H2O and metal atoms of mineral surfaces after adsorption

To assure the strength of interactions between H2O molecules and sulfide surfaces, the changes of H��O��H angle and H��O bond length of H2O molecules on sulfide surfaces before and after adsorption were calculated and the data are presented in Table 2. The change of H��O��H angle of H2O adsorbed on pyrite surface is the largest, and increases from 104.425�� to 107��, while change of H��O bond length of H2O adsorbed on sphalerite surface is the largest, from 0.977  to 0.996
to 0.996  . These results confirm the strong interaction between H2O and the surface of pyrite and sphalerite. The changes of H��O��H angle and H��O bond length of H2O adsorbed on galena, chalcocite, stibnite, and molybdenite surfaces are relatively small, indicating the weak interaction between H2O and the surfaces.
. These results confirm the strong interaction between H2O and the surface of pyrite and sphalerite. The changes of H��O��H angle and H��O bond length of H2O adsorbed on galena, chalcocite, stibnite, and molybdenite surfaces are relatively small, indicating the weak interaction between H2O and the surfaces.
Table 2 Variations of H��O��H angle and H��O bond length of H2O molecule on sulfide surfaces before and after adsorption

3.2 Adsorption energies of H2O molecules on sulfide minerals surfaces
Table 3 shows the adsorption energies of H2O molecules on the surfaces of various sulfide minerals  The data reveal that the adsorption energy of H2O on the pyrite surface is the largest, -71.206 kJ/mol; then sphalerite, -24.218 kJ/mol; galena, -8.973 kJ/mol; stibnite, -8.008 kJ/mol; chalcocite, -2.026 kJ/mol; molybdenite, 2.026 kJ/mol (negative sign represents exothermic reaction). The adsorption energy of H2O on the pyrite surface at low coverage by STIRLING et al [11] is -54.6 kJ/mol (-13 kcal/mol), which is slightly lower than our result (-71.206 kJ/mol). The smaller adsorption energy may be due to different parameters used in the calculations by STIRLING et al [11], such as cut-off energy and vacuum layer. In fact, there also exist interactions between H2O molecules that affect the adsorption of a H2O molecule toward the mineral surfaces. The binding energy between H2O molecules (
The data reveal that the adsorption energy of H2O on the pyrite surface is the largest, -71.206 kJ/mol; then sphalerite, -24.218 kJ/mol; galena, -8.973 kJ/mol; stibnite, -8.008 kJ/mol; chalcocite, -2.026 kJ/mol; molybdenite, 2.026 kJ/mol (negative sign represents exothermic reaction). The adsorption energy of H2O on the pyrite surface at low coverage by STIRLING et al [11] is -54.6 kJ/mol (-13 kcal/mol), which is slightly lower than our result (-71.206 kJ/mol). The smaller adsorption energy may be due to different parameters used in the calculations by STIRLING et al [11], such as cut-off energy and vacuum layer. In fact, there also exist interactions between H2O molecules that affect the adsorption of a H2O molecule toward the mineral surfaces. The binding energy between H2O molecules ( ) was calculated to be -19.297 kJ/mol (Table 3). The data show that the adsorption energies of H2O molecules on pyrite and sphalerite surfaces are larger than binding energy between H2O molecules (
) was calculated to be -19.297 kJ/mol (Table 3). The data show that the adsorption energies of H2O molecules on pyrite and sphalerite surfaces are larger than binding energy between H2O molecules ( <0), while the adsorption energies of H2O on the surfaces of galena, chalcocite, stibnite and molybdenite are smaller than binding energy between H2O molecules
<0), while the adsorption energies of H2O on the surfaces of galena, chalcocite, stibnite and molybdenite are smaller than binding energy between H2O molecules 

 >0). These results suggest that H2O molecule prefers to stay with pyrite and sphalerite surfaces rather than water, whereas for galena, chalcocite, stibnite, and molybdenite, H2O molecule prefers to stay with water rather than the minerals surfaces. These results confirm that pyrite and sphalerite surfaces are hydrophilic, while galena, chalcocite, stibnite, and molybdenite surfaces are hydrophobic. It is obvious that hydrophobic galena, chalcocite, stibnite, and molybdenite provide natural floatability.
>0). These results suggest that H2O molecule prefers to stay with pyrite and sphalerite surfaces rather than water, whereas for galena, chalcocite, stibnite, and molybdenite, H2O molecule prefers to stay with water rather than the minerals surfaces. These results confirm that pyrite and sphalerite surfaces are hydrophilic, while galena, chalcocite, stibnite, and molybdenite surfaces are hydrophobic. It is obvious that hydrophobic galena, chalcocite, stibnite, and molybdenite provide natural floatability.
Table 3 Adsorption energies of H2O molecules on sulfides surfaces (Negative sign represents exothermic reaction)

To find out whether hydrophilic pyrite and sphalerite are naturally floatable, we selected inert gas N2 as adsorption molecule and studied the adsorption energies of N2 on the surfaces of pyrite and sphalerite (Table 4). Adsorption energy of N2 on pyrite surface (-125.913 kJ/mol) was larger than that of H2O on pyrite surface (-71.206 kJ/mol). The results suggest that pyrite surface favors gas (N2) more than water; hence, pyrite surface is aerophilic. In other words, pyrite also offers natural floatability. The adsorption energy of N2 on sphalerite surface is 12.370 kJ/mol (positive value), indicating that N2 cannot be adsorbed on the surface of sphalerite. It suggests that sphalerite surface is hydrophilic but aerophilic. As a result, sphalerite does not provide natural floatability. In summary, pyrite, galena, chalcocite, stibnite, and molybdenite have natural floatability, while sphalerite shows no natural floatability, which is in fair agreement with the experimental results presented in Table 5 [9].
Table 4 Adsorption energies of H2O and N2 molecules on sulfides surfaces

The flotation recovery results in Table 5 reveal that the recoveries of pyrite, galena, chalcocite, stibnite, and molybdenite are greater than 80%, indicating that the surfaces of these sulfides are more hydrophobic than hydrophilic, that is, they provide natural floatability. On the other hand, the recovery of sphalerite is smaller than 60%, indicating that the surface of sphalerite is hydrophilic. According to FUERSTENAU and SABACKY [9], one of the reasons for the differences of these results with those obtained by other researchers is the content of oxygen. Our results confirm this viewpoint.
The reason why pyrite and galena, chalcocite, stibnite, and molybdenite provide natural floatability, while sphalerite is hydrophilic, according to the slab model (Fig. 1), is that galena, stibnite, and molybdenite have layered structure, so their interactions in the direction perpendicular to the surfaces is very weak, which leads to the weak adsorption strength of H2O on the surfaces of these sulfides, as shown in Table 3. Therefore, galena, stibnite, and molybdenite are naturally floatable. As for pyrite, chalcocite, and sphalerite, we will analyze them later using the density of states (DOS) of adsorption atom for sulfide surfaces.
Table 5 Flotation recovery of sulfides from various sources (Conditions: particle size of 100��200 mesh, pH = 6.8, no collector, no frother [9])

3.3 Density of states of sulfides surface atoms
Figure 3 shows DOS results of metal atoms of sulfide surfaces. The position of Fermi level is 0. For metal and semiconductor, significantly physical processes occur in the vicinity of Fermi level. In other words, DOS at Fermi level represents atomic reaction activity. So, we only considered the details near Fermi surface. It is observed from Fig. 4 that near Fermi level, electrons of MoS2, Sb2S3, Cu2S, and PbS surfaces are mainly from the Mo 4d, Sb 5p, Cu 3d, and Pb 6p orbitals. However, their DOS is small at Fermi level, especially for MoS2, Sb2S3, and PbS, which indicates that these sulfides surfaces are inactive. However, it is not easy for them to react with H2O or absorb H2O on the surface. As a result, they are hydrophobic in character. In the case of FeS2, compared with other four sulfide minerals, DOS of Fe 3d at Fermi level is large, indicating that pyrite surface is active. Hence, it is easy for pyrite to absorb water, which is in good agreement with its hydrophilicity. However, it is probable that the interaction of FeS2 with N2 to be stronger than that of FeS2 with H2O, which leads to its natural floatability. To confirm this viewpoint, DOS of surface Fe atom after H2O and N2 adsorption on pyrite surface was studied. The results are presented in Fig. 4. As for ZnS, the reaction activity does not confirm the rule of semiconductors because of being an insulator [18]; therefore, sphalerite is not discussed here.

Fig. 3 DOS results of sulfides surfaces atoms

Fig. 4 DOS results of surface Fe atom for pyrite after N2 and H2O adsorption
According to plot of Fig. 4, Fe 3d DOS curve of H2O�CFeS2 is close to that of pure FeS2 mineral near Femi level, while Fe 3d DOS curve of N2�CFeS2 shows obvious difference compared with pure FeS2 mineral. On one hand, the shape of DOS curve of Fe 3d changes, and on the other hand, peak position of Fe 3d DOS shifts to a lower energy level. The results indicate that the reaction of pyrite with H2O is weaker than that of pyrite with N2. In other words, pyrite tends to be aerophilic (N2) relative to H2O, which confirms the natural floatability of pyrite.
To know why sphalerite is hydrophilic and not aerophilic, DOS values of surface Zn atom after H2O and N2 adsorption on sphalerite surface were calculated, as shown in Fig. 5. For the convenience of comparison, DOS of Zn atom for pure ZnS mineral surface is also shown in Fig. 5. Plots in this figure show that Zn 3d DOS of N2-ZnS surface approximates to the pure ZnS surface while Zn 3d DOS peak of H2O-ZnS surface clearly shifts to a higher energy level. The results suggest that the interaction between H2O and sphalerite surface is stronger than that between N2 and sphalerite. As a result, sphalerite tends to be hydrophilic.

Fig. 5 DOS results of Zn surface atom for sphalerite after N2 and H2O adsorption
4 Conclusions
1) After adsorption, the distance between O of H2O molecules and Fe atom of pyrite surface obviously decreases, the distance between O atoms of H2O molecules and Zn atom of sphalerite surface slightly changes. Whereas for galena, chalcocite, stibnite, and molybdenite, the distances between O atoms of H2O molecules and adsorption atoms of all sulfide surfaces increase.
2) H2O molecule prefers to stay with pyrite and sphalerite surfaces rather than water, whereas for galena, chalcocite, stibnite, and molybdenite, H2O molecule prefers to stay with water rather than the minerals surfaces, which shows that pyrite and sphalerite surfaces are hydrophilic, while galena, chalcocite, stibnite, and molybdenite surfaces are hydrophobic.
3) Pyrite surface favors N2 more than water, while sphalerite surface cannot adsorb N2 and cannot be adsorbed on the surface of sphalerite. Therefore, pyrite surface is aerophilic, while sphalerite surface is hydrophilic but aerophilic.
4) The reaction of pyrite with H2O is weaker than that of pyrite with N2, while the interaction between H2O and sphalerite surface is stronger than that between N2 and sphalerite,
References
[1] RAVITZ S F. Oxygen-free flotation. II��Further experiments with galena [M]. New York: AIME Tech Pub, 1940: 1147-1168.
[2] SUTHERLAND K L, WARK I W. Principles of flotation [M]. Melbourne: Australas Inst Min Metall, 1955.
[3] KLASSEN V I, MOKROUSOV M A. An introduction to the theory of flotation [M]. 2nd ed. London: Butterworths, 1963.
[4] FINKELSTEIN N P, ALLISON S A, LOVELL V M, STEWART B V. Natural and induced hydrophobicity in sulfide mineral systems [C]//SOMASUNDARAN P, GRIEVES R B. Advances in Interfacial Phenomena of Particulate/Solution/Gas Systems. New York: Am Inst Chem Eng, Symp Ser, 1975: 165-175.
[5] LEPETIC V M. Flotation of chalcopyrite without a collector after dry, autogenous grinding [J]. Can Inst Metall Bull, 1974, 67: 71-74.
[6] HEYES G W, TRAHAR W J. The natural floatability of chalcopyrite [J]. Int J Miner Process, 1977, 4: 317-344.
[7] GARDNER J R, WOODS R. An electrochemical investigation of the natural flotability of chalcopyrite [J]. Int J Miner Process, 1979, 6: 1-16.
[8] YOON R H. Collectorless flotation of chalcopyrite and sphalerite ores by using sodium sulfide [J]. Inter J Miner Process, 1981, 8: 31-48.
[9] FUERSTENAU M C, SABACKY B J. On the natural floatability of sulfides [J]. Inter J Miner Process, 1981, 8: 79-84.
[10] ZHAO Cui-hua, CHEN Jian-hua, LONG Xian-hao, GUO Jin. Study of H2O adsorption on sulfides surfaces and hermokinetic analysis [J]. Journal of Industrial and Engineering Chemistry, 2014, 20(2): 605-609.
[11] STIRLING A, BERNASCONI M, PARRINELLO M. Ab initio simulation of water interaction with the (100) surface of pyrite [J]. The Journal of Chemical Physics, 2003, 118: 8917-8926.
[12] GUEVREMONT J M, STRONGIN D R, SCHOONEN M A A. Photoemission of adsorbed Xenon, X-ray photoelectron spectroscopy, and temperature-programmed desorption studies of H2O on FeS2 (100) [J]. Langmuir, 1998, 14(6): 1361-1366.
[13] WRIGHT K, HILLIER I H, VAUGHAN D J, VINCENT M A. Cluster models of the dissociation of water on the surface of galena (PbS) [J]. Chemical Physics Letters, 1999, 299(6): 527-531.
[14] ROSSO K M, BECKER U, HOCHELLA M F. The interaction of pyrite {100} surfaces with O2 and H2O: Fundamental oxidation mechanisms [J]. American Mineralogist, 1999, 84: 1549-1561.
[15] PAYNE M C, TETER M P, ALLAN D C, ARIAS T A, JOANNOPOULOS J D. Iterative minimization techniques for ab initio total energy calculation: Molecular dynamics and conjugate gradients [J]. Reviews of Modern Physics, 1992, 64: 1045-1097.
[16] PERDEW J P, WANG Y. Accurate and simple analytic representation of the electron-gas correlation energy [J]. Phys Rev B, 1992, 45: 13244-13249.
[17] VANDERBILT D. Soft self-consistent pseudopotentials in a generalized eigenvalue formalism [J]. Phys Rev B, 1990, 41: 7892-7895.
[18] de GIUDICI G, VOLTOLINI M, MORET M. Microscopic surface processes observed during the oxidative dissolution of sphalerite [J]. European Journal of Mineralogy, 2002, 14(4): 757-762.
�Դ仪1,2,3���½���1,4���Ⲯ��2�������1
1. ������ѧ ��ѧ����ѧԺ������ 530004��
2. �����������Źɷ�����˾������ 545006��
3. ������ѧ ���Ͽ�ѧ�빤��ѧԺ������ 530004��
4. ������ѧ ��Դ��ұ��ѧԺ, ����530004
ժ Ҫ�������ܶȷ��������о���ˮ���������������Լ��������Ȼ�ɸ��ԡ�Ϊ���ų��������������ص�Ӱ�죬���еļ���ģ�Ͷ�������ջ����½����ġ�ˮ�������ڻ���������п��ı��棬��������ˮ����ڷ�Ǧ��ͭ�����ͻ����ˮ��������ˮ�����������Щ��ı��档��һ���棬�������������������ˮ������п����治������ˮ�������������Ǧ�����ͭ���������ˮ�ģ�����п������ˮ�ġ����������һ������ˮ�ԣ���������������������������Ϊ��������ˮ������Ҫ���뵪��������������ˣ�������Ǧ��ͭ������������Ȼ�ɸ��ԡ�
�ؼ��ʣ���ˮ��������Ȼ�ɸ��ԣ��ܶȷ�������
(Edited by Hua YANG)
Foundation item: Project (51164001) supported by the National Natural Science Foundation of China; Project (NCET-11-0925) supported by New Century Excellent Talents in University, China
Corresponding author: Jian-hua CHEN; Tel: +86-771-3232200; E-mail: jhchen1971@sina.com
DOI: 10.1016/S1003-6326(14)63087-9

