J. Cent. South Univ. (2017) 24: 1-8
DOI: 10.1007/s11771-017-3402-3
Galvanic corrosion behavior of plain carbon steel-B4C composite in 3.5% NaCl solution with electrochemical noise
S. H. Ettefagh Far1, A. Davoodi2
1. Young Researchers and Elites Club, North Tehran Branch, Islamic Azad University, Tehran, Iran;
2. Department of Materials Science and Engineering, Faculty of Engineering,
Ferdowsi University of Mashhad (FUM), Mashhad, Iran
 Central South University Press and Springer-Verlag Berlin Heidelberg 2017
Central South University Press and Springer-Verlag Berlin Heidelberg 2017
Abstract: The galvanic corrosion behavior of metal-matrix composite plain carbon steel/boron carbide (B4C) in 3.5% NaCl solution was studied. The composite was locally produced as a weld band on carbon steel by means of the gas tungsten arc welding process and using nickel as the wetting agent. Samples from the weld band, heat-affected zone and parent metal region were extracted precisely and DC/AC electrochemical tests in combination with techniques such as scanning electron microcopy and energy dispersive spectrometry were conducted. The results of the electrochemical tests show that the corrosion resistance of the parent metal sample is higher than that of the welded composite and the HAZ samples. However, as the corrosion potential (ECorr) of the parent metal is more positive than other two samples, this becomes the cathode in galvanic couples with two other samples. On the other hand, the weld composite sample is also cathodic due to its more positive ECorr compared to HAZ sample. This means that the HAZ can be particularly at risk of preferential dissolution. The approach can be used in specific areas on plain carbon steel to locally increase hardness and resistance to abrasion and reduce manufacturing costs.
Key words: composites; scanning electron microscopy; potentiodynamic polarization; electrochemical noise
1 Introduction
Adding hard ceramic particles to a metal matrix (MMC) increases resistance to abrasion and hardness and can generate suitable lubrication in metal matrix composites [1].
B4C is useful properties such as a relatively high elastic modulus, high melting point and low density [2-4]. It also has very high stiffness (445 GPa) and high hardness (3700 Hv). The high hardness of boron carbide is attributed to the presence of B and C which forms covalently bonded solids [5]. B4C is considered attractive due to the high effective cross section, stability in corrosive solutions [6] and good electrical conductivity (2��107 S/m) of its microparticles [7]. The desirable chemical and thermal stability of B4C is due to its covalent bonding and it is used in the manufacture of sandblast nozzles [8] and cutting tools and with high neutron absorption capacity [9].
Plain carbon steel has abundant applications in the industry because of its high strength, ease of welding, flexibility, availability, excellent physical properties and low price [10-11], but its use has been constrained under some conditions due to its low resistance to abrasion and low surface hardness. However, the surface properties of plain carbon steel can be transformed by means of a composite layer [12].
The present research attempted to utilize locally-manufactured plain carbon steel composite using B4C under wetting by nickel using gas tungsten arc welding (GTAW) on plain carbon steel and to study its corrosive behavior in different conditions. As the B4C composite was generated in-situ on the plain carbon steel surface, its galvanic corrosion behavior becomes important when being exposed to corrosive conditions such as environments containing chloride ions [13].
2 Experimental
2.1 Materials and methods
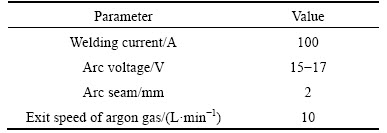
The B4C composite was generated on plain carbon steel under wetting by Ni using GTAW. A sample of plain carbon steel with dimensions of 10 mm��30 mm�� 100 mm was cut and its surface was polished with a polishing machine to remove grease and surface oxide. The sample was washed with nitric acid, alcohol and distilled water to prepare the surface for creating the stable arc. Optimized parameters were used to obtain a desirable composite on surface [14]. Polyvinyl acetate (PVA) glue was saturated with 60% (volume fraction) of B4C powder with 115 ��m, 40% (volume fraction) of Ni powder with 150 ��m and the mixture was dispersed on the parent metal with the width of 10 mm. Then, the prepared sample was placed inside an electric furnace at 140 ��C for 0.5 h in order for the glue mixture on the surface to be dried. Thereafter, the sample was exposed to surface modification operations using the GTAW device. No supplementary filler was used during welding but the melt created by the arc produced by tungsten electrode was used to produce the composite during the surface modification operation. A photograph and schematic diagram of the GTAW method of fabrication of composite are given in Figs. 1(a) and (b), respectively. Table 1 lists the parameters of the GTAW process used in this work [15]. After welding and making the composite on parent metal, the welded sample was transected and cold-mounted. The sample was polished using an 80 to 3000 polishing pad and was etched using an etchant solution to facilitate microstructural studies.
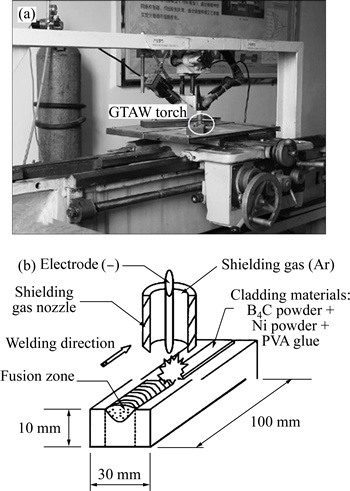
Fig. 1 Image (a) and schematic diagram (b) of GTAW method in fabrication of composite of carbon steel/B4C composite
Table 1 Parameters relating to GTAW method [14]
2.2 Electrochemical measurements
The samples with dimensions of 1 mm'1 mm'10 mm were cut from three zones of the welded area: parent metal, weld bead and heat-affected zone (HAZ), using a wire cutting device. Images of the corrosion test samples that were cut using a wire cutting device are given in Fig. 2.
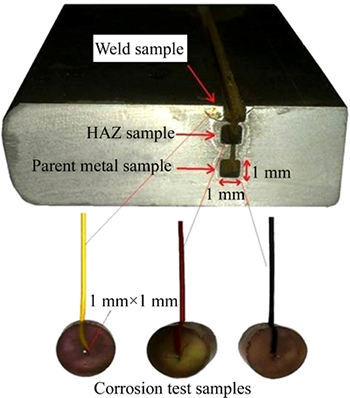
Fig. 2 Sample extraction from three regions: parent metal, weld metal and HAZ
An ��Ivium Compact Stat��, made in the Netherlands, was used to perform electrochemical tests. The temperature of the electrolyte solution was 25 ��C and a temperature controller was used to keep the temperature constant during the tests. The pH of the 3.5% NaCl solution was 5.9 and the solution contained air. The classic corrosion cell included a saturated calomel electrode (SCE) as the reference electrode, a working electrode (corrosion test sample) and a graphite counter electrode.
Parent metal, weld metal and HAZ samples were mounted for the corrosion tests and were polished using 600-1000 grit emery papers followed by a polishing pad, and were then were washed with distilled water and alcohol and dried in air. The open circuit potential (EOCP) was monitored for 3600 s (1 h) for each sample, with the interval of 1 h from the next sample.
The balance time for starting potentiodynamic polarization test was 10 s after determining EOCP. The scan speed and ��Step were 0.0001 V/s and 1 mV, respectively. The scan range was �� 150 mV relative to the ��OCP.
In order to study galvanic corrosion behavior of the composite made on plain carbon steel using electrochemical noise (EN) measurements, corrosion samples of the HAZ-parent metal, HAZ-weld metal and parent metal-weld couples were exposed in a corrosive solution. It should be mentioned that the time for each test was 1 h and 25 data per second were recorded by the instrument.
3 Results
3.1 Microstructural characterization
SEM images of parent metal, weld and HAZ samples after being polished and etched in the etchant solution are presented in Fig. 3. As can be observed, the structure of the HAZ sample in Fig. 3(b) was more coarse-grained than that in the parent metal in Fig. 3(a), which meant that grain boundary in HAZ sample was more evident than in the parent metal. Therefore, more grain boundary can affect the corrosion situation of the sample related to the HAZ zone.
An SEM image was taken from the composite in the structure were analyzed. The point analyzed using energy dispersive spectrometry (EDS) is specified in Fig. 3(c). The results of the EDS analysis relating to this point are given in Fig. 3(d). Accordingly, particles of boron, carbon, nickel and iron constitute the chemical composition of the composite at this point.
3.2 Electrochemical measurements
The potentiodynamic polarization diagram for the corrosion samples after 1 h of immersion in 3.5% NaCl solution is given in Fig. 4 and the data relating to this diagram are presented in Table 2.
As can be observed, in addition to the corrosion potential of the parent metal sample, which was more positive than the weld composite and the HAZ samples, the corrosion current (ICorr) of this sample also was less than that of two other samples. As can be observed in Fig. 5, the galvanic corrosion current and potential values were obtained from intersection of the cathodic branch of the cathode and anodic branch of the anode of the polarization curves for coupled parent metal/weld metal/B4C composite samples.
The galvanic corrosion current and potential values in respect of galvanic coupling of the parent metal sample with HAZ sample were 21.12 ��A��cm-2 and -583.41 mV; also, for its parent metal sample galvanic coupling with weld composite sample, these values were 17.18 ��A��cm-2 and -576.53 mV. Moreover, the weld composite sample was cathodic with respect to the HAZ sample in galvanic coupling and values of the galvanic corrosion current and its potential were 3.22 ��A��cm-2 and -603.21 mV. In this work, the EN data were processed in the time and frequency domains. The diagram of potential and current in the time domain for the coupled samples is represented in (Fig. 5). Using Eq. (1), standard deviation (��) of data on potential and current over the time of the EN test was calculated:
 (1)
(1)
where �� is standard deviation indicator, xi is data with any value,  is mean of the data and N is the total number of data. By dividing the obtained corrosion standard deviation from the potential to current data, noise resistance (Rn=(��E/��I)) related to each of the coupled samples was determined and all the data are presented in Table 3.
is mean of the data and N is the total number of data. By dividing the obtained corrosion standard deviation from the potential to current data, noise resistance (Rn=(��E/��I)) related to each of the coupled samples was determined and all the data are presented in Table 3.
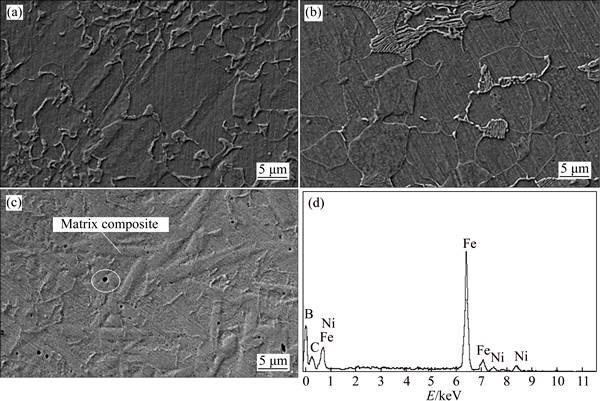
Fig. 3 SEM images of base (a), HAZ (b) and weld samples (c) after being polished and etched in etchant solution and EDS spectrum (d) relating to dark point in Fig. 3(c)
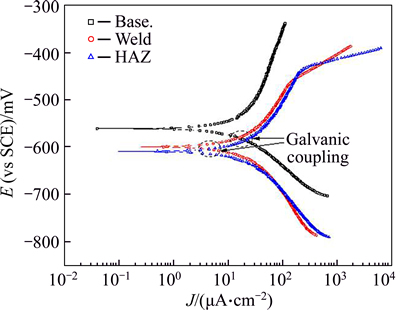
Fig. 4 Potentiodynamic polarization diagram for samples after 1 h of immersion in 3.5% NaCl solution
Table 2 Potentiodynamic polarization extracted data of all three samples after 1 h of immersion in 3.5% NaCl solution
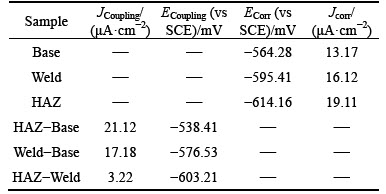
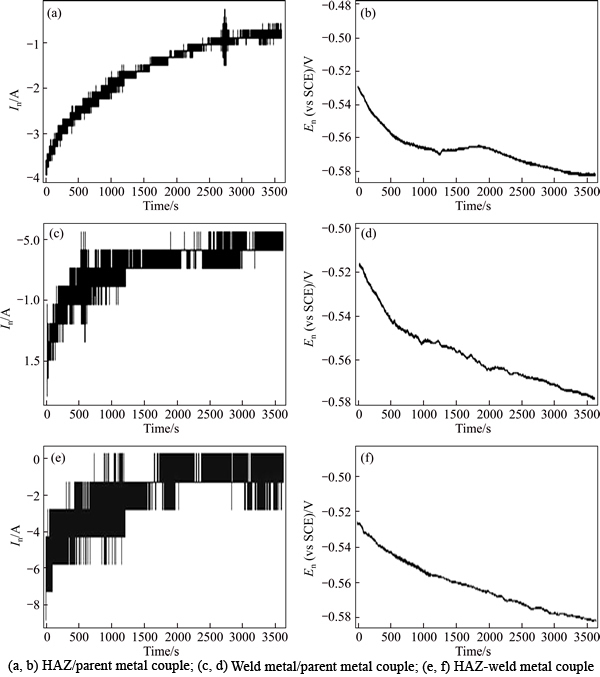
Fig. 5 Current and potential plots in time domain during 1 h immersion in 3.5% NaCl solution:
Table 3 EN extracted data in time domain for coupled samples after 1 h of immersion in 3.5% NaCl solution

As can be observed, the Rn of the coupled HAZ/parent metal samples was smaller than that of the coupled weld/parent and HAZ-weld corrosion samples. Considering the corrosion potential values obtained from the polarization test, it was observed that the value of ��corr for the parent metal samples was more positive (less negative) than that of the weld and HAZ samples. The corrosion potential difference of the parent metal-HAZ metal samples was higher than that of parent-weld and HAZ-weld samples. As a result, value of Rn of the coupled parent metal-HAZ was smaller than that of the parent metal-weld and HAZ-weld corrosion couples.
In order to analyze the electrochemical noise data in the frequency domain, a power spectral density (PSD) curve was obtained in terms of frequency for each couple, based on Fast Fourier Transformation (FFT) [16]. Current PSD, Rn and potential PSD plots in frequency domain during 1 h immersion in 3.5% NaCl solution for HAZ-parent metal couple, weld metal-parent metal couple and HAZ-weld metal couple is given in Figs. 6-8.
3.3 Microstructural studies after corrosion measurements
SEM images of structure of the parent metal, HAZ and weld metal samples were prepared after the corrosion tests and are presented in Fig. 9. As can be observed, there were fewer corrosion products on the surface of the parent metal sample than on the other samples and its surface was less affected by the 3.5% NaCl solution. In contrast, considering the surface appearance of the weld and HAZ samples, it can be observed that the surface of the HAZ sample was intensively affected by the NaCl solution and the weld sample was less severe. Thus, the SEM images confirmed the higher resistance to corrosion of the parent metal sample compared to the weld metal and HAZ samples.
4 Discussion
As has been observed, the microstructure of the HAZ sample was more coarse-grained than that of the base sample, which meant that there were fewer grain boundaries in the HAZ than in the parent metal sample. Considering the higher proportion of boron than carbon in the EDS trace, it can be predicted that B4C particles will be available in the weld zone. In general, the B4C particles with irregular geometrical shape, along with nickel, were dispersed non-uniformly in the steel matrix.
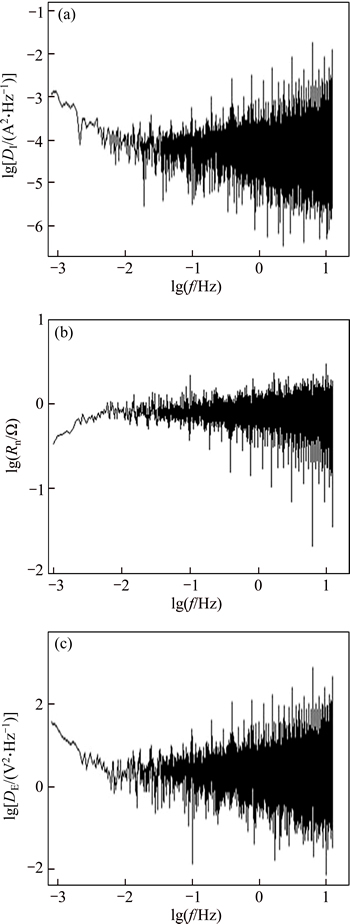
Fig. 6 Current PSD (a), Rn (b) and potential PSD (c) plots in frequency domain during 1 h immersion in 3.5% NaCl solution for HAZ-parent metal couple
Considering the EN results, the value of Rn concerning of HAZ-parent metal couple was smaller than that of both the weld metal-parent metal and HAZ-weld metal couples, respectively. Thus, the highest amount of galvanic corrosion was related to the HAZ-parent metal couple, where the HAZ sample was the anode. Also, the narrow HAZ on either side of the weld is highly susceptible to excessive galvanic attack because of the very large cathodic area provided by the parent plate. In summary, both the HAZ and weld metal samples underwent anodic attack in the corrosive solution when coupled with the parent metal, but the amount of corrosion of HAZ sample was more than thaton the weld metal because the HAZ was more anodic.
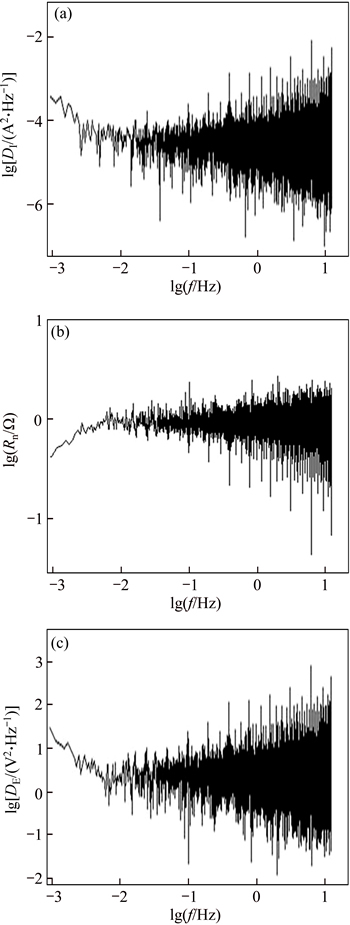
Fig. 7 Current PSD (a), Rn (b) and potential PSD (c) plots in frequency domain during 1 h immersion in 3.5% NaCl solution for weld metal-parent metal couple
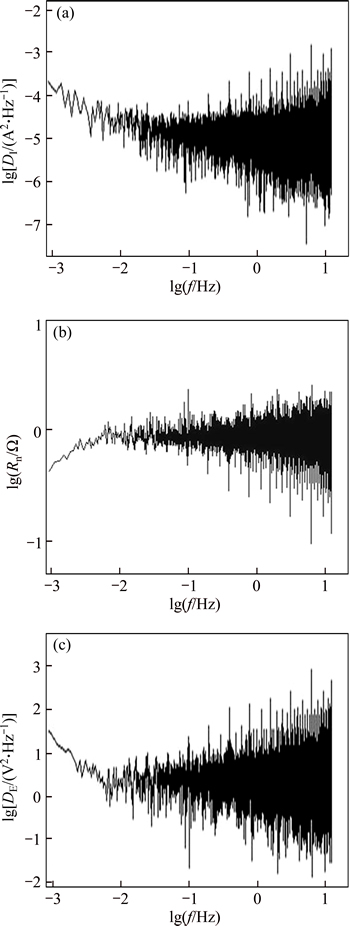
Fig. 8 Current PSD (a), Rn (b) and potential PSD (c) plots in frequency domain during 1 h immersion in 3.5% NaCl solution for HAZ-weld metal couple
From PSD curve in EN results, it was concluded that the slope of the curve was approximately zero in all PSD curves related to different corrosion couples and no high spectral power was observed at high and especially low frequencies. Thus, the anodic samples sustained uniform corrosion in each couple when undergoing galvanic corrosion. However, for all three corrosion couples, very similar Rn values were obtained at low frequencies from Rn curve in frequency domain.

Fig. 9 SEM images of corrosion damage on parent metal (a), HAZ (b) and weld metal (c) samples after corrosion tests in 3.5% NaCl solution
In summary, the following electrochemical reactions occurred during the corrosion process of all three samples. It should be noted that, because carbide particles such as B4C in the weld sample have a more positive electrochemical potential than the steel matrix, galvanic corrosion will locally occur around the B4C particles in the composite surface [17]. Therefore, area surrounding B4C particles are suitable places for anodic reactions to take place [14].
5 Conclusions
Samples from the weld band, heat-affected zone and parent metal region were extracted precisely and DC/AC electrochemical tests in combination with other techniques such as SEM-EDS were used for characterization.
1) Characterization results showed non-uniform distribution of B4C and nickel particles in the composite��s steel matrix.
2) The highest amount of galvanic corrosion was related to the parent metal sample as cathode with two other samples as anode. However, the weld composite sample had little potential difference from the HAZ sample with parent metal sample, which caused more galvanic corrosion of the heat affected sample. In addition, structure of the HAZ sample was more affected by the corrosive ions compared with other samples.
Acknowledgements
Authors would like to acknowledge Young Researchers and Elites Club, North Tehran Branch, Islamic Azad University, Tehran, Iran for providing experimental facilitates.
Nomenclature:
E
Potential (V)
f
Frequency (Hz)
I
Current (A)
D
Power spectral density
DE
Power spectral density of potential (V2/Hz)
DI
Power spectral density of current (A2/Hz)
Rn
Noise resistance (��)
In
Noise current (A)
��n
Noise potential (V)
Rp
Polarization resistance (��)
��E
Standard deviation of potential (V)
��I
Standard deviation of current (A)
References
[1] GLADKOVAS M, MEDELIENE V, SAMULEVICIENE M, JU ZELIUNAS E. Corrosion study of electroplated nickel metal-matrix composites B4C, Al2O3 and SiC [J]. Chemija, 2002, 13(1): 36-40.
[2] RADEV D D, ZAKHARIEV Z T, MARINITCH M A. Corrosion resistance of B4C-MexBy composite materials [J]. Journal of Alloys and Compounds, 1993, 196(1/2): 93-96.
[3] SINIAWASKI M T, HARRIS S J, WANG Q, LIU S. Wear initiation of 52100 steel sliding against a thin boron carbide coating [J]. Tribology Letters, 2003, 15(1): 29-41.
[4] MINGCHAO W, ZUOGUANG Z, ZHIJIE S. Effect of fiber type on mechanical properties of short carbon fiber reinforced B4C composites [J]. Ceramics International, 2009, 35(4): 1461-1466.
[5] KENNEDY A R. The microstructure and mechanical properties of Al-Si-B4C metal matrix composites [J]. Journal of Material Science, 2002, 37(2): 317-323.
[6] ALIZADEH A, TAHERI-NASSAAJ E, HAJIZAMANI M. Hot extrusion process effect on mechanical behavior of stir cast al based composites reinforced with mechanically milled B4C nanoparticles [J]. Jornal of Mater Science and Technology, 2011, 27(12): 1113- 1119.
[7] MEDELIENE V. The influence of B4C and SiC additions on the morphological, physical, chemical and corrosion properties of Ni coatings [J]. Coating Technology, 2002, 154(1): 104-111.
[8] ECKARDTA, BEWILOGUA K, VAN DER KOLK G, HURKMANS T, TRINH T, FLEISCHER W. Improving tribological properties of sputtered boron carbide coatings by process modifications [J]. Surface and Coating Technology, 2000, 126(1): 69-75.
[9] ZHANG W, GAO L, LIA J, YANG J, YIN Y. TiAl/B4C marine material-Fabrication, mechanical and corrosion properties [J]. Ceramic International, 2012, 37(3): 783-789.
[10] SINGH S K, MUKHERJEE A K. Kinetics of mild steel corrosion in aqueous acetic acid solutions [J]. Journal of Material Science and Technology, 2010, 26(3): 264-269.
[11] MARCO R D, JIANG Z T, JOHN D, SERCOMBE M, KINSELLA B. An in situ electrochemical impedance spectroscopy/synchrotron radiation grazing incidence X-ray diffraction study of the influence of acetate on the carbon dioxide corrosion of mild steel [J]. Electrochemical Acta, 2007, 52(11): 3746-3750.
[12] ARAGHI A, PAYDAR M H. Electroless deposition of Ni-P-B4C composite coating on AZ91D magnesium alloy and investigation on its wear and corrosion resistance [J]. Material Design, 2010, 31(6): 3095-3099.
[13] DING H, HAWTHORN G A, HIHARA L H. Inhibitive effect of seawater on the corrosion of particulate-reinforced aluminum-matrix composites and monolithic aluminum alloy [J]. Journal of Electrochemical Society, 2009, 156(10): C352-C359.
[14] ETTEFAGH FAR S H, DAVOODI A. Microstructure and corrosion behaviour of plain carbon steel�CB4C composite produced by GTAW method in 3.5% NaCl solution [J]. Corrosion Engineering Science and Technology, 2014, 14(1): 55-65.
[15] WANG X H, SONG S L, ZOU Z D, QU S Y. Fabrication TiC particles reinforced Fe-based coating produced by GTAW multi-layers melting process [J]. Material Science Engineering A, 2006, 441: 60-67.
[16] MANSFEILD F, XIAO H. Electrochemical noise analysis of iron exposed to NaCl solution of different corrosively [J]. Journal of the Electrochemical Society, 1993, 140(8): 2205-2209.
[17] HAN Y, GALLANT D, CHEN X G. Investigation on corrosion behavior of the Al-B4C metal matrix composite in a mildly oxidizing aqueous environment [J]. Corrosion, 2011, 67(11): 1150051-11.
(Edited by YANG Bing)
Cite this article as: S. H. Ettefagh Far, A. Davoodi. Galvanic corrosion behavior of plain carbon steel-B4C composite in 3.5% NaCl solution with electrochemical noise [J]. Journal of Central South University, 2017, 24(1): 1-8. DOI: 10.1007/s11771-017-3402-3.
Received date: 2015-11-12; Accepted date: 2016-04-11
Corresponding author: S. H. Ettefagh Far; E-mail: hosseinett@gmail.com