
Microstructure and electrochemical properties of low-temperature hydrogen storage alloy used in Ni/MH batteries
DONG Gui-xia (董桂霞), WU Bo-rong (吴伯荣), ZHU Lei (朱 磊), DU Jun (杜 军)
General Research Institute for Nonferrous Metals, Beijing 100088, China
Received 15 July 2007; accepted 10 September 2007
Abstract: AB5 hydrogen storage alloys La0.54Ce0.28Pr0.18Ni4-xCo0.6Mn0.35Alx (x=0.1, 0.2, 0.3) were prepared by arc melting method under an Ar atmosphere. The results show that the contents of Ni and Al have obvious influences on the microstructure and electrochemical properties of the alloys. Both the lattice parameters and the cell volumes decrease with decreasing x value. Moreover, the discharge capacity at different temperatures, the high rate discharge property, and the cycling life of the alloy electrode are also in close relationship with the x value. When x value increases from 0.1 to 0.3, the discharge capacities with a discharge current density of 60 mA/g slightly decreases at 25 ℃, but evidently deteriorates at -40 ℃, the high-rate property gravely decreases, and the cycle life of the alloy electrode is improved in some extent. Therefore, it is meaningful to control Al content for the AB5 hydrogen storage alloys used in Ni/MH batteries.
Key words: Ni/MH batteries; AB5 hydrogen storage alloy; electrochemical properties
1 Introduction
The hydrogen storage alloy is a key negative- electrode material used in MH/Ni battery, and it is crucial for the capacity, cycling life, high-rate discharge, high and low temperature performance, self-discharge, and environment-friendly property[1-3]. At present, the negative-electrode material used in second MH/Ni battery is mainly the AB5 hydrogen storage alloy, of which the electrochemical properties are usually stable in a temperature range from -10 ℃ to 40 ℃, but considerably deteriorates when temperature is higher than 45 ℃ or lower than -20 ℃[4-6]. In order to make MH/Ni battery used as vehicle power source in some cold places, such as northeast and northwest of China, the high-rate discharge abilities and low temperature discharge performance for the commercialized AB5 hydrogen storage alloys still need to be further improved so as to meet the requirements of the extended applications. ZHANG et al[7] have reported that the low-temperature discharge capacities of alloy electrode can be improved by increasing Mg content in La0.85MgxNi4.5Co0.35Al0.15 alloys. ZHANG et al[8] have reported that AB5-5% LaMg3 composite has higher low-temperature discharge capacity than the AB5 alloy. Mo and V also were used as additives in AB5 electrode alloy that operates at low temperature[5, 9-10]. According to the mechanism of Al in AB5 alloy, it has great possibility to influence the discharge capability of electrode alloy at relatively low temperature, but few investigations are related to this subject. In this paper, we describe the effects of Al content on the microstructure and electrochemical properties of La0.54Ce0.28Pr0.18Ni4-x- Co0.6Mn0.35Alx alloys. It will be of great assistance in producing AB5-type hydrogen storage alloy with high capacity in wide-range temperature and with high rate discharge capability.
2 Experimental
The raw materials were based on purified metals, of which the purities of the rare earth (RE) are 99.9% and those of Ni, Co, Mn and Al are all higher than 99% (mass fraction). The alloys were prepared by arc melting method under an Ar atmosphere into button forms, which were re-melted 4 times in order to improve the homogeneity. The resulting ingots were mechanically pulverized into powder with size below 74 μm. And the average diameter measured by using a laser particle size analyzer is 53 mm. The crystallographic structure of the alloy was investigated by using X-ray diffraction (XRD) with the diffraction power of 36 kV, 20 mA and Cu Ka anticathode. The scan manner was step scanning with a step of 0.02? and a scan rate of 2(?)/min. The scan scope is from 10? to 90?.
The electrode pellets were prepared by using the cool-pressure method, that is, the alloy powders of 0.4 g and Ni powders of 1.6 g were evenly mixed and rapped by Ni foams, and then pressed under a pressure of 12 MPa to be a pellet of 18 mm?18 mm?1.5 mm. The “sandwich” method was used to examine the electrochemical properties, and the measured temperatures were -40 ℃ and 25 ℃, respectively.
3 Results and discussion
3.1 Structure characterization
Fig.1 shows the XRD patterns of La0.54Ce0.28Pr0.18Ni4-xCo0.6Mn0.35Alx alloys. All of the three alloys have the single CaCu5-type phase. But there are slight differences between the relative intensities of some Bragg peaks and small shifts between the patterns because of the different x values of them. The lattice parameters and cell volumes of compounds are listed in Table 1. It can be found that both the lattice parameters and the cell volumes decrease with decreasing x value.
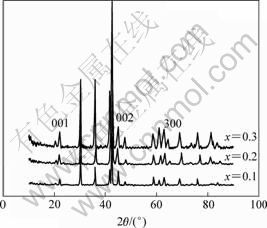
Fig.1 XRD patterns of La0.54Ce0.28Pr0.18Ni4-xCo0.6Mn0.35Alx alloy
Table 1 Lattice parameter and cell volume for La0.54Ce0.28Pr0.18Ni4-xCo0.6Mn0.35Alx alloy
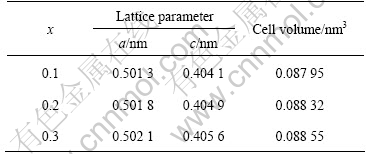
This is likely because that the radius of Al is larger than that of Ni.
3.2 Electrochemical properties
The typical detail capacity―potential curves at the third cycle of La0.54Ce0.28Pr0.18Ni4-xMn0.35Alx alloys at -40 ℃ and 25 ℃ during discharging processes are shown in Figs.2(a) and (b), respectively. It can be clearly seen that at room temperature (25 ℃) the discharge capacity of the alloy has no obvious difference when x value varies from 0.1 to 0.3. The maximum discharge capacity emerges at x = 0.1. But at low temperature (-40 ℃) the discharge capacity decreases from 251.5 mA?h/g to 140.4 mA?h/g when x value increases from 0.1 to 0.3, indicating an intensive influence of x value on the discharge capacity.
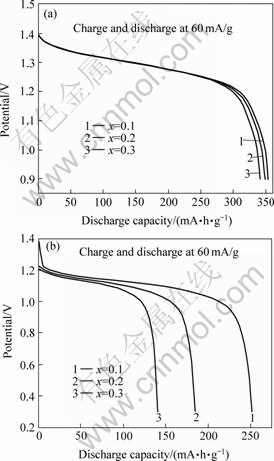
Fig.2 Discharge capacity―potential curves at 3rd cycle of La0.54Ce0.28Pr0.18Ni4-xCo0.6Mn0.35 Alx alloys at 25 ℃(a) and -40 ℃(b)
To elucidate this phenomenon, the chemical elemental characteristics of Ni and Al must be considered. As both the atomic radius and atom volume of Al are greater than those of Ni, increasing x value will inevitably cause the increase of the cell volumes, and therefore increase the interstitial space of the lattice cell, which will be favorable to the hydrogen-storage process. At the same time, the lattice distortions are also enlarged with increasing Al, and this will be detrimental to the hydrogen-storage ability of alloys. The comprehensive effects of the above two opposing factors generate the voltage-capacity dependence curves at 25 ℃ like those in Fig.2(a). Nevertheless, the discharge capacity of an alloy at low temperatures, especially lower than -20 ℃, is mainly controlled by dynamic processes of the electrode reaction[9, 11]. In this case, the smaller the cell volume, the higher the hydrogen-equilibrium pressure of the metal hydride, which would accelerate the diffusion rate of hydrogen in the metal matrix, and therefore favorite to discharge. Additionally, element Ni in the alloy can promote its electrocatalytic activation. Thus, the discharge capacity of alloy La0.54Ce0.28Pr0.18- Ni4-xCo0.6Mn0.35Alx at -40 ℃ decreases seriously with increasing x value. The effects of elements Al and Ni on the high rate performance of La0.54Ce0.28Pr0.18- Ni4-xCo0.6Mn0.35Alx are as the same as those on the discharge capacity at low temperature, and the results are shown in Fig.3.
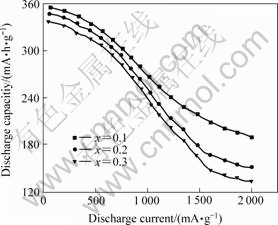
Fig.3 High-rate discharge ability of La0.54Ce0.28Pr0.18Ni4-x- Co0.6Mn0.35Alx alloys
Aluminum appears in all commercial AB5 alloys. It is believed that the incorporation of Al in the alloys substantially reduces the electrode corrosion and attributes this to the formation of protective surface oxides [12-13]. The presence of a even small amount of Al substantially decreases VH (the molar volume of hydrogen), n (the number of H atoms in the charged electrode) and, consequently, both the lattice expansion and corrosion[13]. These are beneficial to the cycling stability. The cycle life of La0.54Ce0.28Pr0.18Ni4-xCo0.6- Mn0.35Alx at 25 ℃ with different x values is shown in Fig.4. It can be seen that the cycle life increases with increasing x value.
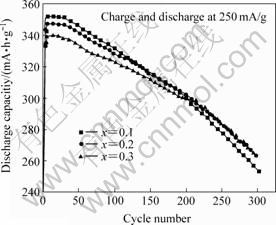
Fig.4 Cycle life of La0.54Ce0.28Pr0.18Ni4-xCo0.6Mn0.35Alx alloys
4 Conclusions
1) Both the lattice parameters and cell volume decrease with the decrease of Al content in La0.54Ce0.28Pr0.18Ni4-xCo0.6Mn0.35Alx (x=0.1, 0.2, 0.3).
2) The discharge capacity of La0.54Ce0.28Pr0.18Ni4-x- Co0.6Mn0.35Alx at 25 ℃ has no obvious difference with x value varying from 0.1 to 0.3, but at -40 ℃ it is intensively influenced by x value. When x value increases from 0.1 to 0.3, the discharge capacity of the alloys at -40 ℃ decreases from 251.5 mA?h/g to 140.4 mA?h/g.
3) The La0.54Ce0.28Pr0.18Ni3.9Co0.6Mn0.35Al0.1 alloy is capable of performing high-rate discharge and has excellent electrochemical properties as negative electrode materials used in Ni/MH battery at low temperature, even at -40 ℃.
References
[1] TLIHA M, MATHLOUTHI H, LAMLOUMIA J, PERCHERON-GUEGAN A. AB5-type hydrogen storage alloy used as anodic materials in Ni-MH batteries[J]. Journal of Alloys and Compounds, 2007, 436: 221-225.
[2] CUNSHENG W, MARIZA M R, DANIEL A, SERAFINI, JACK H, BARICUATRO, MANUEL P, SORIAGA, SUPRAMANIAM S. The self-discharge mechanism of AB5-type hydride electrodes in Ni/MH batteries[J]. International Journal of Hydrogen Energy,2006, 31(5): 603-611.
[3] YANG H B, FUKUNAGA H, OZAKI T, TANSE S, SAKAI T. Investigation on substrates of MmNi5-based alloy electrodes for high power applications[J]. Journal of Power Sources, 2004, 133: 286-292.
[4] SAKAI T, MIYAMURA H, KURIYAMA N, UEHARA, MUTA M, TAKAGI A, KAJIYAMA U, KINOSHITA K, ISOJAI F. Nickel-metal hydride battery for electric vehicles[J]. Journal of Alloys and Compounds, 1993, 192(1/2): 158-160.
[5] RONG Li, JIANMIN W, SHAOXIONG Z, JIUHONG Q. Effects of cobalt content and preparation on electrochemical capacity of AB5-type hydrogen storage alloys at different temperature[J]. Journal of Rare Earth, 2006, 24(3): 341-345.
[6] SAKAI T, MIYAMURA H, KURIYAMA N, KATO A, OGURO K, ISHIKAWA H. Metal hydride anodes for nickel-hydrogen secondary battery[J]. Journal of Electrochemical Society, 1990, 137(3): 795-799.
[7] XINBO Z, YUJUN C, WENYA Y, MINSHOU Z. Crystal structure and electrochemical properties of rare earth non-stoichiometric AB5-type alloy as negative electrode material in Ni-MH battery[J]. Journal of Solid State Chemistry, 2004, 177: 2373-2377.
[8] ZHONG Z, SHUMIN H, YUAN L, TIANFU J, XIAOTIE W. The effects of sintering temperature on the phase structure and electrochemical properties of AB5-5mass% LaMg3 composite alloy[J]. Journal of Alloys and Compounds 2006, 421: 289-293.
[9] IWAKURA C, SENOH H, MORIMOTO K, HARA Y, INOUE H. Influence of temperature on discharge process of mischmetal-based hydrogen storage alloy electrodes[J]. Electrochemistry, 2002, 70(1): 2-7.
[10] SENOH H, HARA Y, INOUE H, IWAKURA C. Charge efficiency of mischmetal-based hydrogen storage alloy electrodes at relatively low temperatures[J]. Electrochimica Acta, 2001, 46(7): 967-971.
[11] VALOEN L O, ZALUSKA A, ZALUSKI L, TANAKA H, KURIYAMA N, STROM-OLSEN J O, TUNLD R. Structure and related properties of (La, Ce, Nd, Pr)Ni5 alloys[J]. Journal of Alloys and Compounds, 2000, 306(1/2): 235-244.
[12] MARSHALL H M, DIETER M G. LaNi5-xAlx is a versatile alloy system for metal hydride applications[J]. Nature, 1977, 269(9): 45-47.
[13] REILLY J J, ADZIC G D, JOHNSON J R, VOGT T, MUKERJEE S, MCBREEN J. The correlation between composition and electrochemical properties of metal hydride electrodes[J]. Journal of Alloys and Compounds, 1999, 293/295: 569-582.
(Edited by HE Xue-feng)
Foundation item: Project (2001AA322050) supported by the National Hi-tech Research and Development Program of China
Corresponding author: DONG Gui-xia; Tel: +86-10-82241245; E-mail: dgxdgx01@163.com