
Performance of La1-xSrxCr1-yMnyO3-δ anode materials for intermediate temperature solid oxide fuel cell
CHEN Xiu-hua(陈秀华)1, MA Wen-hui(马文会)2, 3, YANG Bin(杨 斌)2, 3, DAI Yong-nian(戴永年)2, 3
1. Faculty of Physical Science and Technology, Yunnan University, Kunming 650091, China;
2. Faculty of Materials and Metallurgical Engineering, Kunming University of Science and Technology,Kunming 650093, China;
3. National Engineering Laboratory for Vacuum Metallurgy, Kunming 650093, China
Received 15 July 2007; accepted 10 September 2007
Abstract: La1-xSrxCr1-yMnyO3-δ(LSCM) anode materials were synthesized by glycine nitrate process(GNP). Thermo-gravimetric analysis(TGA) and differential scanning calorimetric(DSC) methods were adopted to investigate the reaction process of LSCM anode materials. The oxides prepared were characterized via X-ray diffraction(XRD), scanning electron microscope and energy dispersive spectroscopy(SEM-EDS), direct current four-electrode and temperature process reduction(TPR) techniques. XRD patterns indicate that perovskite phase created after the precursor was sintered at 1 000 ℃ for 5 h, and single perovskite-type oxides formed after the precursor were sintered at 1 200 ℃ for 5 h. The powders are micrometer size after sintering at 1 000 ℃ and 1 200 ℃, respectively. The conductivities of LSCM samples increase linearly with increasing the temperature from 250 ℃ to 850 ℃ in air and the maximum value is 32 S/cm for La0.7Sr0.3Cr0.5Mn0.5O3-δ. But it is lower about two orders of magnitude in pure hydrogen or methane than that of the same sample in the air. TPR result indicates that LSCM offers excellently catalytic performance.
Key words: La1-xSrxCr1-yMnyO3-δ; anode materials; glycine nitrate process; intermediate temperature solid oxide fuel cells; properties
1 Introduction
Solid oxide fuel cells(SOFCs) are thought to be one of the most promising power generation facilities in future for their distinguishing feature of highly efficiency, highly energy conversion rate in a wide range of fuels and pollution-free. However, high operating temperature (about 1 000 ℃) of the SOFC results in some problems, including thermal expansion mismatches between components, and sealing difficulty between cells with flat plate configurations. Besides the high operating temperature places severe constraints on materials selection, and results in difficulty for fabrication processes[1]. Therefore, intermediate temperature solid oxide fuel cells(ITSOFCs) that could operate at 500-850 ℃ should be developed to solve the problems mentioned above, and research on new materials attracted growing attentions as the important foundation of SOFC technology.
Anode material is one of the key components of solid oxide fuel cell(SOFC)[2]. The precious metals such as platinum and silver were used as anode material at first, but which have few applications now for some reasons such as high price and evaporation of silver[3-5]. Then, the cheaper nickel was applied to be anode materials, but the porous nickel anode materials become dense and the cell’s performance be deteriorated after a long operating at high temperature. Now, the nickel and electrolyte composite materials are often used as anode materials, although many problems such as the serious agglomeration of anode, the carbon deposition at anode when using fuel containing carbon and the poor tolerance of sulfur existed[3, 6].
LaCrO3-based La1-xSrxCr1-yMnyO3-δ(LSCM) has high electronic-ionic conductivity, excellent catalytic and reforming properties[7-8]. Maybe, the above problems such as carbon deposition existing in traditional anode can be resolved when LSCM is used as anode materials.
In this work, the synthesis of LSCM and its performance are studied to promote its application as anode for ITSOFCs.
2 Experimental
2.1 Preparation of materials
La1-xSrxCr1-yMnyO3-δ was prepared by using the La(NO3)3・6H2O, Sr(NO3)2, Cr(NO3)3 and Mn(NO3)2 with a designed molar ratio and distilled water. Glycine 1.5 times to the total molar amount of metal was then added to the above solution. The solutions were then mixed and stirred continuously and heated on an electric cooker until the solutions boiled and evaporated and left over precursor materials.
The calcined precursors were isostatically pressed into pellets (d13 mm×3 mm) under 120 MPa and sintered at 1 400 ℃ for 20 h. Silver electrodes were fabricated on both sides of the discs, respectively.
2.2 Crystal structure characterization and property measurement
Thermogravimetric analysis(TGA) and differential scanning calorimetric(DSC) method were adopted to confirm the synthesis temperature. Crystal structure of the powder was confirmed via X-ray diffraction(XRD). The size of synthesized materials was observed via scanning electron microscopy(SEM). The elements and their percentages in sintered material were characterized via energy dispersive spectroscopy(EDS). The four- probe direct current technique was adopted to measure the total conductivity (ionic and electronic) of the electrode pellets among 250 ℃ and 850 ℃ in air, hydrogen and methane, respectively. Temperature process reducing(TPR) experiment using La0.7Sr0.3Cr0.5- Mn0.5O3-δ(LSCM7355) as typical sample was performed in order to explore the relationship between catalytic performance and atmosphere.
3 Results and discussion
3.1 TGA- DSC analysis
The TGA-DSC curves of the precursors indicate oxides with pervoskite type structure created after 941 ℃. The synthesis process has been discussed in detail in Ref.[9].
3.2 XRD characterization and EDS pattern analysis
Fig.1 shows the XRD patterns of LSCM7355 powder sintered at 1 000℃ and 1 200 ℃ respectively in air. There are two phases including perovskite structure and SrCrO4 sintered at 1 000 ℃, which indicates that the perovskite phase created after the precursor sintered at 1 000 ℃ for 5 h, together with a little secondary phase
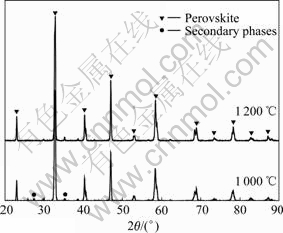
Fig.1 XRD patterns of LSCM7355 powder
SrCrO4. There is only one phase of single-phased perovskite-type oxide sintered at 1 200 ℃, which shows that the single-phased perovskite-type oxide has been obtained after the precursor having been sintered at 1 200 ℃ for 5 h. The peak intensity of sample sintered at 1 200 ℃ is stronger than that of it sintered at 1 000 ℃. This illustrates that the crystallization grain has grown with increasing temperature from 1 000 ℃ to 1 200 ℃. Comparing with the solid state method[10], the minimum sintering temperature and time are both reduced. The feasible kinetic condition can be obtained for the synthesizing of LSCM7355 perovskite powder phase by glycine nitrate process.
According to EDS pattern of LSCM7355 material by glycine nitrate process (GNP) sintered at 1 200 ℃ for 5 h, it can be seen that there is no impurity element other than La, Sr, Cr, Mn and O. The molar ratio of La, Sr, Cr, Mn and O is about 0.7?0.29?0.53?0.45?2.18, which are in good agreement with the designed value, except a little oxygen shortage because of detection. It is also suggested that the oxygen vacancies exist in the oxides.
3.3 SEM images observation
Fig.2 shows the SEM images of the LSCM7355 oxide ((a) and (b) corresponding to the powders sintered at 1 000 ℃ and 1 200 ℃ for 5 h, respectively, and corresponding to the pellets sintered at 1 400 ℃for 20 h). LSCM7355 powders offered nanometer size after sintering at 1 000 ℃ for 5 h. The powder size becomes bigger and is about 1 ?m after sintering at 1 200 ℃ for 5 h. These small size particles are very active, and it is beneficial for decreasing the fabrication temperature of the anode film and enhancing the catalytic properties. When the LSCM7355 pellet is sintered at 1 400 ℃ for 20 h, the most grains increase to 1.5-2 ?m with octahedron shape.

Fig.2 SEM images of LSCM7355 sintered at 1 000 ℃ (a), 1 200 ℃ (b) and 1 400 ℃ (c) by GNP
3.4 Conductivity properties analysis
Fig.3 shows the conductivity of LSCM anode materials at different temperature conditions in the air. It can be seen that the transformation trend of total conductivities in LSCM samples increases linearly with increasing the temperature from low temperature to high temperature, corresponding to the small proton principle. The reason is that the energy of small proton contributing on electric effect increases with increasing the temperature, along with the increasing of the movement speed and the ability of transport electric charges. Fig.4 shows the conductivity of LSCM anode materials at different atmosphere conditions. The maximum value is 32 S/cm for La0.7Sr0.3Cr0.5Mn0.5O3-δ from 250 ℃ to 850 ℃ in the air, which decreases about two orders of magnitude when the atmosphere changes from air to pure
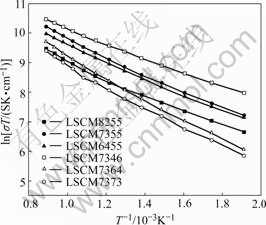
Fig.3 Conductivity lnσT vs 1/T of LSCM in air
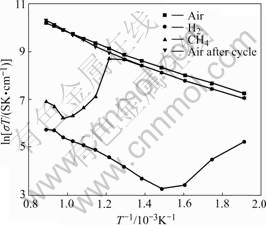
Fig.4 Conductivity of LSCM7355 in different atmospheres
hydrogen and methane. The conductivity of LSCM7355 becomes smaller when the atmosphere changes from air to pure hydrogen and methane at the same temperature. The reason may be that the high-valence ionic such as Cr4+or Mn4+ is reduced to the low-valence one in reductive atmosphere, then the quantity of the small polaron becomes smaller and its contribution to conductivity decreases. The conductivity of LSCM7355 in methane is bigger than that of in hydrogen for its weaker reductive property. The conductivity of LSCM7355 sample in air returns to the initiate value after the cycle, including the excellent reversibility. The transformation law of the conductivity in LSCM7355 sample in methane is some complex. The conductivity of LSCM7355 sample in methane is the same as that of in the air before 400 ℃ due to the reductive degree of high-valence ion is little and the amount of small proton changes small. And it decreases rapidly after 400 ℃ due to the reductive degree of high-valence ion increases rapidly and the amount of small proton decreases. And it increases linearly with the temperature after 600 ℃ due to the reductive reaction of methane to LSCM7355 tends to equilibrium and the amount of small proton does not change anymore. The change of conductivity is only relative to the temperature.
Fig.5 shows the temperature process reduction(TPR) result of LSCM7355 in hydrogen. It can be seen that the consumption rate of hydrogen is very slow beginning from 200 ℃, corresponding to the absorbed oxygen in the LSCM powder surface. Maximum consumption of hydrogen emerges peak point at about 500 ℃, corresponding to the reductive of high-valence metal ion. So, the LSCM7355 has reduced completely at about 400 ℃, and the conductivity of LSCM7355 increases from this temperature again, corresponding to the small proton principle.
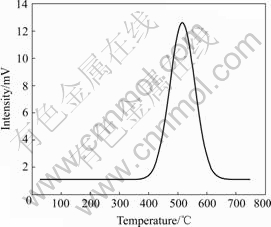
Fig.5 H2-TPR profile of LSCM7355 materials
4 Conclusions
1) A single-phase perovskite is identified after sintering at 1 200 ℃ for 5 h. The molar ratio of the elements in LSCM7355 material as typical sample agrees well with the designed value.
2) LSCM7355 powder offers nanometer size after sintering at 1 000 ℃ for 5 h. The powder size becomes bigger and about 1 ?m after sintering at 1 200 ℃ for 5 h. When the LSCM7355 pellet is sintered at 1 400 ℃ for 20 h, the most crystal grains with octahedron shape are 1.5-2 ?m in size.
3) The total conductivity of LSCM7355 increases linearly with increasing the temperature in air at the range from 250 ℃ to 850 ℃ and the maximum value is 32 S/cm which becomes smaller when the atmosphere being changed from air to pure hydrogen. The conductivity of LSCM7355 in methane is larger than that of in hydrogen for its weaker reducing property and it shows excellent reversibility.
References
[1] EG & G Technical Services Inc. Fuel cell handbook [M]. (7th ed). West Virginia: U.S. Department of Energy Office of Fossil Energy & National Energy Technology Laboratory, 2004: 7-5.
[2] YASHIRO K, TAKEDA K. In situ observation of deposited carbon on anode for solid oxide fuel cell [C]// SINGHAL S C, DOKIYA M, ed. Proceedings of the Eighth International Symposium on Solid Oxide Fuel Cells (SOFCVIII). Paris: The Electrochemical Society, 2003: 714-719.
[3] GORTE R J, VOHS J M. Novel SOFC anodes for the direct electrochemical oxidation of hydrocarbons [J]. Journal of Catalysis, 2003, 216: 477-486.
[4] HUANG Xian-liang, ZHAO Hai-lei, WU Wei-jiang, QIU Wei-hua. Recent process of anode materials for solid oxide fuel cells [J]. Journal of the Chinese Ceramic Society, 2005, 23(11): 1407-1413. (in Chinese)
[5] MINH N Q. Ceramic fuel cells [J]. J Am Ceram Soc, 1993, 76: 563-588.
[6] KOH J H, YOO Y S, PARK J W. Carbon deposition and cell performance of Ni-YSZ anode support SOFC with methane fuel [J]. Solid State Ionics, 2002, 149: 157-166.
[7] ZHA S W, TSANG P, CHENG Z. Electrical properties and sulfur tolerance of La0.75Sr0.25Cr1-xMnxO3 under anodic conditions [J]. Journal of Solid State Chemistry, 2005, 178: 1844-1850.
[8] PLINT S M, CONNOR P A, TAO S. Electronic transport in the novel SOFC anode material La1-xSrxCr0.5Mn0.5O3±δ [J]. Solid State Ionics, 2006, 177: 2005-2008.
[9] LIU Rong-hui, MA Wen-hui, WANG Hua, YANG Bin, MA Xue-ju, DAI Yong-nian. Synthesis and properties of LSCM anode materials by GNP [J]. Acta Scientiarum Naturalium Universitatis Sunyatseni, 2007, 46(suppl.): 293-295.
[10] MA Xue-ju, MA Wen-hui, DAI Yong-nian, YANG Bin, LIU Rong-hui. Preparation and characterization of LSCM anode material for SOFC by solid state reaction [J]. Acta Scientiarum Naturalium Universitatis Sunyatseni, 2007, 46(suppl.): 300-302.
(Edited by LAI Hai-hui)
Foundation item: Project(50204007) supported by the National Natural Science Foundation of China; Project(2005PY01-33) supported by the Talent Foundation of Yunnan Province, China
Corresponding author: MA Wen-hui; Tel: +86-871-5161583; E-mail: mwhui@kmust.edu.cn