Bi��Sr����Al-7Si-0.4Mg�Ͻ�����֯��Ӱ��
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2011���7��
�������ߣ�S. FARAHANY A. OURDJINI M. H. IDRIS L. T. THAI
����ҳ�룺1455 - 1464
�ؼ��ʣ�Al-Si �Ͻ�; ��; ��; ����; ��������
Key words��Al-Si alloy; bismuth; strontium; modification; eutectic solidification
ժ Ҫ���о���������Bi����������Bi��Sr�ڲ�ͬ���������¶�Al-7Si-0.4Mg�Ͻ��й�����ṹ��Ӱ�졣�������������Bi�Թ�������ϸ�����ã���ϸ����������Bi��������(��<0.5%��Χ��)�����Ӷ���ǿ����Bi���ӵ�Sr���ԵĺϽ���ʱ, Ϊ�˵õ���ֵĹ�������ԣ��ϸߵ�Sr/Bi������(����0.45)�DZ�Ҫ�ġ�
Abstract:
The effects of bismuth and the combination of bismuth and strontium on the eutectic silicon structure in Al-7Si-0.4Mg alloys were investigated under different solidification conditions. The results show that bismuth has a refining effect on the eutectic silicon and its refinement behavior increases with increasing Bi content up to 0.5% (mass fraction). When bismuth is added into the molten alloy modified with strontium, a higher Sr/Bi mass ratio of at least 0.45 is required to attain full modification of the eutectic silicon.

S. FARAHANY, A. OURDJINI, M. H. IDRIS, L. T. THAI
Department of Materials Engineering, Faculty of Mechanical Engineering,
Universiti Teknologi Malaysia, 81310, Malaysia
Received 27 August 2010; accepted 29 November 2010
Abstract: The effects of bismuth and the combination of bismuth and strontium on the eutectic silicon structure in Al-7Si-0.4Mg alloys were investigated under different solidification conditions. The results show that bismuth has a refining effect on the eutectic silicon and its refinement behavior increases with increasing Bi content up to 0.5% (mass fraction). When bismuth is added into the molten alloy modified with strontium, a higher Sr/Bi mass ratio of at least 0.45 is required to attain full modification of the eutectic silicon.
Key words: Al-Si alloy; bismuth; strontium; modification; eutectic solidification
1 Introduction
Melt modification treatment in Al-Si cast alloys leads to a change in the morphology of eutectic silicon from large brittle flake or acicular to fine ?brous morphology, resulting in improved mechanical properties [1-3]. Chemical or impurity modification can be achieved by the addition of certain elements to the melt prior to solidification [1, 4]. These added elements can be divided into two major categories: 1) elements such as Na, Sr, Ca, Ba and Cs that show marked effect on the eutectic silicon when added to molten Al-Si alloys by altering its morphology and producing fibrous eutectic silicon structure; 2) elements such as Sb,Y, Yb and Cd that are not capable of producing dramatic changes in the silicon morphology but only refine it into lamellar structure [5-7].
Since bismuth is in group V in the periodic table, it can have a similar effect on the silicon morphology to antimony. However, there is very limited research on the effect of individual bismuth on the microstructure of Al-Si cast alloys. According to Ref. [8], bismuth could be used as an effective modifier in the range of 0.2%-0.25% (mass fraction). On the other hand, MACHOVEC et al [9] reported that with a Sr/Bi mass ratio above 0.45, the Si structure was found to be modified into fine fibres in 319 Al alloy. CHO and LOPER [10] also reported that with the addition of 0.1% Bi in the melt containing Sr, the modification effect of Sr was completely eliminated in A356 alloy. In previous work by EL-HADAD et al [11] the modification effect of strontium was found to diminish continuously with addition up to 0.225% Bi for the 319 alloy, whilst adding 0.5% Bi to the modified and grain-refined Al-10.8% Si alloy led to a noticeable coarsening of the eutectic Si particles [12].
Bismuth is usually added to improve the machinability of aluminium wrought alloys [13]. However, with increasing the awareness of environmental issues, recycling of metal alloys, in particular aluminium alloys, has been increased in recent times. Since the recycled aluminium alloys may contain impurity elements such as Bi, a better understanding of the interaction between Bi and Sr may be necessary. The aim of this work is to present experimental results of an investigation on the influence of Bi addition on the microstructure of unmodified and strontium-modified Al-7Si-0.4Mg alloy.
2 Experimental
2.1 Melt preparation
A commercial Al-Si-0.4Mg ingot with the chemical composition listed in Table 1 was used. The melt was prepared by charging pieces of the alloy into a silicon carbide crucible using an electrical resistance furnace under argon. The melt temperature was maintained at 750-760 ��C for a time long enough to allow for complete melt homogenization. The molten alloy was then poured at the temperature of (730��5) ��C into two different molds: sodium silicate bonded CO2 sand mold and permanent metal mold, so as to produce bars solidified with two different cooling rates (Fig. 1).
Table 1 Chemical composition of base alloy (mass fraction, %)
![]()
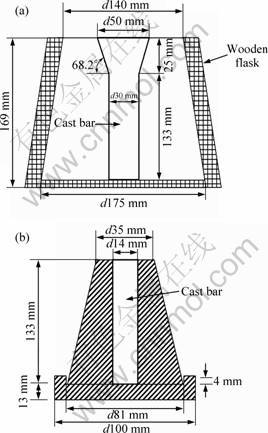
Fig. 1 Schematic of CO2 sand mold (a) and cone-shaped permanent mold (b) with their corresponding dimensions
Upon thermal equilibrium, Bi in the form of metallic shots and Sr as an Al-10Sr master alloy were wrapped in aluminium foil and introduced into at different concentrations according to Table 2. Both melts that were treated with Bi alone and by combination of Bi and Sr were stirred twice for about 30 s each time to allow for complete homogenization. Prior to molten metal pouring the surface of the melt was skimmed to remove the oxide layer. In order to investigate the effect of Bi on the level of Sr modification, the concentrations of both Bi and Sr were adjusted to obtain different Sr/Bi mass ratios. Table 2 lists the chemical analyses of all samples obtained from chemical analysis using glow discharge spectrometer (LECO GDS-850A).
Table 2 Chemical compositions of samples

2.2 Thermal analysis
Thermal analysis was performed by attaching a highly sensitive K-type thermocouple located in the middle of each cast bar. The temperature-time data were obtained using Dewetron high speed data acquisition system linked to a computer at a rate of 20 records per second. Flexpro.8 software was used to plot the cooling and first derivative curves in order to determine the cooling rate, nucleation temperature TN, minimum (Tmin) and growth temperature TG of each transformation during melt solidification. The cooling rates obtained from the curves, prior to nucleation of primary Al, were found to be 0.8 ��C/s and 4 ��C/s in sodium silicate bonded CO2 sand mold and permanent metal mold, respectively. A typical cooling and first derivative curves of Al-7Si-0.4Mg alloy based on thermal analysis for sodium silicate bonded CO2 sand mold is illustrated in Fig. 2.

Fig. 2 Cooling curve and first derivative curve of Al-7Si-0.4Mg alloy (Tc means temperature of cooling curve; dTc/dt refers to first derivative curve of cooling curves)
2.3 Sample characterization
Specimens from each casting were taken out for microstructural examination. Those specimens were sectioned from the centre of each casting bar, mounted and ground with abrasive silicon carbide papers. Those ground specimens were then subjected to a final polishing with silica suspension (0.5 ��m). The microstructures were analyzed and their images were captured with an optical microscope at various magnifications. Selected specimens were deep etched using a solution of 10% hydrochloric acid and 90% ethanol to reveal the 3D morphology of the eutectic silicon phase, which were analyzed using field emission scanning electron microscope (Supra-35VP, Carl Zeiss, Germany).
3 Results and discussion
3.1 Effect of bismuth
3.1.1 Eutectic silicon morphology
Very limited work on the effect of individual Bi on the Si eutectic phase in Al-Si cast alloys has been made over the years. However, since some effect of poisoning was reported when Bi was added to Sr-containing alloys [9, 11-12], it is probable that Bi present in unmodified Al-Si molten alloys may induce some effect on the morphology and/or size of the Si phase. In an attempt to investigate this possibility, Bi was added with increasing concentration to determine whether there is any degree of modification, refinement or coarsening of silicon flakes during solidification.
For specimens solidified under slow cooling rate conditions, there was a significant change in the eutectic silicon size when the mass fraction of Bi was increased to 0.5%, as shown in Fig. 3. A coarse plate-like silicon and large amount of angular silicon can be observed within the eutectic region of untreated alloy in Fig. 3(a). The microstructure of treated alloy with 0.1% Bi (Fig. 3(b)) is similar to that of the untreated alloy, showing that the structure of silicon still remained plate-like. With increasing the mass fraction of Bi to 0.3%, the size of silicon decreased, as shown in Fig. 3(c). When the mass fraction of Bi reached 0.5% in the melt, a refinement of Si flakes can be observed (Fig. 3(d)), resulting in the formation of a lamellar structure. There is also evidence that the interflake spacing decreased with increasing the Bi content, an indication of refinement.

Fig. 3 Optical microstructures of slow-cooled specimens without Bi (a), with 0.1% Bi (b), 0.3% Bi (c) and 0.5% Bi (d)
In order to obtain more conclusive results, the cross-sections of specimens were subjected to deep etching to remove the aluminium phase for revealing the 3D morphology of the Si phase. Indeed, the SEM micrographs shown in Fig. 4 confirm that the eutectic Si changed from coarse plate morphology (Fig. 4(a)) in the unmodified state into a refined structure with increasing Bi addition (Fig. 4(d)). From those fast-cooled specimens with different Bi contents, similar results were obtained, as shown in Fig. 5. In addition, it must be noted that the silicon eutectic is finer compared with those slowly cooled specimens. This fact is attributed to the increased solidification rate which intensifies the refinement of the structure. As can be seen from Figs. 5(a) and (c), the silicon eutectic morphology changed into very refined structure with 0.5% Bi content compared to the untreated sample. This was confirmed by the SEM micrographs shown in Fig. 6, which refined silicon structure when Bi mass fraction increases to 0.5%.

Fig. 4 SEM micrographs of slow-cooled specimens without Bi (a), with 0.1% Bi (b), 0.3% Bi (c) and 0.5% Bi (d)

Fig. 5 Optical microstructures of fast- cooled specimens without Bi (a), with 0.1% Bi (b) and 0.5% Bi (c)

Fig. 6 SEM micrographs of fast-cooled specimens with 0.5% Bi in low (a) and high magnification (b)
Both slow and fast-cooled specimens show similar behaviours of Bi with its tendency to refine the silicon eutectic, which is clearly observable at 0.5% Bi addition. Similar to other modifiers and refiners, the growth of silicon is most possibly affected. The refining ability of Bi does not come as a surprise since Bi has been found to reduce the surface tension of aluminium alloys according KURDYUMOV and INKIN [14] and PAPWORTH and FOX [14-15]. By reducing the liquid surface tension within the eutectic phase, which is a lamellar structure of Al and Si, Bi can reduce the contact angle between Al and Si, resulting in the aluminium being able to wet the silicon phase more easily and encapsulate the silicon growth front, thus limiting its growth. Since Bi and Sb are in Group V in the periodic table, they may share similar chemical properties.
Thus, it is reasonable that Bi refines the silicon particles in ways similar to Sb refinement, a fact in agreement with CHO and LOPER [10]. Therefore, it can be deduced that Bi does not modify the eutectic structure in the same way as Na and Sr do, but rather only refines the silicon structure, much more like the refinement effect from Sb with increase in mechanical properties of Al-Si alloys [16] compared with Sr-modified alloys [17]. Since Bi/Si radius ratio is 1.39, it could hardly fulfill the size factor requirement of the impurity-induced twinning mechanism suggested by LU and HELLAWELL [1]. The silicon structure seen in Fig. 4(d) could have been due to refinement rather than modification with the addition of 0.5% Bi.
3.1.2 Thermal analysis
Figure 7 shows the cooling curves of the slow- cooled untreated and Bi-treated alloys with Bi addition of 0.05%, 0.1%, 0.3% and 0.5%. The first derivative curve (dTC/dt) was also plotted in addition to cooling curve to be used as an indicator of phase transformation during solidification, as shown in Fig. 2. The difference between TG and Tmin describes the recalescence of eutectic transformation. Growth temperature is defined as the maximum reaction temperature after recalescence. The eutectic growth temperature and recalescence were measured from the characteristic temperature of cooling curves for each sample. It is well-established that modification of the Al-Si eutectic is accompanied by a depression in the TG [18-19]. Depression of TG was suggested to reveal modification automatically without microstructure analysis [7, 20].

Fig. 7 Cooling curves of slowly cooled and Bi-treated Al-7Si-0.4Mg alloys with different Bi additions
The eutectic growth temperature of the unmodi?ed alloy was measured to be 574.1 ��C, whereas by adding 0.05% Bi the TG decreased slightly to 573.6 ��C. The TG decreased to the lowest eutectic growth temperature of 572.4 ��C when the mass fraction of Bi increased to 0.5%. It is seen that the eutectic growth temperature decreased by 1.7 ��C, which can be related to the refinement of silicon structure, as shown in Fig. 4(d) and Fig. 6. The variation of eutectic growth temperature as a function of Bi concentration is shown in Fig. 8(a). It is clear that with increasing Bi content the TG decreased continuously. Furthermore, increasing Bi content in the molten alloy led to considerable recalescence (TG-Tmin) prior to eutectic growth, while negligible recalescence was observed in the untreated alloy (~0.2 ��C), as shown in Fig. 8(b). Recalescence is an indication of nucleation effects, while a reduced growth temperature indicates an effect on growth [7] .
On the other hand, bismuth not only affects the growth but also has an impact on the nucleation of the eutectic silicon.
3.2 Interaction between bismuth and strontium
3.2.1 Eutectic silicon morphology
In order to investigate the effect of Bi-Sr interaction on the eutectic silicon, the Bi content was fixed at 0.015% and Sr content gradually increased from 0.001% to 0.031%. This produced specimens with a range of Sr/Bi mass ratio from 0.09 to 2.07. The microstructures of alloys with different Sr/Bi mass ratios for slowly cooled bars are shown in Fig. 9. It is clear that at a low Sr/Bi mass ratio (i.e. 0.20 and less), the eutectic silicon remains in a rather unmodified state consisting of flakes or acicular morphology. As the mass ratio increases, however, the eutectic silicon gradually changes from partially modified (at m(Sr)/m(Bi) of 0.27) into modified fibrous silicon structure at the m(Sr)/m(Bi) of 0.60. Specimens with Sr/Bi mass ratios over 0.60 present fully modified silicon structure. Figure 10(a) shows the unmodified eutectic silicon while Figs.10(b) and (c) show fully modified eutectic silicon at higher magnifications.

Fig. 8 Eutectic growth temperature (a) and recalescence (b) as function of Bi content

Fig. 9 Optical microstructures of slow-cooled specimens with Sr/Br mass ratios of 0.10 (a), 0.20 (b), 0.27 (c), 0.60 (d) and 1.33 (e)
Several sand-cast (slow-cooled) specimens with different Sr/Bi mass ratios were also prepared in order to better understand the possible interaction between Bi and Sr. These specimens were SrBi17aS, SrBi17bS, SrBi5S and SrBi8S, with the mass ratios of 0.15, 0.16, 0.45 and 0.72, respectively. The letter ��S�� at the end of each sample label indicates slow-cooled sample. Figure 11 clearly shows that all specimens, except SrBi17aS, attained a modified silicon structure. For SrBi17bS, it is interesting to note that the eutectic silicon is modified even though the Sr/Bi mass ratio is less than 0.20 and is almost the same as the ratio in SrBi17aS with unmodified structure. In SrBi17bS, the Bi content is considerably higher than that of the rest of the specimens (exceeding 0.2%). However, at the same time, the Sr addition is the highest (i.e. 0.04%). This may indicate that at high strontium level, a wide range of bismuth concentration could be tolerated while still attaining a modified state.

Fig. 10 Optical microstructures of slow- cooled specimens with Sr/Bi mass ratios of 0.2 (a), 0.60 (b), and 1.33 (c) at higher magnification

Fig. 11 Optical microstructures of slow-cooled specimens with higher Sr and Bi concentrations but at different Sr/Bi mass ratios of 0.15 (SrBi17aS) (a), 0.16 (SrBi17bS) (b), 0.45 (SRBi5S) (c) and 0.72 (SRBi8S) (d)
As noticed in this experiment, 0.037% Sr could still impose its modifying ability with Bi mass fraction up to 0.235%, while for 0.018% Sr, 0.04% Bi does not counteract the Sr modification. The eutectic silicon structures of SrBi2S, SrBi5S and SrBi1.33S can be confirmed through SEM analyses shown in Fig. 12. The interaction between Bi and Sr in Al-7Si-0.4Mg can be represented in Fig. 13.
The microstructures of fast-cooled specimens with the same Sr/Bi mass ratios and compositions show similar trend with those of the sand-cast specimens. As shown in Figs. 14(a) and (b) under fast cooling, specimen with Sr/Bi mass ratio as low as 0.15 was found to exhibit a partially modified silicon structure, whereas under slow cooling condition, partial modification could be observed only at a mass ratio of 0.20. Specimens with higher Sr/Bi mass ratios (0.6 and above) clearly show the condition where silicon modification is attained. Thus, the diminishing effect of modification is more significant at slower cooling rates as the Sr/Bi mass ratio decreases. This may be due to the fast solidification effect that could have quench-modified the eutectic silicon.

Fig. 12 SEM micrographs of slow-cooled specimens with Sr/Bi mass ratios of 0.20 (a), 0.45 (b), and 1.33 (c)

Fig. 13 Effect of Bi and Sr interaction on modification of slow- cooled specimens
3.2.2 Thermal analysis
It is evident from Fig. 15 that, below Sr/Bi mass ratio of 0.2, the eutectic growth temperature (TG) decreases slightly compared with the untreated alloy which is constant around 573.2 ��C. By increasing the Sr addition and also Sr/Bi mass ratio, TG decreases continuously to 568.7 ��C for sample with m(Sr)/m(Bi)= 0.6. The results show that the presence of bismuth, even as low as 0.015%, could interfere with the modification effect of strontium. This effect is more obvious at slow cooling rates. It was reported that Sr modification effect was eliminated with 0.1% Bi [10] and 0.225% Bi addition [11]. However, MOHAMED et al [12] did not identify bismuth intermetallic compounds, but suggested there was a possibility that binary Bi3Sr, BiSr and Bi2Sr3 or ternary Bi-Mg-Sr compounds might form before eutectic nucleation, leading to a reduction in the amount of free strontium available for modification.
The present results seem to imply that there may be some interaction between bismuth and strontium, as it requires higher strontium concentration to exert complete silicon eutectic modification when bismuth is present in the melt. Modification could be attained only when more strontium is added, preferably with Sr/Bi mass ratio of more than 0.45 which reconciles well with the previous findings for 319 type alloy [9].

Fig. 14 Optical microstructures of fast- cooled specimens with Sr/Bi mass ratios of 0.1 (a), 0.15 (b), 0.27 (c), 0.60 (d) and 0.72 (e)

Fig. 15 Cooling curves of eutectic growth for untreated, Sr-modified with 0.004% and different Sr/Bi mass ratios (The cooling curves have been displaced along time axis for clear presentation)
4 Conclusions
1) At both slow and fast cooling rates, bismuth refines plate-like eutectic silicon in Al-7Si-0.4Mg alloys. The refinement efficiency of Bi increases continuously with increasing the addition of Bi up to 0.5%.
2) The refinement behavior of Bi causes a continuous decrease in the eutectic growth temperature and also an increase in the recalescence with increasing Bi addition up to 0.5%.
3) In order to retain full modification, Sr/Bi mass ratio higher than 0.45 is required when bismuth is present in the melt.
Acknowledgements
The authors would like to acknowledge Universiti Teknologi Malaysia for providing research facilities and the Ministry of Science and Technology of Malaysia for financial support under the vote 79352.
References
[1] LU S Z, HELLAWELL A. The mechanism of silicon modification in aluminum-silicon alloys: Impurity induced twinning [J]. Metall Trans A, 1987, 18: 1721-1733.
[2] DAHLE A K, NOGITA K, McDONALD S D, DINNIS C, LU L. Eutectic modi?cation and microstructure development in Al-Si alloys [J]. Mater Sci Eng A, 2005, 413-414: 243-248.
[3] NOGITA K, YASUDA H, YOSHIYA M, McDONALD S D, UESUGI K, TAKEUCHI A, SUZUKI Y. The role of trace element segregation in the eutectic modi?cation of hypoeutectic Al-Si alloys [J]. J Alloys Compd, 2010, 489: 415-420.
[4] LU L, DAHLE A K. Effects of combined additions of Sr and AlTiB grain refiners in hypoeutectic Al-Si foundry alloys [J]. Mater Sci Eng A, 2006, 435-436: 288-296.
[5] LU L, NOGITA K, McDONALD S D, DAHLE A K. Eutectic solidification and its role in casting porosity formation [J]. JOM, 2004, 56(11): 52-58.
[6] ZHANG X H, SU G C, JU C W, WANG W C, YAN W L. Effect of modification treatment on the microstructure and mechanical properties of Al-0.35%Mg-7.0%Si cast alloy [J]. Mater Des, 2010, 31(9): 4408-4413.
[7] KNUUTINEN A, NOGITA K, McDONALD S D, DAHLE A K. Modi?cation of Al-Si alloys with Ba, Ca, Y and Yb [J]. Light Met, 2001, 1: 229-240.
[8] PILLAI N P, ANANTHARAMAN T R. Elements of V group as modifiers of aluminium-silicon alloys [J]. Trans Metall Soc AIME, 1968, 24(2): 2025-2027.
[9] MACHOVEC C J, BYCZYNSKI G E, ZINDEL J W, GODLEWSKI L A. Effect of Bi-Sr interactions on Si morphology in a 319-type aluminum alloy [J]. AFS Trans, 2000, 108: 439-444.
[10] CHO J I, LOPER C R. Limitation of bismuth residual in A356.2 Al [J]. AFS Trans, 2000, 108: 359-367.
[11] EL-HADAD, SAMUEL A M, SAMUEL F H, DOTY H W, VALTIERRA S. Effects of Bi and Ca additions on the microstructure of Sr-modified 319 type alloys under variable cooling conditions [J]. AFS Trans, 2004, 112: 141-153.
[12] MOHAMED, SAMUEL F H, SAMUEL A M, DOTY H M. Effects of individual and combined additions of Pb, Bi, and Sn on the microstructure and mechanical properties of Al-10.8Si-2.25Cu- 0.3Mg alloy [J]. Metall Mater Trans A, 2009, 40: 240-254.
[13] ZANDER J, SANDSTR?M R. Modelling technological properties of commercial wrought aluminium alloys [J]. Mater Des, 2009, 30: 3752-3759.
[14] KURDYUMOV A V, INKIN S V. Influence of bismuth and antimony on the structure and surface tension of alloy AL2 [J]. Liteinoe Proizvodstvo, 1986, 6: 28-29.
[15] PAPWORTH A, FOX P. The disruption of oxide defects within aluminium alloy castings by the addition of bismuth [J]. Mater Lett, 1998, 15: 202-206.
[16] PAN E N, CHERNG Y C, LIN C A, CHIOU H S. Roles of Sr and Sb on silicon modification of A356 aluminium alloys [J]. AFS Trans, 1994, 102: 609-629.
[17] SHABESTARI S G, SHAHRI F. Influence of modi?cation, solidification conditions and heat treatment on the microstructure and mechanical properties of A356 aluminum alloy [J]. Mater Sci, 2004, 39: 2023-2032.
[18] GRUZLESKI J E, CLOSSET B M. The treatment of liquid aluminium-silicon alloys [M]. Des Plains: Americans Foundrymen��s Society Inc, 1990: IL 95-157.
[19] BACKUERUD L, CHAI G, TAMMINEN J. Solidification characteristics of aluminum alloys [M]. Vol. 2. Stockholm: Foundry Alloys, AFS/ Skanaluminium, 1990.
[20] DJURDJEVIC M, JIANG H, SOKOLOWSKI J. On-line prediction of aluminum�Csilicon eutectic modi?cation level using thermal analysis [J]. Materi Charact, 2001, 46: 31-38.
S. FARAHANY, A. OURDJINI, M. H. IDRIS, L. T. THAI
Department of Materials Engineering, Faculty of Mechanical Engineering,
Universiti Teknologi Malaysia, 81310, Malaysia
ժ Ҫ���о���������Bi����������Bi��Sr�ڲ�ͬ���������¶�Al-7Si-0.4Mg�Ͻ��й�����ṹ��Ӱ�졣�������������Bi�Թ�������ϸ�����ã���ϸ����������Bi��������(��<0.5%��Χ��)�����Ӷ���ǿ����Bi���ӵ�Sr���ԵĺϽ���ʱ, Ϊ�˵õ���ֵĹ�������ԣ��ϸߵ�Sr/Bi������(����0.45)�DZ�Ҫ�ġ�
�ؼ��ʣ�Al-Si �Ͻ�; ��; ��; ����; ��������
(Edited by YANG Hua)
Corresponding author: S. FARAHANY; Tel: +60127686357; E-mail: fsaeed2@live.utm.my; saeedfarahany@gmail.com
DOI: 10.1016/S1003-6326(11)60881-9

