
Preparation of nickel nanoparticles in emulsion
ZHANG You-xian(张有贤)1, FU Wen-jie(付文杰)1, AN Xue-qin(安学勤)1, 2
1. College of Resources and Environment, Lanzhou University, Lanzhou 730000, China;
2. School of Chemistry and Environment Science, Nanjing Normal University, Nanjing 210097, China
Received 2 March 2007; accepted 8 October 2007
Abstract: The nickel nanoparticles with different sizes and spherical shape were prepared by the reduction of nickel sulfate with sodium borohydride in the water-in-oil emulsions of water/SDBS(sodium dodecylbenzene sulfonate)/n-pentanol/n-heptane. The effects of aging time, molar ratio of water to SDBS(R) and the concentration of nickel sulfate on the size of particles were studied. The samples were characterized by transmission electron microscopy(TEM) and inductively coupled plasma spectrometry(ICP). The results show that the average particle size changes from 20 to 40 nm by adjusting aging time (15-30 min) and R (9-11.5). The concentration of nickel sulfate of 1.0 mol/L is the favorite condition.
Key words: W/O emulsions; SDBS; nickel nanoparticles
1 Introduction
In recent years, the preparation of metal nanoparticles has received increasing attention from many researchers. As a kind of nanosized metallic materials, nano-nickel materials exhibit unusual electronic, magnetic, and chemical properties significantly different from the bulk materials due to their extremely small sizes and large specific surface areas. They have various potential applications, such as battery materials, catalyzer, magnetic materials, nanometer coating materials and hard alloy adhesive agent[1]. For example, the catalyzer made of nickel nanoparticles can make the velocity of organic hydrogenation and dehydrogenation enhanced by several times compared with traditional nickel catalyzer[2]. It is reported that solid rocket propellant with 1% nickel nanoparticles could increase its heat of combustion twice[3]. Recently, with the fast development of mobile telephone, computer and many other telecommunication equipments, the demand of nano-nickel materials increases continually [4].
A number of techniques have been used for the preparation of nickel nanomaterials, such as chemical vapor deposition(CVD)[5], wet chemical method[4, 6], laser-driven aerosol[7], hydrothermal method[8], and microemulsion[9-11]. But the use in industry with the above method is limited. One way to solve the problem is using the regular W/O emulsions as reaction medium. Although emulsion is unstable thermodynamics system, it can keep dynamic stability for a long time in proper condition in the presence of rigid film at the interface[12]. It was reported that spherical yttrium-barium-copper oxide (Y2BaCuO5, Y211) nanoparticles were synthesized using the emulsion method and the solution methods[13]. Making use of the nonionic and cation surfactants as emulsifier, HUANG et al[14] prepared ZnS nanoparticles successfully in the regular emulsion. However, the preparation of nickel nanoparticles with this method has not been reported.
In this work, the synthesis of nickel nanoparticles in the water-in-oil emulsions of SDBS/n-pentanol/ n-heptane/water by the reduction of nickel sulfate with sodium borohydride was described. The factors of emulsion system influencing the size of particles were discussed.
2 Experimental
2.1 Reagent and instrument
Commercially pure anionic surfactant sodium dodecylbenzene sulfonate (SDBS) (>95%), sodium borohydride (NaBH4) (analytic reagent, >98%), n-pentanol (analytic reagent, >99%), n-heptane (analytic reagent, >99%), acetone (analytic reagent, >99%), ethanol (analytic reagent, >99%), commercially pure nickel sulfate (NiSO4. 6H2O) (>95%) and deionized water (conductance <2.1 μS/cm) were utilized.
Emulsion droplets were determined by light scattering on a laser light scattering apparatus (BI-200SM). The particle sizes were determined by transmission electron microscopy (TEM, Hitachi H-600). Inductively coupled plasma spectrometry (ICP IRIS X7) was used to determine the composition of the products.
2.2 Preparation of nickel nanoparticles
SDBS (4.075 g) was mixed with n-pentanol (10.0 mL) and n-heptane (26.0 mL) in a beaker. Then a certain amount of nickel sulfate aqueous solution (1.0 mol/L) was added, and the solution was stirred until emulsion turned transparent. A certain amount of sodium borohydride covered with n-heptane was added into the nickel sulfate emulsion by magnetically stirring at constant temperature. After nickel nanoparticles were formed, acetone and ethanol were used to recover the product. Then the mixture was centrifuged and washed with 1?1 mixture of ethanol and H2O several times to remove the contaminated oil, surfactant and other inorganic substances from the particles.
3 Results and discussion
3.1 Stability of emulsion
The size of droplets in the emulsion was measured by light scattering at various aging time and results are listed in Table 1 ([NiSO4]=1.0 mol/L, mass ratio of water/SDBS/n-pentanol/n-heptane is 4?8?16?35). The samples 1-3 were measured at about 25℃, the samples 4-6 were measured at about 20 ℃, the samples 7-9 were measured at about 15 ℃, and the samples 11-12 were measured at about 30 ℃. From these results we can see that the change of droplets size is small when the temperature is increased from 15 to 30 ℃. The size of droplets in the emulsion mainly depends on the aging time. It is revealed from Fig.1 and Table 1 that, the shorter the time is, the smaller the size of “microreactors” is, and the better the uniformity of droplet diameter is.
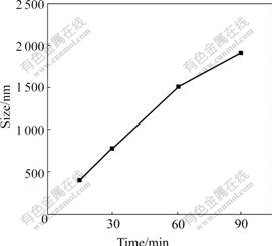
Fig.1 Average size of droplets at various aging time ([NiSO4]= 1.0 mol/L, 25℃, mass ratio of water/SDBS/n-pentanol/ n-heptane 4?8?16?35)
Table 1 Size of droplets in emulsion at various aging time (nm)

The sizes of droplets in the emulsion have better uniformity from 15 to 30 ℃ according to a lot of experiments. The results of laser light scattering test at different temperatures are shown in Figs.2 and 3. The droplet diameters are (408±8) nm and (288±5) nm, respectively. In this system the surfactant SDBS is adsorbed at interface of n-heptane/water, then forms the interfacial film of a certain intensity. Dispersed phase droplets get definite protective action, and gather difficultly.
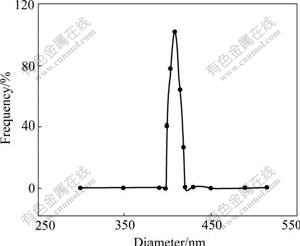
Fig.2 Size distribution of droplets (25 ℃, 15 min, [NiSO4]=1.0 mol/L, mass ratio of water/SDBS/n-pentanol/n-heptane 4?8?16?35)
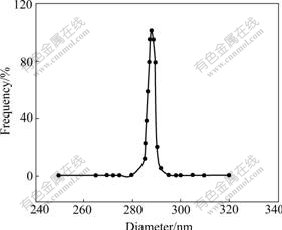
Fig.3 Size distribution of droplets (15 ℃, 15 min, [NiSO4]=1.0 mol/L, mass ratio of water/SDBS/n-pentanol/n-heptane 4?8?16? 35)
NI et al[15] looked into the stability of emulsion and revealed that single surfactant couldn’t form the stable emulsion. While co-surfactant (n-pentanol) presents in the system of water/SDBS/n-heptane, n-heptane/water interface absorbs not only SDBS molecules but also n-pentanol molecules. both mutually affect the adsorption layer, and form compact interfacial film possessing the character of “compound”, enhancing the strength of interfacial film. Meanwhile, the existence of n-pentanol decreases clearly the interfacial tension of n-heptane/water, thereby leading to the more advanced stability of emulsion. This system is similar to that of cholesterol/oil/water/cetyl sodium sulfate.
3.2 Effect of aging time on size of nickel particles
The experimental results show that the size of nano-nickel particles depends on the size of droplets, which is influenced by the aging time. In order to probe the relationship between the aging time and size of nickel particles, the size of droplets of emulsion were measured by light scattering at various aging time. From Fig.4, the nickel particle diameters differ at various aging time of emulsion. While the aging time of emulsion is 15 min, the nickel particle diameter is about 20 nm. At the aging time of emulsion of about 30 min, the nickel particle diameter is about 30 nm. But the shape does not change. It is revealed that the reaction is limited in a droplet, and aggregation hardly occurs with the collision of the droplets. Therefore, particle shape is relative to initial size of droplets of emulsion.
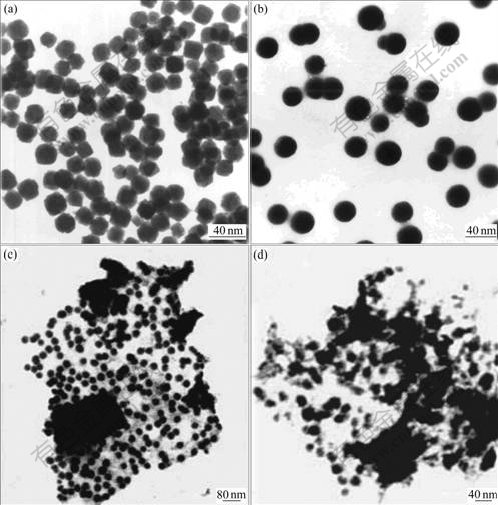
Fig.4 TEM images of nickel nanoparticles (25 ℃, mass ratio of SDBS/n-pentanol/n-heptane of 4?8?16?35): (a) [NiSO4]=1.0 mol/L, R=9.5, 15 min; (b) [NiSO4]=1.0 mol/L, R=9.5, 30 min; (c) [NiSO4]=1.0 mol/L, R=11.5, 15 min; (d) [NiSO4]=1.5 mol/L, R=9.5, 15 min
3.3 Effect of molar ratio of water to surfactant on size of nickel particles
When the aging time and reaction concentration were fixed, the effect of molar ratio of water to SDBS on the products was studied. As shown in Fig.4, when the molar ratio of water to SDBS changes from 9.5 to 11.5, the size of nickel particles increases from 20 nm to 40 nm. The size of product is very uniform. The experiment results show that with the increase of water content, the diameter of products is increased. This also proves that the particle size is mainly related to the initial size of droplets of emulsion, namely so-called “microreactors effect”. The diameter of emulsion droplets increases with the increase of the molar ratio of water to SDBS, then the larger water nuclei supplies bigger space for the growth of particles, which is responsible for the preparation of larger particles.
Meanwhile, the concentration of dispersed phase (droplets) increases with the increase of surfactant concentration, which is attributed to the increase of collision frequency. In this research, the diameter of resultant nickel nanoparticles is controlled by adjusting the size of “microreactors”.
3.4 Effect of concentration of nickel ion on size of nickel particles
It is found from the experiment that the nickel size prepared from emulsion also depends on the concentration of nickel ion in emulsion system. The emulsion would turn turbid very soon when the concentration of NiSO4 is small (less than 1.0 mol/L). When the concentration of NiSO4 is 1.0 mol/L, the stability of emulsion is much better. However, when the concentration of NiSO4 is increased to 1.5 mol/L, the system becomes turbid again. The products obtained on this condition aggregate seriously (Fig.4(d)). It is thought that the salt effect makes the stability of liquid membrane decrease, so the droplets of emulsion get large and are not uniform. So in this research 1.0 mol/L NiSO4 water solution is adopted to prepare the nickel nanoparticles.
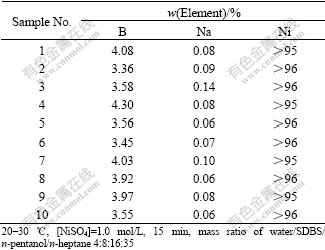
3.5 Element analysis
Element analysis was used to check the purity of products. The results are listed in Table 2. The products contain a little of boron element. Maybe the Ni-B alloy is formed on this condition[16].
Table 2 Element analytical results
4 Conclusions
1) Simple and industrial way to synthesize nickel nanoparticles is presented. In the thermodynamically unstable emulsion system, nickel nanoparticles are prepared by the reduction of NiSO4 with NaBH4. The size of nickel particles is related closely with the constitution of emulsion, aging time, concentrations of water and surfactant.
2) The size of nickel particles becomes larger with the increase of water-to-surfactant molar ratio (R). The size of product is about 20 nm and very uniform when R is 9.5.
3) The longer the aging time of emulsion is, the bigger the droplet is, and the size of nickel particles gets larger and distributes non-uniformly.
4) The concentration of nickel ion ranges from 1.0 mol/L to 1.5 mol/L, the size of nickel particles gets larger, and the uniformity is less.
Acknowledgements
The authors thank the College of Chemistry and Chemical Engineering and the College of Resources and Environment of Lanzhou University for their financial support.
References
[1] KNECHT M R, GARCIA-MARTINEZ J C, CROOKS R M. Synthesis, characterization, and magnetic properties of dendrimer-encapsulated nickel nanoparticles containing <150 atoms [J]. Chem Mater, 2006, 18(21): 5039-5044.
[2] PATEL J D, CANAR O, JONES J. Preparation and characterization of nickel nanoparticles for binding to his-tag proteins and antigens [J]. Pharmaceutical Research, 2007, 24(2): 343-352.
[3] TAN Ling-hua, LI Feng-shang, LIU Lei-li. A review of research on preparation of nano-nickel powder [J]. Materials Review, 2003,17(S1): 41-43. (in Chinese)
[4] CHEN Rui-ying, ZHOU Kang-gen. Preparation of ultrafine nickel powder by wet chemical process [J]. Trans Nonferrous Met Soc China, 2006, 16(5): 1223-1227.
[5] VLADIMIR V B, VALENTIN N M, NIKOLAY V G. Mechanism of Ni film CVD with a Ni(Ktfaa)2 precursor on a silicon substrate [J]. Chem Vap Deposion, 2005, 11: 368-374.
[6] QIN Zhen-ping, GUO Hong-xia, LI Dong-sheng, SHI Xue-ping. Preparation and characterization of nickel nanopowders in diethyleneglycol by liquid-phase reduction [J]. Journal of Functional Materials and Devices, 2004, 10(1): 95-97. (in Chinese)
[7] HE Yuan-qing, LI Xue-geng, SWIHART M T. Laser-driven aerosol synthesis of nickel nanoparticles [J]. Chem Mater, 2005, 17(5): 1017-1026.
[8] LIU Zhao-ping, LI Shu, YANG You, PENG Sheng, HU Zhao-kang, QIAN Yi-tai. Complex-surfactant-assisted hydrothermal route to ferromagnetic nickel nanobelts [J]. Adv Mater, 2003, 15: 1946-1848.
[9] LAN Yun-jun, LUO Wei-ping, WANG Xian-ni. Emulsion theory and its direction on developing leather fatliquor [J]. West Leacher, 2002, 2: 20-25. (in Chinese)
[10] GAO Bao-jiao, GAO Jian-feng, ZHOU Jia-qi. Study on preparation of ultrafine nickel particles by the method of W/O microemulsion [J]. Chinese Journal of Inorganic Chemistry, 2001, 17(4): 491-495. (in Chinese)
[11] CHEN D H, WU S H. Synthesis of nickel nanoparticles in water-in-oil microemulsions [J]. Chem Mater, 2000, 12: 1354-1360.
[12] CUI Zheng-gang, YIN Fu-shan. Microemulsion technology and application [M]. Beijing: Chinese Light Industry Press, 1999.
[13] LI Fang, VIPULANANDAN C. Characterization of Y2BaCuO5 nanoparticles synthesized by nano-emulsions method [J]. Journal of Nanoparticle Research, 2007, 9: 841-852.
[14] HUANG Xiao-bin, MA Ji-ming, CHENG Hu-min, ZHAO Zhen-guo, QI Li-min. Preparation of nanosized ZnS particles in emulsions [J]. Chinese Journal of Applied Chemistry, 1997, 14(1): 117-118. (in Chinese)
[15] NI Liang, JIANG Wen-hua, HAN Shi-jun. Stability of emulsion composed of nitrobenzene/water/sodium dodecyl sulfate by conductance measurement [J]. Journal of Chemical Industry and Engineering, 2001, 52(12): 1104-1108. (in Chinese)
[16] YAN HONG. Study of metallization on surface of BaTiO3 ceramics [J]. Chinese Journal of Rare Metals, 1999, 23(6): 461-465. (in Chinese)
Corresponding author: AN Xue-qin; Tel: +86-13919038681; E-mail: zhangyx@lzu.edu.cn
(Edited by YANG Bing)