
Fabrication and characteristics of spindle Fe2O3@Au core/shell particles
SHEN Hong-xia(���ϼ)1, 2, YAO Jian-lin(Ҧ����)1, GU Ren-ao(���ʰ�)1
1. College of Chemistry, Chemical Engineering and Materials Science, Suzhou University,
Suzhou 215123, China;
2. Department of Biology and Chemical Engineering, Jiaxing University, Jiaxing 314001, China
Received 14 November 2008; accepted 19 March 2009
Abstract: The fabrication and characteristics of spindle Fe2O3@Au core/shell particle were investigated, and the effect of the core/shell nanoparticles as the surface enhanced Raman spectroscopy (SERS)-active substrates was studied. By using the seed-catalyzed reduction technique, anisotropic Fe2O3@Au core/shell particles with spindle morphology were successfully prepared. The Fe2O3 particles with spindle morphology were initially prepared as original cores. The Au nanoparticles of 2 nm were attached onto the Fe2O3 particles through organosilane molecules. Uniform Au shell formed onto Fe2O3 core modified by Au nanoparticles through the in-situ reduction of HAuCl4. The shell thickness was controlled through regulating the concentration of HAuCl4 solution. The results of TEM, XRD and UV-vis characterization show that the core/shell particles with the original shape of the Fe2O3 particles are obtained and these surfaces are covered by Au shell completely. The surface enhanced Raman spectrum of the probe molecules adsorbed on these core/shell substrates is strong and the intensity is enhanced with the increase of the thickness of Au shell or the aspect ratio of particles. The spindle Fe2O3@Au core/shell particles exhibit optimum (SERS) activity.
Key words: surface enhanced Raman spectroscopy; Fe2O3@Au particle; spindle shape; thiophenol
1 Introduction
The nanoparticles with core/shell structure have attracted extensive attention in materials science[1-2], since these particles have many unique physical or chemical properties compared with their single-component counterparts, such as monodispersity, stability, maneuverability, and self-assembly property. Thus, core/shell nanoparticles exhibit some novel properties in electronics[3], magnetics [4], optics[5-7], thermal conductivity[8] and catalysis[9] by adjusting their size, chemical composition and structure order. In particular, structure consisting of a dielectric core and a metal shell exhibits important application in various technological fields such as photonics[10], catalysis[11], medical therapy[12] and sensing[13-14]. Recently, the hybrid particles were severed as the surface enhanced Raman spectroscopy (SERS) substrate due to the tunable optical properties of the hybrid materials[15]. It is well known that SERS is originated from extremely high surface electromagnetic field, which results from the localized surface plasmon resonance(LSPR). As a consequence, SERS intensities are critically dependent on the excitation of LSPR, and hence it is worthy of controlling.
During the last few years, the synthesis and the characterization of anisotropic metal nanoparticles have been intensified. Previous studies showed that the plasmon optical resonance of Au nanoshells could be selectively tuned to any wavelength across the visible and the infrared regions of spectrum by simply varying the thickness ratio of the core to metal shell. In this work, to obtain the convenient and tunable ratio of the core to shell, we paid attention to prolate spheroid-nanorice that bears a remarkable resemblance to a grain of rice. Then, after the investigation of the fabrication of anisotropic dielectric core/metallic shell nanoparticle with tunable thickness and surface roughness, the SERS spectra of probe molecules adsorbed on these particles substrates were studied.
2 Experimental
2.1 Materials
Ferric chloride (FeCl3��6H2O), formaldehyde solution (37%), potassium dihydrogen phosphate (KH2PO4), (3-aminopropyl)trimeth-oxysilane (APTMS, 97%), ethanol, chloroauric acid tetrahydrate (HAuCl4? 4H2O), tetrakis hydroxymethyl phosphonium chloride (THPC), thiophenol(TP) were purchased from Aldrich. Ultrapure water with 18.2 M�� in resistivity and all these chemicals were used as received without further purification.
2.2 Preparation of spindle Fe2O3 particles
Monodisperse spindle-shaped Fe2O3 particles with controllable aspect ratios were fabricated by forced hydrolysis of ferric chloride (FeCl3��6H2O) in 100 mL of aqueous solution containing KH2PO4 for 3 d[16]. The perfect spindle-shaped Fe2O3 particles with aspect ratio of 6.2 (500 nm��80 nm) were prepared. As one of reactants, KH2PO4 prolonged the processes of core formation and induced Fe2O3 to form rice-shaped or spindle-shaped particles. The precipitate was centrifuged and washed several times with water and ethanol. Finally, the precipitate was redispersed in ethanol for the further treatment.
2.3 Surface modification of spindle Fe2O3 cores
Through the self-assembly monolayer technique organosilane molecules (3-aminopropyl trimethoxysilane) were modified onto the surface of spindle Fe2O3 cores[17]. The modified particles were centrifuged and redispersed in ethanol several times to remove the APTMS in the solution. The amine terminated on the surface of particles attached the small THPC-capped Au nanoparticles (about 2 nm in diameter) through the gold-amine interactions[18]. Fe2O3 cores modified with small Au nanoparticles were served as the seeds for the continuous formation of Au shell[19].
2.4 Preparation of spindle Fe2O3@Au core/shell particles
A typical preparation of spindle Fe2O3@Au core/shell particles was carried out as follows: the former seed particles were disturbed in 10 mL of aqueous solution containing HAuCl4 and then reduced by formaldehyde solution. The continuous Au shell was formed due to the small Au nanoparticles as nucleation sites[20]. The thickness of Au shell can be controlled by changing the ratio of Au3+ to reducing agent.
2.5 Measurement
Transmission electron microscopy(TEM) photographs were taken on a Tecnai G220 from USA FEI Company. X-ray powder diffraction(XRD) patterns of the products were obtained on a X��pert-pro MPD from Panalytical Company, Holand. A rotation anode X-ray diffractometer was equipped with graphite monochromatized Cu K�� radiation (��=1.541 78 ?). Ultraviolet-visible (UV-vis) spectra were measured with an ultraviolet-visible spectrophotometer of TU-1810 DSPC model. SERS spectra were measured on micro-confocal Raman system of LabRam HR800 from Horiba Jobin-Yvon with the excitation line of 633 nm.
3 Results and discussion
3.1 Morphology of particles
The perfect spindle-type Fe2O3 particles could be synthesized through hydrolysis in a flask. And the small Au nanoparticle can be attached to the core through Au-amine interaction, which contributes to the formation of continuous Au shell. All these can be inferred from TEM photographs. Fig.1 shows the morphologies of spindle Fe2O3 particles and the core/shell particles with different shell thicknesses. Fig.1(a) shows that monodisperse spindle particles with a mean edge length of 500 nm and edge width of 80 nm are formed. Transmission electron micrograph in Fig.1(b) illustrates that the seed particles are formed by small Au nanoparticles attached to the Fe2O3 core. Micrographs in Figs.1(c) and (d) provide further evidence to prove the formation of core/shell particles due to a sharp contrast between the dark edge and the pale center. Two different thicknesses of Au shell, determined from the TEM image, are 28 nm and 49 nm, respectively.

Fig.1 TEM micrographs: (a) Spindle Fe2O3 particles; (b) Seed particles; (c), (d) Fe2O3 coated with different thicknesses of Au shell of 28 nm and 49 nm, respectively
3.2 Crystal parameters of particles
The XRD patterns of the products are shown in Fig.2. In Fig.2(a), all the peaks match well with the standard JCPDS data of ��-Fe2O3 (JCPDS file, No.01-072-0469). When the ��-Fe2O3 particles are modified with amine and attached by small Au nanoparticles, the characteristic diffraction peaks of ��-Fe2O3 gradually disappear while the characteristic diffraction peaks of Au (111, 200, 220) appear in Fig.2(b). The above phenomena are apparent in Figs.2(c) and (d) with the increase of the thickness of Au shell, and these peaks are well accordant with the standard card (JCPDS file, No.01-089-3697). And the XRD results are well consistent with the above TEM images.
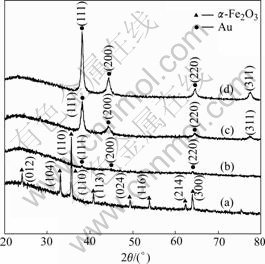
Fig.2 X-ray diffraction patterns for spindle particles: (a) Fe2O3 core; (b) Seed particle; (c) Fe2O3@Au with thinner shell; (d) Fe2O3@Au with thicker shell
3.3 Growth process of Fe2O3@Au core/shell structure
In order to observe the influence of the thickness of the shell on SERS, hybrid particles with different thicknesses of Au shell were also prepared. Two major factors can control the thickness of Au shell in the growth process of Fe2O3@Au core/shell structure: one is the concentration of Au3+ and the other is the concentration of the seeds.
The typical TEM images (Fig.3) show the anisotropic core/shell nature of the products resulted from the change of the concentration of Au3+ in solution. With increasing Au3+ concentration, the shell becomes thicker and thicker, which is similar to that with decreasing the concentration of seed. Once the seeds with small Au particles have been obtained (the particles are shown in Fig.1(b)), further growth can be promoted by a slow reduction of HAuCl4 with a weak reducing agent that can only lead metal Au to form on the surface of the small Au particles. This has been previously reported for the growth of Au spheres[21] or for the growth of Au nanoshells on silica spheres[20-22].

Fig.3 TEM micrographs of Fe2O3@Au core/shell structures with different thickness of Au shell: (a) 4 nm; (b) 11 nm; (c) 29 nm; (d) 59 nm
The TEM micrographs indicate that the Au steadily grows on the surface of seed like islands, which is attributed to the preference chemical reduction of HAuCl4 onto the small Au nanoparticles attached to Fe2O3 cores. And they eventually form a Au shell. However, because of the independent growth of isolated islands, the final structure is relatively rough, which can be readily distinguished from the smooth surface morphology of the Fe2O3 spindles. Rough surface obtained is very useful for SERS[23]. So, In this study, large surface area and roughness are desirable.
3.4 SERS of probe molecules adsorbed on ��-Fe2O3@ Au core/shell structure
A large variability of SERS intensities for different hybrid particles was observed experimentally as shown in Fig.4 in which thiophenol(TP) was used as the probe molecule. It can be seen that the surface Raman signals are extremely strong. The strong SERS signals at 999, 1 020, 1 072, and 1 573 cm-1 are from TP and signal at 900-1 000 cm-1 are from Si substrate[24]. With increasing the thickness and roughness of Au shell, the SERS intensity increases because of the electromagnetic enhancement and the coupling between the hybrid particles.
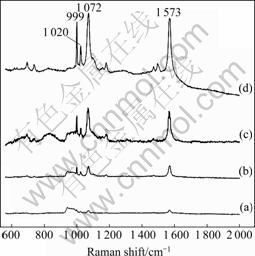
Fig.4 SERS spectra of Fe2O3@Au core/shell particles with different thicknesses of Au shell: (a) 4 nm; (b) 11 nm; (c) 29 nm; (d) 59 nm
3.5 UV-vis spectra of Fe2O3@Au core/shell structure
In Fig.5 the evolution of the UV-vis spectra of Fe2O3@Au core/shell structure is plotted with the growth of the Au particles on the surface of seed. The poor smoothness of the Au structure obtained is responsible for broad absorption bands[25]. And the deposition of the Au nanoparticles leads to an obvious increase in the absorption because of the Au plasmon absorption band.
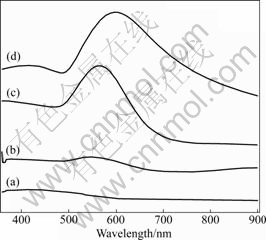
Fig.5 UV-vis spectra of Fe2O3@Au core/shell particles with different thicknesses of Au shell: (a) 4 nm; (b) 11 nm; (c) 29 nm; (d) 59 nm
Although the seeds display a weak plasmon resonance, absorbance largely increases and red-shift happens as Au nanoparticles agglomerate on the Fe2O3 surface owing to increased particle size and interparticle interactions.
4 Conclusions
1) The spindle Fe2O3@Au core/shell particles are successfully prepared by seed-catalyzed method.
2) The strong SERS signals of the probe molecules on the surface of the spindle Fe2O3@Au core/shell nanoparticles are observed because of the located surface plasmon resonance of its rough surface and unique shape.
References
[1] KAMATA K, LU Y, XIA Y N. Synthesis and characterization of monodispersed core-shell spherical colloids with movable cores [J]. J Am Chem Soc, 2003, 125: 2384-2385.
[2] LIN S C, CHEN S Y, CHEN Y T, CHENG S Y. Electrochemical fabrication and magnetic properties of highly ordered silver-nickel core-shell nanowires [J]. J Alloy Compd, 2008, 449: 232-236.
[3] XIANG J, LU W, HU Y J, WU Y, YAN H, LIEBER C M. Ge/Si nanowire heterostructures as high performance field-effect transistors [J]. Nature, 2006, 441: 489-493.
[4] PARK J I, CHEON J. Synthesis of ��solid solution�� and ��core-shell�� type cobalt-platinum magnetic nanoparticles via transmetalation reactions [J]. J Am Chem Soc, 2001, 123: 5743-5746.
[5] OW H, LARSON D R, SRIVASTAVA M, BAIRD B A, WEBB W W, WIESNER U. Bright and stable coreshell fluorescent silica nanoparticles [J]. Nano Lett, 2005, 5(1): 113-117.
[6] HU J W, LI J F, REN B, WU D Y, SUN S G, TIAN Z Q. Palladium-coated gold nanoparticles with a controlled shell thickness used as surface-enhanced Raman scattering substrate [J]. J Phys Chem C, 2007, 111: 1105-1112.
[7] BAER R, NEUHAUSER D, WEISS S. Enhanced absorption induced by a metallic nanoshell [J]. Nano Lett, 2004, 4(1): 85-88.
[8] YANG R, CHEN G, DRESSELHAUS M S. Thermal conductivity modeling of coreshell and tubular nanowires [J]. Nano Lett, 2005, 5(6): 1111-1115.
[9] TIAN N, CHEN W, SUN S G. The characterization spectrum and electric catalysis of Au-Pt core-shell nanoparticles [J]. Acta Phys��Chim Sin, 2005, 21: 74-78.
[10] FLEISCHHAKER F, ZENTEL R. Photonic crystals from core-shell colloids with incorporated highly fluorescent quantum dots [J]. Chem Mater, 2005, 17: 1346-1351.
[11] CARUSO F, LICHTENFELD H, GIERSIG M, MOHWALD H. Electrostatic self-assembly of silica nanoparticle-polyelectrolyte multilayers on polystyrene Latex particles [J]. J Am Chem Soc, 1998, 120(33): 8523-8524.
[12] HUANG X H, EL-SAYED I H, QIAN W, EL-SAYED M A. Cancer cell imaging and photothermal therapy in the near-infrared region by using gold nanorods [J]. J Am Chem Soc, 2006, 128: 2115-2120.
[13] JENSEN T R, MALINSKY M D, HAYNES C L, van DUYNE R P. Nanosphere lithography: Tunable localized surface plasmon resonance spectra of silver nanoparticles [J]. J Phys Chem B, 2000, 104(45): 10549-10556.
[14] LEI Ting. Preparation of novel core-shell nanoparticles by electrochemical synthesis [J]. Trans Nonferrous Met SOC China, 2007, 17(6): 1343-1346.
[15] LI J F, YANG Z L, REN B, LIU G K, FANG P P, JIANG Y X, WU D Y,TIAN Z Q. Surface-enhanced Raman spectroscopy using gold-core platinum-shell nanoparticle film electrodes: Toward a versatile vibrational strategy for electrochemical interfaces[J]. Langmuir, 2006, 22(25): 10372-10379.
[16] OZAKI M, KRATOHVIL S, MATIJEVIC E. Formation of monodispersed spindle-type hematite particles [J]. J Colloid Interf Sci,1984, 102(1): 146-151.
[17] HALL S R, DAVIS S A, MANN S. Cocondensation of organosilica hybrid shells on nanoparticle templates: A direct synthetic route to functionalized core-shell colloids [J]. Langmuir, 2000, 16: 1454-1456.
[18] WESTCOTT S L, OLDENBURG S J, LEE T R, HALAS N J. Formation and adsorption of clusters of gold nanoparticles onto functionalized silica nanoparticle surfaces [J]. Langmuir, 1998, 14: 5396-5401.
[19] JIX J, SHAO R P, ELLIOTT A M, STAFFORD R J, ESPARZA C E, BANKSON J A, LIANG G, LUO Z P, PARK K, MARKERT J T, LI C. Bifunctional gold nanoshells with a superparamagnetic iron oxide-silica core suitable for both MR imaging and photothermal therapy [J]. J Phys Chem C, 2007, 111: 6245-6251.
[20] OLDENBURG S J, AVERITT R D, WESTCOTT S L, HALAS N J. Nanoengineering of optical resonances [J]. Chem Phys Lett, 1998, 288: 243-247.
[21] BROWN K R, LYON L A, FOX A P, REISS B D, NATAN M J. Hydroxylamine seeding of colloidal Au nanoparticles (3): Controlled formation of conductive Au films [J]. Chem Mater, 2000, 12(2): 314-323.
[22] GRAF C, BLADEREN A V. Metallodielectric colloidal core-shell particles for photonic applications [J]. Langmuir, 2002, 18: 524-534.
[23] TALLEY C E, JACKSON J B, OUBRE C, GRADY N K, HOLLARS C W, LANE S M, HUSER T R, NORDLANDER P, HALAS N J. Surface-enhanced Raman scattering from individual Au nanoparticles and nanoparticle dimer substrates [J]. Nano Lett, 2005, 5(8): 1569-1574.
[24] CARRON K T, HURLEY L G. Axial and azimuthal angle determination with surface-enhanced Raman spectroscopy: Thiophenol on copper, silver, and gold metal surfaces [J]. J. Phy. Chem, 1991, 95(24): 9979�C9984.
[25] SPUCH-CALVAR M, PEREZ-JUSTE J, LIZ-MARZAN L M. Hematite spindles with optical functionalities: Growth of gold nanoshells and assembly of gold nanorods [J]. J Colloid Interf Sci, 2007, 310: 297-301.
Foundation item: Projects(20573076, 20503019, 20773091) supported by the National Natural Science Foundation of China; Project(BK2005032) supported by the Natural Science Foundation of Jiangsu Province, China; Project(20050285019) supported by the Specialized Research Fund for the Doctoral Program of Higher Education (SRFDP), China
Corresponding author: GU Ren-ao; Tel: +86-512-65880399; E-mail: ragu@suda.edu.cn
DOI: 10.1016/S1003-6326(08)60328-3
(Edited by YANG Hua)