
Dielectric properties of BiFeO3-PbTiO3 thin films prepared by PLD
CHEN Rui((陈 蕊), YU Sheng-wen(俞圣雯), ZHANG Guan-jun(张冠军), CHENG Jin-rong(程晋荣), MENG Zhong-yan(孟中岩)
Department of Electronic Information Material, School of Materials Science and Engineering,
Shanghai University, Shanghai 200072, China
Received 10 April 2006; accepted 25 April 2006
Abstract: BiFeO3-PbTiO3 (BFO-PT) thin films were prepared on Pt/TiO2/SiO2/Si substrates by pulsed-laser deposition (PLD) technique under different oxygen pressures. The structures of the films were characterized by means of XRD. The current densities were performed to check the conductivity of the films. The dielectric constant and loss factor (tanδ) of the films were measured. The results show that the BFO-PT layers are mainly perovskite structured; the film deposited under 6.665 Pa exhibits low leakage current, low dielectric loss (0.017-0.041) and saturated hysteresis loop with polarization (Pr) value and coercive field (Ec) of 3 ?C/cm2 and 109 kV/cm.
Key words: dielectric; ferroelectric; BiFeO3-PbTiO3; thin Film, PLD
1 Introduction
Since 1960s, bismuth ferrite, BiFeO3, was reported as simultaneously exhibiting ferroelectric ordering with Curie temperature of 850 ℃ and magnetic ordering with Néel temperature of 310 ℃. It has attracted much attention for the potential application in multifunctional devices [1-4]. However, for decades, the obtained dielectric properties of BiFeO3 and BiFeO3-based materials were limited comparing to the theoretical values. It was reported that the relative high electrical conductivity in BiFeO3 caused by the existence of small amounts of Fe2+ ions and oxygen vacancies prevented proper exploration of low-frequency dielectric properties [5]. Recent studies have been focused on solid solutions of BiFeO3 with other perovskite materials to suppress the formation of oxygen vacancies, such as BaTiO3(BT), PbTiO3(PT). Cheng et al [6] have successfully developed La3+ modified 0.6BiFeO3-0.4PbTiO3 (BFO-PT) solid solutions at the morphotropic phase boundary (MPB) which gives strong ferroelectricity.
In this work, we prepared BFO-PT thin films on Pt/TiO2/SiO2/Si substrates by PLD and studied the effects of deposition pressure on the crystalline structure, dielectric and ferroelectric properties at room temperature [7].
2 Experimental
Tuilaser system (ThinFilmStar-100) was used for ablation process. La3+ modified 0.6BiFeO3-0.4PbTiO3 target was sintered by solid-state reaction [6]. BFO-PT thin film was deposited on Pt/TiO2/SiO2/Si substrates. The detailed deposition conditions of BFO-PT thin films are listed in Table 1.
The structures of the films were determined by X-ray diffraction with a D/MAX-RC diffractometer using Cu Kα radiation (40 kV/100 mA) with step of 4(°)/min. The thicknesses of BFO-PT thin films are all about 200 nm which are measured by STYLUS profiler. Pt was deposited on the surface of the films by sputtering through a shadow mask with a diameter of 0.2 mm as upper electrode. The frequency dependences of permittivity and dielectric loss of BFO-PT thin films were measured by an impedance analyzer HP 4192A and the current-density-applied voltage (J-V) curves were detected with HP 4140B PA meter/DC voltage. The polarization-electric-field measurement was conducted with RT6000 HVS high voltage test system using 1 kHz triangular wave form.
Table 1 Deposition conditions of BFO-PT thin films by PLD

3 Results and discussion
The XRD patterns of BFO-PT thin films grown on Pt/TiO2/SiO2/Si substrates with various deposition pressures are shown in Fig.1. It shows that the films consist of both perovskite structure (black triangular symbol) and impurity phase (black circle symbol) which is identified as Bi2Fe4O9 (orthorhombic structure). With the increasing of gas pressure, the intensities of the characteristic peaks of perovskite structure become stronger while those of Bi2Fe4O9 peaks become weaker. The presence of Bi2Fe4O9 can be ascribed to the loss of Bi through volatilization and reaction in the process of deposition, which destroys the stoichiometry[8]. And high gas pressure (oxygen ambient) can lower the loss of Bi to abstain from the impurity phase. As the deposition pressure increases from 0.666 Pa to 1.333 Pa, the peak of (100) in the BFO-PT films is enhanced as shown in Fig.1. This means that the deposition pressure is one important factor controlling the orientation of the thin films.
Fig.2 and Fig.3 present the frequency dependences

Fig.1 Conventionalθ-2θ X-ray diffraction measurements of BFO-PT thin films on Pt/TiO2/SiO2/Si under different deposition pressures: (a) 0.666 Pa; (b) 6.665 Pa; (c) 13.332 Pa
of permittivity and dielectric loss of BFO-PT thin films. The dielectric constant (εr) of thin film deposited under gas pressure of 13.332 Pa is about four times comparing to those deposited under gas pressures of 0.666 and 6.665 Pa. With the frequency increases, the permittivity of the thin film deposited under gas pressure of 13.332 Pa decreases gradually below 600 kHz and increases gradually when frequency is above 600 kHz, while the others decrease slightly from 1 kHz to 1 MHz. At high-frequency region, the dielectric losses of the three samples all increase, which are extrinsic in nature due to the resonance of the equivalent circuit. Further more, the losses of the thin films deposited under gas pressure of 0.666 and 13.332 Pa increase more sharply. At 100 kHz, the loss of thin film deposited under gas pressure of 0.666 Pa is much lower (0.021) compared to the films deposited under gas pressure of 0.666 Pa (0.0815) and 1.332 Pa (0.04).
Fig.4 shows the J-V curves of BFO-PT thin films. It indicates that the current density of the BFO-PT thin
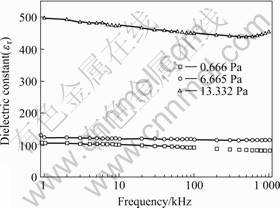
Fig.2 Frequency dependence of dielectric constant of BiFeO3-PbTiO3 (-200 nm) thin films deposited under gas pressures of 0.666, 6.665 and 13.332 Pa
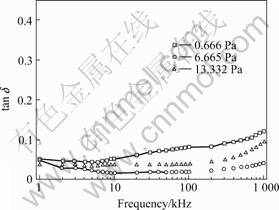
Fig.3 Frequency dependence of dissipation factor (tan δ) of BiFeO3-PbTiO3 (-200 nm) thin films deposited under gas pressures of 0.666, 6.665 and 13.332 Pa
films depends greatly on the deposition pressure. Since higher O2 pressure can suppress the mobilization of the activated electrons from depressing deep-trap energy levels created by oxygen vacancies, while much higher gas pressure makes it hard to obtain thin films with a compact structure and therefore contribute to a higher current density [9, 10]. As a result, in this work, the current density of film deposited under gas pressure of 6.665 Pa is much lower than those deposited under gas pressure of 0.666 and 13.332 Pa. Accordingly, the film prepared under gas pressure of 6.665 Pa has a much lower dielectric loss.
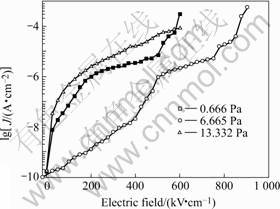
Fig.4 lgJ-E curves of BiFeO3-PbTiO3 thin films deposited under gas pressures of 0.666, 6.665 and 13.332 Pa
Ferroelectric polarization-electric-field (P-E) hysteresis curves of BFO-PT thin films were measured at room temperature. As shown in Fig.5, the film deposited under gas pressures of 0.666 Pa shows a poor loop, which can be due to the effect caused by a relative high leakage current shown in Fig.4. And the film deposited under 6.665 Pa exhibits saturated hysteresis loop, with the remnant polarization (Pr) of 3 μC/cm2, coercive field (Ec) of 109 kV/cm and the maximum applied electric field of 900 kV/cm. This also can be attributed to the greatly reduced leakage current and less ohmic behavior in the film deposited under gas pressures of 0.666 Pa. While for the film deposited under gas pressures of 6.665 Pa, P-E loop is not detectable although its dielectric constant is large in Fig.2. As far as now we are unclear about the mechanism in the film prepared under gas pressures of 13.332 Pa according to its results. More detail investigation is in progress.
4 Conclusions
In summary, we have prepared BFO-PT thin films on Pt/TiO2/SiO2/Si substrate by pulsed-laser deposition technique. The films were crystallized mainly in perovskite structure. Gas pressure (oxygen ambient) is critical to the structure, the dielectric and ferroelectric properties of BFO-PT thin films. In this work, BFO-PT thin film deposited under 6.665 Pa shows a reasonable dielectric properties and detectable ferroelectric properties.
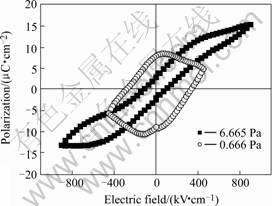
Fig.5 Room temperature ferroelectric P-E hysteresis loops of BiFeO3-PbTiO3 (-200 nm) thin films deposited under gas pressures of 0.666 and 6.665 Pa
References
[1] Yun K Y, Noda M, kuyama M. Structural and multiferroic properties of BiFeO3 thin films at room temperature[J]. Journal Of Applied Physics, 2004, 96: 3399.
[2] Yun K Y, Noda M, Okuyama M. Prominent ferroelectricity of BiFeO3 thin films prepared by pulsed-laser deposition[J]. Applied Physics Letters, 2003, 83: 3981.
[3] WANG J, NEATON J B, RAMESH R. Epitaxial BiFeO3 multiferroic thin film heterostructures[J]. Science, 2003, 299: 1719.
[4] Palkar V R, John J, Pinto R. Observation of saturated polarization and dielectric anomaly in magnetoelectric BiFeO3 thin films[J]. Applied Physics Letters, 2002, 80: 1628.
[5] QI X D, DKO J H, TOMOV R, BLAMIRE M G, MACMANUS-DRISCOLL J L. Greatly reduced leakage current and conduction mechanism in aliovalent-ion-doped BiFeO3 [J]. Applied Physics Letter, 2005, 86: 062903.
[6] Cheng J R, Cross L. Effects of La substituent on ferroelectric rhombohedral/tetragonal morphotropic phase boundary in(1-x)(Bi,La)(Ga0.05Fe0.95)O3-xPbTiO3 piezoelectric ceramics[J]. Journal Of Applied Physics, 2003, 94: 5188.
[7] Yun K Y, Noda M, Okuyama M. Structural and multiferroic properties of BiFeO3 thin films at room temperature[J]. Journal of Applied Physics, 2004, 96: 3399.
[8] Teague J R, Gerson R, James W J. Dielectric hysteresis in single crystal BiFeO3[J]. Solid State Communications, 1970(8): 1073-1074.
[9] Kim J S, Cheon C, Jang P W. Weak ferromagnetism in the ferroelectric BiFeO3-ReFeO3-BaTiO3 solid solutions (Re=Dy,La)[J]. Journal of Applied Physics, 2004, 96: 468.
[10] Harrop P J. Dielectrics Butterworths[M]. London: Butterworths, 1972. 51-53.
(Edited by HE Xue-feng)
Foundation item: Project(04A1B18) supported by Shanghai Municipal Education Commission; Project(50472098) supported by the National Natural
Science Foundation of China; Project(04qmx1440) supported by Shanghai Rising Star Program, China
Corresponding author: YU Sheng-wen; Tel: +86-021-56332704; E-mail: yusw@staff.shu.edu.cn