Trans. Nonferrous Met. Soc. China 22(2012) 3059-3065
Calculation of thermodynamic properties in liquid phase for ternary Al-Ni-Zn alloys
 1, Y. DU2, N. TALIJAN3, A. KOSTOV4,
1, Y. DU2, N. TALIJAN3, A. KOSTOV4,  1
1
1. Technical Faculty, University of Belgrade, Bor, Serbia;
2. State Key Laboratory of Powder Metallurgy, Central South University, Changsha 410083, China;
3. University of Belgrade, Institute of Chemistry, Technology and Metallurgy, Belgrade, Serbia;
4. Mining and Metallurgy Institute, Bor, Serbia
Received 2 February 2012; accepted 25 May 2012
Abstract: The results of the calculation of thermodynamic properties in liquid state for ternary Al-Ni-Zn alloys using the newest version of the general solution model for thermodynamic prediction are presented. Nine sections with different molar ratios of Ni to Zn, Zn to Al and Al to Ni were investigated in a temperature interval of 1800-2000 K. Partial and integral molar thermodynamic properties in liquid phase for the Al-Ni-Zn ternary system are determined and discussed.
Key words: ternary system; Al-Ni-Zn alloys; thermodynamics; calculation; general solution model
1 Introduction
ZA alloys present a group of alloys whose main constituents are zinc and aluminum [1,2]. Originally, they were alloyed with copper and magnesium and developed for gravity casting [3]. But, nowadays, ZA alloys alloyed with other metals, such as Ni, Ga, Ge, Sb, Fe, have a wide variety of demanding applications as engineering materials in different branches of technology: lead-free solders for die attach [4-7], shape memory alloys [8], dental alloys [9,10], coatings for steel sheets [11-13], or as commercial Al bronzes [14,15].
Among numerous ZA alloys, ternary Al-Ni-Zn system due to its structural, mechanical and corrosion resistance properties is used for hot-dip zinc protective coatings on steel exposed to corrosive environments [16,17]. In spite of technological interest for Al-Ni-Zn alloys, this system has not been investigated completely from thermodynamic point of view till now. Insufficient experimental information, mostly due to existing difference between melting temperatures of Ni vs Al and Zn and a high vapour pressure of zinc, caused that mentioned system is still not thermodynamically described [7] although constitutive binary systems Al-Ni [18], Al-Zn [19] and Ni-Zn [20] are assessed and given in COST531 database [21].
On the other hand, there are more data on Al-Ni-Zn phase equilibria investigations [1,7,16,22-27], but still there is no complete definition. Some literature data on isothermal sections of investigated system at different temperatures are given in Fig. 1.
Having in mind given literature review, available thermodynamic data and difficulties in experimental research of the Al-Ni-Zn system, the purpose of this work was to contribute to better knowledge of its thermodynamic properties in liquid state at high investigating temperatures, using thermodynamic prediction according to the newest version of general solution model.
2 Theoretical fundamentals
The general solution model for calculation of thermodynamic properties for ternary systems based on known binary thermodynamic data has been provided by CHOU and WEI [28,29]. It breaks down the boundary between symmetrical and asymmetrical models, and has already been proved in practical examples as the correct and accurate model [30,31].

Fig. 1 Isothermal sections of Al-Ni-Zn system at 1123 K [26] (a) and 613 K [16] (b)
This model has been derived for multicomponent systems and its basic equations are given as follows [28]:
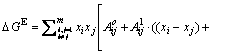
 (1)
(1)
where  ,
,  ,
,  are regular-solution parameters for binary system ij independent of composition, only relying on temperature:
are regular-solution parameters for binary system ij independent of composition, only relying on temperature:

 (2)
(2)
where Xi and Xj indicate the mole fraction of component i and j in ij binary system, expressed as
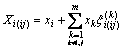 (3)
(3)
and where the coefficient signed as 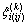 in Eq. (1) presents the similarity coefficient of component k to component i in ij system, defined as
in Eq. (1) presents the similarity coefficient of component k to component i in ij system, defined as
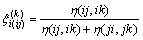 (4)
(4)
where h(ij, ik) is the function related to the excess Gibbs free energy of ij and ik binaries, given as
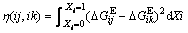 (5)
(5)
Recently, the newest version of general solution model based on Redlich-Kister type parameters was presented in Ref. [32]. The calculations using this innovated model substituted a series of integration processes in older model version with simplified calculation. Since a large number of real systems can be approximately fitted by a Redlich�CKister polynomial [33], a new formalism presents an advantage. The whole integration process is avoided by using algebraic operations, thereby simplifying the calculation.
Main changes in comparison with old model [28] are related to determination of  function, as follows [32]:
function, as follows [32]:
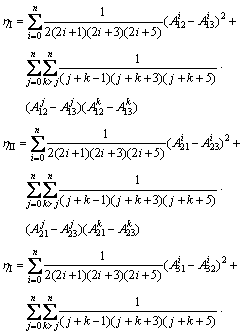
 (6)
(6)
where 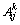 are the Redlich-Kister polynomials. It should be pointed out that
are the Redlich-Kister polynomials. It should be pointed out that  . Based on Eq. (6), main Eq. (1) presenting can be written as follows [32]:
. Based on Eq. (6), main Eq. (1) presenting can be written as follows [32]:

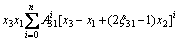 (7)
(7)
In all equations given, ��GE and  respond to the integral molar excess free energies for multicomponent and binary systems, respectively, while x1, x2, x3 respond to mole fraction of components in investigated multicomponent system.
respond to the integral molar excess free energies for multicomponent and binary systems, respectively, while x1, x2, x3 respond to mole fraction of components in investigated multicomponent system.
3 Results and discussion
Thermodynamic calculations in ternary system Al-Ni-Zn were carried out in nine sections along the lines of constant molar ratio of Ni to Zn of 1:3, 1:1, 3:1, sections from aluminium corner; molar ratio of Zn to Al of 1:3, 1:1, 3:1, sections from nickel corner; and molar ratio of Al to Ni of 1:3, 1:1, 3:1, sections from zinc corner. Basic data necessary for the calculation have been taken from Refs. [18-21]. The Redlich-Kister polynomials for the constitutional binaries in the investigated ternary Zn-Ni-Al system are presented in Table 1.
The prediction is done according to the fundamentals of the newest version of general solution model [32]. Based on starting data in Table 1, similarity coefficients are determined and further calculations are carried out for 81 alloys in all selected cross sections in the investigated ternary Al-Ni-Zn system in temperature interval of 1800-2000 K, as given by Eqs. (1-7).
The results of thermodynamic predictions, including the values of ternary integral molar excess Gibbs energy and the activities of all three components in liquid phase, are calculated for all investigated sections at all investigated temperatures, and presented in Table 2, Figs. 2 and 3, respectively. Calculated activity values for all three components are used for construction of isoactivity diagrams at 1900 K and shown in Fig. 4.
Negative values of integral molar excess Gibbs energies are obtained for whole concentration range at all investigated temperatures (Fig. 2). The most negative value of about -35 kJ/mol is present in the section from zinc corner with molar ratio of Al to Ni of 1:1 for low zinc concentration, while other minimal values of about -25 kJ/mol are noticed for equiatomic contents in the n(Ni):n(Zn)=3:1 section from aluminum corner and in the n(Zn):n(Al)=3:1 section from nickel corner.
Negative deviation from Raoult law is obtained for all investigated sections (Fig. 3), except in narrow composition ranges: for sections from zinc corner, x(Zn)<0.7 in n(Al):n(Ni)=1:1 and x(Zn)<0.9 in n(Al):n(Ni)=3:1; for all three sections from nickel corner, x(Ni)>0.9, where a very slight positive deviation can be seen. The strongest negative deviation is noticed for section n(Ni):n(Zn)=3:1 from aluminum corner.
Table 1 Redlich-Kister parameters for liquid phase in constitutional binaries in Al-Ni-Zn system

Table 2 Characteristic dependencies of ternary integral molar excess energies on composition for ternary Al-Ni-Zn alloys expressed as ��GE=Ax2+Bx+C at investigated temperatures

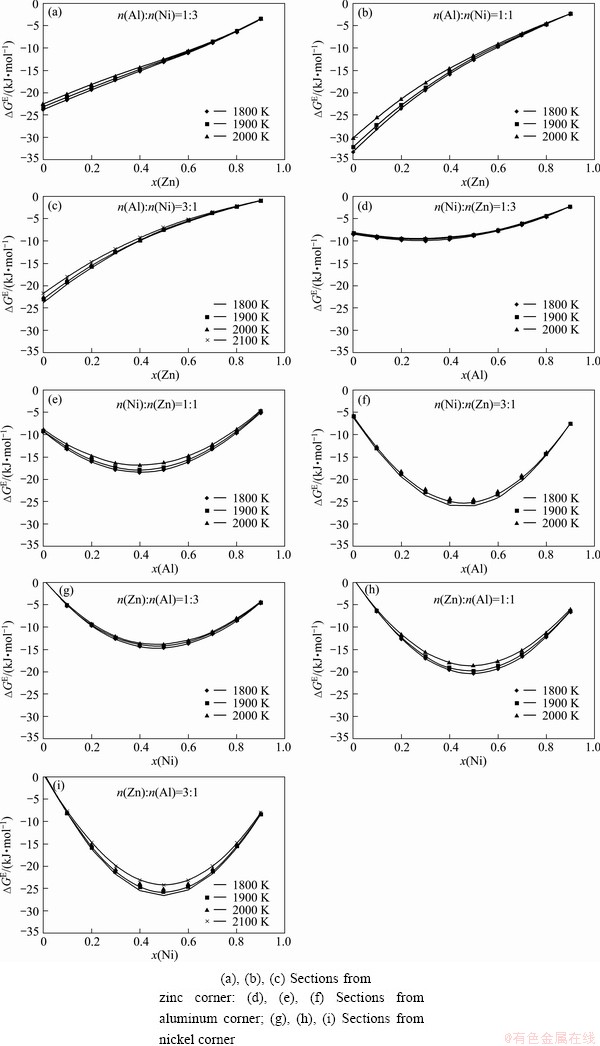
Fig. 2 Graphical presentation of integral molar excess energy dependence on composition and temperature in investigated Al-Ni-Zn system

Fig. 3 Graphical presentation of activity dependence on composition and temperature in investigated Al-Ni-Zn system

Fig. 4 Isoactivity diagrams for constitutive elements in ternary Al-Ni-Zn system at 1900 K
Temperature influence on calculated thermo- dynamic properties is not significant in investigated interval of 1800-2000 K.
Described tendencies indicate to prevalent existence of strong mutual mixing tendencies between constitutive components in Al-Ni-Zn system at investigated temperatures, where aluminum and nickel exhibit more significant mixing tendency compared with zinc.
4 Conclusions
1) The calculation of thermodynamic properties in ternary Al-Ni-Zn system has been done applying the newest version of general solution model for multicomponent systems based on Redlich-Kister type parameters. According to starting thermodynamic data for constituent binary subsystems, ternary integral molar excess Gibbs energies and activities for all three components were calculated in the temperature range of 1800-2000 K in nine sections from zinc, aluminum and nickel corner. Obtained data showed mostly negative deviation from Raoult law, indicating strong mutual mixing tendencies in the investigation system.
2) Due to the fact that experimental determination at selected temperatures is rather difficult and that there is a lack of relevant literature concerning this particular system, these calculated results can be a good substitution for thermodynamic data of this multi- component ZA-based system. That is more important, having in mind already proven accuracy of calculation model applications in different cases.
Acknowledgment
The results of this paper are in the frame of Project OI 172037 financed by the Ministry of Science and Technological Development, Republic of Serbia and bilateral scientific and technological cooperation project between Republic of Serbia and China.
References
[1] RAYNOR G V, FAULKNER C R, NODEN J D, HARDING A R. Ternary alloys formed by aluminium, transitional metals and divalent metals [J]. Acta Metallurgica, 1953, 1(6): 629-648.
[2] SAVAS M A, ALTINTAS S. The microstructural control of cast and mechanical properties of zinc-aluminium alloys [J]. Journal of Materials Science, 1993, 28: 1775-1780.
[3] VARGA B, SZAVA I. Phase transformations in the heat treated and untreated Zn-Al alloys [J]. Acta Universitatis Sapientiae Electrical and Mechanical Engineering, 2009, 1: 207-213.
[4]  L A, RDZAWSKI Z M, LABISZ K. Cooling rate influence on microstructure of the Zn-Al cast alloy [J]. Archives of Materials Science and Engineering, 2010, 43(1): 13-20.
L A, RDZAWSKI Z M, LABISZ K. Cooling rate influence on microstructure of the Zn-Al cast alloy [J]. Archives of Materials Science and Engineering, 2010, 43(1): 13-20.
[5] RETTENMAYR M, LAMBRACHT P, KEMPF B, TSCHUDIN C. Zn-Al based alloys as Pb-free solders for die attach [J]. Journal of Electronic Materials, 2002, 31(4): 278-285.
[6] 
 Calorimetric investigations and thermodynamic calculation of Zn-Al-Ga system [J]. Journal of Thermal Analysis and Calorimetry, 2011, 103(3): 1055-1061.
Calorimetric investigations and thermodynamic calculation of Zn-Al-Ga system [J]. Journal of Thermal Analysis and Calorimetry, 2011, 103(3): 1055-1061.
[7]  D,
D,  G. Experimental and theoretical study of thermodynamic properties and phase equilibria in ternary Al-Zn-X alloys [M]. COST MP0602 Book (Volume 3 Chapter 6) (in Press).
G. Experimental and theoretical study of thermodynamic properties and phase equilibria in ternary Al-Zn-X alloys [M]. COST MP0602 Book (Volume 3 Chapter 6) (in Press).
[8] http://core.materials.ac.uk/search/?filter=375,243
[9] GUASTALDI A C, ADORNO A T, BEATRICE C R, MONDELLI J, ISHIKIRIAMA A, LACEFIELD W. Effect of heat treatment on the structure of a Cu-Zn-Al-Ni system dental alloy [J]. Revista de Odontologia da Universidade de Sao Paulo, 1990, 4(4): 304-308.
[10] Materials for medical devices database [M]. ASM International - http://products.asminternational.org/meddev/index.aspx
[11] SEMOROZ A, HENRY S, RAPPAZ M. Application of the phase-field method to the solidification of hot-dipped galvanized coatings [J]. Metallurgical and Materials Transactions A, 2000, 31: 487-495.
[12] YAMASHITA M, INAGAKI J, YOSHIDA K, YAMAJI T, ISHIKAWA H, OKUMA T. Development of new 55%Al-Zn alloy coated steel sheets with excellent corrosion resistance and formability [J]. NKK Technical Review, 2002, 87: 7-11.
[13] WESELOWSKI J, GLUCHOWSKI W, CIURA L. New lead free zinc alloy unit mode hot-dip galvanising of steel products [J]. Archives of Metallurgy and Materials, 2006, 51(2): 283-288.
[14] KARPAGAVALLI R, BALASUBRAMANIAM R. Development of novel brasses to resist dezinfication [J]. Corrosion Science, 2007, 49(3): 963979.
[15] ZAID H R, ���֧�ڧ�ѧߧ�-���ݧڧ�ߧ� HATAB A M, IBRAHIM A M H. Properties enhancement of Al-Zn-Mg alloy by retrogression and re-aging heat treatment [J]. Journal of Mining and Metallurgy Section B, 2011, 47(1): 31-35.
[16] XU H, DU Y, ZHOU Y, ZHENG X, XIONG W, LIU S, SU X, YIN F. Phase equilibria of the Al-Ni-Zn system at 340 ��C [J]. International Journal of Materials Research, 2008, 99(6): 644-649.
[17] DU Y, LIU S, ZHANG L, XU H, ZHAO D, WANG A, ZHOU L. An overview on phase equilibria and thermodynamic modeling in multicomponent Al alloys: Focusing on the Al-Cu-Fe-Mg-Mn- Ni-Si-Zn system [J]. Calphad, 2011, 35: 427-445.
[18] ANSARA I, DUPIN N, LUKAS H L, SUNDMAN B. Thermodynamic assessment of the Al-Ni system [J]. Journal of Alloys and Compunds, 1997, 247: 20-30.
[19] An MEY S. Re-evaluation of the aluminum-zinc system [J]. Zeitschrift F��r Metallkunde, 1993, 84: 451-455.
[20] MIETTINEN J. Thermodynamic description of the Cu-Ni-Zn system above 600 ��C [J]. Calphad, 2003, 27: 263-274.
[21] DINSDALE A T, KROUPA A, VIZDAL J,  J, WATSON A, ZEMANOVA A. COST 531 database for lead-free solders, Ver. 3.0 (2008)
J, WATSON A, ZEMANOVA A. COST 531 database for lead-free solders, Ver. 3.0 (2008)
[22] BHAN S, LAL A. [R] Aluminium-nickel-zinc, MSIT ternary evaluation program [C]// MSIT Workplace, MSI. Materials Science International Services GmbH, Stuttgart; Document, 1993.
[23] FUSS V. On the constitution of ternary alloys of aluminium [J]. Zeitschrift F��r Metallkunde, 1924, 16: 24-25.
[24] MAYER A. Osservazioni sul sisterna zinco-altumlnio-nichel [J]. Gazzetta Chimica Italiana, 1947, 77: 326-330.
[25] FRIEBEL V R, LANTZ W J, ROE W P. Liquid solubilities of selected metals in Zinc-4% aluminium [J]. Transactions of the Metallurgical Society AIME, 1963, 56: 90-100.
[26] KURIKI Y, OCHIAI S O, YODOGAWA M, SUZUKI T. The effect of zinc addition on the mechanical properties of Ni3Al [J]. Journal of the Japan Institute of Metals, 1985, 26: 213-214.
[27] KISELEVA N N. The stability of Heusler phases [J]. Izvestia Akademie Nauk SSSR, Metally, 1987, 2: 213-218.
[28] CHOU K.C, WEI S K. A new generation solution model for predicting thermodynamic properties of a multicomponent system from binaries [J]. Metallurgical and Materials Transactions B, 1997, 28: 439-445.
[29] CHOU K C. A general solution model for predicting ternary thermodynamic properties [J]. Calphad, 1995, 19: 315-325.
[30]  LIU Y H. Comparative study of thermodynamic predicting methods applied to the Pb-Zn-Ag system [J]. Journal of Alloys and Compounds, 1998, 265: 176-184.
LIU Y H. Comparative study of thermodynamic predicting methods applied to the Pb-Zn-Ag system [J]. Journal of Alloys and Compounds, 1998, 265: 176-184.
[31]  L,
L,  R. Comparative thermodynamic study and phase equilibria of the Bi-Ga-Sn ternary system [J]. International Journal of Materials Research, 2007, 98(10): 1025-1030.
R. Comparative thermodynamic study and phase equilibria of the Bi-Ga-Sn ternary system [J]. International Journal of Materials Research, 2007, 98(10): 1025-1030.
[32] ZHANG G H, CHOU K C. General formalism for new generation geometrical model: application to the thermodynamics of liquid mixtures [J]. Journal of Solution Chemistry, 2010, 39: 1200-1212.
[33] REDLICH O, KISTER A T. Algebraic representation of thermodynamic properties and the classification of solutions [J]. Industrial and Engineering Chemistry, 1948, 40: 345-348.
Һ����ԪAl-Ni-Zn�Ͻ������ѧ���ʼ���
 1���� ��2��N. TALIJAN3��A. KOSTOV4��
1���� ��2��N. TALIJAN3��A. KOSTOV4�� 1
1
1. Technical Faculty, University of Belgrade, Bor, Serbia;
2. ���ϴ�ѧ ��ĩұ������ص�ʵ���ң���ɳ 410083;
3. University of Belgrade, Institute of Chemistry, Technology and Metallurgy, Belgrade;
4. Mining and Metallurgy Institute, Bor, Serbia
ժ Ҫ������������ѧԤ������°汾��ͨ��ģ����������Һ����ԪAl-Ni-Zn�Ͻ������ѧ���ʡ���9�����в�ͬNi��Zn, Zn��Al��Al��NĦ���ȵĽ�����1800~2000 K������ѧ���ʽ����о�����Һ����ԪAl-Ni-Zn�Ͻ�ľֲ��������Ħ������ѧ���ʽ����˲ⶨ�����Խ�����������ۡ�
�ؼ��ʣ���Ԫϵ��Al-Ni-Zn�Ͻ�����ѧ�����㣻ͨ����Һģ��
(Edited by YANG Hua)
Corresponding author:  ; Tel/Fax: +381-30-424-555/421-078; E-mail: dzivkovic@tf.bor.ac.rs
; Tel/Fax: +381-30-424-555/421-078; E-mail: dzivkovic@tf.bor.ac.rs
DOI: 10.1016/S1003-6326(11)61571-9