Trans. Nonferrous Met. Soc. China 24(2014) 4053-4058
Formation mechanism of periodic layered structure in Ni3Si/Zn system
Ya LIU1,2, Zhen DONG1,2, Yuan-yuan SONG1,2, Xu-ping SU1,2, Hao TU1,2
1. School of Materials Science and Engineering, Changzhou University, Changzhou 213164, China;
2. Jiangsu Key Laboratory of Materials Surface Science and Technology, Changzhou University, Changzhou 213164, China
Received 13 July 2014; accepted 9 October 2014
Abstract: The formation of periodic layered structure in Ni3Si/Zn diffusion couples with Zn in vapor or liquid state was investigated by SEM-EDS, FESEM and XRD. The results show that the diffusion path in solid-liquid reaction is Ni3Si/(T+γ)/γ/…T/γ/Ni4Zn12Si3/γ/…Ni4Zn12Si3/γ/Ni4Zn12Si3/δ…/Ni4Zn12Si3/δ/liquid-Zn, and the diffusion path in solid-vapor reaction is Ni3Si/θ/ (T+γ)/γ/…/T/γ/…T/γ/vapor-Zn. With increasing Zn diffusion flux, the diffusion reaction path moves toward the Zn-rich direction, and the distance from the Ni3Si substrate to the periodic layer pair nearest to the interface decreases. In the initial stage of both reactions, γ phase nucleates and grows within T matrix phase at first, and then conjuncts together to form a band to reduce the surface energy. Based on the experimental results and diffusion kinetics analysis, the microstructure differences were compared and the formation mechanism of the periodic layered structure in Ni3Si/Zn system was discussed.
Key words: Ni3Si; Zn; periodic layered structure; interface; diffusion
1 Introduction
Four types of morphologies may form during a solid-state reaction: simple-layered structure, rod-aggregate structure, interwoven-aggregate structure and periodic layered structure. Periodic layered structure was firstly discovered by OSINSKI et al in 1982 [1], where single-phase layer of FeSi and single-phase layer of FeZn10 displayed periodically in the reaction zone. Since then, this kind of structure has been found in several systems, including AgTi/Si [2], Co2Si/Zn [3], Fe3Si/Zn [1], GaAs/Co [4], NiCoFe(or NiCo)/Mg [5], SiC/Ni [6], SiC/Pt [7], SiO2/Mg [8], CuTi/Zn [9], Sn/(Ni-7%V) [10], (Sn-Cu)/(Ni-V) [11], Ni3Si/Zn [12-13], (U,Mo)/Al [14] and Au-12Ge/Ni [15].
Regarding to the morphology of the patterns, both continuous and discontinuous layer structures were observed. The latter consisted of precipitates that were periodically distributed in the reaction zone and the layers were discontinuous. The periodic pattern can be the assembly of the layer with a single phase α and the layer with a single phase β, such as in the systems SiC/Ni [6] and Sn/(Ni-7%V) [10]. And it can also be composed by two-phase layer of α+β and single-phase layer of α, as identified in the systems SiO2/Mg [8] and CuTi/Zn [9].
Several different models have been put forward to explain possible formation mechanisms of this interesting structure. Considering “cracks” shown in the periodic layer, OSINSKI et al [1] and CHEN et al [8,12] explained this phenomenon on the approach of the diffusion-induced stresses. However, in some systems such as SiC/Ni [6] and Sn/(Ni-7%V) [10], the periodic layered structures were composed of continuous layers, and no cracks were observed. From thermodynamic view, KAO and CHANG [16] proposed a chemical-potential instability model. It described the periodic thermodynamic instability of the phases at interface and the consequent nucleation of new reaction layers due to the thermodynamic driving force. This model has been employed for the periodic system (Au-12Ge)/Ni [15]. In this system, the periodic layered structure was the assembly of the layer with a single phase NiGe and the layer with a single phase (Au,Ni,Ge) alternated within the reaction zone. For the formation of two-phase layer in the initial reaction stage of period-layered structure formation, SU et al [13] suggested the diffusion- controlled precipitation and growth model. Due to the mobility differences of species, slow diffusion species will accumulate at the reaction front, leading to another phase precipitate within the matrix phase. However, no obvious evidence can be found from the etched samples. So, the formation mechanism of periodic layer structure composed of two-phase layer and single-phase layer remains controversial.
In the present work, two kinds of diffusion reactions (solid-Ni3Si/vapor-Zn and solid-Ni3Si/liquid-Zn) in Ni3Si/Zn system were designed to study the impact of Zn diffusion flux on the morphology of periodic layered structure, and the possible formation mechanism was proposed.
2 Experimental
Raw materials were Ni rod (purity 99.99%), Si powders (purity 99.99%) and Zn bulk (purity 99.99%). The Ni3Si alloy was prepared by the vacuum arc melting. This alloy was annealed in vacuum at 800 °C for 15 d to complete the homogenizing treatment. After that, it was sliced and polished, sealed together with Zn bulk in the vacuum quartz tube. The solid-liquid diffusion couple was annealed at 450 °C for 4-24 h in this study.
To prepare the solid-Ni3Si/vapor-Zn diffusion couple, Zn bulks were placed into the bottom of a quartz tube, and the corundum crucible was placed on zinc bulks with a quartz particle layer between Zn and crucible. The quartz tubes were vacuumed and kept at 450 °C for 5-60 d. Zn bulks were melted at this temperature. Due to the high vapor pressure of Zn, the Ni3Si alloy was surrounded by Zn vapor, so the solid- vapor diffusion couple formed. The diffusion couples after annealing were quenched into water at room temperature.
The microstructures and chemical composition were examined by a JSM-6510 scanning electron microscope (SEM) equipped with an energy dispersive X-ray spectroscope (EDS). The reaction products were identified by X-ray diffraction (XRD). Some samples were directly broken apart and the fresh features were studied by high-resolution field emission SEM (SUPRA55).
3 Results
3.1 Microstructure of solid-liquid diffusion couple
The cross-section micrograph of the solid-liquid diffusion couple annealed at 450 °C for 4 h is shown in Fig. 1(a), revealing that alternating reaction layers formed in the reaction zone. EDS and XRD analysis (Fig. 1(b)) indicated that the alternating pairs at the reaction front comprised bright layers (γ-NiZn3) and gray layers (T-Ni2Zn3Si). The samples were prepared by standard metallographic method without etching. According to CHEN et al [12], when the periodic layers were composed of nanoparticles, polishing during traditional samples’ preparation might have a negative effect on the morphology examination, and small second phase might be neglected. Drawing on experience of CHEN et al [12], the samples were also directly broken apart and the fracture surfaces were characterized further. Seen from high-resolution field emission SEM images of the fracture surface (Fig. 2(a)), three zones (namely L1, L2 and L3) with different features and thickness can be observed.
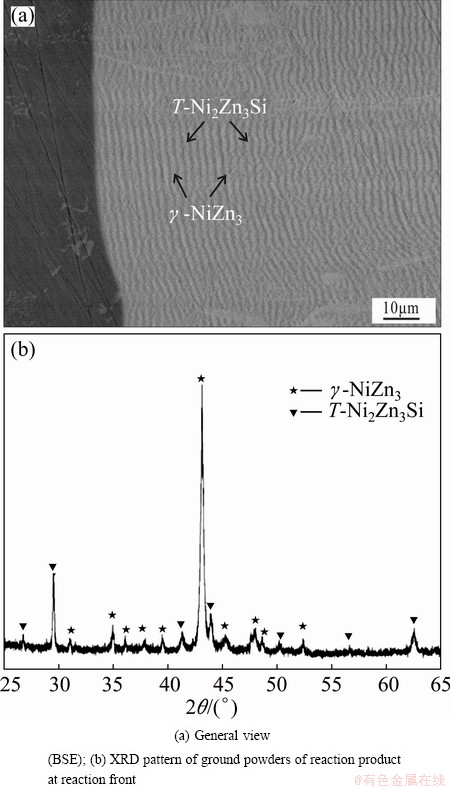
Fig. 1 Reactive diffusion zone in solid-Ni3Si/liquid-Zn diffusion couple annealed at 450°C for 4 h
In zone L1 (see Fig. 2(b)), the first layer close to Ni3Si was a two-phase layer, consisting of column-like T and nanoparticles γ. Some as-grown γ nanoparticles emerged between the first and the second two-phase layer. In the following, a single-phase layer consisting of γ equiaxed crystals and a two-phase layer of T+γ were delineated clearly. In zone L2 (see Fig. 2(c)), periodic pattern consisted of single-phase layer of Ni4Zn12Si3 and single-phase layer of γ, while in zone L3 (see Fig. 2(d)), single-phase layer of γ transformed into single-phase layer of δ-Ni3Zn22, and the single-phase layer of Ni4Zn12Si3 remained. The length of periodic-layer pair (λ) in zone L1 was about 500 nm, and those in zones L2 and L3 got close to 1 μm. Though the phase transformation in these three zones is similar to the previous observation [12], the morphology evolution of the γ phase from precipitated nanoparticles to a stripe composed by equiaxed crystals was not reported by them. This new result supported the previously proposed diffusion- controlled precipitation and growth mechanism on formation of periodic layered structure [13].

Fig. 2 SEM images of fracture surface of solid-Ni3Si/liquid-Zn diffusion couple annealed at 450 °C for 4 h

Fig. 3 SEM images of diffusion region in solid-vapor reaction at 450 °C for 1440 h (sample was un-etched)
3.2 Microstructure of solid-vapor diffusion couple
Figure 3 shows the SEM images of the solid-Ni3Si/vapor-Zn diffusion couple that reacted at 450 °C for 60 d. From Fig. 3(a), periodic layered structure formed in the reaction zone. From the zoomed image in Fig. 3(b), a transformation layer existed at the Ni3Si/ reaction zone interface. For its composition (Ni61.8Zn11.4Si26.8) was not convinced with the reported phases [17], we name this phase layer as θ phase. Next to θ phase was a two-phase area composed of T and γ phases. The white phase (γ) dispersed within the dark grey phase (T) matrix at first, then a compound layer with conjunctive γ particles emerged, at last a continuous single-phase layer of γ phase formed and existed periodically within the two-phase layer of T+γ. As diffusion went on, seen from Figs. 3(c) and (d), small γ grains merged into large grains to reduce the interfacial energies, γ particles in the two-phase region decreased and the periodic layered structure was finally composed of single-phase layer of T phase and single-phase layer of γ phase. In the solid-vapor reaction, distance (L) from Ni3Si substrate to the periodic layer pair nearest to the interface was around 30 μm. From Ni3Si substrate to the outside of reaction zone, the wavelength of modulation (λ) decreased from 5 to 3.5 μm progressively.
In solid-vapor diffusion couples that reacted for 120, 240 and 480 h, similar morphologies were observed: alternating reaction layers formed at a few micrometers away from the interface. With the decrease of annealing time (t), distance (L) from Ni3Si substrate to the periodic pair nearest to the interface decreased, as shown in Fig. 4. The relation between L and t can be fitted to
L=11.75+0.01t (1)
When t is decreased to 4 h, distance L is close to 11.8 μm, which is about 12 times of the distance examined in solid-liquid diffusion couple that reacted for the same time.
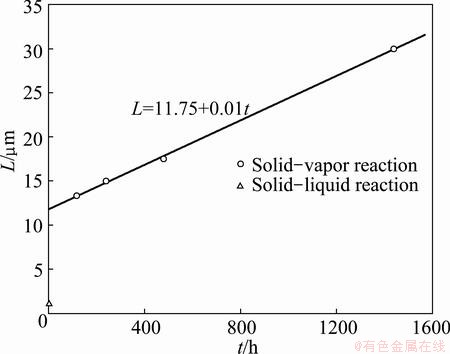
Fig. 4 Distance from Ni3Si substrate to periodic layer pair nearest to interface (L) vs annealing time (t)
4 Discussion
Compared solid-vapor reaction with solid-liquid reaction mentioned above, physical state of Zn has no effect on the formation of periodic layered structure, but some differences are evident. To explain the microstructure evolution during diffusion reaction, diffusion path theory is useful [18,19], and phase stability diagram can explain the structure from thermodynamic consideration [16,20]. However, due to lack of thermodynamic description of this system and the 450 °C isothermal section of the Zn-Ni-Si ternary system, simplified schematic diagrams with phases emerging in the reaction zone are presented in Fig. 5, in which continuous thin line is the tie-line linking neighboring two phases. In solid-liquid reaction, the reaction path (path I, blue double-dot line) is Ni3Si/ (T+γ)/γ/…T/γ/Ni4Zn12Si3/γ/…Ni4Zn12Si3/γ/Ni4Zn12Si3/δ…/ Ni4Zn12Si3/δ/liguid-Zn. However, for reactions in solid- vapor diffusion couple, the diffusion path across the interface is Ni3Si/θ/(T+γ)/γ/…/T/γ/…T/γ/vapor-Zn, indicated as path II (orange dash and dot line). The development of interfacial alternation layer relies on Zn diffusion flux from the end-member. In solid-vapor reaction, Zn atom deposits on the Ni3Si substrate by the vaporization of molten zinc, and the amount of Zn supplied to reaction zone is limited by the vaporization process. While in solid-liquid reaction, abundant Zn can be supplied from molten Zn. Hence, the reaction path moves toward the Zn-rich direction with the increase of Zn diffusion flux. Seen from Fig. 5, Zn diffusion flux has impact on the distance from the Ni3Si substrate to the periodic layer pair nearest to the interface. In solid- liquid reaction, Zn diffusion flux is large and Zn can be supplied to the interface quickly, then Ni can only diffuse within a short range, so periodic layer pair forms near the substrate alloy. In solid-vapor reaction, Zn diffusion flux is limited by Zn deposition process, so Ni gets more time to diffuse away, leading to periodic layer pair formed at a few micrometers away of the substrate alloy.
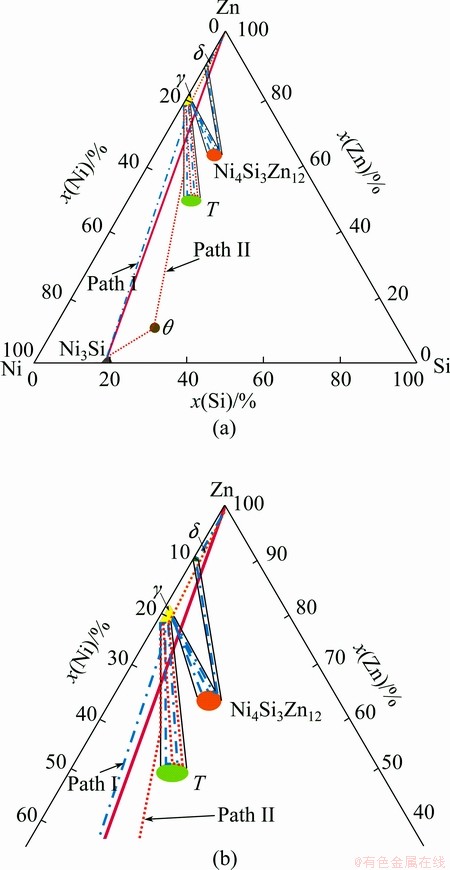
Fig. 5 Diffusion path in Ni3Si/Zn diffusion couple at 450 °C (a) and enlarged schematic diagram (b)
Beyond the difference of the reaction path formed in Ni3Si/Zn system with Zn in solid or liquid state, there are apparent similarities in the observed microstructure. In the initial stage of periodic layered structure formation, periodic layer pair is the assembling of two-phase layer and single-phase layer. As diffusion goes on, two-phase layer transforms into single-phase layer. Another important similarity is that in the formation of periodic layered structure, the single-phase layer of γ phase is not an immediate product, but progressive. For the distance (L) from the substrate to the first periodic pair in solid-vapor reaction is about 12 times of that in solid-liquid reaction, the morphology evolution of the γ phase from particles is dispersed within the T phase matrix to a continuous layer emerging periodical within the two-phase layer of γ+T can be evidenced clearly.
In the diffusion reaction zone with periodic patterns, the growth of the total reaction layers follows the parabolic relationship [1,13], indicating diffusion- controlled kinetics. The diffusivity of element X in the diffusion zone should be much higher than that of A or B in the A-B-X ternary system [17]. In the Ni3Si/Zn system, the mobilities of Zn, Ni and Si are quite different. Ni has low mobility, Si is relatively immobile, and Zn is not only the most mobile component in the product layer, but also has much higher mobility than Ni. CHEN et al [12] explained the formation of periodic layered structure based on mechanical-stress consideration. The difference between mobilities causes phases of T and γ to grow at different growth rates. Therefore, the slowly growing phase (T) will be under tensions as the γ phase grows rapidly, and the T phase finally splits off from the reaction front. In this model, single-phase layer of γ and two-phase layer of T+γ are assumed to form simultaneously at the reaction front, which is contradict to the present experimental observation, where single-phase layer of γ phase is formed by conjunction precipitated γ particles in the two-phase region.
Figure 6 schematically depicts the initial stage of the reactions in the Ni3Si/Zn couples that reacted at 450 °C. Zn is the dominant diffusion species in the system. The growth of the periodic layered structure involves long range diffusion of Zn, short range diffusion of Ni and local re-arrangement of Si. The growth of T phase is supported by the diffusion of Zn from the Zn side to the Ni3Si/(T+γ) interface, and the growth of γ phase is sustained by the diffusion of Ni from the Ni3Si substrate and the diffusion of Zn from the other side (Fig. 6(a)). When the two-phase layer of T+γ phase grows thicker and thicker, Ni diffusion will be retarded by the thicker two-phase layer, thus γ phase nucleates and grows within the short-range with Ni diffusion. In the ripening process, small γ grains merge into large grains to reduce the interfacial energies (Fig. 6(b)). When grew γ particles conjunct together to form a band, the two-phase (T+γ) region is divided into two parts. Thus, a new periodic pair is formed (Fig. 6(c)). The process is periodic, which results in the formation of the periodic layered structure (Fig. 6(d)). As diffusion goes on, γ particles within the two-phase layer merge into single-phase of γ gradually, leading to the periodic layer pair composed of a single-phase layer of T phase and a single-phase layer of γ phase. In solid-liquid reaction, diffusion flux of Zn is sufficient, then T phase transforms into Ni4Si3Zn12 and the single-phase layer of γ phase transforms to the single-phase layer of δ phase. In addition, a transformation layer θ phase emerges between Ni3Si substrate and two-phase layer of T+γ.
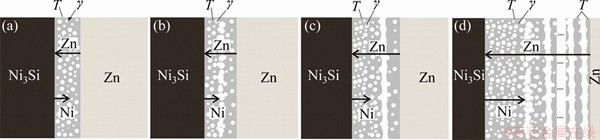
Fig. 6 Schematic drawings showing initial stage of formation of periodic layered structure in Ni3Si/Zn system
5 Conclusions
1) Physical state of Zn has no effect on the formation of periodic layered structure, which is identified in both solid-liquid reaction and solid-vapor reaction of Ni3Si/Zn. The diffusion reaction path moves toward the Zn-rich direction in solid-liquid reaction.
2) In the initial stage of periodic layered structure formation, the morphology evolution of γ phase from particles dispersing within the T phase matrix to a continuous layer emerging periodical within the two-phase layer of γ+T is observed. The periodic layered structure finally consists of single-phase layer.
3) New experimental results support the proposed diffusion-controlled precipitation and the growth mechanism on the formation of periodic layered structure.
References
[1] OSINSKI K, VRIEND A W, BASTIN F, LOO F. Periodic formation of FeSi bands in diffusion couples Fe(15 wt.% Si)-Zn [J]. Z Metallkd, 1982, 73: 258-261.
[2] RIJNDERS M R, van BEEK J A, KODENTSOV A A, van LOO F J. Solid state reactions in the Ag-Ti-Si system-periodic layer formation [J]. Z Metallkd, 1996, 87: 732-739.
[3] CHEN Y C, ZHANG Y G, CHEN C Q. Quantitative descriptions of periodic layer formation during solid state reactions [J]. MaterialsScienceandEngineering A, 2003, 362: 135-144.
[4] SHIAU F Y, CHANG Y A, LIN J C. Reactions between cobalt and gallium arsenide in bulk and thin-film forms [J]. MaterialsChemistryand Physics, 1992, 32: 300-309.
[5] DUNAEV S, ZVER’KOV S. Influence of high pressure on the formation of periodic regular structures in multicomponent diffusion zones [J]. Journal of the Less-Common Metals, 1989, 153: 143-150.
[6] BHANUMURTHY K, SCHMID-FETZER R. Interface reactions between silicon carbide and metals (Ni, Cr, Pd, Zr) [J]. Composites, 2001, 32: 569-574.
[7] RIJNDERS M R, KODENTSOV A A, van BEEK J A, VANDEN- AKKER J, van LOO F J J. Pattern formation in Pt-SiC diffusion couples [J]. Solid State Ionics, 1997, 95: 51-59.
[8] CHEN Y C, XU J, FAN X H, ZHANG X F, HAN L, LIN D Y, LI Q H, UHER C. The mechanism of periodic layer formation during solid-state reaction between Mg and SiO2 [J]. Intermetallics, 2009, 17: 920-926.
[9] WU Chang-jun, ZHU Chen-lou, SU Xu-ping, LIU Ya, WANG Jian-hua, PENG Hao-ping. Thermodynamical and kinetic investigation of the formation of periodic layered structure in TiCu/Zn interface reaction [J]. Acta Metallurgic Sinica, 2014, 50(8): 930-936. (in Chinese)
[10] CHEN S W, CHEN C C, CHANG C H. Interfacial reactions in Sn/Ni-7wt.%V couple [J]. Scripta Materialia, 2007, 56(6): 453-456.
[11] CHEN C C, CHEN S W, CHANG C H. Solid/solid interfacial reactions between Sn-0.7 wt% Cu and Ni-7 wt% V [J]. Journal of Material Research, 2008, 23: 1895-1901.
[12] CHEN Y C, ZHANG X F, RENY K, HAN L, LIN D Y, WANG Q P. Microstructure evolution of periodic layers formed during solid state reaction between Zn and Ni3Si [J]. Intermetallics, 2013, 36: 8-11.
[13] SU X P, GAO C P, LI Z F, LIU Y. The mechanism of periodic layered structure formation in Ni3Si/Zn system [J]. Journal of Phase Equilibria and Diffusion, 2013, 34(5): 416-420.
[14] MAZAUDIER F, PROYE C, HODAJ F. Further insight into mechanisms of solid-state interactions in UMo/Al system [J]. Journal of Nuclear Materials, 2008, 377: 476-485.
[15] LIN S K, TSAI M Y, TSAI P H, HSU B H. Formation of alternating interfacial layers in Au-12Ge/Ni joints [J]. Scientific Reports, 2014, 4: 1-6.
[16] KAO C, CHANG Y. A theoretical analysis for the formation of periodic layered structure in ternary diffusion couples involving a displacement type of reactions [J]. ActaMetallurgica et Materialia, 1993, 41: 3463-3472.
[17] XU H H, HU B, SUN W H, WANG P S, DU Y, XIONG X, JIN Z P. Phase equilibria of the Ni-Si-Zn system at 600°C [J]. Intermetallics, 2011, 19: 1089-1095.
[18] SU Xu-ping, LI Zhi, YIN Fu-cheng, HE Yue-hui, PAN Shi-wen. A study of the silicon reactivity in galvanizing [J]. Acta Metallurgica Sinica, 2008, 44: 718-722. (in Chinese)
[19] YIN F C, ZHAO M X, LIU Y X, HAN W, LI Z. Effect of Si on growth kinetics of intermetallic compounds during reaction between solid iron and molten aluminum [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(2): 556-561.
[20] ZHANG Chao, DU Yong. New method for calculating phase stability diagram of metal-Si-C system [J]. The Chinese Journal of Nonferrous Metals, 2006, 16(12): 1999-2004. (in Chinese).
Ni3Si/Zn体系中周期型层片组织的形成机理
刘 亚1,2,董 振1,2,宋媛媛1,2,苏旭平1,2,涂 浩1,2
1. 常州大学 材料科学与工程学院,常州 213164;
2. 常州大学 江苏省材料表面科学与技术重点实验室,常州 213164
摘 要:利用SEM-EDS、FESEM和XRD研究Ni3Si/Zn固-气扩散偶和Ni3Si/Zn固-液扩散偶中周期型层片组织的形成。结果表明,固-液反应中的扩散通道为Ni3Si/(T+γ)/γ/…T/γ/Ni4Zn12Si3/γ/…Ni4Zn12Si3/γ/Ni4Zn12Si3/δ…/ Ni4Zn12Si3/δ/Zn(液);固-气反应中的扩散通道为Ni3Si/θ/(T+γ)/γ/…/T/γ/…T/γ/Zn(气)。随着锌扩散流量的增大,扩散通道向富锌方向移动,Ni3Si基体与其最近邻的周期型层片对之间的距离减小。在周期型层片组织形成的初始阶段,γ相在T相基体中不断形核并长大,为降低表面能,最后变为连续的带状组织。根据实验结果和扩散动力学理论分析,探讨Ni3Si/Zn体系中周期型层片显微组织差异的形成原因和形成机理。
关键词:Ni3Si;Zn;周期型层片组织;界面;扩散
(Edited by Wei-ping CHEN)
Foundation item: Projects (51271040, 51171031) supported by the National Natural Science Foundation of China; Project supported by the Priority Academic Program Development of Jiangsu Higher Education Institutions, China
Corresponding author: Xu-ping SU; Tel: +86-13401569578; E-mail: sxping@cczu.edu.cn
DOI: 10.1016/S1003-6326(14)63568-8