Electrochemical oxidation of reactive brilliant orange X-GN dye on boron-doped diamond anode
��Դ�ڿ������ϴ�ѧѧ��(Ӣ�İ�)2018���8��
�������ߣ�κ��ƽ ���� ����ȫ ����� ÷���� �ܲ� YU Zhi-ming(��־��)
����ҳ�룺1825 - 1835
Key words��reactive brilliant orange X-GN; boron-doped diamond; film thickness; electrochemical oxidation
Abstract: In this study, the electrochemical oxidation of reactive brilliant orange X-GN dye with a boron-doped diamond (BDD) anode was investigated. The BDD electrodes were deposited on the niobium (Nb) substrates by the hot filament chemical vapor deposition method. The effects of processing parameters, such as film thickness, current density, supporting electrolyte concentration, initial solution pH, solution temperature, and initial dye concentration, were evaluated following the variation in the degradation efficiency. The microstructure and the electrochemical property of BDD were characterized by scanning electron microscopy, Raman spectroscopy, and electrochemical workstation; and the degradation of X-GN was estimated using UV-Vis spectrophotometry. Further, the results indicated that the film thickness of BDD had a significant impact on the electrolysis of X-GN. After 3 h of treatment, 100% color and 63.2% total organic carbon removal was achieved under optimized experimental conditions: current density of 100 mA/cm2, supporting electrolyte concentration of 0.05 mol/L, initial solution pH 3.08, and solution temperature of 60 ��C.
Cite this article as: MA Li, ZHANG Ming-quan, ZHU Cheng-wu, MEI Rui-qiong, WEI Qiu-ping, ZHOU Bo, YU Zhi-ming. Electrochemical oxidation of reactive brilliant orange X-GN dye on boron-doped diamond anode [J]. Journal of Central South University, 2018, 25(8): 1825�C1835. DOI: https://doi.org/10.1007/s11771-018-3872-y.

J. Cent. South Univ. (2018) 25: 1825-1835
DOI: https://doi.org/10.1007/s11771-018-3872-y
MA Li(����)1, ZHANG Ming-quan(����ȫ)1, ZHU Cheng-wu(�����)1, MEI Rui-qiong(÷����)1,
WEI Qiu-ping(κ��ƽ)1, ZHOU Bo(�ܲ�)2, YU Zhi-ming(��־��)1
1. State Key Laboratory of Powder Metallurgy, School of Materials Science and Engineering,Central South University, Changsha 410083, China;
2. School of Engineering and Materials Science, Queen Mary, University of London, United Kingdom
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2018
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2018
Abstract: In this study, the electrochemical oxidation of reactive brilliant orange X-GN dye with a boron-doped diamond (BDD) anode was investigated. The BDD electrodes were deposited on the niobium (Nb) substrates by the hot filament chemical vapor deposition method. The effects of processing parameters, such as film thickness, current density, supporting electrolyte concentration, initial solution pH, solution temperature, and initial dye concentration, were evaluated following the variation in the degradation efficiency. The microstructure and the electrochemical property of BDD were characterized by scanning electron microscopy, Raman spectroscopy, and electrochemical workstation; and the degradation of X-GN was estimated using UV-Vis spectrophotometry. Further, the results indicated that the film thickness of BDD had a significant impact on the electrolysis of X-GN. After 3 h of treatment, 100% color and 63.2% total organic carbon removal was achieved under optimized experimental conditions: current density of 100 mA/cm2, supporting electrolyte concentration of 0.05 mol/L, initial solution pH 3.08, and solution temperature of 60 ��C.
Key words: reactive brilliant orange X-GN; boron-doped diamond; film thickness; electrochemical oxidation
Cite this article as: MA Li, ZHANG Ming-quan, ZHU Cheng-wu, MEI Rui-qiong, WEI Qiu-ping, ZHOU Bo, YU Zhi-ming. Electrochemical oxidation of reactive brilliant orange X-GN dye on boron-doped diamond anode [J]. Journal of Central South University, 2018, 25(8): 1825�C1835. DOI: https://doi.org/10.1007/s11771-018-3872-y.
1 Introduction
Synthetic dyes are widely used in textile, leather, paper, food, and other industries [1]. However, many dye effluents are released in the industrial production process, which have become a refractory pollutant because of their high concentration, high chroma, and good chemical stability. Azo dyes are characterized by the presence of one or more ��N=N�� chromophoric groups [2]. Azo dyes account for over 70% of commercial dyestuff [3]; thus, they are rated as one of the most important class of synthetic dyes.
The reactive brilliant orange X-GN dye (hereafter referred to as X-GN) is a typical reactive azo dye that is widely used in the dyeing and printing industries [4] and needs to be treated urgently. However, X-GN is difficult to degrade using traditional physical, chemical, and biological methods [5] for various reasons. For example, secondary pollution arises because of the additives used during chemical treatment [6]; ozone and hypochlorite oxidation are not cost-effective due to the high cost of equipment and operation [7]; and the potential toxicity of reactive dyes show high resistance to microbial degradation [8]. Recently, advanced oxidation processes (AOPs) have attracted great attention for the removal of organic pollutants by generating strong oxidants in the effluents [3]. According to the previous reports, the research works on X-GN treatment concentrated on one of the AOPs, called the Fenton method. CHEN et al [9] investigated photocatalytic degradation of X-GN using Fe-Mt/H2O2 as heterogeneous photo- Fenton catalysis. WU et al [10] accomplished the treatment of X-GN by a heterogeneous Fenton system using activated carbon-FeOOH catalyst and H2O2. LI et al [11] studied Fenton degradation of X-GN using activated carbon-supported zero-valent iron catalyst. However, in this homogeneous Fenton process, iron ions acting as a catalyst are dissolved in water, which results in sludge disposal problems [12].
Electrochemical oxidation, as one of the AOPs, has been widely studied and applied because effluents can be effectively degraded without additional chemicals [13]. There are two methods of conducting the electrochemical oxidation process. One is direct oxidation, in which the organic matter is oxidized directly on the anode��s surface, and the other is indirect oxidation, in which the oxidation of organic matter is mediated by electrogenerated oxidants, such as hydroxyl radicals (��OH), active chlorine, and peroxide [14]. An anodic material is the key element in the degradation effect of organic effluent treatments; thus, various anodes, such as graphite [15], noble metal [16], and metal oxide [17, 18], have been studied. However, these anodes have the corresponding disadvantages of activity, cost, and service life [19�C21]. The boron-doped diamond (BDD) anode has attracted great interest due to its excellent properties, such as high overpotential of oxygen evolution, low adsorption, and good stability [22�C24]. Further, it has been extensively studied for dye effluent treatments. YAVUZ et al [25] studied anodic oxidation of the reactive black 5 dye on a BDD electrode in a bipolar trickle tower reactor; 97% color, 51% chemical oxygen demand (COD), and 29.3% total organic carbon (TOC) removals were achieved. SOLANO et al [26] studied decontamination of real textile effluents on a BDD electrode; color and COD removal reached the Brazilian legal requirements after 12 h of treatment.
According to the previous reports, very few studies are conducted on electrochemical oxidation of X-GN using the BDD anode, and few studies reported the effect of BDD properties on dye effluent treatments. Electric energy consumption is the primary focus of electrochemical oxidation processes [27]; thus, the best operating parameters and performance of BDD electrodes need to be investigated for attaining a highest degradation efficiency. In this study, BDD electrodes were prepared with three different deposition time by hot filament chemical vapor deposition (HFCVD). The effects of varying operating parameters, such as film thickness, current density, supporting electrolyte concentration, initial solution pH, temperature, and initial dye concentration were investigated. Further, the UV-Vis spectrophotometer, TOC, and energy consumption were used for the characterization of the treatment effect.
2 Experiment
2.1 Preparation and characterization of BDD/Nb
A batch of BDD films deposited on Nb substrates (30 mm��20 mm��1 mm) was prepared by HFCVD. The HFCVD process and the pretreatment of substrates have been discussed in previous literatures [28, 29]. The mixture of 1.0 mL/min CH4, 99 mL/min H2, and 0.2 mL/min B2H6 (diluted to 5% with hydrogen) fed for boron addition was used as a reaction gas system. The substrate temperature measured by a thermocouple was set at around 800 ��C, and the total pressure of BDD deposition was maintained at 3 kPa. The BDD films were deposited with deposition durations of 3, 6 and 12 h (named as 3 h BDD, 6 h BDD, and 12 h BDD) to obtain films with the thicknesses of 899 nm, 2.8 ��m and 3.6 ��m, respectively.
The morphology and grain size of the BDD films were observed using a FEI Sirion200 field emission SEM system. The boron doping level and phase purity were evaluated by Raman spectroscopy with LabRAM ARAMIS (484 nm and 10 mW). Cyclic voltammetric measurements were performed using the CHI 660E (Shanghai CH Instruments, China) to characterize the overpotential of BDD oxygen evolution.
2.2 Preparation of X-GN solution
X-GN (Figure 1) with the molecular formula C19H9Na3Cl2N6O10S3, was purchased from Shanghai Eighth Dyestuff Chemical Plant and used without further purification. The structural formula is shown as follows: The dye solution used in all trials was prepared using deionized water. NaSO4 was added to the dye solution as a supporting electrolyte due to the poor conductivity of the dye solution. H2SO4 and NaOH were used for pH adjustment. The pH value of the dye solution was tested with the pH meter (ST20, OHAUS Company, American). All reagents were bought from Sinopharm Chemical Reagent Co., Ltd, Shanghai.
2.3 Electrochemical oxidation
The bulks of the electrochemical oxidation experiments were conducted in a glass electrolytic cell containing 500 mL X-GN solution by direct- current power supply (RD-3020, Suzhou Wanruida Electric Company, China). The electrolytic cell was placed on a magnetic stirrer (WH220-HT, WIGGENS Company, Germany), which adjusts the stirring rate and temperature of the solution. The BDD/Nb samples were encapsulated as anode and the stainless steel plates were used as cathode. The distance between anode and cathode was maintained at 1 cm. Every experiment in this study was repeated three times to avoid errors, and the average of the three experiments was considered as the final result.
2.4 Analyses
A batch of 10 mL dye solution samples was taken from the electrolytic cell, at regular intervals of 30 min during the experiments, for analyses. A UV-Vis spectrophotometer (UV-2600, Shimadzu, Japan) was used to measure the concentration of X-GN because of the variations in X-GN concentration determined by absorbance at the maximum absorption wavelength of 479 nm. The degradation efficiency of the X-GN dye solution was calculated by the following equation [30]:
Color removal=(A0-At)/A0��100% (1)
where A0 and At denote the absorbances before electrochemical oxidation and at time t, respectively.
The mineralization degree of the X-GN dye solution was evaluated by the variation in TOC concentration using a TOC analyzer (TOC-L, Shimadzu, Japan). The specific energy consumption per volume of the treated effluent was estimated in kW��h/m3 by the following equation [31]:
Ec=UIt/(1000V) (2)
where U denotes the mean applied voltage (V); I is the current (A); t is the duration of treatment (h); and V is the volume of the treated effluent (m3).
3 Results and discussion
3.1 Effect of electrode with different deposition time
Figure 2 show the SEM images of 3 h BDD,6 h BDD, 12 h BDD, respectively. The continuous grains and clear grain boundaries without cracks or voids can be observed clearly in these images. Meanwhile, the grain size increases and tends to be more homogeneous as the deposition time increases. This can be explained by the fact that crystals are composed of irregular grains in the initial nucleation stage of growth and the grain size increases through columnar growth mode with time [32].
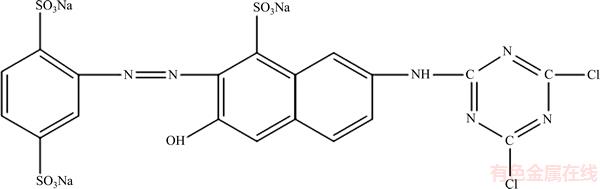
Figure 1 Structural formula of X-GN

Figure 2 SEM images of BDD films with different deposition time:
Figure 3 displays the Raman spectra of the BDD films. All the samples show a sharp diamond peak at 1331 cm�C1, and any obvious peak can be hardly observed around 1580 cm-1 related to the non-diamond carbon phase, indicating that the samples are of high quality [33]. In addition, the two bands assigned to the boron atoms doping appear at approximately 500 cm�C1 and 1220 cm�C1 [34]. At 1331 cm�C1, the peak intensity gradually decreases with an increase in the deposition time, while the peak intensities of 500 cm�C1 and 1220 cm�C1 increase with an increase in the deposition time. This could be attributed to the Fano effect of boron doping, indicating an increase in boron atoms doped in the BDD film with an increase in thickness. Further, this result is consistent with the previous reports [35, 36], and will affect the electrocatalytic properties of the BDD electrode. The ratios of boron in 3 h BDD, 6 h BDD, and 12 h BDD films were calculated using Eq. (3) [37], and they are 5.27��1019/cm3, 1.71��1020/cm3 and 1.97��1020/cm3, respectively.
[B]=8.44��1030exp(�C0.048W) (3)
where W is the wave number of the Lorentzian component of the 500 cm�C1 broad peak, and it can be obtained from the Raman spectroscopy results.

Figure 3 Raman spectra of BDD films with different deposition time
The cyclic voltammogram of the 3 h BDD, 6 h BDD, and 12 h BDD films in 0.1 mol/L H2SO4 solution is shown in Figure 4. The higher overpotential of oxygen evolution than that of other anode materials guarantees the high current efficiency during the degradation process. The oxygen evolution potentials of 3 h BDD, 6 h BDD and 12 h BDD are 2.6, 3.5 and 3.6 V, respectively. The overpotential increases with increasing deposition time; however, the overpotentials of oxygen evolution of 6 h BDD and 12 h BDD are close.
Figure 5 shows the changes in the UV-Vis spectra and color of the X-GN solution during the degradation process using the 6 h BDD electrode. X-GN can be characterized by the absorption peak at 479 nm attributed to the N=N chromophoric group in the visible region, and by the absorption peak at 298 nm attributed to the ultraviolet region [12]. The peak at 479 nm gradually became weak with time and almost reached zero absorbance values after 1.5 h of treatment, indicating that the N=N chromophoric groups were completely destroyed. The peak at 298 nm also declined with time and nearly disappeared after 2.5 h, indicating that the naphthalene ring also can be destroyed effectively by electrochemical oxidation. Moreover, the absorbance between 200�C250 nm is related to the intermediate products, such as phenol, fumaric acid, and oxalic acid [2]. Their values decreased slowly with time, indicating that the dye molecule was breakdown first, and then the intermediate products were mineralized [12].

Figure 4 Cyclic voltammograms of BDD films with different deposition time at scan rate of 100 mV/s in 0.1 mol/L H2SO4 solution
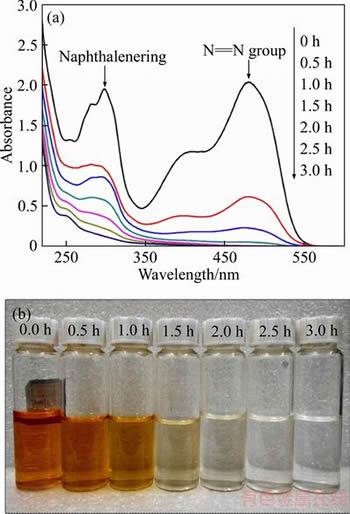
Figure 5 UV-Vis spectra of X-GN during degradation process (a) and color variations during degradation process (b) (The experimental conditions: 100 mg/L X-GN, J=100 mA/cm2, 0.05 mol/L of NaSO4, pH 6.47(nature value), and room temperature)
The degradation effects of X-GN using the 3 h BDD, 6 h BDD, and 12 h BDD films were investigated. The degradation efficiency is affected by the performance parameters of the BDD electrodes, such as film thickness, boron concentration, and grain size. For example, FENG et al [21] and BOGDANOWICZ et al [38] observed that high boron concentration can improve film conductivity, catalytic activity, and degradation efficiency of effluent treatments. KROTOVA et al [39] reported that the catalytic activity can be improved with an increase in the crystallite size. As shown in Figure 6, the degradation efficiency of 3 h BDD is lower than that of the others and the degradation efficiency of 6 h BDD and 12 h BDD are close.
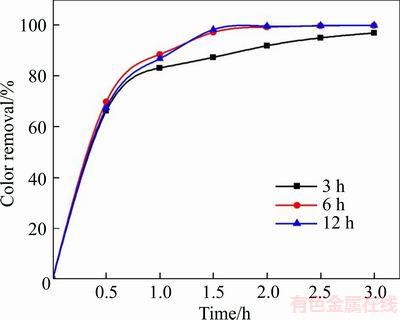
Figure 6 Variation of degradation efficiency with film thickness (The experimental conditions: 100 mg/L X-GN, J=100 mA/cm2, 0.05 mol/L of NaSO4, pH nature, and room temperature)
In conclusion, the electrocatalytic properties of the BDD electrodes are enhanced due to the growth of grain, the increase of boron dopant, and overpotential oxygen evolution when the deposition time of the BDD films increases from 3 h to 12 h, while the properties and the degradation efficiency of 12 h BDD were very closed to those of 6 h BDD. It can be speculated that the electrocatalytic properties are enhanced with the film thickness of BDD increasing; however, the degradation efficiency of X-GN is almost unchanged when the film thickness reaches a certain value. Thus, 6 h BDD is chosen to conduct the following experiments considering cost.
3.2 Effect of current density
The influence of current density on the degradation efficiency was investigated using applied current densities at 50, 100 and 150 mA/cm2, and the results are depicted in Figure 7. It can be observed that the degradation efficiency was fairly high at the initial stage of degradation; however, it went down gradually as degradation continues. For instance, 80.4% of color removal can be reached after the first half hour, while the color removal increased from 80.4% to 97% during degradation for the next half hour at current density of 150 mA/cm2. In the initial period of electrolysis, the electrolysis is under current-limited control due to the high dye concentration, and nearly all the applied current is used to attack the dye molecules. However, the electrolysis is then gradually controlled by mass transport as the dye concentration reduces, resulting in the decrease of degradation efficiency, which can be attributed to the appearance of side reactions, such as oxygen evolution (reaction (4)) [40].
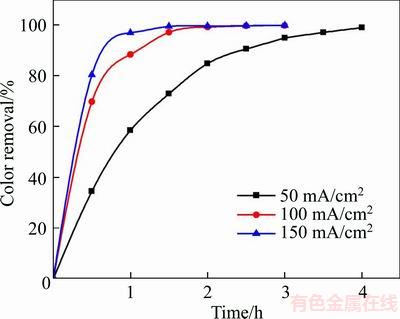
Figure 7 Variation of degradation efficiency with current density (The experimental conditions: 100 mg/L X-GN, 0.05 mol/L of NaSO4, pH nature, and room temperature)
Furthermore, the increase of degradation efficiency with current density can be obviously observed when other conditions were identical. This can be explained that the rate of electron and mass transfer and production of ��OH increased significantly in higher current density, which is beneficial to indirect oxidation of effluents (reactions (5) and (6), R denoted organic effluent). Further, the formation of mediated oxidants, such as S2O82�C, H2O2, and O3, can be considerably enhanced by increasing the current density using reactions (7)�C(9) [41]. To be specific, 58.5%, 88.5% and 97% of color removal were obtained under current densities of 50, 100 and 150 mA/cm2, respectively. However, there is no great improvement of degradation efficiency by increasing the current density from 100 to 150 mA/cm2. This case can be attributed to the mass transport limitation [40]. Additionally, under the current density of 100 and 150 mA/cm2, the color removal reached 99% after 2 h of degradation and the energy consumption values were 22.9 and 47.8 kW��h/m3, respectively. Considering the above analyses, the current density of 100 mA/cm2 was considered as the best parameter.
BDD(��OH)��BDD+1/2O2+H++e (4)
BDD+H2O��BDD(��OH)+H++e (5)
BDD(��OH)+R��BDD+mCO2+nH+ (6)
2SO42�C��S2O82�C+2e (7)
2H2O��H2O2+2e+2H+ (8)
3H2O��O3+6e+6H+ (9)
3.3 Effect of supporting electrolyte concentration
Electrolysis experiments of 0.025, 0.05 and 0.100 mol/L NaSO4 concentration were conducted to investigate the effect of supporting electrolyte concentration on degradation efficiency as shown in Figure 8. It was evidently found that increasing supporting electrolyte concentration lead to the raise of degradation efficiency. The conductivity of solution is enhanced by adding supporting electrolyte, thus the electron transfer and the electrolysis reaction are accelerated [42]. Moreover, the strong oxidant S2O82�C is generated more as reaction (5), thus, stimulating the oxidation reaction further and improving the degradation efficiency.
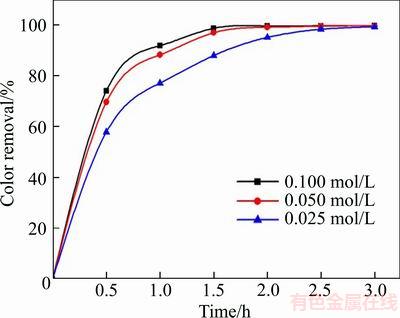
Figure 8 Variation of degradation efficiency with current density (The experimental conditions: 100 mg/L X-GN, J=100 mA/cm2, pH nature, and room temperature)
As shown in Figure 8, after 1.5 h of electrolysis, 88.21% of color removal occurred under 0.025 mol/L electrolyte concentration, while the color removal reached to 97.2% and 98.9%, under 0.05 mol/L and 0.1 mol/L electrolyte concentration, respectively. We can assume that the influence of electrolysis concentration weakens gradually or even disappears when the electrolysis concentration reaches a certain level. Therefore, 0.05 mol/L was chosen as the optimum electrolyte concentration to reduce the cost of chemical without sacrificing the degradation efficiency.
3.4 Effect of initial solution pH
A set of experiments were conducted to evaluate the effect of initial solution pH between 3.05 and 10.94, and the results are shown in Figure 9. The degradation efficiency is favored at acidic initial pH values and inhibited at alkali conditions. Specifically, 94.8%, 92.8%, 88.5%, 85% and 83.1% of color removal were obtained at initial pH of 3.05, 5.08, 6.47 (nature value, without adjustment), 8.95, and 10.94, respectively, after 1 h of treatment. This is because the formation of ��OH and S2O82�C can be promoted at acidic conditions, while they might be inhibited at an alkali condition [29]. The initial solution pH of 3.05 was the optimal pH value of this experiment.
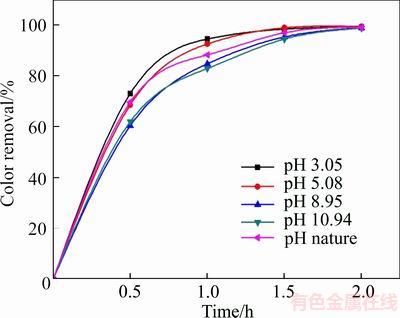
Figure 9 Variation of degradation efficiency with initial pH value (The experimental conditions: 100 mg/L X-GN, J=100 mA/cm2, 0.05 mol/L of NaSO4, and room temperature)
3.5 Effect of solution temperature
Figure 10 illustrates the influence of solution temperature on the degradation efficiency by varying the temperature of solution between 30 and 60 ��C. As shown in Figure 10, a higher solution temperature led to higher degradation efficiency. For instance, the color removal reached 69.9%, 73.9%, 76.8% and 82.9% at 30, 40, 50 and 60 ��C, respectively, after half an hour of electrolysis. This is because the production rate of ��OH is improved at elevated temperature and this causes an acceleration of the electrolysis reaction [43]. In addition, the applied voltage declined with the increasing rate of mass transfer increased at higher temperature; thus, the energy consumption was reduced. Consequently, the ideal temperature of this experiment is 60 ��C.
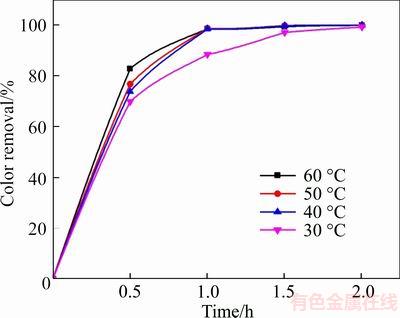
Figure 10 Variation of degradation efficiency with solution temperature (The experimental conditions:100 mg/L X-GN, J=100 mA/cm2, 0.05 mo/L of NaSO4, and pH nature)
3.6 Effect of X-GN concentration
Various studies were conducted by varying the X-GN concentration between the 50�C150 mg/L range to elucidate the effect of X-GN concentration on the degradation efficiency, as depicted in Figure 11. It can be observed that higher degradation efficiency can be achieved with a lower X-GN solution. For example, 99.45% of color removal occurred for 50 mg/L X-GN after 1 h of electrolysis, compared to 88.5% and 79% of color removal for 100 mg/L and 150 mg/L, respectively. This could be ascribed to the fact that the same amount of oxidants is required in the same experiment conditions; thus, it takes longer time to reach the same color removal for X-GN of higher concentration [25,44].
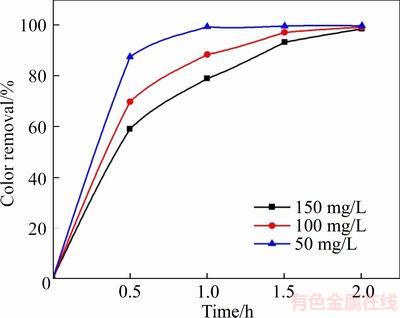
Figure 11 Variation of degradation efficiency with X-GN concentration (The experimental conditions: 100 mg/L X-GN, J=100 mA/cm2, 0.05 mol/L of NaSO4, and 3.05 pH)
It is also worth noting that the mass of X-GN reduced to 24.86, 44.26 and 59.24 mg in the X-GN concentration of 50, 100 and 150 mg/L, respectively after 1 h of treatment. It can be concluded that more X-GN molecules are destroyed at the same time for high X-GN concentration, which can be attributed to the lower limitation of mass transport in higher X-GN concentration [45].
3.7 Energy consumption and TOC removal
The optimal experimental conditions have been determined through a series of experiments. The electrochemical oxidation of 100 mg/L X-GN was conducted under these conditions, and the energy consumption and TOC concentration were estimated. The results demonstrate that 99% of color removal is obtained with 10.6 kW��h/m3 of energy consumption after 1 h of treatment and 63.2% of TOC removal is reached with 31.2 kW��h/m3 of energy consumption after 3 h of electrolysis. The curve of TOC concentration as a function of time is displayed in Figure 12.
4 Conclusions
1) The 3 h BDD, 6 h BDD, and 12 h BDD films were prepared on Nb substrates by the HFCVD method. The performance of the BDD electrodes was characterized, and the electrolysis of X-GN using three types of BDD electrodes was conducted. The results show that the electrocatalytic properties are enhanced with the film thickness of BDD increasing; however, the degradation efficiency of X-GN is almost unchanged when the film thickness reaches a certain value.
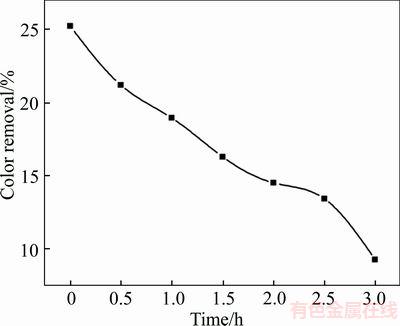
Figure 12 Variation of TOC concentration with time (The experimental conditions: 100 mg/L X-GN, I=ted to the ultraviolet region 100 mA/cm2, 0.05 mol/L of NaSO4,pH nature, and 60 ��C)
2) The influence of the varying operating parameters on the degradation efficiency was investigated based on color removal using 6 h BDD. Finally, the degradation results degradation of the 100 mg/L X-GN solution indicated that the optimum experimental conditions were as follows: current density 100 mA/cm2, 0.05 mol/L supporting electrolyte concentration, initial solution pH 3.08, and 60 ��C of solution temperature. Under these conditions, 100% of color removal and 63.2% of TOC removal were achieved with 31.2 kW��h/m3 after 3 h of electrolysis. These results prove that the X-GN solution can be degraded effectively on the BDD anode. Considering the energy consumption and high efficiency in the initial stage, this technique can be used as a pretreatment in other processes.
References
[1] FORGACS E, CSERH TI T, OROS G. Removal of synthetic dyes from wastewaters: A review [J]. Environment International, 2004, 30(7): 953�C971. DOI: 10.1016/j.envint. 2004.02.001.
TI T, OROS G. Removal of synthetic dyes from wastewaters: A review [J]. Environment International, 2004, 30(7): 953�C971. DOI: 10.1016/j.envint. 2004.02.001.
[2] BARROS W R P, STETER J R, LANZA M R V, MOTHEO A J. Degradation of amaranth dye in alkaline medium by ultrasonic cavitation coupled with electrochemical oxidation using a boron-doped diamond anode [J]. Electrochimica Acta, 2014, 143(10): 180�C187. DOI: 10.1016/j.electacta.2014. 07.141.
[3] EL-GHENYMY A, CENTELLAS F, GARRIDO J A, RODRIGUEZ R M, SIRES I,CABOT P L,BRILLAS E. Decolorization and mineralization of Orange G azo dye solutions by anodic oxidation with a boron-doped diamond anode in divided and undivided tank reactors [J]. Electrochimica Acta, 2014, 130: 568�C576. DOI: 10.1016/j.electacta.2014.03.066.
[4] ZHONG Deng-jie, YANG Ji, XU Yun-lan, JIA Jin-ping, WANG Ya-lin,SUN Tong-hua. De-colorization of reactive brilliant Orange X-GN by a novel rotating electrochemical disc process [J]. Journal of Environmental Sciences, 2008, 20(8): 927�C932. DOI: 10.1016/S1001-0742(08)62188-9.
[5] MONDAL S. Methods of dye removal from dye house effluent: An overview [J]. Environmental Engineering Science, 2008, 25(3): 383�C396. DOI: 10.1089/ees.2007. 0049.
[6] KOPARAL A S, YAVUZ Y, BAKIR  TVEREN U B. Electroadsorption of Acilan Blau dye from textile effluents by using activated carbon-perlite mixtures [J]. Water Environment Research A: 2002, 74(6): 521�C525. DOI: 10.2175/106143002X140314.
TVEREN U B. Electroadsorption of Acilan Blau dye from textile effluents by using activated carbon-perlite mixtures [J]. Water Environment Research A: 2002, 74(6): 521�C525. DOI: 10.2175/106143002X140314.
[7] CHEN Jun-shui, LIU Mei-chuan, ZHANG Ji-dong, XIAN Yue-zhong, JIN Li-tong. Electrochemical degradation of bromopyrogallol red in presence of cobalt ions [J]. Chemosphere, 2003, 53(9): 1131�C1136. DOI: 10.1016/ S0045-6535(03)00581-2.
[8] PETRUCCI E, MONTANARO D. Anodic oxidation of a simulated effluent containing Reactive Blue 19 on a boron-doped diamond electrode [J]. Chemical Engineering Journal, 2011, 174(2, 3): 612�C618. DOI: 10.1016/j.cej.2011. 09.074.
[9] CHEN Qiu-qiang, WU Ping-xiao, LI Yuan-yuan, ZHU Neng-wu, DANG Zhi. Heterogeneous photo-Fenton photodegradation of reactive brilliant orange X-GN over iron-pillared montmorillonite under visible irradiation [J]. Journal of Hazardous Materials, 2009, 168(2, 3): 901�C908. DOI: 10.1016/j.jhazmat.2009.02.107.
[10] WU Jin-hua, LIN Guang-hui, LI Ping, YIN Wei-zhao, WANG Xiang-de, YANG Bo. Heterogeneous Fenton-like degradation of an azo dye reactive brilliant orange by the combination of activated carbon-FeOOH catalyst and H2O2 [J]. Water Science & Technology A: Journal of the International Association on Water Pollution Research, 2013, 67(3): 572�C578. DOI: 10.2166/wst.2012.596.
[11] LI Dong-yu, ZHU Ji-shu, WU Jin-hua, YIN Wei-zhao, LIANG Hao, LIN Guang-hui. Development of an activated carbon-supported zero-valent iron catalyst (AC-Fe-0) for enhancing degradation of reactive brilliant orange and reducing iron sludge production [J]. Environmental Progress & Sustainable Energy, 2016, 35(4): 949�C956. DOI: 10.1002/ep.12298.
[12] CHEN Qiu-qiang, WU Ping-xiao, ZHI Dang, ZHU Neng-wu, LI Ping, WU Jin-hua, WANG Xiang-de. Iron pillared vermiculite as a heterogeneous photo-Fenton catalyst for photocatalytic degradation of azo dye reactive brilliant orange X-GN [J]. Separation & Purification Technology, 2010, 71(3): 315�C323. DOI: 10.1016/j.seppur.2009.12.017.
[13]  G, COMNINELLIS C. Kinetic modelling of the electrochemical mineralization of organic pollutants for wastewater treatment [J]. Journal of Applied Electrochemistry, 2008, 38(1): 7�C16. DOI: 10.1007/s10800- 007-9365-6.
G, COMNINELLIS C. Kinetic modelling of the electrochemical mineralization of organic pollutants for wastewater treatment [J]. Journal of Applied Electrochemistry, 2008, 38(1): 7�C16. DOI: 10.1007/s10800- 007-9365-6.
[14] COMNINELLIS C. Electrocatalysis in the electrochemical conversion/combustion of organic pollutants for waste water treatment [J]. Electrochimica Acta, 1994, 39(11, 12): 1857�C1862. DOI: https://doi.org/10.1016/0013-4686(94) 85175-1.
[15] ABUZAID N S. Electrochemical oxidation of phenol using graphite anodes [J]. Separation Science and Technology, 1999, 34(4): 699�C708. DOI: 10.1081/SS-100100675.
[16] CHEN Guo-hua. Electrochemical technologies in wastewater treatment [J]. Separation & Purification Technology, 2004, 38(1): 11�C41. DOI: 10.1016/j.seppur.2003.10.006.
[17] AWAD H S, GALWA N A. Electrochemical degradation of Acid Blue and Basic Brown dyes on Pb/PbO2, electrode in the presence of different conductive electrolyte and effect of various operating factors [J]. Chemosphere, 2005, 61(9): 1327�C1335. DOI: 10.1016/j.chemosphere.2005.03.054.
[18] WU W Y, HUANG Z H, LIM T T. Recent development of mixed metal oxide anodes for electrochemical oxidation of organic pollutants in water [J]. Applied Catalysis A: General, 2014, 480: 58�C78. DOI: 10.1016/j.apcata.2014.04.035.
[19] KRAFT A, STADELMANN M, BLASCHKE M. Anodic oxidation with doped diamond electrodes: A new advanced oxidation process [J]. Journal of Hazardous Materials, 2003, 103(3): 247�C261. DOI: 10.1016/j.jhazmat.2003.07.006.
[20] BENISEK M, MARCANO G, BETZLER C. Recent developments of electro-oxidation in water treatment��A review [J]. Journal of Electroanalytical Chemistry, 2001, 37(2): 127�C134. DOI: 10.1016/j.jelechem.2015.06.016.
[21] FENG Yu-jie, LV Jiang-wei, LIU Jun-feng, GAO Na, PENG Hong-yan, CHEN Yu-qiang. Influence of boron concentration on growth characteristic and electro-catalytic performance of boron-doped diamond electrodes prepared by direct current plasma chemical vapor deposition [J]. Applied Surface Science, 2011, 257(8): 3433�C3439. DOI: 10.1016/ j.apsusc.2010.11.041.
[22] PANIZZA M, CERISOLA G. Application of diamond electrodes to electrochemical processes [J]. Electrochimica Acta, 2005, 51(2): 191�C199. DOI: 10.1016/j.electacta.2005. 04.023.
[23] TSANTAKI E, VELEGRAKI T, KATSAOUNIS A, MANTZAVINOS D. Anodic oxidation of textile dyehouse effluents on boron-doped diamond electrode [J]. Journal of Hazardous Materials, 2011, 207�C208(12): 91�C96. DOI: 10.1016/j.jhazmat.2011.03.107.
[24] DENG Ze-jun, LONG Hang-yu, WEI Qiu-ping, YU Zhi-ming, ZHOU Bo, WANG Yi-jia, ZHANG Long, LI Sha-sha, MA Li, XIE You-neng, MIN Jie. High-performance non-enzymatic glucose sensor based on nickel- microcrystalline graphite-boron doped diamond complex electrode [J]. Sensors and Actuators B-Chemical, 2017, 242: 825�C834. DOI: 10.1016/j.snb.2016.09.176.
[25] YAVUZ Y, SHAHBAZI R. Anodic oxidation of Reactive Black 5 dye using boron doped diamond anodes in a bipolar trickle tower reactor [J]. Separation & Purification Technology, 2012, 85(6): 130�C136. DOI: 10.1016/ j.seppur.2011.10.001.
[26] SOLANO A M S, ARA JO C K C D, MELO J V D, PERALTA-HERNANDEZ J M, da SILVA D R, MARTINEZ-HUITLE C A. Decontamination of real textile industrial effluent by strong oxidant species electrogenerated on diamond electrode: Viability and disadvantages of this electrochemical technology [J]. Applied Catalysis B: Environmental, 2013, 130�C131: 112�C120. DOI: 10.1016/ j.apcatb.2012.10.023.
JO C K C D, MELO J V D, PERALTA-HERNANDEZ J M, da SILVA D R, MARTINEZ-HUITLE C A. Decontamination of real textile industrial effluent by strong oxidant species electrogenerated on diamond electrode: Viability and disadvantages of this electrochemical technology [J]. Applied Catalysis B: Environmental, 2013, 130�C131: 112�C120. DOI: 10.1016/ j.apcatb.2012.10.023.
[27] AQUINO J M, PEREIRA G F, ROCHAFILHO R C, BOCCHI N, BIAGGIO S R. Electrochemical degradation of a real textile effluent using boron-doped diamond or ��-PbO2 as anode [J]. Journal of Hazardous Materials, 2011, 192(3): 1275�C1282. DOI: 10.1016/j.jhazmat.2011.06.039.
[28] LONG Fen, WEI Qiu-ping, YU Zhi-ming, LUO Jia-qi, ZHANG Xiong-wei, LONG Hang-yu, WU Xian-zhe. Effects of temperature and Mo2C layer on stress and structural properties in CVD diamond film grown on Mo foil [J]. Journal of Alloys & Compounds, 2013, 579(23): 638�C645. DOI: 10.1016/j.jallcom.2013.06.146.
[29] ZHOU Bo, YU Zhi-ming, WEI Qiu-ping, LONG Hang-yu, XIE You-neng, WANG Yi-jia. Electrochemical oxidation of biological pretreated and membrane separated landfill leachate concentrates on boron doped diamond anode [J]. Applied Surface Science, 2016, 377: 406�C415. DOI: 10.1016/j.apsusc.2016.03.045.
[30] BRILLAS E, MART NEZ-HUITLE C A. Decontamination of wastewaters containing synthetic organic dyes by electrochemical methods. An updated review [J]. Applied Catalysis B: Environmental, 2015, 166�C167(3): 603�C643. DOI: 10.1016/j.apcatb.2014.11.016.
NEZ-HUITLE C A. Decontamination of wastewaters containing synthetic organic dyes by electrochemical methods. An updated review [J]. Applied Catalysis B: Environmental, 2015, 166�C167(3): 603�C643. DOI: 10.1016/j.apcatb.2014.11.016.
[31] ROCHA J H B, GOMES M M S, DOS SANTOS E V, de MOURA E C M, DA SLIVA D R, QUIROZ M A, MARTINEZ-HUITLE C A. Electrochemical degradation of Novacron Yellow C-RG using boron-doped diamond and platinum anodes: Direct and indirect oxidation [J]. Electrochimica Acta, 2014, 140: 419�C426. DOI: 10.1016/ j.electacta.2014.06.030.
[32] QI Yao, LONG Hang-yu, MA Li, WEI Qui-ping, LI Si-te, YU Zhi-ming, HU Jing-yuan, LIU Pei-zhi, WANG Yi-jia, MENG Ling-cong. Enhanced selectivity of boron doped diamond electrodes for the detection of dopamine and ascorbic acid by increasing the film thickness [J]. Applied Surface Science, 2016, 390: 882�C889. DOI: 10.1016/ j.apsusc.2016.08.158.
[33] LIU Xue-zhang, LUO Hao, SU Xu, YU Zhi-ming. Preparation of diamond/Cu microchannel heat sink by chemical vapor deposition [J]. Journal of Central South University, 2015, 22(3): 835�C841. DOI: 10.1007/s11771- 015-2590-y.
[34] LI Hong-dong, ZHANG Tong, LI Liu-an, LUE, Xian-yi, LI Bo, JIN Zeng-sun, ZOU Guang-tian. Investigation on crystalline structure, boron distribution, and residual stresses in freestanding boron-doped CVD diamond films [J]. Journal of Crystal Growth, 2010, 312(12): 1986�C1991. DOI: 10.1016/j.jcrysgro.2010.03.020.
[35] LONG Hang-yu, LUO Hao, LUO Jia-qi, XIE You-neng, DENG Ze-jun, ZHANG Xiong-wei, WANG Yi-jia, WEI Qiu-ping, YU Zhi-ming. The concentration gradient of boron along the growth direction in boron doped chemical vapor deposited diamond [J]. Materials Letters, 2015, 157: 34�C37. DOI: 10.1016/j.matlet.2015.05.069.
[36] LIU Na, ZHU He-kang, WEI Qiu-ping LONG Hang-yu, DENG Ze-jun, YU Zhi-ming, XIE You-neng, WANG Jian, MA Li, ZHOU Ke-chao. A niobium and nitrogen co-doped DLC film electrode and its electrochemical properties [J]. Journal of the Electrochemical Society, 2017, 164(14): H1�CH8. DOI: 10.1149/2.1001714jes.
[37] BERNARD M, DENEUVILLE A, MURET P. Non- destructive determination of the boron concentration of heavily doped metallic diamond thin films from Raman spectroscopy [J]. Diamond and Related Materials, 2004, 13(2): 282�C286. DOI: 10.1016/j.diamond.2003.10.051.
[38] BOGDANOWICZ R, FABIA SKA A, GOLUNSKI L,, SOBASZEK M, GNYBA M, RYL J, DAROWICKI K, OSSOWSKI T, JANSSENS S D, HAENEN K, SIEDLECKA E M. Influence of the boron doping level on the electrochemical oxidation of the azo dyes at Si/BDD thin film electrodes [J]. Diamond & Related Materials, 2013, 39: 82�C88. DOI: 10.1016/j.diamond.2013.08.004.
SKA A, GOLUNSKI L,, SOBASZEK M, GNYBA M, RYL J, DAROWICKI K, OSSOWSKI T, JANSSENS S D, HAENEN K, SIEDLECKA E M. Influence of the boron doping level on the electrochemical oxidation of the azo dyes at Si/BDD thin film electrodes [J]. Diamond & Related Materials, 2013, 39: 82�C88. DOI: 10.1016/j.diamond.2013.08.004.
[39] KROTOVA M D, PLESKOV Y V, VARNIN V P, TEREMETSKAYA I G.. The effect of CVD-diamond film thickness on the electrochemical properties of synthetic diamond thin-film electrodes [J]. Journal of Applied Electrochemistry, 2010, 40(10): 1839�C1844. DOI: 10.1007/ s10800-010-0111-0.
[40] PANIZZA M, MICHAUD P A, CERISOLA G, COMNINELLIS C. Anodic oxidation of 2-naphthol at boron-doped diamond electrodes [J]. Journal of Electroanalytical Chemistry, 2001, 507(1, 2): 206�C214. DOI: 10.1016/S0022-0728(01)00398-9.
[41] MICHAUD P A, PANIZZA M, OUATTARA L, DIACO T, FOTI G, COMINELLIS C. Electrochemical oxidation of water on synthetic boron-doped diamond thin film anodes[J]. Journal of Applied Electrochemistry, 2003, 33(2): 151�C154. DOI: 10.1023/A:1024084924058.
[42]  . Oxidative degradation of Basic Black 3 by electro-generated Fenton��s reagent using carbon fiber cathode [J]. Clean Technologies and Environmental Policy, 2016, 18(5): 1�C10. DOI: 10.1007/s10098-016-1134-y.
. Oxidative degradation of Basic Black 3 by electro-generated Fenton��s reagent using carbon fiber cathode [J]. Clean Technologies and Environmental Policy, 2016, 18(5): 1�C10. DOI: 10.1007/s10098-016-1134-y.
[43] SUN Jian-hui, SUN Sheng-peng, FAN Mao-hong, GUO Hui-qin, LEE Yi-fan, SUN Rui-xia. Oxidative decomposition of p-nitroaniline in water by solar photo- Fenton advanced oxidation process [J]. Journal of Hazardous Materials, 2008, 153(1, 2): 187�C193. DOI: 10.1016/j.jhazmat. 2007.08.037.
[44] VAHID B, KHATAEE A. Photoassisted electrochemical recirculation system with boron-doped diamond anode and carbon nanotubes containing cathode for degradation of a model azo dye [J]. Electrochimica Acta, 2013, 88(2): 614�C620. DOI: 10.1016/j.electacta.2012.10.069.
[45] HAMZA M, ABDELHEDI R, BRILLAS E, SIRES I. Comparative electrochemical degradation of the triphenylmethane dye Methyl Violet with boron-doped diamond and Pt anodes [J]. Journal of Electroanalytical Chemistry, 2009, 627(1, 2): 41�C50. DOI: 10.1016/j.jelechem. 2008.12.017.
(Edited by YANG Hua)
���ĵ���
����ӽ��ʯ�����绯ѧ�������Գ�X-GNż��Ⱦ�Ϸ�ˮ
ժҪ�������о�������ӽ��ʯ������BDD���Ի�����X-GNȾ�ϵĵ绯ѧ������Ӱ�졣BDD�缫�Dz��ò�����˿��ѧ�����������HFCVD�����꣨Nb���ĵױ����Ʊ��ġ��о��˹��ղ�������Ĥ�ĺ�ȡ������ܶȡ����ҺŨ�ȡ���ҺpHֵ����Һ�¶ȡ�Ⱦ�ϳ�ʼŨ�ȣ��Խ���Ч�ʵ�Ӱ�졣����ɨ��羵��SEM�����������͵绯ѧ����վ������BDD���۽ṹ�͵绯ѧ���ܣ���������C�ɼ��ֹ��ȼƲ�����X-GN�������ܡ����������BDD��Ĥ��ȶԵ绯ѧ����X-GN������Ӱ�졣���Ż���ʵ�������£������ܶ�Ϊ100 mA/cm2�������Ũ��Ϊ0.05 mol/L����Һ��ʼpHֵΪ3.08����Һ�¶�Ϊ60 ��C��������ˮ3 h��ɵõ�100%��ɫ���Ƴ��ʺ�63.2%�����л�̼����TOC��ȥ���ʡ�
�ؼ��ʣ�������X-GN������ӽ��ʯ����Ĥ��ȣ��绯ѧ����
Foundation item: Project(2016YEB0301402) supported by the National Key Research and Development Program of China; Project(51601226) supported by the National Natural Science Foundation of China; Project supported by the Open-End Fund for the Valuable and Precision Instruments of Central South University, China; Project supported by State Key Laboratory of Powder Metallurgy, China
Received date: 2017-02-20; Accepted date: 2017-04-13
Corresponding author: WEI Qiu-ping, PhD, Associate Professor; Tel: +86�C731�C88876692; E-mail: qiupwei@csu.edu.cn; ORCID: 0000- 0002-8109-5982

