Trans. Nonferrous Met. Soc. China 26(2016) 2518-2522
Effects of magnesium and cooling rate on titanium phase transformation for production of TiO2
Yi-jie WANG1,2, Shu-ming WEN1,2, Qi-cheng FENG1,2, Jian LIU1,2, Wei-cheng REN2
1. State Key Laboratory of Complex Nonferrous Metal Resources Clean Utilization, Kunming University of Science and Technology, Kunming 650093, China;
2. Faculty of Land Resource Engineering, Kunming University of Science and Technology, Kunming 650093, China
Received 26 August 2015; accepted 27 April 2016
Abstract: X-ray diffraction (XRD) and crystal structure analysis were used to study the effects of Mg content and cooling rate on the titanium phase transformation of three types of titanium slag. The results indicate that in the rapid cooling process, the titanium phase is anosovite, whose chemical formula is MgnTi3-nO5 (0<N<1). In the slow cooling process, when the Mg content is high, anosovite transforms into karrooite MgTi2O5 structure; when the Mg content is low, karrooite MgTi2O5 and rutile TiO2 both exist. The stability of karrooite MgTi2O5 is better than that of anosovite MgnTi3-nO5. Slow cooling contributes to the doping of Mg into the anosovite crystal and stabilises the anosovite crystal structure.
Key words: anosovite; karrooite; rutile; crystal structure; stability
1 Introduction
Titanium is known as a modern metal. It has been widely used because of its unique physical and chemical properties [1-4]. In the current titanium industry, the production of titanium sponge and titanium dioxide, produced by the chlorination process, requires the use of high grade titanium product, whose content of CaO and MgO must be less than 1.5%. The titanium slag produced by the electric furnace smelting process using the generic quality of ilmenite concentrate contains high contents of CaO and MgO. This slag is only suitable for the production of titanium dioxide via a sulphuric acid process. If the slag is used to produce titanium dioxide via a chloride process or is used to produce titanium sponge, additional technical and cost problems arise [5]. With the advent of new technologies, new titanium-rich product is produced, such as direct reduction and smelting of titanium slag, which is produced in a process in which titanomagnetite concentrate is directly reduced with coal, and then is smelted in an electric furnace. The titanium slag produced by direct reduction and smelting process contains TiO2 with a content of 40%-50% and CaO and MgO with a content that is in the range of 13%-20%. The comprehensive utilisation of smelting titanium slag in the new process is a key issue. Therefore, the research on determining how to remove impurities, such as Ca and Mg, and how to improve the TiO2 grade, has great significance for promoting the development of the titanium industry.
However, some impurities in titanium slag are difficult to be removed through leaching, flotation and other methods, especially the magnesium. Studies have found that magnesium existing as independent minerals, such as spinel and olivine, also exist in titanium minerals. The primary titanium mineral in titanium slag is anosovite solid solution, which can be expressed as m(AO・TiO2)・n(B2O3・TiO2), where A is the divalent titanium, magnesium, iron, manganese, etc., B is the trivalent titanium, iron, aluminium, chromium, etc., and m and n are constant coefficients [6-8]. Magnesium always dopes into the anosovite crystal by isomorphism. Thus, the study of the effects of magnesium on titanium phase composition will contribute to the development of impurity removal technology. Studies on the preparation of high quality synthetic rutile using titanium slag containing a high content of impurities also indicate that changing the phase composition is the key to improve the quality of titanium slag [9-11].
The anosovite crystal structure is affected by magnesium content and the cooling method of titanium slag. Therefore, the effects of magnesium content and slow cooling rate on the phase transformation behaviour of three types of titanium slag were investigated in the present work. In addition, the related mechanisms for these effects were also discussed.
2 Experimental
2.1 Materials
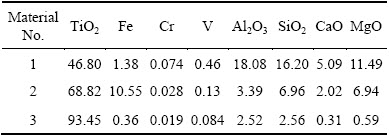
The No. 1 and No. 2 titanium slag materials used in the experiment were provided by Panzhihua Iron and Steel Group Limited Liability Company in China; the No. 3 titanium slag material came from Yunnan Province in China. The No. 1 material is smelting titanium slag which is produced by the direct reduction and electric furnace smelting process, and the raw materials of the process are titanomagnetite concentrates. The No. 2 and No. 3 materials are titanium slag produced by the electric furnace smelting process, and the raw materials of the process are ilmenite concentrates. The chemical compositions of the three materials are presented in Table 1. As seen in Table 1, the TiO2 grades of the No. 1, No. 2 and No. 3 materials are 46.80 %, 68.82 % and 93.45 %; the MgO grades of the No. 1, No. 2 and No. 3 materials are 11.49 %, 6.94 % and 0.59 %, respectively.
Table 1 Chemical composition of materials (mass fraction, %)
2.2 Experimental procedure and product characterisation
The experiments were performed in a box-type resistance furnace. The heating elements are molybdenum disilicide U-shaped bars, and the temperature was measured by a platinum-rhodium thermocouple. Temperature was controlled by an AI intelligent digital temperature controller with an accuracy of 2 K.
The materials were crushed to 2 mm by using a crusher. Approximately 80 g of the sample was transferred into a graphite crucible and placed in a resistance furnace. The temperature of the furnace was increased to 1723 K, and then maintained for 30 min to melt the materials. Afterward, the resistance furnace was cooled to 1423 K at cooling rates of 4 K/min and 2 K/min, as shown in Fig. 1. Then the resistance furnace was closed, and the materials in resistance furnace together with the furnace were cooled to room temperature slowly. Finally, when the temperature dropped to room temperature, the materials were taken out and XRD analysis was conducted.
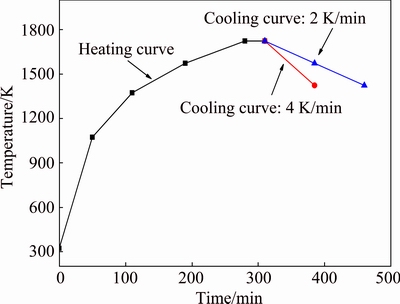
Fig. 1 Heating and cooling curve
The crystal phases composition of the titanium slag before and after slow cooling treatment were characterised by XRD (D/Max 2200, Rigaku, Japan) using Cu Kα radiation (λ=0.154056 nm, 40 kV, 200 mA) over the 2θ range of 10° to 90° at a scanning rate of 3 (°)/min.
3 Results and discussion
3.1 XRD characterisation
Figure 2 and Table 2 present the XRD results of the three types of materials before and after the samples were subjected to different cooling rates. From Fig. 2 and Table 2, it can be seen that the Mg content and the cooling rate have a significant effect on the titanium phase in titanium slag.
No. 1, No. 2 and No. 3 materials were cooled to room temperature directly from the molten state after electric furnace smelting. As crystallisation occurs in the melt, a fast cooling rate limits crystal growth. As a result, crystallisation completed in a short time, and numerous small crystals contain irregular granular aggregates. As seen from the XRD patterns, there are many minor peaks of anosovite, which indicates that the crystal developed incompletely. A smaller amount of Mg was doped into anosovite lattice; therefore, the anosovite chemical formula of the No. 1, No. 2 and No. 3 materials are Mg0.6Ti2.4O5, Mg0.75Ti2.25O5 and Mg0.09Ti2.91O5, respectively.
Anosovite crystals grew slowly and developed completely during the slow cooling process. To stabilise the anosovite crystal structure, Mg separated from the minerals which contain Mg and then transferred into the anosovite crystal during the slow cooling process. Thus, with the decrease of the cooling rate, the Mg content in anosovite solid solution increased gradually. As shown in Table 2, the titanium phase crystal structure of the three titanium slag materials finally transformed into MgTi2O5 at a cooling rate of 2 K/min. MgTi2O5 was named as karrooite by KNORRING and COX in 1961 [12]. The main characteristic peaks of karrooite are evident, as shown in Figs. 2(a) and (b).

Fig. 2 XRD patterns of three materials before and after slow cooling treatment
The low Mg content (0.59% MgO) in the No. 3 material is not sufficient to cause the entire anosovite crystal to transform into karrooite MgTi2O5 structure, so only a part of the crystal had been transformed. The structure of the remaining anosovite crystal is of Ti3O5 type, which is unstable. As a result, the remaining anosovite crystals were oxidised to rutile TiO2 by oxygen during the slow cooling process. The XRD patterns in Fig. 2(c) show that the main characteristic peaks of the No. 3 material are anosovite. However, the main characteristic peaks of 4 K/min and 2 K/min are rutile; also, there are some characteristic peaks of karrooite and a small characteristic peak of anatase. As shown in Table 2, the titanium phase of the No. 3 material, the material cooled at 4 K/min, and the material cooled at 2 K/min in Fig. 2(c) are anosovite Mg0.09Ti2.91O5, anosovite Mg0.9Ti2.1O5, and karrooite MgTi2O5, respectively.
Table 2 XRD results of titanium phase component

3.2 Mechanism
The crystal structures of Ti3O5 and MgTi2O5 were created using Materials Studio 7.0 software, as shown in Figs. 3 and 4.
As seen from Fig. 3, the [TiO6] octahedron forms a characteristic row extending along the c-axis by sharing faces, and a characteristic row extending along the b-axis by sharing corners. The radii of 6 coordination Mg2+, Ti3+ and Ti4+ are 0.0720, 0.0670 and 0.0605 nm, respectively. The radius of 4 coordination O2+ is 0.138 nm [13]. Thus, r(Mg2+)/r(O2+)=0.522, r(Ti3+)/r(O2+)=0.486 and r(Ti4+)/r(O2+)=0.438, all in the range of octahedral coordination 0.414-0.732 [14]. As a result, Mg2+, Ti3+ and Ti4+ easily form an octahedral coordination when combined with O2+. In the crystal structure of the anosovite, O2+ forms an octahedron and cations fill the octahedral voids, thereby forming an [MO6] octahedron.
According to Pauling’s rules [15], the sharing of the faces by two [TiO6] octahedrons decreases the stability of the ionic structure. The effect is larger for Ti3+ and Ti4+ cations of high charge and low coordination number. When adjacent [TiO6] octahedrons share a common face, it brings the cations closer, thus increasing electrostatic repulsions and leading to an unstable structure. As a result, Ti3+ and Ti4+ cations will be as far apart from each other as possible and are thus separated by other low charged cations.
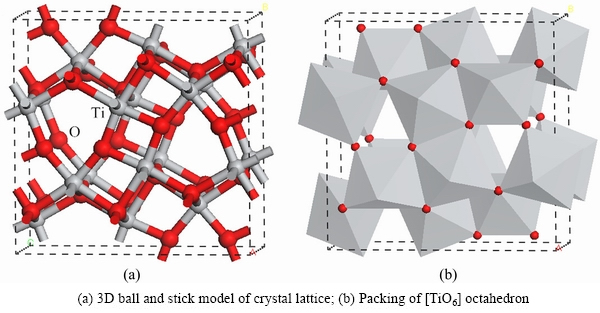
Fig. 3 Crystal structure of modification Ti3O5
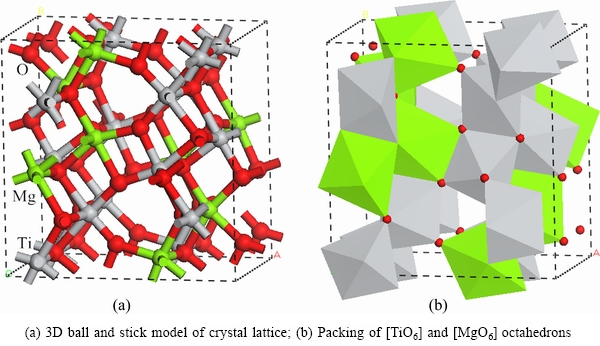
Fig. 4 Crystal structure of modification MgTi2O5
In the actual crystal, a [TiO6] octahedron is linked only by sharing a corner that does not exist because its stabilisation energy is negative [16]. If the [TiO6] octahedrons attempt to link with each other by sharing a corner, some cations must be involved. As a result, the pure Ti3O5 crystal is unstable. Therefore, Mg, with an ionic radius similar to Ti, instead of Ti dopes the anosovite crystal and form [MgO6] octahedrons. A [TiO6] octahedron tends to link with an [MgO6] octahedron or link with each other by sharing corners and edges, rather than by sharing faces, as shown in Fig. 4. Thus, [MgO6] octahedrons are used to separate [TiO6] octahedrons. In addition, because of the doping of Mg, the stability of the anosovite crystal is enhanced [17-20].
When titanium slag cools rapidly, Mg does not have sufficient time to dope the anosovite crystal. Thus, the anosovite crystal of poor stability with less Mg formed, such as the No. 1-No. 3 materials. When titanium slag cools slowly, with anosovite crystal growth, Mg gradually dopes the anosovite crystal and increases the stability. XRD patterns indicate that when a sufficient amount of Mg has doped into anosovite, anosovite will transform into karrooite MgTi2O5 structure, which has good stability. However, if there is insufficient Mg in titanium slag to make all of the anosovite Ti3O5 to transform into karrooite MgTi2O5 structure, Ti3O5 will be partially oxidised into rutile TiO2. Therefore, during the slow cooling process, part of the anosovite Mg0.09Ti2.91O5 in No. 3 material transformed into karrooite MgTi2O5 structure, and the others were oxidised into rutile TiO2.
4 Conclusions
The Mg content and the cooling rate have a great influence on the titanium phase transformation of titanium slag. The titanium phase mainly consists of MgnTi3-nO5 (0<N<1), MgTi2O5 and rutile TiO2. Using a rapid cooling process, the titanium phase is anosovite, and its chemical formula is MgnTi3-nO5 (0<N<1). Using a slow cooling process, when the Mg content is high, anosovite transforms into karrooite MgTi2O5 structure; when the Mg content is low, part of anosovite transforms into karrooite MgTi2O5 structure, and the rest are oxidised into rutile TiO2. By crystal structure analysis, it is found that the titanium phase transformation relies on the change of crystal stabilization.
References
[1] WANG Xiao-hua, LI Jin-shan, HU Rui, KOU Hong-chao. Mechanical properties and pore structure deformation behaviour of biomedical porous titanium [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(5): 1543-1550.
[2] MIDDLEMAS S, FANG Z Z, FAN P. A new method for production of titanium dioxide pigment [J]. Hydrometallurgy, 2013, 131: 107-113.
[3] ARIFVIANTO B, SUYITNO, MAHARDIKA M. Surface modification of titanium using steel slag ball and shot blasting treatment for biomedical implant applications [J]. International Journal of Minerals Metallurgy and Materials, 2013, 20(8): 788-795.
[4] VALIGHAZVINI F, RASHCHI F, NEKOUEI R K. Recovery of titanium from blast furnace slag [J]. Industrial and Engineering Chemistry Research, 2013, 52(4): 1723-1730.
[5] ZHANG Wen-sheng, ZHU Zhao-wu, CHENG Chu-yong. A literature review of titanium metallurgical processes [J]. Hydrometallurgy, 2011, 108(3-4): 177-188.
[6] GRASS V, ISTOMIN P, NAZAROVA L. X-ray diffraction refinement of the crystal structure of anosovite prepared from leucoxene [J]. Crystal Research and Technology, 2009, 44(1): 117-122.
[7] GREY I E, CRANSWICK L M D, LI C, WHITE T J, BURSILL L A. New M3O5-anatase intergrowth structures formed during low-temperature oxidation of anosovite [J]. Journal of Solid State Chemistry, 2000, 150(1): 128-138.
[8] XIROUCHAKIS D, SMIRNOV A, WOODY K, LINDSLEY D H, ANDERSEN F J. Thermodynamics and stability of pseudobrookite- type MgTi2O5 (karrooite) [J]. American Mineralogist, 2002, 87(5-6): 658-667.
[9] LIU Shui-shi, GUO Yu-feng, QIU Guan-zhou, JIANG Tao, CHEN Feng. Preparation of Ti-rich material from titanium slag by activation roasting followed by acid leaching [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(4): 1174-1178.
[10] DONG Hai-gang. Study on quality synthetic rutile preparation using high calcium and magnesium furnace titanium slag [D]. Changsha: Central South University, 2010: 14-15. (in Chinese)
[11] CHEN Guo, SONG Zeng-kai, CHEN Jin, SRINIVASAKANNAN C, PENG Jin-hui. Investigation on phase transformation of titania slag using microwave irradiation [J]. Journal of Alloys and Compounds, 2013, 579: 612-616.
[12] von KNORRING O, COX K G. Kennedyite, a new mineral of the pseudobrookite series [J]. Mineralogical Magazine, 1961, 32: 676-682.
[13] ZHANG Si-yuan. Structure, composition and properties of inorganic crystal [M]. Beijing: Science, 2013. (in Chinese)
[14] PAN Zhao-lu. Crystallography and Mineralogy [M]. 3rd ed. Beijing: Geological Press, 1993. (in Chinese)
[15] PAULING L. The principles determining the structure of complex ionic crystals [J]. Journal of the American Chemical Society, 1929, 51: 1010-1026.
[16] ZHONG Wei-zhuo, HUA Su-kun. Crystal growth morphology [M]. Beijing: Science Press, 1999. (in Chinese)
[17] HAZEN R M, YANG He-xiong. Increased compressibility of pseudobrookite-type MgTi2O5 caused by cation disorder [J]. Science, 1997, 277: 1965-1967.
[18] MATTEUCCI F, CRUCIANI G, DONDI M, GASPAROTTO G, TOBALDI D M. Crystal structure, optical properties and colouring performance of karrooite MgTi2O5 ceramic pigments [J]. Journal of Solid State Chemistry, 2007, 180(11): 3196-3210.
[19] YANG H X, HAZEN R M. Comparative high-pressure crystal chemistry of karrooite, MgTi2O5, with different ordering states [J]. American Mineralogist, 1999, 84(1-2): 130-137.
[20] SUZUKI Y, SHINODA Y. Magnesium dititanate (MgTi2O5) with pseudobrookite structure: A review [J]. Science and Technology of Advanced Materials, 2011, 12(3): 1-6.
TiO2生产中镁和冷却速度对钛物相转变的影响
王伊杰1,2,文书明1,2,丰奇成1,2,刘 建1,2,任伟成2
1. 昆明理工大学 省部共建复杂有色金属资源清洁利用国家重点实验室,昆明 650093;
2. 昆明理工大学 国土资源工程学院,昆明 650093
摘 要:以三种钛渣为研究对象,采用X射线衍射和晶体结构分析,研究镁含量和冷却速度对钛渣中钛物相转变的影响。结果表明,急速冷却的钛渣中,生成的钛物相为黑钛石,化学式为MgnTi3-nO5(0<N<1);缓慢冷却的钛渣中,当镁含量较高时,黑钛石转变为板钛镁矿,化学式为MGTI2O5;当镁含量较低时,板钛镁矿MgTi2O5和金红石型TiO2同时存在。板钛镁矿MgTi2O5的稳定性比黑钛石MgnTi3-nO5的稳定性好,缓慢冷却有助于镁掺杂进入黑钛石晶体,稳定其晶体结构。
关键词:黑钛石;板钛镁矿;金红石;晶体结构;稳定性
(Edited by Yun-bin HE)
Foundation item: Project (51090385) supported by the National Natural Science Foundation of China
Corresponding author: Shu-ming WEN; Tel: +86-871-65153445; E-mail: shmwen@126.com
DOI: 10.1016/S1003-6326(16)64374-1