
Photocatalytic activity enhancing for TiO2 photocatalyst by doping with La
WEN Chen (文 晨), DENG Hua (邓 桦), TIAN Jun-ying (田俊莹), ZHANG Ji-mei (张继梅)
Institute of Material and Chemical Engineering, Tianjin Polytechnic University, Tianjin 300160, China
Received 10 April 2006; accepted 25 April 2006
Abstract: La doped nanocrystalline TiO2 photocatalyst was developed by sol-gel method. The prepared La-TiO2 photocatalysts with anatase phases were characterized by X-ray diffractometry (XRD), UV-Vis absorption spectroscopy, and photoluminescence spectra (PL). The photocatalytic activity was evaluated by the photocatalytic degradation of phenol in solution under sunlight irradiation. The results show that the crystallinity of anatase is improved by La doping. Moreover, La not only suppresses phase transition from anatase to rutile but also exhibits an absorption in the λ≥ 400 nm range. The photocatalytic activity of La-doped TiO2 photocatalysts exceeds that of pure TiO2 photocatalyst prepared by the same method when the molar ratio of La to Ti is kept at 0.3%.
Keywords: La doping; TiO2 photocatalyst; photocatalytic activity; sunlight
1 Introduction
The photoassisted catalytic degradation of organic pollutants in water and wastewater employing semiconductors as photocatalysts has been a promising method[1, 2]. Many photocatalysts have been attempted for the degradation of pollutants in wastewater. Among the semiconductors employed, anatase phase of TiO2 is the most preferable material for the photocatalytic process due to its high photosensitivity, non-toxic nature, large bandgap and stability[3]. Despite the positive attributes of TiO2, there are a few drawbacks associated with its use: 1) charge carrier recombination occurs within nanoseconds [4]; 2) the band edge absorption threshold does not allow the utilization of visible light [5]. To circumvent these particular limitations, a number of strategies have been proposed to improve the light absorption features and lengthen the carrier life time characteristics of TiO2. It has been shown that the photocatalytic activity of a catalyst can be influenced by its crystal structure, surface area, size distribution, porosity, band gap and surface hydroxyl group density [6]. In recent years, many attempts to dope transitional metals into TiO2 have been made in order to absorb light in the visible region of the solar spectrum [7]. According to the pioneering works of ASAHI et al[8], some groups demonstrated the substitution of anion atoms such as fluorine (F), carbon (C) and sulfur (S) for oxygen [9-11]. DOMEN et al[12] reported the La and N codoped TiO2 as visible light photocatalysts. Therefore, the synergistic effects of rare earth elements such as La and Y in TiO2 will enhance the photocatalytic properties.
In this study, we studied the prepared process and characterization of the La-doped TiO2, the catalytic effects of the La-TiO2 particles on the degradation of phenol were also demonstrated under a simulated sunlight irradiation.
2 Experimental
All reagents were standard grade and used without further purification. Tetrabutylorthotitanate was used as a titanium source. La(NO3)3 as a precursor of the dopant was dissolved in 10 mL deionized water. The synthesis of the La-TiO2 particles is as follows. The mixture containing 10 mL tetrabutylorthotitanate and 40 mL absolute ethyl alcohol was agitated for 30 min and was added dropwise to a mixted solution containing 10 mL La(NO3)3-H2O, 10 mL absolute ethylalcohol and 2 mL HNO3 with vigorous stirring for 2 h at room temperature. The molar ratios of La to Ti, which hereafter was designated as RLa, were 0.1%, 0.3% and 0.4%. After being aged for 5 h, the samples were dried at 70 ℃ for 48 h to vaporize water and alcohol and then ground to fine particles to obtain xerogel samples. The xerogel samples were calcined at 773, 873 and 973 K in air for 2 h, respectively. The pure TiO2 without any doping was obtained also using the same procedure.
The X-ray diffraction (XRD) patterns were obtained on a Burker D8 GADD X-ray diffractometer using Cu Kα radiation. A UV-Vis spectrophotometer (JASCO V-570) was used to obtain the absorption spectra of samples. A Hitachi F-2500 fluorescence spectrophotometer (PL) was used to investigate the photogenerated electrons and holes information about the surface of samples.
A total of 100 mL phenol solution (20 mg/L) was put into an open reactor and air was bubbled at a flow rate of 100 mL/min through the solution in the dark for 30 min at 30 ℃. The La-TiO2 particles used for each experiment were kept at 1.0 g / L. After the adsorption of phenol on the particles reached equilibrium, the solution was irradiated with a 250 W metal halide lamp as a simulated sunlight source. The intensity of light entered the phenol solution measured with a UV-Vis radiometer was (13.4±10) mW/cm2 for 310-550 nm. The concentration of phenol was monitored by a Waters 1525 HPLC system. The photocatalytic activity of pure TiO2 particles was also examined under the same conditions.
3 Results and discussion
The crystalline phase of each sample was determined by powder XRD, and the calcination temperature on phase changes of the as-prepared pure TiO2 and La-TiO2 are shown in Fig.1. It can be seen that, in Fig.1(a), as the calcination temperature increases, the phase changes form pure anatase 773 K to mixed anatase /rutile (873 K and 973 K), but at 973 K, rutile is the main phase when doping La in TiO2. The peak intensities of anatase increase and the width of the (101) plane diffraction peak of anatase becomes narrower, indicating that the as-prepared La-doped TiO2 samples (773 K and 873 K) have a main anatase structures. As shown in Fig.1(b), as the calcination temperature increases, the prepared pure TiO2 are an anatase /rutile mixture (773 and 873 K), and the rutile phase starts to appear at 973 K. However, the diffraction peak (La2O3) of La doping in the XRD patterns is not observed, probably owing to a lower La doping content (0.3 %). The fact also implies that the La2O3 is well dispersed within TiO2 phase. In addition, it is also clear that the temperature of phase transition in the La-doped TiO2 is increased compared with that of pure TiO2.
Fig.2 shows the effect of RLa on phase structures of TiO2 particles. With increasing RLa in the system, the peak intensities of anatase increase, and La-doped TiO2 contains only anatase phase over the calcinations temperature of 773 K for 2 h. Interestingly, RLa=0.3% TiO2 sample has a smaller particles size than other samples because the width of the (101) plane diffraction peak of anatase (2θ=25.4?) becomes wider. In view of these facts, it is easy to understand the improvement of the crystallinity of anatase of TiO2 particles by La

Fig.1 XRD patterns of TiO2 samples calcined at 773 K, 873 K and 973 K for 2 h: (a) RLa = 0.3%TiO2; (b) Pure TiO2

Fig.2 XRD patterns of TiO2 samples with different RLa calcined at 773 K for 2 h
doping. TiO2 contains three polymorphs: anatase, rutile, and brookite. Each phase exhibits different properties and has different applications.
Anatase with lower agglomeration and smaller
particles size usually shows higher photocatalytic activity than that of rutile or brookite TiO2 since rutile is normally prepared by calcination of anatase at high temperatures. The results presented here clearly show that the phase and the phase composition of TiO2 can be controlled by simply doping with La in the synthesis, making it very useful to the study of the photocatalytic activity of TiO2 particles. In addition, these results also imply that the La-doping not only suppresses the formation of any impurity phase but also prevents phase transition of anatase to rutile.
The optical absorption spectra of the samples are shown in Fig.3. Compared to the undoped TiO2, La doping expands the wavelength response range of TiO2 to visible range. The absorption of La-doped TiO2 in visible light region is stronger than that of pure TiO2, which is probably caused by the color of the La-doped TiO2.
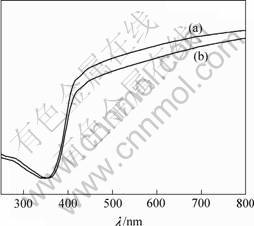
Fig. 3 UV-Vis reflection spectra of pure(a) and RLa=0.3%La doped(b) TiO2 samples calcined at 773 K for 2 h
Fig.4 indicates the fluorescence spectra (PL) of pure and La-doped TiO2 samples. The spectra of samples are somewhat different, that is, a small decrease in the fluorescence of RLa = 0.3% TiO2 particles were observed by comparing the spectrum of pure TiO2. This indicates that an appropriate amount of La doping may slow the radiative recombination process of photogenerated electrons and holes in TiO2. This may be due to the introduction of new defect sites (or recombination centers) that enhance the recombination of photogenerated electrons and holes. Our PL measurement results confirm that the higher the photocatalytic activity, the lower the intensity of PL spectrum is.

Fig.4 PL spectra (λex = 250 nm) of TiO2 samples with RLa=0.3%(a) and pure TiO2(b) calcined at 773 K for 2 h
Fig.5 depicts the results of the degradation of phenol solution photocatalyzed by pure and RLa=0.3% TiO2 samples at various calcination temperatures. It is apparent that the RLa=0.3% TiO2 (773 K) shows significantly higher photocatalytic activity compared to other samples and pure TiO2, it achieves a degradation of about 71 % for the phenol solution, but RLa=0.3% TiO2 (873 K) and pure TiO2 (773 K) degrades only 59 % and 31 % in 4 h. However, RLa=0.3% TiO2 (973 K) shows a poor photocatalytic activity which is probably due to the presence of rutile phase.

Fig.5 Effects of La-doped TiO2 (RLa=0.3%) and pure TiO2 samples with various calcination temperatures on degradation of phenol under conditions of bubbling air and sunlight, phenol concentration 20 mg/L, reaction temperature 30±2 ℃ and catalyst amount 1.0 g/L
The degradation of phenol by the La-doped TiO2 particles was compared with that using pure TiO2 under the conditions of bubbling air and sunlight. The experimental results are shown in Fig.6. It is clear that the La-doped TiO2 particles have higher photocatalytic activity for the degradation of phenol than pure TiO2 and the degradation rate follows the order: RLa= 0.3% TiO2 >RLa=0.4% TiO2>RLa=0.1% TiO2 >pure TiO2. This is due to the presence of enhanced anatase phase caused by La doping because anatase TiO2 is believed to exhibit the highest photocatalytic activity. Our experiment results are in good agreement with the XRD, UV-Vis and PL analyses.

Fig.6 Effects of La-doped TiO2 and pure TiO2 particles with various RLa on degradation of phenol under conditions of bubbling air and sunlight, phenol concentration 20 mg/L, reaction temperature (30±2) ℃ and catalyst amount 1.0 g/L
4 Conclusions
La-doped TiO2 nanoparticles with pure anatase and mixed anatase/rutile phases have been prepared. The crystallinity of anatase is improved by La doping. Moreover, with increasing RLa, La not only suppresses the formation of brookite phase but also prevents the phase transition from anatase to rutile. The La-doped TiO2 samples also show effective absorption in the visible light range and enhance the photocatalytic activity for the degradation of phenol solution under a simulated sunlight irradiation.
References
[1] HOFFMANN M R, MARTIN S T, CHOI W, BAHNEMANN D W. Environmental applications of semiconductor photocatalysis [J]. Chem Rev, 1995, 95: 69-96.
[2] MILLS A, HUNTE SL. An overview of semiconductor photocatalysis [J]. Photochem Photobiol A, 1997, 108: 1-35.
[3] SAKTHIVEL S, NEPPOLIAN B, ARABINDOO B, et al. TiO2 catalysed photodegradation of leather dye [J]. Indian J Sci Ind Res, 2000, 59: 556-620.
[4] ROTHENBERGER G, MOSER J, GRATZED M et al. Charge carried trapping and recombination dynamics in small semiconductor particles [J]. Am Chem Soc, 1985, 107: 8054-9000.
[5] SERPONE N, LAWLESS D, DISDIER J, et al. Spectroscopic, photoconductivity, and photocatalytic studies of TiO2 colloids: naked and with the lattice doped with Cr3+, Fe3+ and V5+ cations [J]. Langmuir, 1994, 10: 643-652.
[6] BAHNEMANN D W. Mechanisms of organic transformations on semiconductor particles [A]. Pelizzetti E, Schiavello M. Photochemical Conversion and Storage of Solar energy [M]. Netherlands: Kluwer Academic Publishers, 1991. 76-251.
[7] SAKTHIVEL S, SHANKAR M V, PALANICHAMY M et al. Enhancement of photocatalytic activity by metal deposition: characterization and photonic efficiency of Pt, Au and Pd deposited on TiO2 catalyst [J]. Water Res, 2004, 38, 3001-3008.
[8] ASAHI R, MORIKAWA T. Visible-light photocatalysis in nitrogen-doped titanium oxides [J]. Science, 2001, 293, 269-271.
[9] YU J C, YU J, HO W, et al. Effects of F- doping on the photocatalytic activity and microstructures of nanocrystalline TiO2 powders [J]. Chem Mater, 2002, 14: 3808-3816.
[10] IRIE H, WATANABE Y, HASHIMOTO K. Carbon-doped anatase TiO2 powders as a visible-light sensitive photocatalyst [J]. Chem Lett, 2003, 32: 772-773.
[11] OHNO T, MITSUI T, MATSUMURA M, Photocatalytic activity of S-doped TiO2 photocatalyst under visible light [J]. Chem Lett, 2003, 32: 364-365.
[12] SAKATANI Y, NUNOSHIGE N, ANODO T, OKUSAKO K, KOIDE H, TAKATA T, KONDO J N, HARA M, DOMEN K. Photoocatalytiv decomposition of acetadehyde under visible light irradiation over La3+ and N-doped TiO2 [J]. Chem Lett, 2003, 32: 1156-1157.
(Edited by YUAN Sai-qian)
Foundation item: Project (02009) supported by the National High Technology Research Plan of China
Corresponding author: WEN Chen; Tel: +86-22-81333894; E-mail: wenchen@tjpu.edu.cn