
Leaching of magnesium from desiliconization slag of nickel laterite ores by carbonation process
MU Wen-ning(IJ����)1, ZHAI Yu-chun(����)2, LIU Yan (�� ��)2
1. Department of Materials Science and Engineering, Northeastern University at Qinhuangdao Branch,Qinhuangdao 066004, China;
2. Department of Metallurgical Physical Chemistry, Northeastern University, Shenyang 110004, China
Received 6 July 2009; accepted 30 December 2009
____________________________________________________________________________________________
Abstract: The leaching of magnesium from desiliconization slag of nickel laterite ores by carbonation process was studied. The influence of various parameters was investigated to optimize the conditions and determine the kinetics of the reaction. The results show that with increasing stirring speed, liquid-to-solid ratio and reaction time, and decreasing temperature, the leaching rate of magnesium enhances. The leaching process of the desiliconization slag in the range of 288-298 K is controlled by the surface chemical reaction model. The apparent activation energy is -20.45 kJ/mol, and the kinetics model is obtained.
Key words: desiliconization slag; nickel laterite ores; magnesium; carbonatation; leaching; kinetics
____________________________________________________________________________________________
1 Introduction
Nickel laterite ores are formed by the chemical weathering of nickeliferous peridotite rock under humid climates[1-2], in which nickel is concentrated in different secondary minerals in the form of oxide. Because of the complex mineralogy and low nickel content, the recovery of nickel from the nickel laterite ores is quite difficult directly [3-4], and the technology, which is economically viable, is limited.
In the recent years, there is an increasing focus on the utilization of nickel laterite ores, along with the growing demand for stainless steel and the declining of sulphide ores[5-7]. The traditional methods for treating nickel laterite ores are costly, especially the pyrometallurgical treatment[8], and also generate huge volumes of solid residuals containing valuable elements of silicon and magnesium, and thereby bring enormous burden to the environment.
Recently, a novel process for nickel laterite ores has been developed by Institute of Metallurgical Physical Chemistry, Northeastern University, China[9]. In this process, nickel laterite ore was firstly treated by molten NaOH under atmospheric pressure, then the Na2SiO3 solution obtained was used for preparing silica products by carbonization, and the other elements such as magnesium, iron, nickel were concentrated in the desiliconization slag.
In this study, the desiliconization slag was used as raw material to recover magnesium by carbonization process. The objectives are to investigate the main factors involved in the leaching such as stirring speed, leaching temperature, reaction time and liquid-to-solid ratio, and also to determine the leaching kinetics of the desiliconization slag.
2 Experimental
2.1 Materials
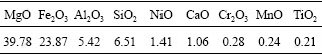
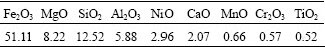
The detailed chemical compositions of the desiliconization slag were examined by ICP-OES, and the results are presented in Table 1.
Table 1 Chemical compositions of desiliconization slag expressed as oxides (mass fraction, %)
The mineralogical analysis investigated by XRD shown in Fig.1 indicates that the major mineralogical constituents of the desiliconization slag are magnesium hydroxide (Mg(OH)2) and hematite (Fe2O3).
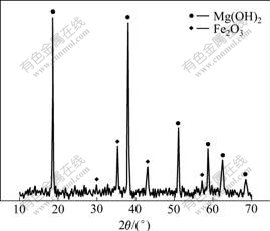
Fig.1 XRD pattern of desiliconization slag
2.2 Leaching procedure
The leaching experiments were performed in a 1 L three-necked flask equipped with a mechanical stirrer and heated in a water bath kept constant with a difference of ��0.5 ��. After putting suitable liquid-to- solid ratio of slag and water into the reactor, the system was heated to the desired temperature under the condition of continuous stirring. When the temperature reached and remained stable, the carbon dioxide gas was blew into the reactor at gas-flow rate of 0.5 L/min. The mixture was intensively stirred under atmospheric pressure. After selected time intervals, approximately 10 mL samples of the reaction mixture were taken out, diluted by 100 mL deionized water and then quickly filtered. The filtrate was analyzed by ICP spectrometry to determine the content of Mg.
3 Results and discussion
3.1 Chemical reactions
The ionization of carbon dioxide dissolved in aqueous medium depends on the concentration of H+ ions during carbonation process[10]. The pH value of desiliconization slag is close to 8.0, so carbonate ionizes in an aqueous medium according to the following reactions:
CO2(g)��CO2(aq) (1)
CO2(aq)+OH-(aq)�� (aq) (2)
(aq) (2)
The reaction of desiliconization slag in aqueous medium is
Mg(OH)2(s)��Mg2+(aq)+2OH-(aq) (3)
The neutralization reaction between Mg2+ and  occurs according to
occurs according to
Mg2+(aq)+2 (aq)��Mg(HCO3)2(aq) (4)
(aq)��Mg(HCO3)2(aq) (4)
Consequently, when carbon dioxide is blew into the aqueous solution containing desiliconization slag, the overall reaction taking place in the medium can be written as follows:
Mg(OH)2(s)+2CO2(aq)��Mg(HCO3)2(aq) (5)
Magnesium hydroxide dissolves into the liquid phase continuously to generate magnesium hydrogen carbonate during the carbonization process.
3.2 Effect of stirring speed
The influence of stirring speed on the magnesium recovery was studied in experiment conducted at 288 K for 36 h with 20:1 of liquid-to-solid ratio. The results are shown in Fig.2.
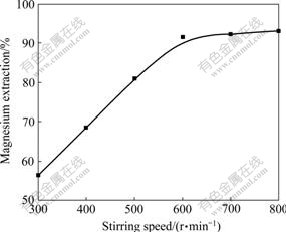
Fig.2 Effect of stirring speed on magnesium extraction
It is evident from Fig.2 that the magnesium extraction increases sharply with stirring speed (ranging from 300-600 r/min) and thereafter remains constant. As expected, after leaching for 36 h, 80.97% Mg at 500 r/min, 91.57% Mg at 600 r/min, and 92.43% Mg at 700 r/min are extracted. This means that there is an adequate suspension of the solid particles and adequate distribution of carbon dioxide when the stirring speed is 600 r/min. Thus all subsequent experiments are carried out at a stirring speed of 600 r/min in order to eliminate the stirring speed as a variable in the rate study.
3.3 Effect of liquid-to-solid ratio
The influence of liquid-to-solid ratio on magnesium extraction was carried out at 288 K, with a stirring speed of 600 r/min and reaction time of 36 h.
The effect of liquid-to-solid ratio on the magnesium extraction is presented in Fig.3. It can be seen that the extraction of magnesium increases with increasing liquid-to-solid ratio. The reason may be that larger liquid-to-solid ratio decreases the solid pulp density, and therefore decreases the mass transfer resistance on the liquid-solid interface, and finally accelerates the carbonation leaching reaction. In order to investigate the other leaching parameters, liquid-to-solid ratio of 20:1 is chosen.
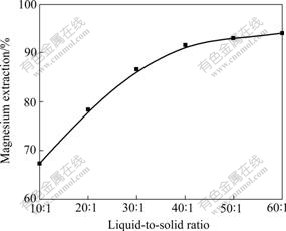
Fig.3 Effect of liquid-to-solid ratio on magnesium extraction
3.4 Effects of temperature and reaction time
The effects of temperature and reaction time (0-36 h) on the extraction of magnesium were studied under the conditions of stirring speed of 600 r/min and liquid-to-solid ratio of 20:1, and the results are shown in Fig.4.
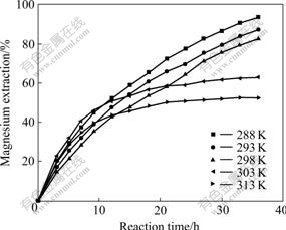
Fig.4 Effects of temperature and reaction time on magnesium extraction
According to the results given in Fig.4, the temperature and the reaction time have large influence on the extraction of magnesium. It can be see that the magnesium extraction increases steadily with increasing reaction time and decreasing temperature. The increase of temperature would decrease the solubility of carbon dioxide, and render the decomposition of magnesium hydrogen carbonate generated during the carbonization leaching. The decomposition reaction of magnesium hydrogen carbonate is presented as follows:
Mg(HCO3)2(aq)+2H2O(aq)��MgCO3?3H2O(s)��+CO2(g)�� (6)
3.5 Kinetics analysis
In order to determine the kinetic parameters and the rate controlling step of the leaching of desiliconization slag in carbonatation process, the experimental data of Fig.4 in the range of 288-298 K were analyzed by the shrinking unreacted-core model.
In the heterogeneous solid-liquid reactions, the soluble reactants diffuse across the interface and/or through the porous solid layer. Afterwards, chemical reactions occur. The reaction rate is controlled either by the diffusion of reactant through the solution boundary layer, or through a solid product layer, or by rate of the chemical reaction at the surface of the core of unreacted particles[11-12].
It is assumed that the particles of desiliconization slag have a spherical geometry, the simplified equations of the shrinking unreacted-core model when either diffusion or the surface chemical reactions are the slowest step can be expressed as follows, respectively.
1-(1-a)1/3 = krt (7)
1-3(1-a)1/3+2(1-a) = kdt (8)
where a is the magnesium fraction reacted at time t (min); kr (m/min) and kd (m/min) are the apparent rate constants.
The rate constant values and their correlation coefficients for each temperature are given for the diffusion through the product layer and the chemical reaction controlled models in Table 2. Furthermore, the linear relation between 1-(1-a)1/3 and the reaction time can be seen in Fig.5 for the reaction temperature. In accordance with these results, the equation representing the kinetics of this process was determined to obey the surface chemical reaction model, which is consistent with the results studied by LIU et al[13].
Table 2 Apparent rate constant correlation coefficient values
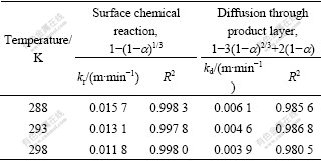
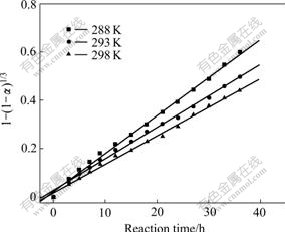
Fig.5 Plots of 1-(1-a)1/3 versus time at different reaction temperatures
From the slope of Fig.5, constants kr at different temperature are calculated. According to the Arrhenius equation, kr =k0exp[-Ea/(RT)], a plot of ln kr versus 1/T is a straight line with a slope of -Ea/(RT) and an intercept of ln k0 (see Fig.6). Using the Arrhenius equation, the following values were calculated, Ea = 20.45 kJ/mol, k0 = 3.08��10-6 s-1.
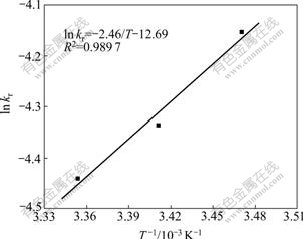
Fig.6 Arrhenius plot for leaching of magnesium
Generally speaking, the surface chemical-reaction- controlling processes have an activation energy above 40 kJ/mol[14-15]. However, if the rate is very sensitive to the temperature variation, chemical reaction may be considered to be the rate-controlling factor[16]. It can be seen from Fig.4 that the temperature has a large influence on the extraction of magnesium, thus the leaching process of desiliconization slag is controlled by the surface chemical reaction and the kinetic model for the leaching process (Eq.(5)) can be expressed as
1-(1-a)1/3=3.08��10-6 exp{[20 450/(RT)]t}
3.6 Analysis of leaching residue
The characterization of the solid residues after the carbonation leaching process includes chemical analysis, XRD analysis and SEM analysis. The leaching residue, obtained at 288 K under the conditions of stirring speed of 600 r/min, liquid-to-solid ratio of 20:1, reaction time of 36 h and gas-flow rate of carbon dioxide of 0.5 L/min, was chosen for chemical compositions, XRD and SEM analyses.
The chemical compositions of leaching residue are listed in Table 3. As expected, the elements of iron and nickel have been further enriched after extracting magnesium from desiliconization slag, of which the content of nickel oxide is improved from 1.41% to 2.96%.
Table 3 Chemical composition of leaching residue expressed as oxides (mass fraction, %)
The XRD pattern of the leaching residue is shown in Fig.7. Fig.7 indicates that the main phase is hematite, and only a small amount of magnesium hydroxide peaks can be seen. Furthermore, it is proved indirectly that the reaction takes place between slag and CO2 as shown in Eq.(5) during the carbonation leaching of the desiliconization slag.
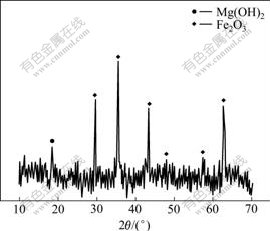
Fig.7 XRD pattern of leaching residue
The morphologies of the desiliconization slag and leaching residue were examined by SEM. It can be seen from Fig.8(a) that the desiliconization slag of the nickel laterite ores presents a rough surface, many small particles are found on the surface of the larger ones. The SEM image of leaching residue shown in Fig.8(b) shows a progressive increase in the roughness and porosity of the solid, and the particle surface does not present a reaction product layer. The morphology of the leaching residue implies that the carbonation leaching process of desiliconization slag is controlled by the surface chemical reaction.

Fig.8 SEM images of desiliconization slag and leaching residue
4 Conclusions
1) The influence of the several parameters on the dissolution rate of magnesium was examined. The magnesium extraction increases with increasing stirring speed, liquid-to-solid ratio and reaction time, and decreasing reaction temperature.
2) The extraction of magnesium is up to 91.57% under the conditions as follows: reaction temperature of 288 K, reaction time of 36 h, liquid-to-solid ratio of 20:1 and stirring speed of 600 r/min. The elements of iron and nickel are further enriched, and the content of nickel oxide is improved from 1.41% to 2.96%.
3) The leaching kinetics of desiliconization slag in carbonatation process was investigated. The carbonatation leaching process of the desiliconization slag is controlled by the surface chemical reaction in the range of 288-298K. The apparent activation energy is found to be 20.45 kJ/mol, and the kinetics model can be expressed as 1-(1-a)1/3=3.24��105 exp{[-20 450/(RT)]t}.
References
[1] PICKLES C A. Drying kinetics of nickeliferous limonitic laterite ores [J]. Minerals Engineering, 2003, 16(12): 1327-1338.
[2] SOLER J M, CAMA J, GAL? S, MEL?NDEZ W, RAM?REZ A, ESTANGA J. Composition and dissolution kinetics of garierite from the Loma de Hierro Ni-laterite deposit, Venezuela [J]. Chemical Geology, 2008, 249(1/2): 191-202.
[3] BRAND N W, BUTT C R M, ELIAS M. Nickel laterites: Classification and features [J]. AGSO Journal of Australian Geology and Geophysics, 1998, 17(4): 81-88.
[4] SWAMY Y V, KAR B B, MOHANTY J K. Physico-chemical characterization and sulphatization roasting of low-grade nickeliferous laterites [J]. Hydrometallurgy, 2003, 69(1/3): 89-98.
[5] PICKLES C A. Microwave heating behaviour of nickeliferous limonitic laterite ores [J]. Minerals Engineering, 2004, 17(6): 775-784.
[6] LEE H Y, KIM S G, OH J K. Electrochemical leaching of nickel from low-grade laterites [J]. Hydrometallurgy, 2005, 77(3/4): 263-268.
[7] MCDONALD R G, WHITTINGTON B I. Atmospheric acid leaching of nickel laterites review: Part I. Sulphuric acid technologies [J]. Hydrometallurgy, 2008, 91(1/4): 35-55.
[8] DEEPATANA A, TANG J A, VALIX M. Comparative study of chelating ion exchange resins for metal recovery from bioleaching of nickel laterite ores [J]. Minerals Engineering, 2006, 19(2): 1280-1289.
[9] LIU Y, ZHANG X, SHEN X Y, ZHAI Y C, XU D. Technology research on green metallurgy of high added value utilization of valuable elements in laterite nickel ore (��)��Preparation of silica white using molten NaOH extracting silicon method [J]. Journal of Chemical Industry and Engineering, 2008, 59(10): 2687-2691. (in Chinese)
[10] HU Q F, LIU S B, SONG L Y. New technological process for production of light magnesium carbonate from dolomite by carbonization [J]. Non-Metallic Mines, 2004, 27(3): 33-35. (in Chinese)
[11] LEVENSPIEL O. Chemical reaction engineering[M]. New York: John Wiley & Sons, 1999: 664.
[12] MAZET N. Modeling of gas-solid reactions 1. Nonporous solids [J]. Int Chem Eng, 1992, 32(2): 271�C284.
[13] LIU B S, QIAO M J, HU Q F, ZHAI X L. Kinetics of the carbonation of dolomite slurry [J]. Journal of Hebei University of Science and Technology, 2005, 26(2): 118�C123. (in Chinese)
[14] ABDEL-AAL E A. Kinetics of sulfuric acid leaching of low-grade zinc silicate ore [J]. Hydrometallurgy, 2000, 55(5): 247�C254.
[15] ASHRAF M, ZAFAR Z I, ANSARI T M. Selective leaching kinetics and upgrading of low-grade calcareous phosphate rock in succinic acid [J]. Hydrometallurgy, 2005, 80(4): 286�C292.
[16] WEN C Y. Noncatalytic heterogeneous solid-fluid reaction models [J]. Ind Eng Chem, 1968, 60(9): 34�C54.
__________________________
Foundation item: Project(2007CB613603) supported by the National Basic Research Program of China
Corresponding author: ZHAI Yu-chun; Tel: +86�C24�C83687731; E-mail: zhaiyc@smm.neu.edu.cn
(Edited by LONG Huai-zhong)