Trans. Nonferrous Met. Soc. China 25(2015) 3967-3973
Morphology and photoluminescence of BaMoO4micro- and nano-crystals synthesized by coprecipitation method
Marziyeh GHAED-AMINI1, Mehdi BAZARGANIPOUR2, Masoud SALAVATI-NIASARI1, Kamal SABERYAN3
1. Institute of Nano Science and Nano Technology, University of Kashan, Kashan, P. O. Box. 87317-51167, Iran;
2. Nanotechnology and Advanced Materials Institute, Isfahan University of Technology, Isfahan, P. O. Box 84156-83111, Iran;
3. Nuclear Fuel Cycle Research School, NSTRI, Tehran, P. O. Box 11365-8486, Iran
Received 25 January 2015; accepted 4 June 2015
Abstract: Barium molybdate (BaMoO4) micro- and nano-crystals were synthesized by the coprecipitation method. Utilizing the water as the solvent provides octahedron-like nanostructures. These nano-crystals were structurally characterized by X-ray diffraction, energy dispersive X-ray micro-analysis, Fourier transform infrared spectra. The size and shape were observed by scanning electron microscopy. The optical properties were studies by ultraviolet-visible diffuse reflectance spectroscopy and photoluminescence measurements at room temperature. The effects of temperature, solvent, surfactant and barium source were considered to obtain a controlled shape. It is found that the morphology, particle size and phase of the final products are extremely affected by these parameters.
Key words: BaMoO4; microcrystals; nanocrystals; coprecipitation; photoluminescence
1 Introduction
The molybdates have fascinated much consideration because of their particular luminescence and structure originated from the MoO42- group [1,2]. Among the molybdate oxides with scheelite-type tetragonal structure, such as AMoO4 (A=Sr2+, Ca2+, Pb2+ and Cd2+), BaMoO4 is a significant material, so that many studies have been performed on the synthesis technique and optical properties of BaMoO4 [3-10]. A current tendency in material synthesis is to investigate new methods to control the architecture and morphology at all dimensions from the nanoscale to macroscopic scale.
Diverse methods have been explored to synthesize these materials, involving solid-state reaction [11], electrochemical method [12], combustion synthesis [13], reverse microemulsion process [14], complex polymerization [15], solvothermal synthesis [16], molten salt route [17] and microwave-assisted citrate complex method [18]. Among all these approaches, the coprecipitation process has demonstrated to be an effective approach to synthesize inorganic nanomaterials with interesting morphologies and regular particle sizes [19-26].
In this work, a new coprecipitation approach was proposed to synthesize homogeneous octahedron-like BaMoO4 nanostructures. Furthermore, the morphology of BaMoO4 nanostructures can be regulated by adjusting the processing parameters.
The reaction was proceeded in an open system at atmospheric pressure. No other additives were utilized. This method has apparent advantages like easy experimental set-up, easy handling, short reaction time, environmental friendliness, and the process is very simple, fascinating and novel by focusing large amount of coprecipitation into the solutions to manufacture pure products. To the best of our knowledge, no such studies have ever been reported.
2 Experimental
2.1 Synthesis of octahedron-like BaMoO4 nanostructures
BaMoO4 nanostructures were prepared by a simple coprecipitation method. In a typical procedure, an aqueous solution of Ba(Sal)2 (Sal=salicylidene) in the presence of different surfactants, such as poly vinyl pyrrolidone (PVP), sodium dodecyl sulfate (SDS) and cetyl trimethyl ammonium bromide (CTAB), was mixed with (NH4)6Mo7O24 aqueous solution and the mixed solution was heated up to 70 ЎгC for 2 h. The white precipitate was centrifuged, washed with distilled water and methanol three times and dried under vacuum at 60 ЎгC. The effects of temperature, solvent, surfactant and barium source on the morphology and particle sizes of BaMoO4 nanostructures were investigated.
2.2 Synthesis of Ba(Sal)2 complex
[Ba(Sal)2] was synthesized as follows: 2 mmol barium(II) nitrate [Ba(NO3)2ЎӨ4H2O] was dissolved in 40 mL distilled water, a solution of 4 mmol salicylaldehyde dissolved in the same volume of methanol was dropwise added to the above solution under magnetic stirring. After the addition of all reagents, the mixture was refluxed for about 3 h.
2.3 Materials and experiments
All the chemicals were of analytical grade and purchased from Merck and used as-received without further purification. X-ray diffraction (XRD) patterns were recorded by a Rigaku D-max C III X-ray diffractometer using Ni-filtered CuKҰБ radiation. Scanning electron microscopy (SEM) images were obtained on a Philips XL-30ESEM scanning electron microscope equipped with an energy dispersive X-ray spectroscopy (EDX). Fourier transform infrared spectroscopy (FT-IR) was recorded with a Shimadzu Varian 4300 spectrophotometer in KBr pellets. EDS analysis was performed on a Philips EM208 energy dispersive spectrometer. Room temperature photo- luminescence (PL) was studied on a PerkinElmer (LS 55) fluorescence spectrophotometer. Transmission electron microscopy (TEM) images were obtained on a JEM- 2100 transmission electron microscope with an accelerating voltage of 200 kV.
3 Results and discussions
3.1 XRD analyses
The crystal structure and composition of the as-prepared products were determined by XRD. The XRD patterns of the barium molybdates obtained under different conditions (surfactant) are presented in Fig. 1. All peaks in Fig. 1 correspond to the reflections of tetragonal phase of BaMoO4. which are in a good agreement with the reported data (JCPDS: 08-0455). No remarkable diffractions of other phases can be found in the XRD patterns, exhibiting that a pure BaMoO4 phase forms.
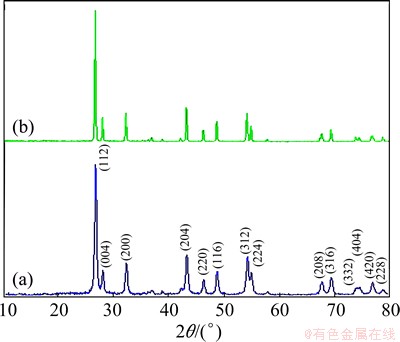
Fig. 1 XRD patterns of BaMoO4 nanostructures obtained in presence of SDS and methanol (a), and CTAB and water (b) at 30 ЎгC
3.2 FT-IR analyses
Figure 2 shows the FT-IR spectra of Ba(Sal)2 precursor and BaMoO4 nanostructures. The vibrations of BaMoO4 are sorted into the internal and external modes [27]. The first belongs to the vibration inside [MoO4]2- units. Their centers of masses are stationary. The second is called lattice phonon mode which corresponds to the motion of Ba2+cations and the rigid molecular units [27-29]. Figure 2(a) exhibits the FT-IR spectrum of BaMoO4 nanostructures acquired in methanol as the solvent and SDS as the surfactant. The spectrum illustrates a band of MoЎӘO stretching vibration in tetrahedral MoO42- clusters [15] at 791-900 cm-1. It is one of the internal modes indicated as antisymmetric stretching vibrations [15] of the three products. Weak MoЎӘO bending vibrations [28] are also discerned at about 403 cm-1 for BaMoO4 nanostructures. In the case of [Ba(Sal)2] (Fig. 2(b)), the peaks at 1525 and 1639 cm-1 belong to the CЎӘO stretching vibrations (vCЎӘO) and the peak at 1436 cm-1 belongs to CЎӘC stretching vibrations (vCЎӘC) of the salicylaldehyde. Free salicylaldehyde vCЎӘO emerges at 1680 and 1660 cm-1, and vCЎӘC emerges at 1490 and 1380 cm-1. Upon complex formation, these stretching vibrations shift to lower regions. The broad absorption band nearby 3332 cm-1 in Fig. 2(a) is appointed to the stretching vibrations of absorbed water. The absorption peaks at 469-939 cm-1 belong to BaЎӘO band, there are no absorption bands nearby this range in salicylaldehyde.
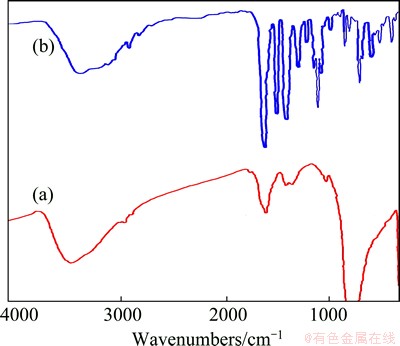
Fig. 2 FT-IR spectra of BaMoO4 (a) and Ba(Sal)2 (b) obtained in presence of SDS and methanol at 30 ЎгC
3.3 EDX analysis
EDX analysis was employed to investigate the chemical composition and purity of as- synthesized BaMoO4 nanostructures. The EDX pattern of BaMoO4 shown in Fig. 3 exhibits that the only existing elements are Ba, Mo and O. No peak of any impurity is observed, indicating the high purity of the product.
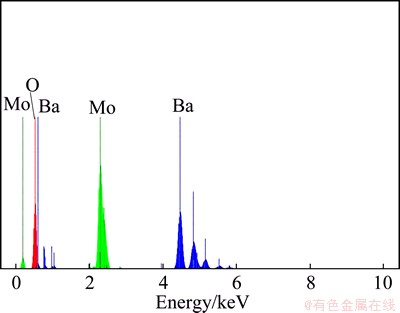
Fig. 3 EDX pattern of BaMoO4
3.4 SEM images
The morphologies of the samples were investigated by SEM. To explore the effect of surfactant on the morphology of the products, the reaction was performed in the presence of three different surfactants in methanol at 30 ЎгC for 2 h. The SEM images of the samples obtained in the presence of CTAB, PVP and SDS are displayed obtained in Figs. 4(a)-(c), respectively. Figure 4(a) exhibits rod-like nanostructures in the presence of CTAB as the surfactant. In the presence of PVP and SDS as the surfactants, only particle nanostructures form (Figs. 4(b) and (c)).
Figure 5 shows the SEM images of the samples synthesized at 30 ЎгC for 2 h with different solvents via the coprecipitation method. In the presence of propylene glycol (PG) as the solvent, rod-like nanostructures are shaped (Fig. 5(a)). The morphology of as-prepared products changes to octahedron-like nanostructures, when water is utilized as the solvent, as exhibited in Fig. 5(b). Probably, the reason is that the PG is more polar agent than water and can protect the as-formed nuclei in three dimensions and restrict them from further growing in one direction and hinder the formation of octahedron- like nanostructures.

Fig. 4 SEM images of BaMoO4 obtained from Ba(Sal)2 and (NH4)6Mo7O24ЎӨ4H2O in methanol via coprecipitation method in presence of CTAB (a), PVP (b) and SDS (c) as surfactant
Figure 6 shows the SEM images of the samples obtained at 30, 50 and 70 ЎгC. It can be observed that with increasing the reaction temperature from 30 to 70 ЎгC, the size of the nanostructures and their agglomeration enlarge.

Fig. 5 SEM images of BaMoO4 obtained from Ba(Sal)2 and (NH4)6Mo7O24ЎӨ4H O via coprecipitation method with PG (a) and H2O (b) as solvents
The effect of the Ba source on the morphology of BaMoO4 nanostructures was explored (Fig. 7). Here, Ba(NO3)2 is used as the Ba source and other reaction parameters remain unchanged. Plate-like barium molybdate nanostructures are obtained from Ba(NO3)2 salt, as shown in Fig. 7. Figure 8 exhibits octahedron-like barium molybdate nanostructures obtained in the presence of CTAB as the surfactant and water as the solvent. It is proved that water as solvent has critical role for the formation of octahedron-like barium molybdate nanostructures.
3.5 TEM images
Figure 9 shows the TEM images and SAED pattern of BaMoO4 nanostructures obtained in the presence of SDS and methanol at 30 ЎгC. The obtained TEM images show exhibit that BaMoO4 nanostructures with an average size of 20 nm and particle-like shape are obtained. The ring-like SAED pattern illustrates that the nanostructures are polycrystalline.
3.6 Optical properties
Figure 10 shows the (ҰБhҰФ)2 versus hҰФ curve of BaMoO4 nanostructures which was calculated from their UVЁCvisible absorbance using the equation proposed by THONGTEM et al [29]:
ҰБhҰФ=(hҰФ-Eg)n (1)
where ҰБ is the absorbance, h is the Planck constant, ҰФ is the photon frequency, Eg is the energy gap and n is the pure number associated with different types of electronic transitions. For n=1/2, 2, 3/2 and 3, the transitions are directly allowed, indirectly allowed, directly forbidden and indirectly forbidden, respectively. Each energy gap is determined by the extrapolation of each linear portion of the curves to ҰБ=0. In the current investigation, the BaMoO4 presents directly allowed electronic transition (n=1/2) [29], and the energy gaps of BaMoO4 nanostructures is 4.27 eV.

Fig. 6 SEM images of BaMoO4 obtained from Ba(Sal)2 and (NH4)6Mo7O24ЎӨ4H2O in methanol via coprecipitation method at 30 ЎгC (a), 50 ЎгC (b) and 70 ЎгC (c)
The photoluminescence properties of the prepared BaMoO4 nanostructures excited at 275 nm at room temperature are shown in Fig. 11. The emission spectra of the metal molybdates are principally due to the charge-transfer transitions within the [MoO4]2- complex [30]. With the excitation at 275 nm, BaMoO4 nanostructures reveal PL emission in the wavelength of 303 nm (Fig. 11). It is usually accepted that the emission spectra of the barium molybdates are mostly influenced by charged transitions in the [MoO4]2- complexes, involving some factors such as the distortions of [MoO4]2- tetrahedron groups happened by the different angles of OЎӘMoЎӘO, particle sizes, crystalline degree, morphology and surface defects [29].

Fig. 7 BaMoO4 nanostructures obtained from Ba(NO3)2 and (NH4)6Mo7O24ЎӨ4H2O

Fig. 8 Octahedron-like BaMoO4 nanostructures obtained in presence of CTAB and water at 30 ЎгC
4 Conclusions
1) Octahedron-like BaMoO4 nanostructure was successfully synthesized from Ba(Sal)2 and ammonium hepta molybdate using a simple coprecipitation method at low temperature and pressure.
2) The coprecipitation method has apparent advantages like easy experimental set-up, easy handling, short reaction time, environmental friendliness, and the process is very simple, fascinating and novel by focusing large amount of coprecipitation into the solutions to manufacture pure products.

Fig. 9 TEM images (a, b) and SAED pattern (c) of BaMoO4 nanostructures obtained in presence of SDS and methanol at 30 ЎгC
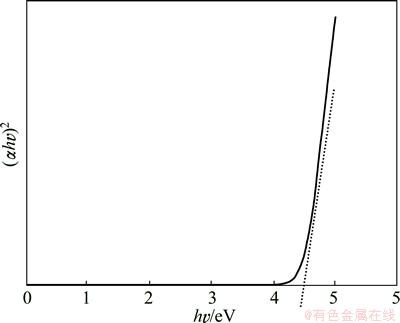
Fig. 10 (ҰБhҰФ)2 vs hҰФ plot of BaMoO4 nanostructures obtained in presence of SDS and methanol at 30 ЎгC
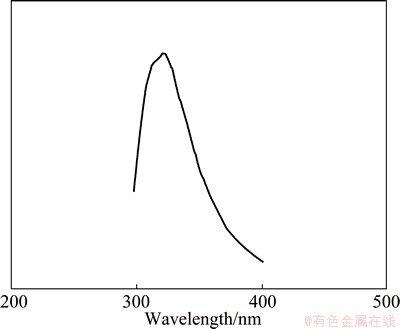
Fig. 11 PL spectrum of BaMoO4 nanostructures obtained in presence of SDS and methanol at 30 ЎгC
Acknowledgment
The authors are grateful to the council of Iran National Science Foundation and University of Kashan for supporting this work by Grant No. 159271/148.
References
[1] CHEN Z X, BU W B, ZHANG N, SHI J L. Controlled construction of monodisperse La2(MoO4)3:Yb, Tm micro-architectures with upconversion luminescent property [J]. J Phys Chem C, 2008, 112: 4378-4383.
[2] SU Y G, LI L P, LI G S. Self-assembly and multicolor emission of core/shell structured CaWO4:Na+/Ln3+ spheres [J]. Chem Commun, 2008, 34: 4004-4006.
[3] LUO Z J, LI H M, SHU H M, SHU K X, XIA J X, YAN Y S. Synthesis of BaMoO4 nestlike nanostructures under a new growth mechanism [J]. Cryst Growth Des, 2008, 8: 2275-2281.
[4] LUO Y S, ZHANG W D, DAI X J, YANG Y, FU S Y. Facile synthesis and luminescent properties of novel flower like BaMoO4 nanostructures by a simple hydrothermal route [J]. J Phys Chem C, 2009, 113: 4856-4861.
[5] ROSA I L V, MARQUES A P A, TANAKA M T S, MELO D M A, LEITE E R, LONGO E, VARELA J A. Synthesis, characterization and photophysical properties of Eu3+ doped in BaMoO4 [J]. J Fluoresc, 2008, 18: 239-245.
[6] XIA Z G, CHEN D M. Synthesis and luminescence properties of BaMoO4:Sm3+ phosphors [J]. J Am Ceram Soc, 2010, 93: 1397-1401.
[7] XIA Z, JIN S, SUN J, DU H, DU P, LIAO L. Facile morphology-controlled synthesis and luminescence properties of BaMoO4:Eu3+ microparticles and micro-rods obtained by a molten-salt reaction route [J]. Journal of Nanoscience and Nanotechnology, 2011, 11: 9612-9620.
[8] CAVALCANTE L S, SCZANCOSKI J C, TRANQUILIN R L, VARELA J A, LONGO E, ORLANDI M O. Growth mechanism of octahedron-like BaMoO4 microcrystals processed in microwave- hydrothermal: Experimental observations and computational modeling [J]. Particuology, 2009, 7: 353-362.
[9] RYU E K, HUH Y D. Synthesis of hierarchical self-assembled BaMoO4 microcrystals [J]. Bull Korean Chem Soc, 2008, 29: 503-506.
[10] CAVALCANTE L S, SCZANCOSKI J C, TRANQUILIN R L, JOYA M R, PIZANI P S, VARELA J A, LONGO E. BaMoO4 powders processed in domestic microwave-hydrothermal: Synthesis, characterization and photoluminescence at room temperature [J]. Journal of Physics and Chemistry of Solids, 2008, 69: 2674-2680.
[11] PUPP C, YAMDAGNI R, PORTER R F. Mass spectrometric study of the evaporation of BaMoO4 and BaWO4 [J]. J Inorg Nucl Chem, 1969, 31: 2021-2029.
[12] SUN Y, MA J, FANG J, GAO C, LIU Z. Synthesis of BaMoO4 high photoluminescent whiskers by an electrochemical method [J]. Ceramics International, 2011, 37: 683-686.
[13] XIA C T, FUENZALIDA V M, ZARATE R A. Electrochemical preparation of crystallized Ba1-xSrxMoO4 solid-solution films at room-temperature [J]. J Alloy Compd, 2001, 316: 250-255.
[14] MI Y, HUANG Z Y, HU F L, LI X Y. Room temperature reverse-microemulsion synthesis and photoluminescence properties of uniform BaMoO4 submicro-octahedra [J]. Mater Lett, 2009, 63: 742-744.
[15] de AZEVEDO MARQUES A P, de MELO D M A, PASKOCIMAS C A, PIZANI P S, JOYA M R, LEITE E R, LONGO E. Photoluminescent BaMoO4 nanopowders prepared by complex polymerization method (CPM) [J]. J Solid State Chem, 2006, 179: 671-678.
[16] ZHANG C, SHEN E H, WANG E B, KANG Z H, GAO L, HU C W, XU L. One-step solvothermal synthesis of high ordered BaWO4 and BaMoO4 nanostructures [J]. Mater Chem Phys, 2006, 96: 240-243.
[17] AFANASIEV P. Molten salt synthesis of barium molybdate and tungstate microcrystals [J]. Mater Lett, 2007, 61: 4622-4626.
[18] RYU J H, YOON J W, LIM C S, SHIM K B. Microwave-assisted synthesis of barium molybdate by a citrate complex method and oriented aggregation [J]. Mater Res Bull, 2005, 40: 1468-1476.
[19] SHAHRI Z, BAZARGANIPOUR M, SALAVATI-NIASARI M. Controllable synthesis of novel zinc molybdate rod-like nanostructures via simple surfactant-free precipitation route [J]. Superlattices Microstruct, 2013, 63: 258-266.
[20] GHOLAMI T, SALAVATI-NIASARI M, BAZARGANIPOUR M, NOORI E. Synthesis and characterization of spherical silica nanoparticles by modified  process assisted by organic ligand [J]. Superlattices Microstruct, 2013, 61: 33-41.
process assisted by organic ligand [J]. Superlattices Microstruct, 2013, 61: 33-41.
[21] SALAVATI-NIASARI M, SHOSHTARI-YEGANEH B, BAZARGANIPOUR M. Facile synthesis of rod-shape nanostructures lead selenide via hydrothermal process [J]. Superlattices Microstruct, 2013, 58: 20-30.
[22] NOORI E, BAZARGANIPOUR M, SALAVATI-NIASARI M, GHOLAMI T. Synthesis and characterization of silica nanostructures in the presence of schiff-base ligand via simple sonochemical method [J]. J Clust Sci, 2013, 24: 1171-1180.
[23] GOUDARZI M, BAZARGANIPOUR M, SALAVATI-NIASARI M. Synthesis, characterization and degradation of organic dye over Co3O4 nanoparticles prepared from new binuclear complex precursors [J]. RSC Adv, 2014, 4: 46517-46520.
[24] MANDIZADEH S, BAZARGANIPOUR M, SALAVATI-NIASARI M. A low-cost and eco-friendly viable approach for green synthesis of barium haxaferrite nanostructures using palm oil [J]. Ceramics International, 2014, 40: 15685-15691.
[25] GHAED-AMINI M, BAZARGANIPOUR M, SALAVATI-NIASARI M. Calcium molybdate octahedral nanostructures, hierarchical self-assemblies controllable synthesis by coprecipitation method: Characterization and optical properties [J]. Journal of Industrial and Engineering Chemistry, 2015, 21: 1089-1097.
[26] GHOLAMREZAEI S, SALAVATI-NIASARI M, BAZARGANIPOUR M, PANAHI-KALAMUEI M, BAGHERI S. Novel precursors for synthesis of dendrite-like PbTe nanostructures and investigation of photoluminescence behaviour [J]. Advanced Powder Technology, 2014, 25: 1585-1592.
[27] BASIEV T T, SOBOL A A, VORONKO Y K, ZVEREV P G. Spontaneous Raman spectroscopy of tungstate and molybdate crystals for Raman lasers [J]. Optic Mater, 2000, 15: 205-216.
[28] THONGTEMA T, PHURUANGRAT A, THONGTEM S. Sonochemical synthesis of MMoO4 (M = Ca, Sr and Ba) nanocrystals [J]. Journal of Ceramic Processing Research, 2008, 9: 189-191.
[29] THONGTEM T, KUNGWANKUNAKORN S, KUNTALUE B, PHURUANGRAT A, THONGTEM S. Luminescence and absorbance of highly crystalline CaMoO4, SrMoO4, CaWO4 and SrWO4 nanoparticles synthesized by co-precipitation method at room temperature [J]. J Alloy Compd, 2010, 506(1): 475-481.
[30] HONG G Y, JEON B S, YOO Y K, YOO J S. Photoluminescence characteristics of spherical YЎх2ЎхOЎх3:ЎхEu phosphors by aerosol pyrolysis [J]. J Electrochem Soc, 2001, 148: H161-H166.
№ІіБөн·ЁЦЖұёBaMoO4ОўГЧәНДЙГЧҫ§өДРОГІәН№вЦВ·ў№в
Marziyeh GHAED-AMINI1, Mehdi BAZARGANIPOUR2, Masoud SALAVATI-NIASARI1, Kamal SABERYAN3
1. Institute of Nano Science and Nano Technology, University of Kashan, Kashan, P. O. Box. 87317-51167, Iran;
2. Nanotechnology and Advanced Materials Institute, Isfahan University of Technology, Isfahan, P. O. Box 84156-83111, Iran;
3. Nuclear Fuel Cycle Research School, NSTRI, Tehran, P. O. Box 11365-8486, Iran
ХӘ ТӘЈәНЁ№э№ІіБөн·ЁЦЖұёовЛбұө(BaMoO4)ОўГЧәНДЙГЧҫ§ЎЈІЙУГЛ®ЧчОӘИЬјБҝЙөГөҪ°ЛГжМеЧҙөДДЙГЧҪб№№ЎЈАыУГXЙдПЯСЬЙдЎўXЙдПЯДЬЖЧПФОў·ЦОцәНёөАпТ¶ұд»»әмНв№вЖЧ¶ФХвР©ДЙГЧҫ§Ҫб№№ҪшРРұнХчЈ¬К№УГЙЁГиөзҫө№ЫІмЖдіЯҙзәНРОГІЈ¬НЁ№эКТОВПВөДЧПНв-ҝЙјы·ҙЙд№вЖЧәН№вЦВ·ў№вІвБҝСРҫҝЖд№вС§РФДЬЎЈСРҫҝОВ¶ИЎўИЬјБЎўұнГж»оРФјБәНұөФҙ¶ФҝЙҝШРОГІөДУ°ПмЈ¬·ўПЦХвР©ІОКэ¶јј«ҙуөШУ°ПмЧоЦХІъОпөДРОГІЎўҝЕБЈіЯҙзәНПаЎЈ
№ШјьҙКЈәBaMoO4Ј»ОўГЧҫ§Ј»ДЙГЧҫ§Ј»№ІіБөнЈ»№вЦВ·ў№в
(Edited by Mu-lan QIN)
Corresponding author: Masoud SALAVATI-NIASARI; Tel: +98-31-55912383; Fax +98-31-55913201; E-mail: salavati@kashanu.ac.ir
DOI: 10.1016/S1003-6326(15)64045-6