反向扩散效应对过冷单相固溶体合金凝固动力学的影响
来源期刊:中国有色金属学报(英文版)2012年第3期
论文作者:王海丰 刘峰 王慷 翟海民
文章页码:642 - 646
关键词:Ni-Cu合金;快速凝固;扩散;过冷
Key words:Ni-Cu alloy; rapid solidification; diffusion; undercooling
摘 要:
基于平均体积方法建立过冷单相固溶体合金的凝固动力学模型并研究反向扩散在凝固动力学过程中的作用。模型在过冷Ni-15%Cu(摩尔分数)合金快速凝固中的应用表明:反向扩散显著影响凝固结束温度但对再辉阶段凝固的固相体积分数影响不明显。与Herlach观点相反,凝固结束温度介于杠杆定律和Scheil方程的预测值之间,且其具体值由反向扩散、初始过冷度和冷却速率决定。
Abstract: Departing from the volume-averaging method, an overall solidification kinetic model for undercooled single-phase solid-solution alloys was developed to study the effect of back diffusion on the solidification kinetics. Application to rapid solidification of undercooled Ni-15%Cu (mole fraction) alloy shows that back diffusion effect has significant influence on the solidification ending temperature but possesses almost no effect on the volume fraction solidified during recalescence. Inconsistent with the widely accepted viewpoint of Herlach, solidification ends at a temperature between the predictions of Lever rule and Scheil’s equation, and the exact value is determined by the effect of back diffusion, the initial undercooling and the cooling rate.

![]()

Trans. Nonferrous Met. Soc. China 22(2012) 642-646
WANG Hai-feng, LIU Feng, WANG Kang, ZHAI Hai-min
State Key Laboratory of Solidification Processing, Northwestern Polytechnical University, Xi’an 710072, China
Received 9 September 2011; accepted 4 January 2012
Abstract: Departing from the volume-averaging method, an overall solidification kinetic model for undercooled single-phase solid-solution alloys was developed to study the effect of back diffusion on the solidification kinetics. Application to rapid solidification of undercooled Ni-15%Cu (mole fraction) alloy shows that back diffusion effect has significant influence on the solidification ending temperature but possesses almost no effect on the volume fraction solidified during recalescence. Inconsistent with the widely accepted viewpoint of Herlach, solidification ends at a temperature between the predictions of Lever rule and Scheil’s equation, and the exact value is determined by the effect of back diffusion, the initial undercooling and the cooling rate.
Key words: Ni-Cu alloy; rapid solidification; diffusion; undercooling
1 Introduction
The major aim of solidification science is to optimize the mechanical and physical properties of solidified alloys by microstructure control. Such an aim can be fulfilled, which provides that the whole solidification process, including liquid-cooling, solidification and solid-cooling, is fully understood. Therefore, modeling the overall solidification kinetics, where the macroscope and microscope models are coupled (i.e. micro-macroscopic approach [1]), becomes quite important, since it gives a more accurate assessment of the quality and ultimately gives the mechanical properties of the solidified product.
An original model was proposed by RAPPAZ and TH?VOZ (RT) [2] for equiaxed dendritic growth under near-equilibrium conditions. Using the volume-averaging method [3], a more general model was subsequently developed by WANG and BECKERMANN (WB) [4]. Since then, many improved works have been carried out [5-7], but limit themselves to near-equilibrium solidification. With the development of non-equilibrium solidification technologies, rapid solidification of undercooled melts has become a hot-spot in solidification science. However, as a successful method to produce novel metastable phases and microstructures [8-10], it did not cause sufficient concern in modeling the overall solidification kinetics. As the first step, an overall solidification kinetic model without considering the effect of back diffusion was proposed for rapid solidification of undercooled single-phase solid-solution alloys [11,12].
Back diffusion is not critical for a good agreement between the predicted and measured cooling curves, but is very important in the overall solidification kinetics (e.g. it influences the solidification ending temperature [11,12]) and should be taken into consideration. In the present work, back diffusion is further incorporated in the model of WANG et al [11] and its effect on the overall solidification kinetics is studied in detail, based on the model calculation results in undercooled Ni-15%Cu (mole fraction) alloy.
2 Model derivation
Following the RT [2] and WB [4] models, the grain envelope is defined as a fictitious spherical surface stretched by (or around) the dendrite tip, and the spherical grain for dendritic growth is divided into three different regions or phases, i.e. the solid dendrite s, the inter-dendritic liquid li, and the extra-dendritic liquid le, where s and le are only in pointwise contact and the corresponding interfacial area is ignored. If the densities of s, li and le are equal and constant, the solute diffusion in the macroscopic scale is omitted, and the solute diffusion in li and le is under quasi-steady states and back diffusion in s is considered, the solute balances for s, li and le, respectively, can be, according to the volume-averaging method [3] obtained as:
 (1)
(1)

![]() (2)
(2)
 (3)
(3)
where ![]() is an averaging operator [3]; gs(
is an averaging operator [3]; gs(![]() ),
), ![]() (
(![]() ) and
) and ![]() (
(![]() ) are respectively the volume fractions (the averaged concentration) of s, li and le;
) are respectively the volume fractions (the averaged concentration) of s, li and le; ![]() and
and ![]() are the averaged concentrations of liquid at the s-li and li-le interfaces; k is the non-equilibrium partition coefficient;
are the averaged concentrations of liquid at the s-li and li-le interfaces; k is the non-equilibrium partition coefficient; ![]() and
and ![]() are respectively the solute diffusion length in s and le at the s-li and li-le interfaces; Ss and Se are the interfacial area concentrations for the s-li and li-le interfaces; Ds and Dl are respectively the solute diffusion coefficients of the solid (s) and the liquid (li and le), and
are respectively the solute diffusion length in s and le at the s-li and li-le interfaces; Ss and Se are the interfacial area concentrations for the s-li and li-le interfaces; Ds and Dl are respectively the solute diffusion coefficients of the solid (s) and the liquid (li and le), and
![]() (4)
(4)
where v is the growth velocity of the grain envelope (i.e., the li-le interface) and vD is the velocity of solute diffusion in the liquid (li and le), which are introduced to account for the effect of non-equilibrium solute diffusion [13,14]. Note that Eqs. (1)-(3) are the same as that in
Ref. [11], except that a term ![]()
is introduced in Eqs. (1) and (2) to account for the effect of back diffusion.
If the latent heat of solidification is constant (ΔHf), the specific heat capacities of s, li and le are equal and constant (cp), heat diffusion in the macroscopic scale is omitted, heat diffusion in le is under a quasi-steady state, heat diffusion of s and li is not considered, and the temperatures of s and li are equal (Ti), the energy balance for the mushy zone (s and li) and the extra-dendritic liquid (le), respectively, can be, according to the volume-averaging method [3] obtained as:
![]() (5)
(5)
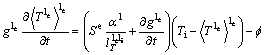 (6)
(6)
where ![]() is the averaged temperature of le;
is the averaged temperature of le; ![]() is the heat diffusion length in le at the li-le interface; αl is the thermal diffusivity; f is the cooling rate.
is the heat diffusion length in le at the li-le interface; αl is the thermal diffusivity; f is the cooling rate.
Depending on the growth kinetics of the s-li interface, dendritic grain growth can be classified into three stages, i.e., 1) purely thermal-controlled growth, 2) mainly thermal-controlled growth and 3) solute-diffusion controlled growth [11]. Since the growth kinetics of the s-li interface can be described by either the solute or the energy balance, e.g., for stages (1) and (2), the energy balance of the mushy zone (i.e. Eq. (5)) is applied; whereas for stage (3), the solute balance at the s-li interface, i.e.,
![]()
![]() (7)
(7)
is adopted. The transition from thermal-controlled to solute-diffusion controlled growth is assumed to happen once the growth rate predicted by Eq. (7) is larger than that by Eq. (5). As to the growth kinetics of the grain envelope, an extended dendrite growth model developed recently [15,16] is applied to obtaining v, Ti, Cl * and k, which is similar to the treatment in the RT [2] and WB [4] models.
For globular grain growth, there is no li any more and the grain envelope coincides with the s-le interface, i.e., ![]() and Ss=Se. In this case, the solute balance for s follows Eq. (1) and the solute balance for le according to the volume-averaging method [3] can be obtained as:
and Ss=Se. In this case, the solute balance for s follows Eq. (1) and the solute balance for le according to the volume-averaging method [3] can be obtained as:
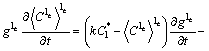
![]() (8)
(8)
As to the growth kinetics of the s-le interface, it can be obtained from the solute balance as
![]()
![]() (9)
(9)
where ![]() and
and ![]() are respectively the solute diffusion length in s and le at the s-le interface.
are respectively the solute diffusion length in s and le at the s-le interface.
Generally, globular grain growth happens in two cases [11]. Case 1: at the initial stage of solidification for sufficient small initial undercooling, ΔT, the predicted volume fraction of s (gs) is larger than that of the mushy zone![]() if the inter-dendritic liquid li is presented; Case 2: at the late stage of solidification, the growth of the s-li interface is controlled by the external heat extraction, and the growth of the grain envelope slows down due to soft impingement. Thus li is consumed gradually until the growth of the remaining le starts with
if the inter-dendritic liquid li is presented; Case 2: at the late stage of solidification, the growth of the s-li interface is controlled by the external heat extraction, and the growth of the grain envelope slows down due to soft impingement. Thus li is consumed gradually until the growth of the remaining le starts with![]() , namely, a globular grain growth happens. For both cases (1) and (2), the undercooling is so small that a uniform temperature by Eqs. (5) and (6) reduces to
, namely, a globular grain growth happens. For both cases (1) and (2), the undercooling is so small that a uniform temperature by Eqs. (5) and (6) reduces to
![]() (10)
(10)
In other words, the globular grain growth is solute- diffusion controlled.
Subjected analogous treatments to nucleation, and thermal and solute diffusion impingements as that in Ref. [11], an extended overall solidification kinetic model incorporating the effect of back diffusion was established for undercooled single-phase solid-solution alloys, where the expressions for the interfacial area concentration, the solute diffusion length and the thermal diffusion length can be found in Ref. [11].
3 Results and discussion
The present model is adopted to describe the overall solidification kinetics of undercooled Ni-15%Cu (mole fraction) alloy. Values of the physical parameters are available in Ref. [12]. From HERLACH [8], the solidification process can be divided into two stages.
1) Recalescence: Once solidification initiates, primary crystallization takes place under non-equilibrium condition and the volume fraction solidified during recalescence gR can be determined by
![]() (11)
(11)
where ΔHhyp is the hypercooling limit.
2) Post- recalescence: The remaining liquid with a volume fraction of 1-gR solidifies under near-equilibrium condition within the melting interval ΔT0=TL(C0)-TS(C0) with TL(C0) and TS(C0) respectively as the liquidus and solidus temperature for the initial solute concentration C0.
In what follows, the effect of back diffusion on both the recalescence and post-recalescence stages was studied. For a better description, two different values of Ds (i.e. Ds=3×10-11 m2/s, Ds/Dl=10-2 and Ds=3×10-13 m2/s, Ds/Dl=10-4) and f (i.e. f=50 K/s and f=225 K/s) were chosen.
Evolution of gR with ΔT predicted by the present model and Eq. (12) is shown in Fig. 1. Similar to the model predictions without considering the effect of back diffusion [12], gR increases linearly with ΔT and the present model predictions almost coincide with that of Eq. (12). This indicates that back diffusion has not enough time to proceed within such a short recalescence stage, and thus it possesses almost no effect on gR. Note that relatively larger derivations between the present model predictions and that of Eq. (12) occur for low ΔT where the solidification rate is smaller and the near-adiabatic condition is less fulfilled during recalescence (see Fig. 1).
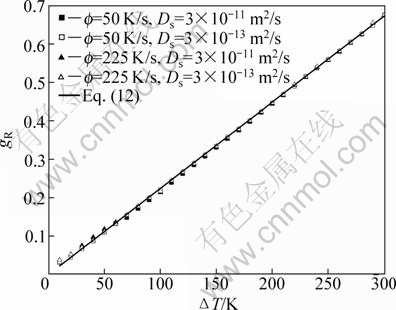
Fig. 1 Volume fraction solidified during recalescence (gR) as function of initial undercooling (ΔT) predicted by the present model and Eq. (12) for undercooled Ni-15%Cu (mole fraction) alloy
Following the above description of HERLACH [8], after recalescence, solidification should end at TS(C0), i.e. Tend=TS(C0) with Tend as the solidification ending temperature. Actually, solidification of undercooled melt is so rapid that solute mixture in the solid and the liquid cannot be performed well, namely, Lever rule is not fulfilled and Tend≠TS(C0). Meanwhile, the cooling rate after recalescence is not quick enough to suppress completely the effect of back diffusion, namely, Scheil’s equation is not fulfilled and ![]() (the melting temperature of pure Cu). On this basis, it seems reasonable and necessary to reexamine Tend, particularly incorporating the effect of back diffusion by the present model. Evolution of Tend with ΔT is predicted by the present model with different Ds and f (see Fig. 2). Independent of ΔT, Tend distributes between TS(C0) and Tm. In other words, if back diffusion is considered, solidification then ends at a temperature between the prediction of Lever rule (TS(C0)) and Scheil’s equation (
(the melting temperature of pure Cu). On this basis, it seems reasonable and necessary to reexamine Tend, particularly incorporating the effect of back diffusion by the present model. Evolution of Tend with ΔT is predicted by the present model with different Ds and f (see Fig. 2). Independent of ΔT, Tend distributes between TS(C0) and Tm. In other words, if back diffusion is considered, solidification then ends at a temperature between the prediction of Lever rule (TS(C0)) and Scheil’s equation (![]() ).
).
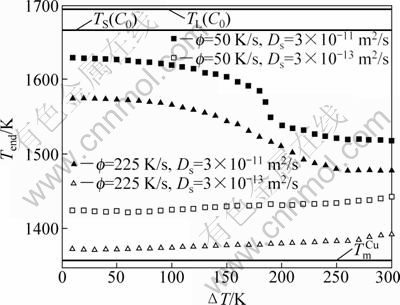
Fig. 2 Solidification ending temperature (Tend) as function of initial undercooling (ΔT) predicted by the present model for undercooled Ni-15%Cu (mole fraction) alloy
The exact value of Tend is determined by the effect of back diffusion, the initial undercooling and the cooling rate (see Fig. 2). The values of Ds and f determine respectively the effect of back diffusion and the solidification time. Considering the effect of back diffusion, the influence of ΔT on Tend should be analyzed from two points of view. On one hand, increasing ΔT equals intensifying the effect of solute trapping, thus reducing the solute gradient in the solid at the solid-liquid interface and weakening the effect of back diffusion (i.e. decreasing Tend). This is denoted as Effect (1). On the other hand, increasing ΔT leads to a linear decrease of the volume fraction solidified during post-recalescence stage (see Fig. 1), thus reducing the release of latent heat that should be extracted to the environment (i.e. increasing Tend for a fixed f). This is denoted as Effect (2). For a high value of Ds (e.g. Ds=3×10-11 m2/s), effect of back diffusion is so strong that Effect 1 and Effect 2 are equally important for Tend. As shown by the filled squares and filled triangles in Fig. 2, Tend decreases slowly at small ΔT, rapidly at medium ΔT and slowly again at high ΔT. This is due to the completion and transition between Effect (1) and Effect (2), namely, for small ΔT, both Effect (1) and Effect (2) are weak, but with increasing ΔT, Effect (1) becomes dominant at medium ΔT and is replaced by Effect (2) at high ΔT. For a small value of Ds (e.g. Ds=3×10-13 m2/s), effect of back diffusion is sufficiently weak and can be neglected, Thus Effect (2) is always dominant and Tend increases with ΔT (See the open squares and open triangles in Fig. 2). Furthermore, if f is high (e.g. f=225 K/s), the substantial decrease of solidification time will suppress the effect of back diffusion, so that solidification will end at relatively high temperature (See the solid and open squares and solid and open triangles in Fig. 2).
Such an examination of the overall solidification kinetics is very important in our understanding of the rapid solidification phenomena and the formation of novel metastable phases and microstructures. For example, the above description for the post-recalescence stage by HERLACH [8] is widely accepted, based on which the plateau duration was measured to predict grain refinement [17,18]. According to the present model, however, solidification generally does not yet complete at the solidus temperature, so that the measured plateau duration in Refs. [17,18] is actually underestimated. On this basis, the grain refinement model of KARMA et al [17,18] should be extended and the present overall solidification kinetic model is very helpful in predicting the plateau duration as well as analyzing the effect of back diffusion on grain refinement [19].
4 Conclusions
1) Due to the short time span of recalescence, back diffusion possesses almost no effect on the volume fraction solidified in this stage.
2) Incorporating back diffusion, solidification should end at a temperature between the prediction of Lever rule and Scheil’s equation, and the exact value is determined by the effect of back diffusion, the initial undercooling and the cooling rate.
References
[1] RAPPAZ M. Modelling of microstructure of formation in solidification process [J]. International Materials Reviews, 1989, 34: 93-123.
[2] RAPPAZ M, TH?VOZ P H. Solute diffusion model for equiaxed dendritic growth [J]. Acta Metallurgica, 1987, 35: 1487-1497.
[3] BECKERMANN C, VISKANTA R. Mathematical modeling of transport phenomena during alloy solidification [J]. Applied Mechanics Reviews, 1993, 46: 1-27.
[4] WANG C Y, BECKERMANN C. Multiphase solute diffusion model for dendritic alloy solidification [J]. Metallurgical and Materials Transactions A, 1993, 24: 2787-2802.
[5] RAPPAZ M, BOETTINGER W J. On dendritic solidification of multicomponent alloys with unequal liquid diffusion coefficients [J]. Acta Materialia, 1999, 47: 3205-3219.
[6] MARTORANO M A, BECKERMANN C, GANDIN CH-A. A solutal interaction mechanism for the columnar-to-equiaxed transition in alloy solidification [J]. Metallurgical and Materials Transactions A, 2003, 34: 1657-1674.
[7] GANDIN CH-A, MOSBAH S, VOLKMANN T H, HERLACH D M. Experimental and numerical modeling of equiaxed solidification in metallic alloys [J]. Acta Materialia, 2008, 56: 3023-3035.
[8] HERLACH D M. Non-equilibrium solidification of undercooled metallic melts [J]. Materials Science and Engineering Reports, 1994, 12: 177-272.
[9] YANG Chang-lin, YANG Geng-cang, LU Yi-ping, CHEN Yu-zeng, ZHOU Yao-he. Phase selection in highly undercooled Fe-B eutectic alloy melts [J]. Transactions of Nonferrous Metals Society of China, 2006, 16(1): 39-43.
[10] YANG Chang-lin, YANG Geng-cang, LIU Feng, ZHOU Yao-he. Purification and hypercooling of Fe-B eutectic alloy [J]. Acta Metallurgica Sinica, 2008, 44: 956-960.
[11] WANG Hai-feng, LIU Feng, YANG Gen-cang, ZHOU Yao-he. Modeling the overall solidification kinetics for undercooled single-phase solid-solution alloys. I. Model derivation [J]. Acta Materialia, 2010, 58: 5402-5410.
[12] WANG Hai-feng, LIU Feng, YANG Gen-cang, ZHOU Yao-he. Modeling the overall solidification kinetics for undercooled single-phase solid-solution alloys. II. Model application [J]. Acta Materialia, 2010, 58: 5411-5419.
[13] GALENKO P K, DANILOV D A. Local nonequilibrium effect on rapid dendritic growth in a binary alloy melt [J]. Physical Letters A, 1997, 235: 271-280.
[14] GALENKO P K, DANILOV D A. Model for free dendritic alloy growth under interfacial and bulk phase nonequilibrium conditions [J]. Journal of Crystal Growth, 1999, 197: 992-1002.
[15] WANG Hai-fang, LIU Feng, CHEN Zheng, YANG Gen-cang, ZHOU Yao-he. Analysis of non-equilibrium dendrite growth in a bulk undercooled alloy melt: Model and application [J]. Acta Materialia, 2007, 55: 497-506.
[16] WANG Hai-fang, LIU Feng, CHEN Zheng, YANG Gen-cang, ZHOU Yao-he. Effect of non-linear liquidus and solidus in undercooled dendrite growth: A comparative study in Ni-0.7 at.% B and Ni- 1 at.% Zr systems [J]. Scripta Materialia, 2007, 57: 413-416.
[17] KARMA A. Model of grain refinement in solidification of undercooled melts [J]. International Journal of Non-equilibrium Processing, 1998, 11: 201-233.
[18] SCHWARZ M, KARMA A, ECKLER K, HERLACH D M. Physical mechanism of grain refinement in solidification of undercooled melts [J]. Physical Review Letters, 1994, 73:1380-1383.
[19] WANG Hai-feng, LIU Feng, TAN Yi-ming. Modeling grain refinement for undercooled single-phase solid-solution alloys [J]. Acta Materialia, 2011, 59: 4787-4797.
王海丰,刘 峰,王 慷,翟海民
西北工业大学 凝固技术国家重点实验室,西安 710072
摘 要:基于平均体积方法建立过冷单相固溶体合金的凝固动力学模型并研究反向扩散在凝固动力学过程中的作用。模型在过冷Ni-15%Cu(摩尔分数)合金快速凝固中的应用表明:反向扩散显著影响凝固结束温度但对再辉阶段凝固的固相体积分数影响不明显。与Herlach观点相反,凝固结束温度介于杠杆定律和Scheil方程的预测值之间,且其具体值由反向扩散、初始过冷度和冷却速率决定。
关键词:Ni-Cu合金;快速凝固;扩散;过冷
(Edited by LI Xiang-qun)
Foundation item: Project (2011CB610403) supported by the National Basic Research Program of China; Project (51125002) supported by the National Science Fund for Distinguished Young Scholars of China; Projects (51101122, 51071127, 50901059) supported by the National Natural Science Foundation of China; Project (111502) supported by the Huo Yingdong Young Teacher Fund, China; Projects (66-QP-2010, 24-TZ-2009) supported by the Free Research Fund of State Key Laboratory of Solidification Processing, China; Projects (JC201008, JC200801) supported by the Fundamental Research Fund of Northwestern Polytechnical University, China; Project (B08040) supported by the Program of Introducing Talents of Discipline to Universities, China
Corresponding author: LIU Feng; Tel: +86-29-88460374; E-mail: liufeng@nwpu.edu.cn
DOI: 10.1016/S1003-6326(11)61226-0

