
Preparation of poly(propylene carbonate)/organophilic rectorite nanocomposites via direct melt intercalation
WAN Chun-jie(万春杰)1,2, YU Jian-ying (余剑英)1, SHI Xiao-jian (石小建)1, HUANG Li-hua (黄丽华)1
1.College of Materials Science and Engineering, Wuhan University of Technology, Wuhan 430070, China;
2.School of Chemical Engineering and Pharmacy, Wuhan Institute of Chemical Technology, Wuhan 430073, China
Received 10 April 2006; accepted 25 April 2006
Abstract: The completely degradable nanocomposites comprised of poly(propylene carbonate)(PPC) and organo-modified rectorite (OREC) were prepared by direct melt intercalation. The structure and mechanical properties of PPC/OREC nanocomposites were investigated. The wide-angle X-ray diffraction (WAXD) results show that the galleries distance of OREC is increased after PPC and OREC melt intercalation, which indicates that PPC molecular chain has intercalated into the layers of OREC. The PPC/OREC nanocomposites with lower OREC content show an increase in thermal decomposition temperature compared with pure PPC. The tensile strength and impact strength of PPC/OREC nanocomposites are improved. When the mass fraction of OREC is 4%, the tensile strength and impact strength of the PPC/OREC nanocomposite increase by 22.86% and 48.58% respectively, compared with pure PPC.
Key words: poly(propylene carbonate); rectorite; degradation; nanocomposites
1 Introduction
In recent years, polymer/layered silicate nanocomposites(PLSNs) have been reported in many papers because they often exhibit remarkable improvement of mechanical properties, enhanced barrier properties, reduced flammability and enhanced biodegrability of a biodegradable polymer[1].
Intercalation methods for nanocomposites include in situ polymerization, melt polymer intercalation, exfoliation adsorption, template synthesis, and direct solution intercalation. The melt intercalation method involves mixing the layered silicates with the polymer matrix above its melting point, which provides a convenient technique for preparation of the nanocomposites, polymer/clay nanocomposites could be fabricated by a direct route in the polymer melt in which the polymer chains are diffused into the space between the silicate layers or galleries[2-4].
Poly(propylene carbonate)(PPC) is a degradable resin. However, the mechanical properties of PPC are poor. In order to improve the mechanical properties, many organic and inorganic fillers have been compounded with the PPC to fabricate composites[5]. In this study, we report the direct melt intercalation of PPC and organo-modified rectorite(OREC). The mechanical properties of PPC/OREC nanocomposites were improved greatly, which was expected to meet the need of practical applications. The mechanism properties and the morphology of PPC/OREC nanocomposites were investigated.
2 Experimental
2.1 Materials
PPC was supplied by Mengxi High-tech Group Company. Purified sample of rectorite was from Celebrities Real Estate Development Group Co, Ltd.
2.2 Preparation of organo-rectorite
The inorganic cations in the rectorite were first ion exchanged with sodium ions as follows: 10 g rectorite (size of 50 μm) was refluxed in about 100 mL of aqueous solution containing 1.2 g NaCl for 48 h. The reaction temperature was controlled at 50-60 ℃, and the slurry was washed with distilled water until no Cl- was detected, which was performed using 0.1 mol/L AgNO3. The Na-exchanged rectorite was then added to a 500 mL plastic flask, containing 12 g cetyltrimethyl ammonium bromide in 200 mL deionized water. The mixture of rectorite and alkyl ammonium salt was stirred at 70 ℃ for 3 d. The resulting organo-rectorite (OREC) was washed with distilled water until no precipitate was observed upon titrating the filtrate with 0.1 mol/L AgNO3. The final OREC in the form of fine powder was kept in a dryer for further use.
2.3 Preparation of PPC/OREC nanocomposites
PPC/OREC nanocomposites were prepared via direct melt mixing PPC, OREC and various processing additives in the mixing chamber of a Haake Rheometer 90. The mixing temperature was set at 120 ℃, the rotor speed was 50 r/min, and the mixing time was 10 min. The PPC was introduced into the chamber of the mixer as quickly as possible, followed by adding OREC. Upon completion of mixing, the nanocomposite samples were removed from the mixing chamber, and the nanocomposites prepared were then molded into sheets of 4 mm in thickness by hot pressing at 120 ℃ and 10 MPa for 10 min, followed by cooling to room temperature at 10 MPa. The sheets were prepared for structure characterization and properties measurements[6].
2.4 Measurements
Infrared absorption spectra were collected on a Perkin-Elmer Paragon 1000 FT-IR Spectrometer.
Wide-angle X-ray diffraction (WAXD) measurement was carried out using a D8-advance X-ray diffractionmeter (provided by Bruker) with Cu Kα radia- tion (λ=0.154 nm) and a scanning rate of 12.0 (?)/min, diffraction angle from 1.7? to 60?. BRAGG’s equation, λ=2dsin θ, was used to calculate the crysta-
llographic spacing. The specimens for WAXD were 15 mm×18 mm cut from molded sheets with 4 mm in thickness.
The TEM micrographs were obtained using a JEM-100CX Ⅱtransmission electron microscope
(produced by JEOL)operating at an accelerating voltage of 100 kV. TEM samples for particle characterization were cryogenically cut into ultra thin sections (30-50 nm in thickness) with a diamond knife at -65 ℃ using a Leica Ultracut ultramicrotome (Leica Inc), and they were collected in a trough filled with water and placed on a gold grids with size of 50 μm for observation.
Thermal transition temperatures of PPC and OREC/PPC nanocomposite were determined using differential scanning calorimetry (DSC) (Model DSC 2920, TA Instrument). The used temperature profile ranged from 50 to 400 ℃ and experiments were performed at a scanning rate of 20 ℃/min under the protection of N2 gas with a flow rate of 90 mL/min.
Tensile strength tests were performed according to ASTM D638. The tensile tests were conducted at a crosshead speed of 50 mm/min. Notched Charpy impact strength tests were performed according to ASTM D256 standard at room temperature.
3 Results and discussion
3.1 Fourier transform infrared spectroscopy
FOURIER transform infrared(FT-IR) spectra, as shown in Fig.1, reveal the chemical groups in REC and OREC. It can be seen that the C―H stretching vibration of alkylammonium chain appears at 2 921 cm-1 and
2 850 cm-1, indicating the increase in hydrophobic pro- perty of resulting OREC[7].
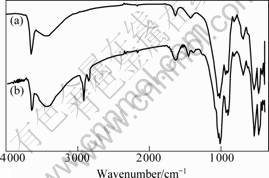
Fig.1 FT-IR spectra of pristine REC (a) and OREC (b)
3.2 WAXD analysis of nanocomposites
WAXD is the most powerful and reliable technique to study the dispersion of organoclay inside the polymer matrix. The X-ray diffraction(XRD) patterns of REC and OREC are shown in Fig.2.
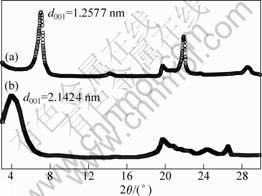
Fig.2 X-ray diffraction patterns of REC (a) and OREC (b)
It can be seen from Fig.2 that the peak corresponding to the basal spacing (d001) of the REC appears at 2θ =7.02? (corresponding d001 spacing is 1.257 7 nm). The d001 peak of the OREC is shifted to low angle corresponding to an increase in spacing from 1.257 7 nm to 2.142 4 nm. The higher basal spacing of OREC in the nanocomposites as compared to virgin REC is due to the intercalation of polymer chains inside the clay layers[8].
Nano-scale structure of the PPC/OREC composites XRD technology has been extensively used to characterize the gallery spacing of original clay and organo-treated clay based on the basal d-spacing. Fig.3 shows the XRD patterns of the OREC and PPC/OREC nanocomposites with different OREC contents. All the (001) plane diffraction peaks of PPC/OREC nanocomposites shift to 2.142 4 nm with a basal spacing of 3.221 8 nm, demonstrating the basal spacing of OREC increased from 2.142 4 nm to 3.221 8 nm. It is believed that the increased spacing results from the intercalation of PPC chains into the gallery of OREC. Because of the great increase in the gallery spacing, an intercalated nano-structure of PPC/OREC composites was obtained. It should be noted that the PPC/OREC nanocomposites with different OREC contents had the same basal spacing[9].
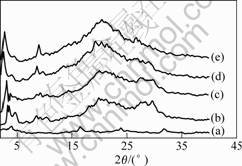
Fig.3 X-ray diffraction patterns of composites: (a) PPC/2%OREC; (b) PPC/4%OREC; (c) PPC/6%OREC; (d) PPC/8%OREC; (e) OREC
3.3 Morphologies of OREC/PPC nanocomposite
Fig.4 shows the typical transmission electron microscope(TEM) image of PPC/OMMT nanocompo-
sites. The result is consistent with the results of XRD. The dark lines in Fig.4 are the OREC layers. It can be seen from area A of Fig.4, that individual OREC layers are well-dispersed in the PPC matrix, and some tactoids or multilayer particles are shown in area B of Fig.4. The observed individual layers of OREC reveal that the nanocomposites have an intercalated exfoliated structure. The delaminated OREC layers are about 20-50 nm in length. For the intercalated particles, the gallery spacings between the tactoids layers are enlarged to about 30 nm. The TEM micrograph clearly reveals the nano-scale dispersion of OREC in PPC matrix together with very large aspect ratios[10].
3.4 Thermal analysis
Fig.5 shows the TGA curves for PPC/OREC nanocomposites. It can be seen that the temperature with 5% mass loss of the PPC/OREC composites is about 25-30 ℃ higher than that of neat PPC. The

Fig.4 TEM image of PPC/4% OREC nanocomposites
improvement of thermal stability can be profited from the shielding effect of OREC layers. The enhanced thermal stability implies the well-dispersed of OREC in PPC matrix and the amorphous PPC chains become more stable within the gallery of OREC. Due to the thermal stable character of OREC, OREC layers exhibit obvious barrier effect to stop the evaporation of the small molecules generated during the thermal decomposition and effectively limit the progress of PPC’s decomposition. For the PPC/OREC composite with high OREC loading level of 8%(mass fraction) OREC, however, the 5% mass loss temperature becomes smaller compared with those containing lower OREC contents. It is believed that the organic ammonium compounds within the OREC result in the poor thermal stability[11].
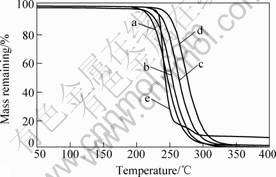
Fig.5 TGA curves of pure PPC and PPC/OREC nanocomposites with various OREC contents: (a) PPC;(b) PPC/2%OREC; (c) PPC/4%OREC; (d) PPC/6%OREC;(e) PPC/8%OREC
3.5 Mechanical properties of PPC/OREC nanocom-
posites
Mechanical properties tests were performed on PPC/OREC nanocomposites with OREC loadings varying from 2% to 10%, and the results are shown in Figs.6 and 7. It is found that the mechanical properties of the four PPC/OREC nanocomposites are improved significantly within 2%-8% of OREC loadings. With 4% OREC loadings, the stiffness and impact strength of the nanocomposites simultaneously reach the maximum, the tensile strength of the PPC/OREC nanocomposite increase by 22.86%, the impact strength of the PPC/OREC nanocomposite increase by 48.58%. Compared with pure PPC, the tensile strength and impact strength of PPC/OREC nanocomposites are improved. Therefore, the intercalated structure of PPC/OREC nanocomposites may be favorable to enhancing mechanical properties[12].
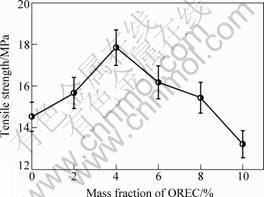
Fig.6 Tensile strength of composites with different OREC contents
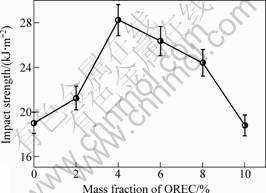
Fig.7 Impact strength of composites with different OREC contents
The improvement of mechanical properties of nanocomposites may be due to several reasons: the content of OREC, the interaction of OREC and PPC, compatible of OREC phase and PPC phase. When the content of OREC is lower than 4%, the mechanical properties of composites are improved with the increase of OREC, because OREC is dispersed in matrix to form nanocomposite. When the content of OREC is higher than 4%, OREC tends to reunite, which leads to the decrease of mechanical properties.
4 Conclusions
1) Intercalated and partially exfoliated PPC/OREC nanocomposites were fabricated via direct melt compounding of OREC and PPC. The nanoscale dispersion of OREC sheets was confirmed by both X-ray diffraction and TEM. However, the mechanical strength of the PPC/OREC nanocomposites is increased with the increase of OREC content to reach a maximum and then decreased.
2) The galleries distance of OREC is increased after PPC and OREC melt intercalation, which indicates that PPC molecular chain has intercalated into the layers of OREC. Thermal decomposition temperature of the nanocomposites increases with the increase of OREC content because of the shielding effect of the nano-scale layers of OREC.
References
[1] Thostenson E T, LI C Y, Chou T W. Nanocomposites in context [J]. Composites Science and Technology, 2005, 65: 491-516.
[2] Ray S S, Yamad K, Okamoto M. New polylactide-layered silicate nanocomposites. 2.Concurrent improvements of material properties, biodegradability and melt rheology[J]. Polymer, 2003, 44: 857-866.
[3] Mishra J K, Hwang K J, Ha C S. Preparation, mechanical and rheological properties of a thermoplastic polyolefin(TPO)/organoclay nanocomposite with reference to the effect of maleic anhydride modified polypropylene as a compatibilizer[J]. Polymer, 2005, 46: 1995-2002.
[4] Wang M, Hsieh A J, Rutledge G C. Electrospinning of poly(MMA-co-MAA) copolymers and their layered silicate nanocomposites for improved thermal properties[J]. Polymer, 2005, 46: 3407-3418.
[5] Xu J, Li R K Y, Xu Y. Preparation of poly(propylene carbonate)/organo-vermiculite nanocomposites via direct melt intercalation[J]. European Polymer Journal, 2005, 41: 881-888.
[6] Ren Jie, Huang Yan-xia, Liu Yan. Preparation, characterization and properties of poly(vinyl chloride)/compatibilizer/organophilic montmorillonite nanocomposites by melt intercalation[J]. Polymer Testing, 2005, 24: 316-323.
[7] Liu T X, Liu Z H, Ma K X. Morphology, thermal and mechanical behavior of polyamide 6/layered-silicate nanocomposites[J]. Composites Science and Technology, 2003, 63: 331-337.
[8] Chen Yong-chun, Zhou Shu-xue, Yang Hai-hua. Preparation and characterization of nanocomposite polyurethane[J]. Journal of Colloid and Interface Science, 2004, 279: 370-378.
[9] Ray S S, YAMAD K. Polymer/layered silicate nanocomposites: a review from preparation to processing[J]. Prog Polym Sci, 2003, 28: 1539-1641.
[10] McNally T, Murphy W R, Lew C Y. Polyamide-12 layered silicate nanocomposites by melt blending[J]. Polymer, 2003, 44: 2761-2772.
[11] Xu J, Li R K Y, Meng Y Z, MAI Y W. Biodegradable poly(propylene carbonate)/montmorillonite nanocomposites prepared by direct melt intercalation[J]. Materials Research Bulletin, 2006, 41: 244-252.
[12] Yoon P J, Hunter D L, Paul D R. Polycarbonate nanocomposites: Part 2. Degradation and color formation[J]. Polymer, 2003, 44: 5341-5354.
(Edited by YANG You-ping)
Corresponding author: YU Jian-ying; Tel: +86-13607134766; E-mail: wrm222@126.com