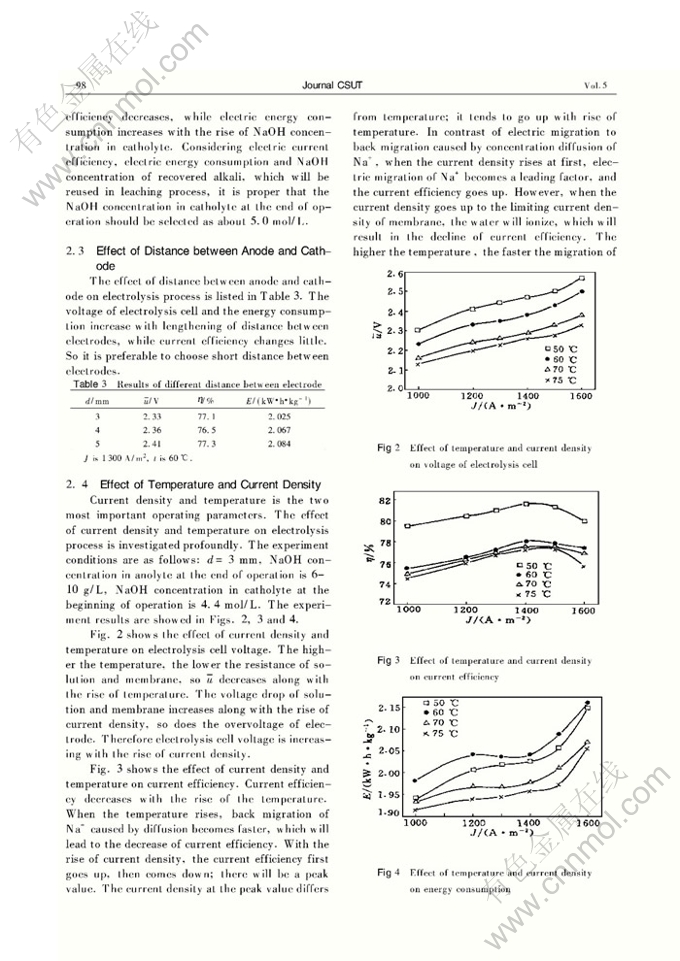
FREE ALKALI RECOVERY FROM NaOH LEACHING LIQUOR OF TUNGSTEN ORE BY ELECTROLYSIS WITH ION-EXCHANGE MEMBRANE
来源期刊:中南大学学报(英文版)1998年第2期
论文作者:Zhang Guiqing Zhang Qixiu
文章页码:23 - 26
Key words:ion-exchange membrane; electrolysis; tungsten; caustic soda
Abstract: Caustic soda leaching liquor of tungsten ore always contains a certain amount of free alkali. General method of removing free alkali in the liquor is to neutralize it by inorganic acid. A new method, i.e. applying electrolysis with ion-exchange membrane to recover free alkali from caustic soda leaching liquor of tungsten ore, was proposed in the paper. In the electrolysis cell the effective area of membrane is 100 mm×100 mm, the anode is β PbO2 plated at screen of titanium net, the cathode is active nickel plated at screen of stainless steel net, and the membrane is a kind of perfluorsulphonic acid membrane. Some effect factors of electrolysis process including the NaOH concentration in anolyte and catholyte, distance between electrodes, temperature, current density, are investigated. The results show that, by choosing proper operating condition, the specific energy consumption can be controlled within 2 kWh・kg-1NaOH. Electrolysis with ion-exchange membrane can not only recover free alkali from caustic soda leaching liquor, reduce the consumption of inorganic acid, but also utilize the hydrogen gas produced in the process to produce tungsten powder.