J. Cent. South Univ. Technol. (2008) 15: 520-525
DOI: 10.1007/s11771-008-0098-4
Separation of aluminosilicates and diaspore from diasporic-bauxite by selective flocculation
HUANG Chuan-bing(�ƴ���)1, ZHANG Lin(�� ��)2, WANG Yu-hua(��ع��)2, LAN Ye(�� Ҷ)2
(1. Institute of Process Engineering, Chinese Academy of Sciences, Beijing 100080, China;
2. School of Resource Processing and Bioengineering, Central South University, Changsha 410083, China)
Abstract: The flocculation tests of four pure minerals (diaspore, kaolinite, illite, pyrophyllite) and bauxite ore were investigated by the sedimentation. The dispersion behavior of the four pure minerals shows a very good consistency with the variation of zeta potential. The concentrate with the mass ratio of Al2O3 to SiO2 (m(Al2O3)/m(SiO2)) 8.90 and the recovery of Al2O3 86.98% is obtained from bauxite ore (m(Al2O3)/m(SiO2)=5.68) in pH range of 9.5-10.0 by using sodium carbonate (5 kg/t) and sodium polyacrylate (7 g/t) as dispersant and flocculant respectively. Sodium carbonate acts as both pH modifier and favorable dispersant for aluminosilicates. The high performance of sodium polyacrylate on flocculation for diaspore is contributed to the carboxyl of sodium polyacrylate that interacts with active Al sites on diaspore by chemical absorption, and the hydrogen bond effects between hydroxyl group of macromolecule and surface Al��OH on diaspore to accelerate the sedimentation of diaspore.
Key words: diasporic-bauxite; sodium carbonate; sodium polyacrylate; ratio of Al2O3 to SiO2; selective flocculation; flocculant; dispersant
1 Introduction
Unlike the bauxite ores in other countries, more than 98% of the bauxite in China is of diasporic-bauxite. The gangue minerals in bauxites mainly consist of kaolinite, pyrophyllite and illite. And the gangue minerals are often presented as finely disseminated inclusions[1-2]. The diasporic-bauxite is characterized by a high content of Al2O3 and SiO2 and a low mass ratio of Al2O3 to SiO2 (m(Al2O3)/m(SiO2), usually 5-6) compared with gibbsite-bauxite and boehmite-bauxite in other countries[3]. Bayer process[4] to treat diasporic bauxite needs bauxite with m(Al2O3)/m(SiO2) larger than 8. The sintering process is extremely energy-intensive and environmentally unfriendly, incurring a costly alumina production[5]. Therefore, it is highly desirable to increase the m(Al2O3)/m(SiO2) of the Chinese diasporic-bauxite ores by a low cost physical separation process so that the Bayer process can be applied directly. Due to its economic importance, the vast diasporic-bauxite resource in China has been extensively studied to develope a flotation-Bayer process[6-9]. But this process also has some problems such as difficulty of dewatering, long and complex flow sheets, large amount of reagents remaining in concentrate.
Selective flocculation is one of the techniques applicable to the enrichment of ultrafine particles in pulp. Some progresses have been achieved in applying this technique for the separation of finely disseminated valuable minerals from various ores[10-14]. In this work, the separation of aluminosilicates and diapore from diasporic-bauxite ore was investigated by using selective flocculation.
2 Experimental
2.1 Materials and reagents
The pure minerals of diaspore, kaolinite, illite and pyrophyllite were obtained from Henan and Zhejiang Deposits in China. All of the pure mineral samples were hand-picked, ground by porcelain mill with agate ball, and screened to the samples with size less than 38 ��m. Due to the discrepancy of grindability, the particle size analysis by Sympatec Helos laser sizer (H1305) showed that the average size of diaspore, kaolinite, illite, pyrophyllite was 13.54, 12.70, 6.95 and 7.24 ��m, respectively. And the chemical compositions of pure minerals are listed in Table 1.
The bauxite ore used in these experiments was sampled from the Henan Deposits in China. Based on the X-ray diffraction(XRD) analysis, the compositions of the bauxite ore are diaspore (53.4%-53.8%), pyrophyllite (14.7%-17.4%), illite (16.5%-15.8%), chlorite (9.3%- 12.6%) and kaolinite (3.0%-3.5%). The chemical analysis (Table 2) indicates the contents of Al2O3 and SiO2 in the bauxite ore are 64.69% and 11.39%, respectively, i.e. m(Al2O3)/m(SiO2) of 5.68. The bauxite ore samples were crushed to the samples with size less than 3 mm and separated into batches with mass of 0.5 kg for the subsequent experiments.
Table 1 Chemical compositions of pure minerals (mass fraction, %)

Table 2 Chemical compositions of bauxite ore (mass fraction, %)

The commercial product of sodium polyacrylate (0.05%, mass fraction) was used as the flocculant; sodium carbonate (5%, mass fraction) was used as dispersant; sodium hydroxide and hydrochloric acid with concentration of 0.1 mol/L were used for pH adjustment.
2.2 Methods
Single-stage selective flocculation tests were performed in a 100 mL glass cylinder with 35 mm in diameter by sedimentation tests method. All experiments were performed at a pulp density of 5% (mass fraction). After adjusting pH, sodium polyacrylate was added, and the cylinder was turned up and down 10 times after each addition. The pulp was allowed to settle for 50 s and the supernatant of the 80% upper part of the cylinder was siphoned off. The sediment of diaspore was dried and weighed to determine the sedimentation yield. In selective flocculation experiments of bauxite ore, tests were carried out in a 5 L graduated cylinder with 20 cm in diameter. Bauxite ore was ground by steel bars to obtain sample with particle size less than 0.074 mm (accounting for 90%). In addition, sodium carbonate was added to adjust pH value of pulp instead of sodium hydroxide. The flow sheet of selective flocculation on diasporic-bauxite is shown in Fig.1.
Zeta potentials of the mineral samples were measured with Delsa 440SX. The infrared spectra of samples with or without reagents pretreated were obtained using the KBr technique by the model FTIR-750 infrared spectrophotometer from Nicolet Co, USA. XRD analysis was performed on Shimadzu D/MAX-rA X-ray diffraction instrument from Japan. DM 4000M digital metallographic microscope made in Germany was used to take photos on morphologies of particles and flocs.

Fig.1 Flow sheet of selective flocculation on diasporic-bauxite ore
3 Results
3.1 Flocculation-dispersion behavior of four pure minerals
Fig.2 shows the flocculation-dispersion behavior of four pure minerals as a function of pH value. Under acidic conditions, the diaspore particles reveal a significant aggregation. Along with increasing pH value, aggregation of the diaspore particles is weakened gradually. When pH value is above 9, the diaspore particles are dispersed very well. For the three types of aluminum silicate minerals, the pH value has less effect on their dispersion behavior. Four minerals show good dispersion behavior under alkaline conditions, which will provide favorable conditions for separation by selective flocculation at acidic pH value.
The effect of pH value on zeta potential of minerals in distilled water is shown in Fig.3. It can be seen from Fig.3 that the dispersion behavior of the four pure minerals shows a very good consistentcy with the variation of zeta potential. With the increase of the pH value, the absolute value of zeta potential on the surface of mineral particles increases, resulting in larger repulsive force among particles and better dispersion.
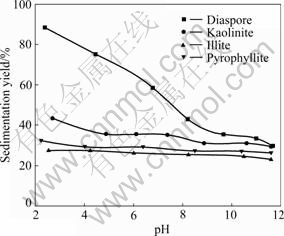
Fig.2 Effect of pH value on dispersion of mineral particles

Fig.3 Effect of pH value on zeta potential of minerals in distilled water
Fig.4 presents the flocculation-dispersion behavior of the four pure minerals by using sodium polyacrylate with a concentration of 2 mg/L at different pH values. It can be seen that in the pH range of 2-12, sodium polyacrylate shows very strong flocculation performance to diaspore and the sedimentation yield of diaspore is above 95%. The sedimentation yield of kaolinite gradually increases with the increase of pH value. Illite and pyrophyllite are almost dispersed at pH above 9 even by using sodium polyacrylate in this pH range. The sedimentation yield difference between diaspore and illite or pyrophyllite can reach 60%, but that between diaspore and kaolinite is 20% at pH above 9. Because the content of kaolinite in bauxite ore is about 3.0%-3.5%, it is possible to separate diaspore from aluminosilicates to achieve the concentrate with higher m(Al2O3)/m(SiO2) by selective flocculation.

Fig.4 Effect of pH value on flocculation of mineral particles with 2 mg/L sodium polyacrylate
3.2 Selective flocculation of diasporic-bauxite ore
It was reported that sodium carbonate can act as both pH modifier and favorable dispersant for aluminosilicates[15]. In selective flocculation of the bauxite ore, sodium polyacrylate was used as an flocculant for diaspore and sodium carbonate used as a dispersant. The results are shown in Table 3. When pH is adjusted to 9.5-10 by 3 kg/t Na2CO3, all aluminosilicates are well dispersed. Then sodium polyacrylate can selectively flocculate diaspore more effectively. It can be seen that the concentrate with m(Al2O3)/m(SiO2) of 8.9 and Al2O3 recovery of 86.98% is obtained after sedimentation three times on the bauxite ore with m(Al2O3)/m(SiO2) of 5.68. The total dosage of sodium carbonate is 5 kg/t, while that of sodium polyacrylate is 7 g/t.
Table 3 Results of selective flocculation separation of diaspore bauxite

The compositions of feed ore, concentrate and tailing after the selective flocculation were examined by XRD analysis, as shown in Fig.5. In the comparison with feed ore, the concentrate is composed mainly of diaspore and a small amount of illite. The intensity of diaspore peaks increases, which means its relative content increases, and the intensity of illite peaks declines, showing a decrease of the content of illite. The intensity of diaspore peaks in tailing distinctively decreases, while peak intensities of the three aluminosilicates obviously increase, indicating a significant decrease of diaspore and enrichment of three silicate minerals. The test results reveal that sodium polyacrylate shows a good capability for the selective flocculation separation of bauxite ore.

Fig.5 XRD patterns of bauxite ores: (a) Tailing; (b) Concentrate; (c) Feed
4 Discussion
Sodium polyacrylate is a water-soluble poly- electrolyte that can be dissociated into low molecular Na+ ions and macro-ions with a large number of carboxyl anions. Its molecular formula is (C3H3NaO2)n. The molecular structure is shown in Fig.6. XIA et al[16] used the electron spin resonance(ESR) combined with spin-labeling technique to investigate the adsorption configuration and molecular motion behavior of sodium polyacrylate at Al2O3/water interface. The results show that the adsorption of sodium polyacrylate on Al2O3 surface, which is induced by electrostatic attraction, is the multipoint adsorption, where most segments of sodium polyacrylate are bound to the surface and a few residual segments are suspended in the solution. The adsorption between sodium polyacrylate and Al2O3 is electrostatic adsorption, and the adsorption layer on the surface of the solid is more compact than the general polymer adsorption layer.
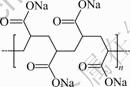
Fig.6 Molecular structure of sodium polyacrylate
In order to understand the selective mechanism of sodium polyacrylate on separating diaspore from aluminosilicates, the micrographs of diaspore flocs were taken, as shown in Fig.7. Sodium polyacrylate is a linear polymer flocculant which contains large amounts of solid pro-active groups in its long-chain molecules. It can be seen from Fig.7(a) that diaspore particles are in well dispersed situation after agitating for 1 min. The particles are in fine and normal distribution. But if 75 mg/L Na2CO3 and 2 mg/L sodium polyacrylate are added in solution, as shown in Fig.7(b), bridge-bonds of macromolecule are formed, resulting in generation of dense diaspore flocs. This accelerates the sedimentation speed of diaspore particles, leading to different sedimentation speeds between diaspore and aluminosilicates.
Infrared spectra of minerals and reagents were recorded to detect the adsorption type of sodium polyacrylate on diaspore particles, as shown in Fig.8. The infrared spectra of sodium polyacrylate and sodium polyacrylate reacted with Al3+ are shown in Figs.8(a) and (b), respectively. For sodium polyacrylate, stretching frequency of 1 440 cm-1 for ��COO-, and stretching frequency of 3 453 cm-1 for ��OH can be observed. A new stretching frequency of 1 624 cm-1 can be seen and stretching frequency of 1 440 cm-1 is weakened in Fig.8(b), which means appearance of a new compound formed by the chemical reaction between Al3+ and sodium polyacrylate. Figs.8(c) and (d) are the infrared spectra of diaspore and diaspore treated by sodium polyacrylate, respectively. It can be seen that there is a new stretching frequency of 1 640 cm-1 in Fig.8(d), corresponding to the new stretching frequency of 1 624 cm-1 found in Fig.8(b), indicating that a chemical adsorption happens between diaspore and sodium polyacrylate. When diaspore is crushed, the broken bonds of diaspore surface are Al��O and Al��OH bonds. The chemical adsorption of polyacrylate ion on diaspore may take place through ��COOH and Al��OH bonding to form ��COO��Al and dehydroxylate. The number of the broken Al��O bonds on the surface of kaolinite, illite and pyrophyllite is much less than that of diaspore. This is the reason why a new stretching frequency cannot be found in the infrared spectra of aluminosilicates treated by sodium polyacrylate.

Fig.7 Micrographs of diaspore particles (Black) under different conditions: (a) No reagent; (b) With 2 mg/L sodium polyacrylate and 75 mg/L Na2CO3
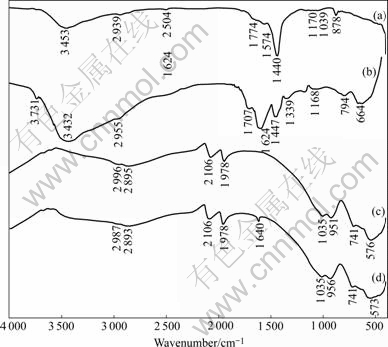
Fig.8 Infrared spectra of minerals and reagents: (a) Sodium polyacrylate; (b) Sodium polyacrylate+Al3+; (c) Dispore; (d) Dispore+ sodium polyacrylate
5 Conclusions
1) The dispersion behavior of four pure minerals shows a very good consistency with the change of zeta potential. With the increase of the pH value, the absolute value of zeta potential on the surface of mineral particles increases, resulting in larger repulsive force among particles.
2) The bauxite ores with m(Al2O3)/m(SiO2) of 5.68 and average particle size less than 0.074 mm (accounting for 90%) and 10% solid concentration are dispersed by adding 5 kg/t Na2CO3, then flocculated by adding 7 g/t sodium polyacrylate. After triple sedimentations, the Al2O3 recovery of concentrate reaches 86.98% with m(Al2O3)/m(SiO2) of 8.9.
3) Sodium carbonate can act as both pH modifier and favorable dispersant for aluminosilicates. The high performance of sodium polyacrylate on flocculation for diaspore is due to that carboxyl of sodium polyacrylate interacts with active Al sites on diaspore by chemical absorption.
References
[1] WANG Yu-hua, HU Yue-hua, LIU Xiao-wen. Flotation de-silicating from diasporic-bauxite with cetyl trimethylammonium bromide [J]. J Cent South Univ Technol, 2003, 10(4): 324-328.
[2] LU Yi-ping, ZHANG Guo-fan, FENG Qi-ming, OU Le-ming. A new collector RL for flotation of bauxite[ J]. J Cent South Univ Technol, 2002, 9(1): 21-234.
[3] WANG Yu-hua, HU Yue-hua, HE Ping-bo, GU Guo-hua. Reverse flotation for removal of silicates from diasporic-bauxite [J]. Minerals Engineering, 2004, 17(1): 63-68.
[4] CHEN Wan-kun, PENG Guan-cai. The digestion technology of diasporic bauxite [M]. Beijing: Metallurgical Industry Press, 1997. (in Chinese)
[5] FAN Xiao-hui, LI Guang-hui, JIANG Tao, QIU Guan-zhou. Sintering properties of high iron gibbsite-type bauxite ores [J]. Metal Mine, 2002(7): 16-18. (in Chinese)
[6] HU Yue-hua, WANG Yu-hua, WANG Dian-zuo. Flotation chemistry of aluminum and silicate and desilication of bauxite [M]. Beijing: Science Press, 2004. (in Chinese)
[7] HU Yue-hua, JIANG Hao, QIU Guan-zhou, WANG Dian-zuo. Solution chemistry of flotation separation of disporic bauxite [J]. The Chinese Journal of Nonferrous Metals, 2001, 11(1): 125-130. (in Chinese)
[8] HUANG Chuan-bing, WANG Yu-hua, CHEN Xing-hua, LAN Ye, HU Ye-min, YU Fu-shun. Review of research on desilication of bauxite ores by reverse flotation [J]. Metal Mine, 2005(6): 21-24. (in Chinese)
[9] HUANG Chuan-bing, WANG Yu-hua, LAN Ye. Mechanism of selective flocculation separation of diasporic bauxite with organic flocculant HSPA [J]. The Chinese Journal of Nonferrous Metals, 2006, 16(7): 1250-1256. (in Chinese)
[10] OZKAN A, USLU Z, DUZYOL S, UCBEYIAY H. Correlation of shear flocculation of some salt-type minerals with their wettability parameter [J]. Chemical Engineering and Processing: Process Intensification, 2007, 46(12): 1341-1348.
[11] JONAS A M. Enhanced flocculation and dewatering of clay mineral dispersions [J]. Powder Technology, 2007, 179 (1/2): 73-78.
[12] KLEIN WOLTERINK J, KOOPAL L K, COHEN STUART M A, van RIEMSDIJ W H. Surface charge regulation upon polyelectrolyte adsorption, hematite, polystyrene sulfonate, surface charge regulation: Theoretical calculations and hematite-poly(styrene sulfonate) system [J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2006, 291/300(1/3): 13-23.
[13] OZKAN A, UCBEYIAY H, AYDOGAN S. Shear flocculation of celestite with anionic surfactants and effects of some inorganic dispersants [J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2006, 281/290(1/3): 92-98.
[14] SOMASUNDARAN P, RUNKANA V. Investigation of the flocculation of colloidal suspensions by controlling adsorbed layer microstructure and population balance modelling [J]. Chemical Engineering Research and Design,2005, 83(7): 905-914.
[15] WANG Yu-hua, HU Yue-hua, HE Ping-bo, GU Guo-hua. Test research on selective desliming of bauxite [J]. Metal Mine, 2004(4): 38-40. (in Chinese)
[16] XIA Xiao-hong, LI Guo-xi, KUANG Ya-fei, LIU Guo-gen. The ESR investigation on adsorption behaviour of sodium polyacrylate at Al2O3/water interface [J]. Ion Exchange and Adsorption, 2003, 19(2): 127-132. (in Chinese)
Foundation item: Project(2005CB623701) supported by the Major State Basic Research Development Program of China
Received date: 2007-11-16; Accepted date: 2007-12-27
Corresponding author: WANG Yu-hua, PhD, Professor; Tel: +86-731-8830545; E-mail: wangyh@mail.csu.edu.cn
(Edited by CHEN Wei-ping)