J. Cent. South Univ. (2012) 19: 1813-1816
DOI: 10.1007/s11771-012-1213-0
Carbothermic reduction of alumina with carbon in vacuum
YU Qing-chun(郁青春), YUAN Hai-bin(袁海滨), ZHU Fu-long(朱富龙),
ZHANG Han(张晗), WANG Chen(王辰), LIU Da-chun(刘大春), YANG Bin(杨斌)
National Engineering Laboratory of Vacuum Metallurgy,
Key Laboratory of Breeding Base of Complex Nonferrous Metal Resources Clear Utilization in Yunnan Province,
State Key Laboratory of Nonferrous Metal Vacuum Metallurgy,
Kunming University of Science and Technology, Kunming 650093, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2012
Abstract: Carbothermic reduction alumina in vacuum was conducted, and the products were analysed by means of XRD and gas chromatography. Thermodynamic analysis shows that in vacuum the initial carbothermic reduction reaction temperature reduces compared with that under normal pressure, and the preferential order of products is Al4O4C, Al4C3, Al2OC, Al2O and Al. Experiment results show that the carbothermic reduction products of alumina are Al4O4C and Al4C3, and neither Al2OC, Al2O or Al was found. During the carbothermic reduction process, the reaction rate of Al2O3 and carbon decreases gradually with increasing time. Meanwhile, lower system pressure or higher temperature is beneficial to the carbothermic reduction of alumina process. Al4O4C is firstly formed in the carbothermic reaction, and then Al4C3 is formed in lower system pressure or at higher temperature.
Key words: alumina; carbothermic reduction; vacuum; aluminum
1 Introduction
Aluminum is currently produced industrially via the Hall-He?roult process by dissolving Al2O3 in fused NaF-AlF3 (cryolite) followed by direct current electrolysis. The main drawbacks of the electrolytic production are its high energy consumption, the release of perfluorocarbons, and the high specific CO2-equiv emissions. The greenhouse gas emission by the electrolytic Al production contributes to 2.5% of the world anthropogenic CO2-equiv emissions [1-3]. During the past years, there were a lot of attempts to produce metal aluminum by nonelectrolysis [4-7]. Carbothermic reduction and chlorination process [8-10] is one of the most effective ways of extracting aluminum in the metallurgical industry. In this process, carbon and aluminum chloride are used as reducing agent and chlorinating agent, respectively, and minerals in the form of metal oxides react with them. The corresponding metal subchlorides formed and separated from minerals in gaseous form are unstable at the temperature of operation and vaporize readily. Subchlorides can be condensed, separated, and further processed to get desired pure metals.
However, during the carbothermic reduction stage, the formation of aluminum carbide and oxycarbide byproducts complicates the carbothermic reduction and chlorination process. The thermodynamic study of chemical reactions provides a basic understanding of the process prior to designing suitable reaction experiments, and provides a useful guideline for the selection of processing conditions [11-12].
Prior to experiments, it is essential to determine the feasibility of the chemical reactions, the nature and amount of the solid and gaseous species present in the system. These can be determined from the calculation of the standard Gibbs free energy changes for chemical reactions and the standard free energy changes at a given set of processing conditions such as reaction temperature, pressure and reactant concentration [13].
The main purpose of this work was to investigate the carbothermic process in vacuum. The resultant samples were investigated by X-ray diffraction, and gas chromatography analysis.
2 Experimental
2.1 Experimental equipment
The schematic diagram of experimental setup is shown in Fig. 1. A reaction zone, composed of cylinder containing sample, is controlled by a temperature controller. The reactor was provided with a central removable tube that was permitted to insert a thermocouple in order to know the temperature in the reaction zone by means of a digital thermometer. A zone for the collection of the reaction products consisted of a box-like collecting plate, where most of the reaction products were condensed, and a washing trapper for the collection of gas products.
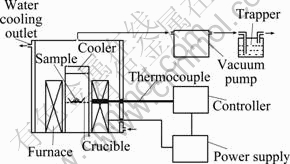
Fig. 1 Schematic diagram of experimental setup
2.2 Materials and sample preparation
Analytical grade of alumina and graphite were used as the raw materials in experiments. The solid samples used in the experiments were mixtures of Al2O3 and graphite with molar ratio of 1:4.
2.3 Experimental procedure
Once the reagent solid sample was placed in the reactor, cooling water and vacuum pump were opened, and the reaction zone was heated as the pressure reached 100 Pa. Vacuum degree is regulated by the corresponding control and measuring devices. The heating rate of system temperature was about 20 K/min. The system temperature is kept constant for 140 min when it reached 1 693, 1 703, 1 753 and 1 853 K. Then, the heating device was turned off and vacuum degree was kept till room temperature.
3 Results and discussion
3.1 Thermodynamic analysis of carbothermic reduction
During this solid-solid reaction, gaseous products are generated when the carbothermic reduction reaction takes place. In vacuum, gaseous products are removed continuously through pump, which can accumulate the chemical reaction. Main formulas of thermodynamic calculations are as follows:
 (1)
(1)
 (2)
(2)
where  (3)
(3)
 (4)
(4)
 (5)
(5)
 (6)
(6)
 (7)
(7)
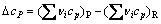 (8)
(8)
Based on the reactions given in Table 1, the initial reaction temperatures of these reactions have been calculated using the thermodynamic data from Refs. [14-16] under the condition of partial pressure of 100 Pa and normal pressure. In the view of reaction kinetics, dismissing carbon monoxide gas under the condition of low partial pressure gives advantages to chemical equilibrium, which makes reaction move toward right side. It can be found in Table 1 that in vacuum the initial reaction temperature reduces, which means that much less energy would be consumed. The relationships between Gibbs free energy change and temperature of these reactions calculated under the condition of partial pressure of 100 Pa are shown in Fig. 2.
Table 1 Initial reaction temperatures of carbothermic reduction of alumina (K)
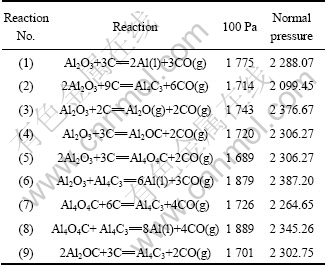
According to Fig. 2, ΔG decreases sharply by increasing temperature. The initial temperatures of nine reactions are 1 775, 1 714, 1 743, 1 720, 1 689, 1 879, 1 726, 1 889 and 1 704 K, respectively. The preferential order of products is Al4O4C>Al4C3>Al2OC>Al2O>Al during the carbothermic reduction of alumina. Aluminum generated by Al2O3-Al4C3 and Al4O4C-Al4C3 should be at higher temperature. The temperature and system pressure in the furnace are important determinants of feasibility of reactions.
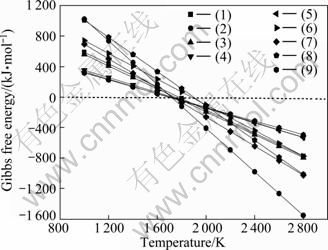
Fig. 2 Gibbs free energy change versus temperature of Al2O3-C system at 100 Pa
3.2 Effect of carbon monoxide change on carbothermic reduction
The volume of carbon monoxide per 20 min and total volume of carbon monoxide were calculated, and the average production rate of carbon monoxide per 20 min was determined. Figure 3 shows the average production rate of carbon monoxide and total volume of carbon monoxide. It can be found that production rate of carbon monoxide decreases, and total volume of carbon monoxide increases with increasing time. After about 80 min, the production rate of carbon monoxide tends to be stable at about 0.02 L/min, and the volume of carbon monoxide increases slowly at about 0.4 L per 20 min. The carbon monoxide change means that reaction rate of alumina and carbon decreases gradually with increasing time.

Fig. 3 Carbon monoxide change versus time
3.3 Effect of temperature on carbothermic reaction
Carbothermic reactions were carried out at different temperatures in pressure range of 40-150 Pa. The XRD patterns of slag are shown in Fig. 4.
The reaction product in the crucible identified by XRD consists of alumina and carbon at 1 693 K, which suggests that no reaction occurs between alumina and

Fig. 4 XRD pattern of carbothermic reduction products at different temperatures
carbon under that condition. The characteristic peaks of Al4O4C and Al4C3 are found clearly over 1 703 K and increases with increasing temperature. Meanwhile, the diffraction intensities of carbon and alumina decrease with increasing temperature, which can be deduced that the reactions (2), (5) and (7) occur in the temperature range of 1 703-1 853 K. There is no diffraction peak of Al2OC found by XRD analysis in the reaction product of carbothermic process and no aluminum is collected in condensation tower of vacuum furnace. If there were Al2O gas, it would disproportionate into aluminum and alumina at low temperature and be found on the cooler. According to the carbothermic reduction analysis, it can be deduced that reactions (1), (3), (4), (6), (8) and (9) do not occur between alumina and carbon at 40-150 Pa.
The initial temperatures of reactions (2), (5) and (7) are 1 714, 1 689 and 1 726 K, respectively. According to the formula of X-ray quantitative analysis [17], the content of certain phase is proportional to its diffracted intensity. That is to say, Al4O4C is firstly formed in the carbothermic reaction, and then Al4C3 is formed at higher temperature. Thus, the diffraction intensity of Al4O4C is stronger than that of Al4C3 with increasing temperature from 1 693 to 1 853 K. This analysis and experimental results are well agreed with QIU [18], who pointed out that the first product Al4O4C is a stable compound in thermodynamics, and another product Al4C3 is produced when reactant further reacts with carbon. Also, FOLOMEIKIN et al [19] pointed out that there is Al2OC during the transformation of Al4O4C to Al4C3, but no Al2OC is found in the experiment.
3.4 Effect of system pressure on carbothermic reaction
Carbothermic reduction alumina in temperature range of 1 773-1 823 K is a process of solid-solid reaction to generate gas. System pressure has a marked effect on carbothermic reaction. The temperature of equilibrium (ΔG=0) decreases notably by reducing the partial pressure. The effect of system pressure on the carbothermic reduction process was investigated under 20 Pa and 100 Pa. XRD patterns of slag at different system pressures are shown in Fig. 5.
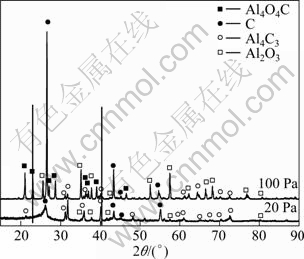
Fig. 5 XRD patterns of residues at different system pressures
It can be seen from Fig.5 that Al4O4C cannot be found in the residues at 20 Pa, but much more Al4C3 is found. Instead, the diffraction intensity of Al4O4C is clearly stronger than that of Al4C3 under 100 Pa. In other words, the diffraction intensity of Al4O4C decreases with increasing system pressure, but Al4C3 is quite opposite. It can be concluded that alumina is changed into Al4O4C which continues to change into Al4C3. The sequence of product is consistent with the reactions (5) and (7). The reaction (7) can occur even at a higher reaction temperature than that of the reaction (5) due to the lower pressure.
4 Conclusions
1) In vacuum, the initial reaction temperature reduces, meaning that much less energy would be consumed. The preferential order of products is Al4O4C, Al4C3, Al2OC, Al2O and Al during the carbothermic reduction of alumina.
2) During the carbothermic reduction process, the reaction rate of alumina and carbon decreases gradually with increasing time.
3) The carbothermic reduction products of alumina are Al4O4C and Al4C3, and neither Al2OC nor Al2O is found.
4) Lower system pressure or higher temperature is beneficial to the carbothermic reduction of alumina process. Al4O4C is firstly formed in the carbothermic reaction, and then Al4C3 is formed in lower system pressure or at higher temperature.
References
[1] KAI G, HALVOR K, LI Qing-feng, QIU Zhu-xian. Metal production by molten salt electrolysis especially aluminum and magnesium [M]. Beijing: China University of Mining and Technology Press, 1998: 73-75.
[2] STEINFELD A, THOMPSON G. Solar combined thermochemical processes for CO2 mitigation in the iron, cement, and syngas industries [J]. Energy, 1994, 19(10): 1077-1081.
[3] LI Jie, FANG Zhao, LAI Yan-qing, L? Xiao-jun, TIAN Zhong-lian. Electrolysis expansion performance of semigraphitic cathode in [K3AlF6/Na3AlF6]-AlF3- Al2O3 bath system [J]. Journal of Central South University of Technology, 2009, 16: 422-428.
[4] MYKLEBUST H, RUNDE P. Greenhouse gas emission from aluminum carbothermic technology compared to Hall-Héroult technology [J]. Light Metals, 2005: 519-522.
[5] CHOATE W, GREEN J. Technoeconomic assessment of the carbothermic reduction process for aluminum production [J]. Light Metals, 2006: 445-450.
[6] WANG Ping-yan, DAI Yong-nian, JIANG Shi-xin, ZHONG Hong-quan. Study on production of aluminum by carbothermic reduction and subhalide decomposition under vacuum [J]. Nonferrous Metals (Metallurgy), 2005(2): 11-13. (in Chinese)
[7] JEAN P M. Aluminum production using high temperature solar process heat [J]. Solar Energy, 1999, 66(2): 133-142.
[8] FABIOLA J A, ANA E B. Direct chlorination of nickel-containing materials. Recovery of the metal from different sources [J]. Industry Engineering Chemistry Research, 2008, 47: 8184-8191.
[9] YANG F, HLAVACEK V. Carbochlorination of tantalum and niobium oxides, Thermodynamic simulation and kinetic modeling [J]. Journal of AIChE, 1999, 45: 581-589.
[10] WANG Ping-yan, YANG Bu-zheng, DAI Yong-nian, LIU Yong-cheng. Test equipment for the production of aluminum by carbothermic reduction of aluminum-bearing materials with the aid of subhalide decomposition under vacuum [J]. Mining and Metallurgical Engineering, 2005, 25(2): 43-45. (in Chinese)
[11] MATTIGOD S V, MCGRAIL B P. Estimating the standard free energy of formation of zeolites using the polymer model [J]. Microporous and Mesoporous Materials, 1999, 27: 41-47.
[12] ALBERTY R A. Use of standard Gibbs free energies and standard enthalpies of adenosine(aq) and adenine(aq) in the thermodynamics of enzyme-catalyzed reactions [J]. Journal of Chemistry Thermodynamics, 2004; 36: 593-601.
[13] CHOY K L. Chemical vapour deposition of coatings [J]. Progress of Material Science, 2003, 48: 57-170.
[14] LIHRMANN J M. Thermodynamic of the Al2O3-Al4C3 system I. Thermochemical functions of Al oxide, carbide and oxycarbides between 298 and 2 100 K [J]. Journal of the European Ceramic Society, 2008, 28: 633-642.
[15] CHASE M W. NIST-JANAF thermochemical tables [M]. 4th Ed. New York: American Chemical Society and the American Institute of Physics for the National Institute of Standards and Technology, 1998: 98-143.
[16] HALMANN M, FREI A, STEINFELD A. Carbothermal reduction of alumina: Thermochemical equilibrium calculations and experimental investigation [J]. Energy, 2007, 32: 2420-2427.
[17] MIAO Chun-sheng. X-ray quantitative analysis method and its application [M]. Beijing: Geography Press, 1988, 12: 10-20.
[18] QIU Zhu-xian. Physical chemistry of aluminum metallurgy [M]. Shanghai: Shanghai Science and Technology Press, 1985: 340-360.
[19] FOLOMEIKIN Y K., DEMONIS I M, KABLOV E N, LOPATIN S I, STOLYAROVA V L. Investigation into the Vaporization of Al2O3 in the Presence of Carbon at High Temperatures [J]. Glass Physics and Chemistry, 2006, 32(2): 191-195
(Edited by YANG Bing)
Foundation item: Project(U0837604) supported by the Natural Science Foundation of Yunnan Province, China; Project(Jinchuan 201114) supported by the Pre Research Foundation of Jinchuan Group Ltd., China; Project(2011148) supported by the Analysis and Testing Funds of Kunming University of Science and Technology, China
Received date: 2011-05-13; Accepted date: 2011-12-20
Corresponding author: YU Qing-chun, Associate Professor; Tel: +86-871-5114017; E-mail: yqcy@163.com