一种新型的SiC-ZrB2涂层包覆C/C复合材料的制备及烧蚀性能
来源期刊:中国有色金属学报(英文版)2015年第11期
论文作者:黄东 张明瑜 黄启忠 王丽平 汤贤 杨鑫 童凯
文章页码:3708 - 3715
关键词:烧蚀;涂层;显微结构;C/C复合材料
Key words:ablation; coating; microstructure; carbon/carbon composites
摘 要:利用两步工艺结合泥浆烧结法和化学气相反应法在C/C复合材料表面制备了一种新型的SiC-ZrB2涂层。SiC-ZrB2涂层由分散的ZrB2相和连续的SiC相组成,涂层与C/C复合材料基体结合较好。与C/C复合材料相比,SiC-ZrB2涂层包覆C/C复合材料试样具有更好的抗烧蚀性能。SiO2-ZrO2陶瓷层的阻氧作用、气态氧化产物的散热以及ZrO2的钉扎作用是涂层试样具有良好抗烧蚀性能的主要原因。
Abstract: A novel SiC-ZrB2 coating was prepared using a two-step technique by slurry-sintering and chemical vapor reaction on carbon/carbon (C/C) composites. The SiC-ZrB2 coating was composed of the scattered ZrB2 phase and the continuous SiC phase. It was observed that a good adhesion was built between the coating and the C/C composites. The SiC-ZrB2 coating samples exhibited a better ablation resistance in comparison with the uncoated C/C composites. The SiO2-ZrO2 barrier layer, the heat dissipation of the gaseous products and the pinning effect of ZrO2 all contributed to the good ablation resistance of the SiC-ZrB2 coated composites.
Trans. Nonferrous Met. Soc. China 25(2015) 3708-3715
Dong HUANG, Ming-yu ZHANG, Qi-zhong HUANG, Li-ping WANG, Xian TANG, Xin YANG, Kai TONG
State Key Laboratory of Powder Metallurgy, Central South University, Changsha 410083, China
Received 9 December 2014; accepted 24 March 2015
Abstract: A novel SiC-ZrB2 coating was prepared using a two-step technique by slurry-sintering and chemical vapor reaction on carbon/carbon (C/C) composites. The SiC-ZrB2 coating was composed of the scattered ZrB2 phase and the continuous SiC phase. It was observed that a good adhesion was built between the coating and the C/C composites. The SiC-ZrB2 coating samples exhibited a better ablation resistance in comparison with the uncoated C/C composites. The SiO2-ZrO2 barrier layer, the heat dissipation of the gaseous products and the pinning effect of ZrO2 all contributed to the good ablation resistance of the SiC-ZrB2 coated composites.
Key words: ablation; coating; microstructure; carbon/carbon composites
1 Introduction
C/C composites have attracted much attention due to their low coefficient of thermal expansion (CTE), good strength at high temperature, high thermal conductivity, and good ablation resistance and thermal shock resistance [1-3]. These excellent properties make C/C composites the promising structural materials used in high temperature environment such as nose tips, leading edges, nozzles of solid rocket motor and other thermal protection systems (TPS) for space vehicles [4,5]. However, poor oxidation resistance (above 773 K) and severe ablation environments (high-pressure gas and high velocity grains) have greatly restricted their high temperature application [6,7]. Therefore, improving ablation resistance at ultrahigh temperature (>2273 K) is crucial to extend applications of C/C composites.
Surface coating technology is one of the effective ways to protect C/C composites from oxidation and ablation at high temperature [8-11]. SiC coatings have attracted extensive attention because of their ability to generate dense SiO2 layer and lower coefficient of thermal expansion (CTE) mismatch than other ceramic materials [12-14]. However, the molten SiO2 glass (melting point 1943 K) due to the oxidation of the SiC is easy to be peeled off by the high velocity gas during ablation because of its low viscosity at an ultra-high temperature (>2273 K), which possibly resulted in the poor ablation resistance of the SiC coating. It has been reported that introducing both SiC and Zr-based ceramics at the same time can further improve the ablation resistance and oxidation resistance of the composites under ultra-high temperature environments (>2273 K) because of the formation of less volatile silica-containing scales [15-21]. So, the introduction of ZrB2 into SiC coating is considered as a promising method to provide ablation protection for C/C composites at ultra-high temperature.
In the present work, a novel SiC-ZrB2 coating for C/C composite was reported, using a two-step technique which combines slurry-sintering with chemical vapor reaction (CVR). The introduction of ZrB2 particles was easy and low cost, and the obtained SiC-ZrB2 coating samples showed a good ablation resistance. The microstructural characteristics and ablation property of the ZrB2-SiC coatings were investigated and the ablation mechanisms were discussed.
2 Experimental
Before coating, the C/C composites (homemade from the 2D needled carbon fiber preform, Center South University, China, density 1.73 g/cm3) were cut into bulks with dimensions of 10 mm × 10 mm × 10 mm. After being ground with 800 grit SiC papers, the samples were ultrasonically cleaned and dried at 373 K for 1 h.
Firstly, the ZrB2-resin carbon was prepared from powder of ZrB2 (15 μm, purity >99.1%, Aladdin Co., Ltd., Shanghai, China) and phenolic resin solution. The phenolic resin powders were dissolved in ethyl alcohol with a ratio of 20 g per 100 mL and used as phenolic resin solution. The ZrB2 powders were added into the dissolved phenolic resin solution to make the slurry. The mass ratio of ZrB2 to resin was 1:2. The slurry was brushed directly on the surface of the C/C composites. The samples were dried at 423 K and then carbonized at 1173 K for 2 h in N2 atmosphere. The thickness of the ZrB2-resin carbon layer was 30-40 μm by 2-3 times of brushing according to Eq. (1). The mass fractions of SiC and ZrB2 in the coating were 71.52% and 18.48%, respectively.
Secondly, the outer SiC coating was prepared by CVR. The SiO generator was produced using SiO2/Si mixture at 1773-2123 K (Eq. (2)). The CVR SiC was produced during the reaction of SiO with the substrate at 2373-2573 K (Eq. (3)) [8,21].
CxHyOz(s)→C(g)+by-product(g) (1)
SiO2(l)+Si(l)→SiO(g) (2)
C(s)+SiO(g)→SiC(s)+CO(g) (3)
The ablation resistance of the SiC-ZrB2 coating for C/C composites was tested using a plasma generator (Multiplaz, 3500) with cylindrical samples (d30 mm × 10 mm, Fig. 1(a)) [15]. The erosion direction of the flame and the axial orientation of the samples were parallel to the Z direction of the carbon felts (needled direction, Fig. 1(b)). The working current and voltage of the plasma generator were 6 A and (160±1) V, respectively. The inner diameter of the plasma gun tip was 2 mm, and the distance between the gun tip and the sample was about 10 mm. The samples were exposed to the flame, and the maximum temperature of the ablation center reached as high as 2573 K, as confirmed by an optical pyrometer. Both linear and mass ablation rates were calculated based on the thickness and mass changes before and after the ablation. More than five tests were made for each sample, and the presented results were the averages of these ablation rates.
The microstructural characterization was carried out using scanning electron microscopy (SEM, FEI Nova Nano SEM230), backscattered electron images (BSE, FEI Nova Nano SEM230), energy dispersive spectrometer (EDS, FEI Nova Nano SEM230) and X-ray diffraction (XRD, Rigaku Dmax/2550VB+18 kW).
3 Results and discussion
3.1 Microstructure of SiC-ZrB2 coating
Figure 2 shows the images of the C/C samples with SiC-ZrB2 coating after brushing and carbonization. The ZrB2-resin carbon covers the surface of the samples completely, and no holes are observed (Fig. 2(a)). Figure 2(b) indicates that ZrB2 particles (white phase) are dispersed uniformly in the resin carbon (gray black phase). The average grain size of ZrB2 is in the region of 10-20 μm micrometers (Fig. 2(c)). It is observed that the ZrB2 particles are combined strongly with resin carbon after the carbonization. Figure 2(d) shows the BSE image of the cross section of the ZrB2-resin carbon. There is no lamination in the ZrB2-resin carbon layer after brushing several times. The thickness of the ZrB2-resin carbon layer is 20-30 μm.

Fig. 1 Images of ablation test

Fig. 2 SEM images and BSE images of samples after brushing and carbonization
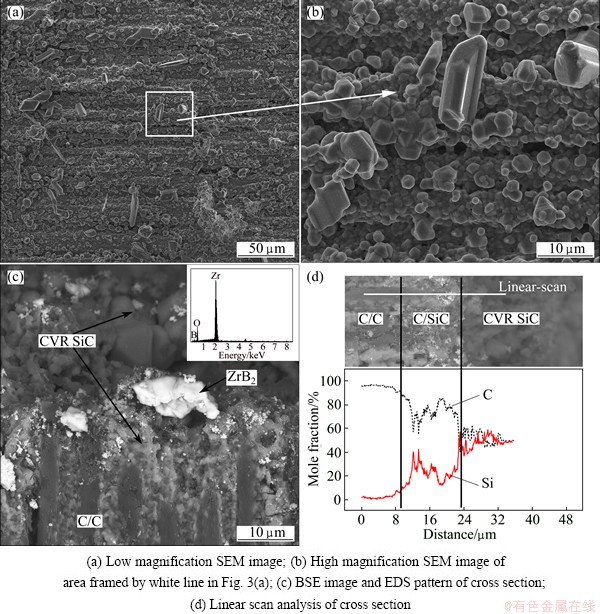
Fig. 3 Microstructures of SiC-ZrB2 coatings after CVR
Figure 3(a) shows the SEM image of the surface of the SiC-ZrB2 coatings after CVR. It can be seen that ceramic grains are coarse, and appear to be rubble-like on the surface of the coating. The XRD pattern (Fig. 4) shows that three obvious characteristic peaks at 35.7°, 60.1° and 71.8°, are corresponding to the diffraction of (111), (220) and (311) crystalline planes of a FCC lattice, respectively, which are in good agreement with the known results for β-SiC. No peaks of ZrB2 or C phase are observed, indicating that the sample surface is completely covered with the CVR SiC coating. Higher magnification image of the surface (Fig. 3(b)) shows that both large and small sizes of the SiC grains have been formed after CVR process. The small ones absorb the C/C composite directly, whereas the large ones are present on the surface of the small SiC grains. Figure 3(c) shows the BSE images of the cross section of the ZrB2-SiC coating. It can be seen that the SiC-ZrB2 coating is mainly composed of the scattered ZrB2 phase and the continuous SiC phase. The ZrB2 phase (white part) is wrapped in the CVR layer (gray part) and confirmed by EDS analysis. It is known that the resin carbon around the ZrB2 powder reacts with SiO to form SiC by the carbothermic reaction (Eq. (3)). It is believed that ZrB2 powders and CVR SiC phase are bonded through mechanical bond and the intermolecular forces between them. Linear scan analysis (Fig. 3(d)) has shown that a C/SiC gradient layer can be formed between the carbon substrate and CVR SiC due to the permeation and reaction of SiO vapor in the carbothermic reaction [8,21]. As a result, a continuous change of the elastic modulus and the CTE of the coating occurs at the transitional interface, which makes the thermal stress release effectively and improves the bonding strength between the coatings and the substrates. It therefore can be concluded that a good adhesion can be built between the CVR SiC-ZrB2 composite coating and C/C composites.
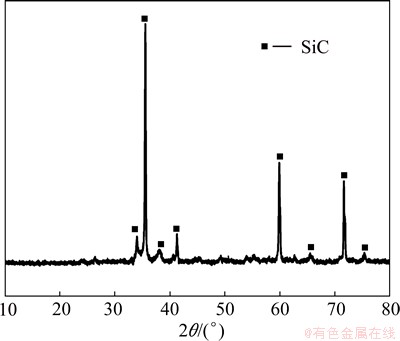
Fig. 4 XRD pattern of SiC-ZrB2 coating
3.2 Ablation resistance of ZrB2-SiC coating
Figure 5 shows the macro-photographs of the C/C composites before and after coating. A compact and gray-green ceramic coating is formed after CVR process. Some shiny grains on the surface of the coatings are proved to be the larger SiC grains according to Fig. 5(b).

Fig. 5 Macro-photographs of C/C composites before (a) and after (b) coating
The photographs of the samples after ablation for 120 s and 240 s are shown in Fig. 6. The coating in the ablation center has been stripped off and the carbon fibers are ablated after ablation for 120 s. However, some ceramic oxides residues are observed on the surface, and the linear ablation is slight. The SiC-ZrB2 coating samples show a better ablation resistance than uncoated C/C composite after 120 s ablation test (Table 1). The mass and linear ablation rates of the coated samples are 0.83 mg/s and 2.37 μm/s, respectively. And those of uncoated samples are 2.23 mg/s and 15.25 μm/s, respectively. In addition, the coating has been destroyed completely after 240 s ablation, and a large ablated hole is present on the surface (Fig. 6(b)). The mass and linear ablation rates of the coating samples are close to those of the uncoated ones (Table 1).

Fig. 6 Macro-photographs of SiC-ZrB2 coating after ablation for 120 s (a) and 240 s (b)
Table 1 Ablation resistance of C/C composite before and after coating
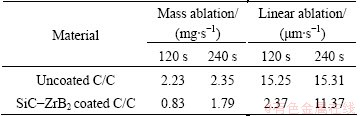
Figure 7 shows the SEM images and EDS analysis of ablation center of coating samples after 120 s. Figure 7(a) indicates that the SiC-ZrB2 coating has been stripped off in the ablation center. The SiC-ZrB2 coating is not destroyed in the ablation brim because the temperature and the combustion gas velocity in the ablation brim are much lower. Figure 7(b) shows the SEM images of the ablation center. Some carbon fibers have been seriously eroded, and appear to be needle-like. Others are damaged slightly under the protection of the ceramic residues. High magnification image (Fig. 7(c)) indicates that some glass layers are left on the surface of un-ablated carbon fibers. EDS analysis indicates that the residues are composed of ZrO2 and SiO2. It can be concluded that the ceramic residues play a positive role in the ablation resistance.
In order to further understand the ablation mechanism, the microstructures of coating after ablation for 60 s and before being stripped off, have been investigated, the results are shown in Fig. 8. There are some cracks and holes on the surface, which could provide channels for the diffusion of O2 into the composite (Fig. 8(a)). A shallow pit is present on the surface of the ablation center (Fig. 8(b)), because the high-speed combustion gas has blew away the molten ceramic oxides at the ultra-high temperature. High magnification image (Fig. 8(c)) shows that some molten ceramic oxides have been formed in the shallow pit, and EDS analysis indicates that the oxides are SiO2 and ZrO2 (Fig. 8(d)). XRD pattern (Fig. 9) indicates that the surface is covered with ZrO2 and SiO2 glassy layer from the oxidation of ZrB2 and SiC, which also illustrates the statement mentioned above. No B2O3 (boiling point 1773 K) has been detected on the ablated surface, which indicates that B2O3 has been evaporated during ablation at ultra-high temperature (above 2573 K). The molten SiO2 glass (melting point 1973 K) suffered from shearing forces resulted from the flame with high temperature, high velocity and pressure. However, the refractory ZrO2 could prevent SiO2 from peeling off and act as a pinning effect for the molten SiO2 due to its high melting point (2953 K). The SiO2-ZrO2 layer acts as an effective barrier which can prevent oxygen from diffusing into the composite due to its low oxygen diffusivity.
3.3 Ablation mechanism
Based on the above analysis, we propose several processes which show the ablation mechanism of the SiC-ZrB2 coatings (Fig. 10).
1) The SiC-ZrB2 coating is rapidly oxidized to produce SiO2, ZrO2 and B2O3 according to Eq. (4) and Eq. (5). The SiC-ZrB2 coating (in situ barrier) and its ceramic oxides layer (low oxygen diffusivity) act as an effective barrier which can prevent oxygen from diffusing into the composite due to its low oxygen diffusivity.
2) The fusion and evaporation of SiO2 (melting point 1943 K) and B2O3 (melting point 723 K) will dissipate the heat, resulting in the temperature on the surface of composite decreasing.
3) The refractory ZrO2 could have a pinning effect for the molten SiO2 due to its high melting point (2953 K), which can prohibit the peeling or exfoliation of the molten SiO2 during ablation. The refractory oxide scales formed may give the composites a capability to withstand the ultra-high temperature ablation.

Fig. 7 Microstructures of SiC-ZrB2 coating after ablation for 120 s
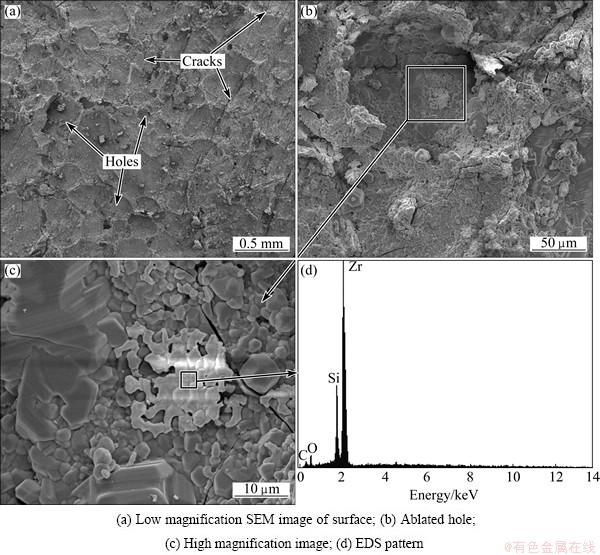
Fig. 8 Microstructures of SiC-ZrB2 coating after ablation for 60 s
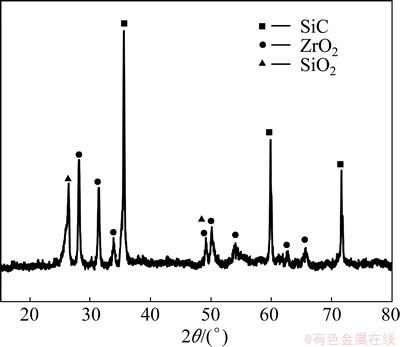
Fig. 9 XRD pattern of SiC-ZrB2 coating after ablation for 60 s
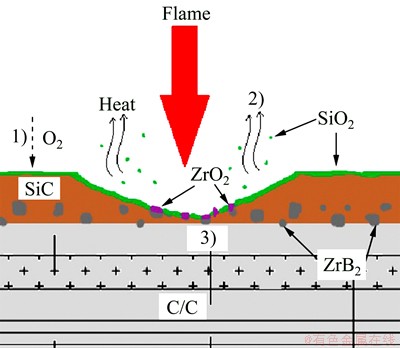
Fig. 10 Ablation mechanism of SiC-ZrB2 coating
As the ZrO2-SiO2 layer has been largely destroyed after ablation for 240 s, the ZrO2 residues cannot act as an intact protective layer. As a result, the carbon fibers are ablated due to the oxidation (Eq. (7)) and scouring, and the ablation rate is increased compared with that for 120 s (Table 1). And then SiC-ZrB2 coating failed in protecting composites from corrosion (Fig. 6(b) and Table 1).
SiC(s)+O2(g)→SiO2(l)+COn(g) (n=1, 2) (4)
ZrB2(s)+O2(g)→ZrO2(s)+B2O3(g) (5)
SiO2(l)→SiO2(g) (6)
C(s)+O2(g)→COn(g) (7)
4 Conclusions
1) A novel SiC-ZrB2 coating was fabricated on the C/C composites using slurry-sintering and chemical vapor reaction. The ZrB2 phases were dispersed uniformly in the SiC-ZrB2 coatings, and a good adhesion can be built between the coating and the C/C composite.
2) The plasma ablation test indicated that the coating samples possessed a good ablation resistance. The linear and mass ablation rates of the SiC-ZrB2 coating samples are 0.83 mg/s and 2.37 μm/s after ablation for 120 s, respectively.
3) The good ablation resistance is attributed to the low oxygen diffusivity ZrO2-SiO2 barrier layer and a pinning effect of refractory ZrO2 phase in the present work. As the ZrO2-SiO2 layer has been largely destroyed after ablation for 240 s, the SiC-ZrB2 coating failed in protecting composites from corrosion.
References
[1] FITZER E. The future of carbon-carbon composites [J]. Carbon, 1987, 25(2): 163-190.
[2] SHEEHAN E, BUESKING W, SULLIVAN J. Carbon-carbon composites [J]. Annual Review of Materials Science, 1994, 24(0): 19-44.
[3] TANG Su-fang, WANG Dao-ling, DENG Jing-yi, LIU Wen-chun, YANG Ke. Ablation properties of C/C composites with various needled preforms prepared by isothermal chemical vapor infiltration [J]. Journal of Central South University of Technology, 2007, 14(1): 13-18.
[4] VIGNOLES L, ASPA Y, QUINTARD M. Modelling of carbon-carbon composite ablation in rocket nozzles [J]. Composites Science and Technology, 2010, 70(9): 1303-1311.
[5] ZAMMAN W, LI K Z, IKRAM S. Morphology, thermal response and anti-ablation performance of 3D-four directional pitch-based carbon/carbon composites [J]. Corrosion Science, 2012, 61: 134-142.
[6] GE Yi-cheng, YANG Ling-yan, WU Shuai, LI Chai, LUO Jian, YI Mao-zhang. Influence of heat-treatment on oxidation-resistance of phosphate-coating for C/C composite [J]. Transactions of Nonferrous Metals Society of China, 2014, 24(2): 455-461.
[7] LIU Chuan-xuan, CHEN Jian-xun, SU Zhe-an, YANG Xin, CAO Liu-xu, HUANG Qi-zhong. Pyrolysis mechanism of ZrC precursor and fabrication of C/C-ZrC composites by precursor infiltration and pyrolysis [J]. Transactions of Nonferrous Metals Society of China, 2014, 24(6): 1779-1784.
[8] HUANG D, ZHANG M Y, HUANG Q Z. Preparation of silicon carbide double layer coating and its anti-oxidation property at 1773 K [J]. Corrosion Science, 2014, 87: 134-140.
[9] LI H J, YAO X Y, ZHANG Y L. Anti-oxidation properties of ZrB2 modified silicon-based multilayer coating for carbon/carbon composites at high temperatures [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(7): 2113-2117.
[10] FU Qian-gang, ZHANG Jia-ping, ZHANG Zheng-zhong, LI Hen-jun, SUN Can. SiC-MoSi2/ZrO2-MoSi2 coating to protect C/C composite against oxidation [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(7): 2094-2099.
[11] WANG K T, CAO L Y, HUANG J F. A mullite-SiC oxidation protective coating for carbon/carbon composites [J]. Journal of the European Ceramic Society, 2013, 33(1): 191-198.
[12] HAGER D, GREIL P, LEYENS C. Self-healing materials [J]. Advanced Materials, 2010, 22(47): 5424-5430.
[13] YANG Xin, ZOU Yang-hong, HUANG Qi-zhong, SU Zhe-an, CHANG Xin, ZHANG Ming-yu, XIAO Yong. Improved oxidation resistance of chemical vapor reaction SiC coating modified with silica for carbon/carbon composites [J]. Journal of Central South University of Technology, 2010, 17(1): 1-6.
[14] CHU Y H, FU Q G, LI H J. SiC coating toughened by SiC nanowires to protect C/C composites against oxidation [J]. Ceramics International, 2012, 38(1): 189-194.
[15] LIU C X, CAO L X, CHEN J X. Microstructure and ablation behavior of SiC coated C/C-SiC-ZrC composites prepared by a hybrid infiltration process [J]. Carbon, 2013, 65: 196-205.
[16] WANG Y G, ZHU X J, ZHANG L T. Reaction kinetics and ablation properties of C/C-ZrC composites fabricated by reactive melt infiltration [J]. Ceramics International, 2011, 37(4): 1277-1283.
[17] YAO X Y, LI H J, ZHANG Y L. A SiC/ZrB2-SiC/SiC oxidation resistance multilayer coating for carbon/carbon composites [J]. Corrosion Science, 2012, 57: 148-153.
[18] ZHAO L Y, JIA D C, DUAN X M. Oxidation of ZrC-30 vol% SiC composite in air from low to ultrahigh temperature [J]. Journal of the European Ceramic Society, 2012, 32(4): 947-954.
[19] MA Y, LI Q G, DONG S M. Microstructures and ablation properties of 3D 4-directional Cf/ZrC-SiC composite in a plasma wind tunnel environment [J]. Ceramics International, 2014, 40(7): 11387-11392.
[20] LI Z Q, LI H J, ZHANG S Y. Microstructure and ablation behaviors of integer felt reinforced C/C-SiC-ZrC composites prepared by a two-step method [J]. Ceramics International, 2012, 38(4): 3419-3425.
[21] YANG Xin, HUANG Qi-zhong, ZOU You-hong, CHANG Xin, SU Zhe-an, ZHANG Ming-yu, XIE Zhi-yong. Anti-oxidation behavior of chemical vapor reaction SiC coating on different carbon materials at high temperatures [J]. Transactions of Nonferrous Metals Society of China, 2009, 19(5): 1044-1050.
黄 东,张明瑜,黄启忠,王丽平,汤 贤,杨 鑫,童 凯
中南大学 粉末冶金国家重点实验室,长沙 410083
摘 要:利用两步工艺结合泥浆烧结法和化学气相反应法在C/C复合材料表面制备了一种新型的SiC-ZrB2涂层。SiC-ZrB2涂层由分散的ZrB2相和连续的SiC相组成,涂层与C/C复合材料基体结合较好。与C/C复合材料相比,SiC-ZrB2涂层包覆C/C复合材料试样具有更好的抗烧蚀性能。SiO2-ZrO2陶瓷层的阻氧作用、气态氧化产物的散热以及ZrO2的钉扎作用是涂层试样具有良好抗烧蚀性能的主要原因。
关键词:烧蚀;涂层;显微结构;C/C复合材料
(Edited by Yun-bin HE)
Foundation item: Projects (51404041, 51304249) supported by the National Natural Science Foundation of China; Project (2015JJ3016) supported by the Natural Science Foundation of Hunan Province, China; Project (2011CB605801) supported by the National Basic Research Program of China; Project supported by the State Key Laboratory for Powder Metallurgy Foundation, Central South University, Changsha, China.
Corresponding author: Ming-yu ZHANG; Tel: +86-731-88877671; Fax: +86-731-88836078; E-mail: csuzmy@csu.edu.cn
DOI: 10.1016/S1003-6326(15)64012-2

