
Internal oxidation thermodynamics and microstructures of Ag-Y alloy
WU Chun-ping(吴春萍)1, YI Dan-qing(易丹青)1, CHEN Jing-chao(陈敬超)2,
LI Jian(李 荐)1, LIU Hui-qun(刘会群)1, WANG Bin(王 斌)1, FANG Xi-ya(方西亚)1
1. School of Materials Science and Engineering, Central South University, Changsha 410083,China;
2. Key Laboratory of Advanced Materials of Yunan Province, Kunming University of Science and Technology, Kunming 650093, China
Received 8 May 2006; accepted 1 November 2006
Abstract: The thermodynamic data of pure Ag and Y were calculated. The phase constitution, composition of micro-region and microstructures of Ag-Y alloy after internal oxidation were investigated by X-ray diffractometry(XRD), energy dispersion spectrometry(EDS) and scanning electron microscopy(SEM). The results show that the internal oxidation behavior of Ag-Y alloy is feasible from the view of thermodynamics. The upper limit of oxygen partial pressure of Ag-Y alloy oxidation is a function of temperature. Two phases (Ag and Y2O3) appear in Ag-Y alloy after the internal oxidation. The surface of Ag-Y alloy is convex because of the volume expansion of oxide in the alloy and the composition of the convex part is Ag. In Ag-Y2O3 sintered bulk Y2O3 particles are distributed inhomogeneously and conglomerated seriously, but they are dispersed uniformly in the Ag matrix after severe plastic deformation.
Key words: Ag-Y alloy; internal oxidation; thermodynamics; microstructure
1 Introduction
The electrical contact materials of silver metal- oxide(Ag-MeO) fabricated by internal oxidation are important electrical contact materials. It is used in middle and low electric current switchgears. At present, widely used electrical contact material is internal oxidation Ag-Cd alloy[1]. However, two instructions made by European Confederation, ROHS 2002/95/EC and WEEE 2002/96/EC, were executed in 2006, the utilization of Cd, Pb, Hg ect, in the electronic and electric products of European Confederation market would be restricted rigidly, which means that European Confederation would completely forbid the utilization of Ag-CdO electrical contact materials[2]. As the placement materials of Ag-CdO, Ag-SnO2 materials have been used widely in business in European Confederation and Japan. However, Ag-SnO2 materials have high temperature rise and are more difficult to process, the researchers also positively study and develop new non-toxic environment-friendly electrical contact materials to compensate the shortcoming of Ag-SnO2 materials when they develop Ag-SnO2 materials[3]. The electrical contact materials contained rare earth element have the following advantages[4]: 1) They can provide stronger alloy as well as maintain the high electrical and thermal conductivity of the matrix materials; 2) They can improve the materials’ stability by restricting the grain growth and increasing the recrystallization temperature; 3) They have low and stable contact resistance; 4) They have good resistance of electrical erosion and welding; 5) They can extinguish the electrical arc; 6) They have no harm to mankind and environment. Therefore, the development of silver-rare earth electrical contact materials have been attracted worldwide researchers. But at present the reports about Ag-Y2O3 materials are few all over the world, and the study of Ag-Y alloy internal oxidation behavior is little. It is found that the addition of a small quantity of the active Y element to high temperature alloy will be prone to form the protected oxide membrane with the structure similar to Al2O3 and Cr2O3, which will enhance the performance of conglutination between Y and matrix metals. The mutual solubility between Ag and Y is small, there are three narrow uniform intermetallic phase regions[5-6] in Ag-Y binary phase diagram, so Ag-Y alloy has two-phase structure in the whole component range. In addition, Ag is inert in oxygen atmosphere, but the oxide of Y is stable. Ag-Y system is a typical one that the oxidation characteristic of two components has large discrepancy[7].Ag-Y binary phase diagram shows that Ag-10.33 Y (mass fraction) alloy is solid solution at internal oxidation temperature[8]. The matrix metal’s (Ag) oxide will decompose when temperature is 300 ℃, Y2O3 formed by internal oxidation is more stable than Ag2O, which satisfies the formation of Ag-Y2O3 material. So in this paper the thermodynamics of internal oxidation, phase constitution and microstructures of Ag-10.33 Y alloy after internal oxidation were studied in order to provide important theoretic basis for internal oxidation of Ag-Y alloys.
2 Experimental
The internal oxidation of Ag-10.33Y (mass fraction) alloy powder and oxidant powder was carried out in electric furnace (SB2-5-12) after fully mixing. The internal oxidation powder was pressed in mould, and then sintered. The process flow diagram of experiment is shown in Fig.1. The phase constitution, composition of micro-region and microstructures of Ag-Y alloy after internal oxidation were investigated by using X-ray diffraction (D8ADVAN XRD), energy diffraction spectrum (PHILIPS-EDAX-PHDENIX EDS) and scanning electron microscope (XL30SEM).
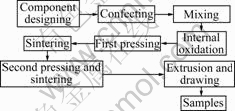
Fig.1 Process flow diagram of experiment
3 Results and discussion
3.1 Thermodynamics of internal oxidation in Ag-Y alloy
Internal oxidation of the alloy means that oxygen diffuses into the interior of the alloy, and then the oxidation reaction occurs between oxygen and the alloying element[9-11]. The essential conditions of internal oxidation are as follows[12]:
1) At internal oxidation temperature, oxidation element and oxygen should be solvable in matrix metal, but the oxide should not be solvable.
2) The oxide of additive element should be more stable than that of the matrix metal.
3) The diffusion of oxygen to matrix metal should be stronger than that of oxygen to additive element.
4) In order to obtain internal oxidation layer thick enough, matrix metal should not form stable oxide at oxidation temperature.
From the viewpoint of thermodynamics, preferential oxidation plays the first role in internal oxidation. The oxygen partial pressure of the working medium is the key factor that controls the preferential oxidation[13]. For Ag-Y alloys, preferential oxidation means Ag free oxidation and Y oxidation, the related formulas of oxidation reactions are as follows:

According to Gibbs’ function ?G=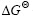 +2.303RT? lg Kp[14], where Kp is reaction equilibrium constant. On the assumption that ?G=0, applying the related thermodynamic data[15] into the Gibbs’ function, the relationship between temperature(T) and the critical oxygen partial pressure for forming or decomposing Ag2O and Y2O3 can be obtained, which is as follows:
+2.303RT? lg Kp[14], where Kp is reaction equilibrium constant. On the assumption that ?G=0, applying the related thermodynamic data[15] into the Gibbs’ function, the relationship between temperature(T) and the critical oxygen partial pressure for forming or decomposing Ag2O and Y2O3 can be obtained, which is as follows:
lg p(O2)=-3 190/T+11.91 (3)
lg p(O2)=-66 342/T+15.35 (4)
According to Eqns.(3) and (4), we can calculate the thermodynamic condition of oxidation and reduction of Ag and Y. The thermodynamic condition of the internal oxidation of Y is as follows:
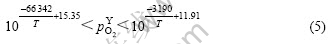
where 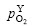 is the required oxygen partial pressure when Y is oxidized.
is the required oxygen partial pressure when Y is oxidized.
The threshold of oxygen partial pressure of oxidation and free oxidation of pure Ag and Y can be calculated by Eqns.(3) and (4), and then thermodynamic condition diagram of oxidation, preferential oxidation and free oxidation of Ag-Y alloys is obtained and shown in Fig.2.

Fig.2 Thermodynamic condition diagram of oxidation, preferential oxidation and free oxidation of Ag-Y alloys
Fig.2 shows that the threshold of oxygen partial pressure of oxidation and free oxidation of pure Y is very low and there is a large area of preferential oxidation, which is advantageous to control the oxygen partial pressure. The internal oxidation (preferential oxidation) of Ag-Y alloy is feasible from the viewpoint of thermodynamics under the oxidation temperature and oxygen partial pressure of preferential oxidation. The upper limit of oxygen partial pressure of internal oxidation is a function of temperature determined by Eqn.(3). If the actual oxygen partial pressure is higher than the upper limit of oxygen partial pressure of internal oxidation, external oxidation of Ag will take place. Whereas, if the medium’s oxygen partial pressure is too low, the driving force of internal oxidation is not enough and the reversed diffusion of Y is strengthened, the external oxidation of Y will occur. As a result, the oxygen partial pressure of internal oxidation should be close to the upper limit as possible. The actual internal oxidation area is small under the upper limit of oxygen partial pressure, through which we can select the reasonable process of internal oxidation.
3.2 Phase constitution of Ag-Y alloy after internal oxidation
The phase analysis of Ag-Y alloy after internal oxidation was performed by XRD. Fig.3 shows the XRD pattern of Ag-Y2O3 fabricated by internal oxidation. It can be seen that Ag-Y alloy after internal oxidation is composed of two phases (Ag and Y2O3) without any impure phases, so it is feasible to fabricate the Ag-Y2O3 materials by internal oxidation method.
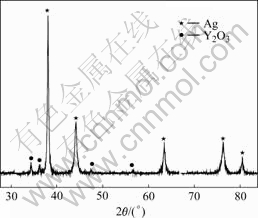
Fig.3 XRD pattern of Ag-Y2O3 material fabricated by internal oxidation
3.3 Morphologies of Ag-Y alloy powder in fore-and- aft internal oxidation
The morphology analysis of the Ag-Y alloy powder particles before and after internal oxidation was carried out by SEM. The SEM images are shown in Fig.4.

Fig.4 SEM images of Ag-Y (a) and Ag-Y2O3 (b) powder particles
Fig.4 shows that the morphology of Ag-Y alloy particles after internal oxidation is changed significantly. Slick and round surface of the particles becomes coarse and scraggy because the oxidation reaction takes place on the surface of the particles. The formation of large amount of Y2O3 particles inside the Ag-Y alloy particles gives rise to the partial stress concentration, which results in the bulge part on the surface of alloy particles. The composition of regions A and B in Fig.4(b) was analyzed using EDS. The results of EDS (Fig.5) show that region B does not contain element Y and Y2O3, while region A does. The fact that Ag content of region B is 100% shows that the convex part is pure Ag. Oxide particles distribute continuously around convex part (region B in Fig.4(b)). The reaction of Y with oxygen results in the impoverishment of Y in region B. The density of the formed oxides is smaller than that of Ag-Y alloy, and the volume expansion of oxides leads to the convex surface of Ag-Y alloy.
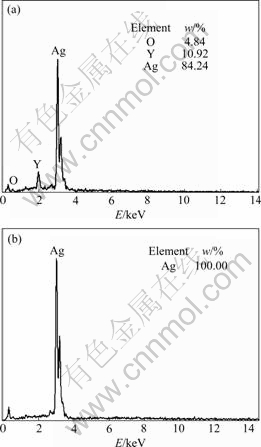
Fig.5 EDS analysis for regions A and B in Fig.4(b):(a) Region A; (b) Region B
After the Ag-Y alloy internal oxidation, the obvious feature on particles surface is the formation of pure silver area, which means that matrix metal element (Ag) diffuses from the interior part to the surface of particles, in general, this process directly relates to the oxide formation. Because the PBR (Pilling-Bedworth Ratio) of oxide is more than 1[16], and Y is enriched in internal oxidation area, when Y2O3 precipitates, it is unavoidable that the volume expansion of matrix alloy leads to stress in alloy. For Ag-Y alloy, the volume expansion after internal oxidation can be calculated through the following equation:

where V is the total volume of alloy after internal oxidation of 1 mol Ag-Y, Valloy is molar volume of Ag-Y alloy,  is the volume of Y2O3 after internal oxidation of 1 mol Y,
is the volume of Y2O3 after internal oxidation of 1 mol Y,  is the mole fraction of Y in the alloy matrix Ag.
is the mole fraction of Y in the alloy matrix Ag.
The inner stress produced by volume expansion should be released in some ways. The diffusion of the matrix metallic element toward the surface is the main manner of the inner stress relaxation. So the convex part on particles surface is pure silver.
3.4 Microstructures of Ag-Y2O3 material
The optical micrograph of the as-sintered Ag-Y2O3 materials is shown in Fig.6(a). The dark and gray parts are Y2O3 particles and Ag matrix, respectively. The Y2O3 particles distribute uniformly inside grains as colony Ag-Y2O3 materials, subsequent processing was carried out. Figs.6(b) and (c) show the SEM images of Ag-Y2O3 materials after severe plastic deformation, indicating that the finer Y2O3 particles distribute dispersedly and more uniformly in Ag matrix. Therefore, the severe plastic deformation can improve the microstructures of the Ag-Y2O3 material.

Fig.6 Microstructures of Ag-Y2O3 material: (a) Sintered bulk; (b) Vertical to extrusion direction; (c) Parallel to extrusion direction
morphology. So the microstructure of Ag-Y2O3 sintered bulk is not uniform. In order to improve the microstructures of
4 Conclusions
1) The thermodynamic calculation of pure Ag and Y shows that the internal oxidation behavior of Ag-10.33Y alloy is feasible from the viewpoint of thermodynamics when using Ag2O as the oxidant. The upper limit of oxygen partial pressure of Ag-Y alloy oxidation that is determined by lg p(O2)=-3 190/T+11.91 is the function of temperature.
2) XRD result shows that two phases (Ag and Y2O3) appear after the internal oxidation of Ag-Y alloy. SEM results show that particles surface of Ag-Y alloy is slick and round before internal oxidation and becomes coarse and scraggy after internal oxidation. The volume expansion of oxide formed inside the alloy leads to surface protrusion of Ag-Y alloy.
3) The distribution of Y2O3 grain is asymmetric and seriously conglomerated in microstructure of sintered Ag-Y2O3 bulks, but it becomes dispersed and uniform in silver matrix after severe plastic deformation.
References
[1] PENG Xiao, LI Tei-pan, WU Wei-wen. Internal oxidation behavior of Ag-Cu alloy [J]. Journal of China Corrosion and Protection, 1997, 17(4): 248-254. (in Chinese)
[2] CHEN Jing-chao, SUN Jia-lin, ZHANG Kun-hua, DU Yan, ZHOU Xiao-long, GAN Guo-you. European Confederation restrictive policy of Ag-CdO materials and the development of other silver metal oxide electrical contact materials [J]. Electric Engineering Materials, 2002(4): 41-44. (in Chinese)
[3] LI Ying-min, WANG Jun-bo, JI Dong, LI Shao-chun. The development status of novel Ag-SnO2 contact material [J]. Electrical Switching, 2003(3): 20-27. (in Chinese)
[4] NING Yuan-tao. Investigation of noble metal-rare earth alloy’s structure, performance and new materials [J]. Nobel Metal, 1994, 15(3): 61-71. (in Chinese)
[5] LANG L E. The Role of Active Elements in the Oxidation Behavior of High Temperature Metals and Alloys [M]. London: Elsevier Applied Science, 1989: 56-67.
[6] MASSALSKI T B, SUBRAMANIAN P R, ORAMOTO H. Binary Alloys Phase Diagrams (Vol.1-3) [M]. 2nd ed. Ohio: ASM International Materials Park, 1990: 32-46.
[7] GESMUNDO F, VIANI F, NIU Y. The internal oxidation of two-phase binary alloys under low oxidant pressures [J]. Oxid Met, 1996, 24(2): 45-51.
[8] YU Jue-qi, YI Wen-zhi, CHEN Bang-di, CHEN Hong-jian. The Phase Diagrams Collection of Binary Alloy State [M]. Shanghai: Shanghai Science and Technology Press, 1987: 127-130. (in Chinese)
[9] FU Guang-yan, NIU Yan, WU Wei. The oxidation of the Cu-Cr alloy fabricated by machine alloy in the 0.1 MPa pure oxygen[J]. Journal of China Corrosion and Protection, 2000, 10(3): 269-273. (in Chinese)
[10] TIAN Su-gui, ZHANG Lu-ting, WANG Gui-hua, QIAO Rui-qing, JIN Shou-ri, LI Tie-fan. Study on internal oxidation of the AlN particles strengthen Cu based compound materials [J]. Materials Project, 2000, 10(1): 25-28. (in Chinese)
[11] ZHANG Yun, WU Jian-yun, LI Guo-bing, SHEN Yu-tian, LEI Qian-quan. Internal oxidation of Cu-Al alloy [J]. Materials Science and Processing, 1999, 6(3): 91-95. (in Chinese)
[12] BIRKS N, MEIER G H. Introduction to high temperature oxidation of metals [J]. Edward Amold, 1983, 34(4): 375-381.
[13] SHEN Yu-tian, CUI Chun-xiang, MENG Fan-bin. Internal oxidation of Cu-Al alloy and thermodynamics analysis of Cu oxidation behavior [J]. Technology of Powder Metallurgy, 2000, 19(1): 28-32. (in Chinese)
[14] DU Qing-zhi, YANG Ji-shun. Physical and Chemistry [M]. Chongqing: Chongqing University Press, 1997: 72-75.
[15] YE Da-lun, HU Jian-hua. Thermodynamic Data Handbook of the Practical Mineral [M]. Beijing: Metallurgical Industry Press, 2002: 26-31.
[16] WU Chun-ping. Internal Oxidation Fabrication and Mechanism Study of the Ag-ReO Electrical Contact Materials [D]. Kunming: Kunming University of Science and Technology, 2005: 49-52.
Foundation item: Project(2006BAE03B03) supported by the National Science and Technology Program of China
Corresponding author: YI Dan-qing; Tel: +86-731-8830263; E-mail: yioffice@mail.csu.edu.cn
(Edited by CHEN Wei-ping)