���Ϳɳ��-������ص����״�״��-MnO2/Pd���������
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2014���1��
�������ߣ��� �� �� ǿ ɣ �� �� �� ���˽� ������
����ҳ�룺164 - 170
�ؼ��ʣ��-������أ����ϴ�������״�п��˿ǽṹ
Key words��lithium-air battery; composite catalyst; nanoneedle-based hollow microsphere; core-shell structure
ժ Ҫ������NaBH4��Һ��ԭPdCl2�ķ����ھ������״�״����Ħ�-MnO2�п����ϳ���Pd���Ʊ�һ�֦�-MnO2/Pd�˿����ϴ�����Ϊ��һ���ɳ��-������صĴ�����TEM��XRD��EDS�ȷ����ķ�������� �����ڴ���������Pd�������ȵطֲ��ڴ�״��-MnO2�п�����棬Pd�ڴ����е���������ԼΪ8.88%����ŵ���Խ�������������״�״�Ħ�-MnO2�п��������ȣ���-MnO2/Pd���ϴ�������˿����缫������ת��Ч�ʺͳ�ŵ�ѭ�����ܡ���Super P̼���Ϻ����Ʊ��Ħ�-MnO2/Pd���ϴ��������ɵĿ����缫��0.1 mA/cm2�����ܶ��µ��״ηŵ���������Դ�1220 mA��h/g����13�γ�ŵ�ѭ���������������Ϊ47.3%�������״�״��-MnO2/Pd�˿����ϴ�����һ����ǰ;�Ŀ�����ش�����
Abstract: The hollow ��-MnO2 nanoneedle-based microspheres coated with Pd nanoparticles were reported as a novel catalyst for rechargeable lithium-air batteries. The hollow microspheres are composed of ��-MnO2 nanoneedles. Pd nanoparticles are deposited on the hollow microspheres through an aqueous-solution reduction of PdCl2 with NaBH4 at room temperature. The results of TEM, XRD, and EDS show that the Pd nanoparticles are coated on the surface of ��-MnO2 nanoneedles uniformly and the mass fraction of Pd in the Pd-coated ��-MnO2 catalyst is about 8.88%. Compared with the counterpart of the hollow ��-MnO2 catalyst, the hollow Pd-coated ��-MnO2 catalyst improves the energy conversion efficiency and the charge-discharge cycling performance of the air electrode. The initial specific discharge capacity of an air electrode composed of Super P carbon and the as-prepared Pd-coated ��-MnO2 catalyst is 1220 mA��h/g (based on the total electrode mass) at a current density of 0.1 mA/cm2, and the capacity retention rate is about 47.3% after 13 charge-discharge cycles. The results of charge-discharge cycling tests demonstrate that this novel Pd-coated ��-MnO2 catalyst with a hierarchical core-shell structure is a promising catalyst for the lithium-air battery.
Trans. Nonferrous Met. Soc. China 24(2014) 164-170
Ming ZHANG1,2, Qiang XU1, Lin SANG2, Fei DING2, Xing-jiang LIU1,2, Li-fang JIAO3
1. School of Chemical Engineering and Technology, Tianjin University, Tianjin 300072, China;
2. National Key Laboratory of Science and Technology on Power Sources, Tianjin Institute of Power Sources, Tianjin 300384, China;
3. Key Laboratory of Advanced Energy Materials Chemistry, Ministry of Education, Nankai University, Tianjin 300071, China
Received 23 November 2012; accepted 20 April 2013
Abstract: The hollow ��-MnO2 nanoneedle-based microspheres coated with Pd nanoparticles were reported as a novel catalyst for rechargeable lithium-air batteries. The hollow microspheres are composed of ��-MnO2 nanoneedles. Pd nanoparticles are deposited on the hollow microspheres through an aqueous-solution reduction of PdCl2 with NaBH4 at room temperature. The results of TEM, XRD, and EDS show that the Pd nanoparticles are coated on the surface of ��-MnO2 nanoneedles uniformly and the mass fraction of Pd in the Pd-coated ��-MnO2 catalyst is about 8.88%. Compared with the counterpart of the hollow ��-MnO2 catalyst, the hollow Pd-coated ��-MnO2 catalyst improves the energy conversion efficiency and the charge-discharge cycling performance of the air electrode. The initial specific discharge capacity of an air electrode composed of Super P carbon and the as-prepared Pd-coated ��-MnO2 catalyst is 1220 mA��h/g (based on the total electrode mass) at a current density of 0.1 mA/cm2, and the capacity retention rate is about 47.3% after 13 charge-discharge cycles. The results of charge-discharge cycling tests demonstrate that this novel Pd-coated ��-MnO2 catalyst with a hierarchical core-shell structure is a promising catalyst for the lithium-air battery.
Key words: lithium-air battery; composite catalyst; nanoneedle-based hollow microsphere; core-shell structure
1 Introduction
Traditional cathode materials used in Li-ion batteries, such as LiCoO2 and LiFePO4, have limited capacities ranging from 130 to 150 mA��h/g owing to the nature of their crystal structures and their electrochemical properties [1]. Recently, lithium-air batteries have attracted much attention because of their extreme high specific energy density, so they have the potential to be the power source for the advanced electric vehicles. Unlike conventional lithium and lithium-ion batteries, the cathode material for lithium-air batteries is not stored inside the batteries, and the theoretical specific energy density of the lithium-air battery excluding oxygen is 11140 W��h/kg [2-4]. The lithium-air battery was firstly reported by ABRAHAM and JIANG [5], that it had an open-circuit potential of about 3 V and an energy density of 250-350 W��h/kg. Such a lithium-air battery comprises a Li-containing anode, an organic electrolyte and an air cathode [5-7].
Although the lithium-air battery is a very attractive system, there are still many challenges that need to be overcome before practical applications, such as the blocking of pores within air electrodes, decomposition of the non-aqueous electrolyte, high overpotential and the poor charge-discharge cycling performance [4,8,9]. The foremost challenge that needs to be focused on is how to improve the cycling performance of rechargeable lithium-air batteries in the non-aqueous electrolyte. To improve the energy conversion efficiency and the cycling performance of lithium-air batteries, various electrochemical catalysts for the air electrode have been studied recently, such as the transitional metal oxides [10], the carbon-supported manganese oxide nanocatalysts [11], the MnO2 nanoflakes coated multi-walled carbon nanotubes [12], the bifunctional electrocatalyst of platinum-gold nanoparticles loaded on the XC-72 carbon [13], the nitrogen-doped carbon nanotubes [14] or graphene nanosheets [15], the compound transitional metal oxides (MnCo2O4 [16] and Ag2Mn8O6 [17]) and the platinum nanoparticle-graphene complexes [18].
Recently, a novel MnO2/Pd composite catalyst of rechargeable lithium-air batteries was prepared by mixing manganese dioxide, Pd and PTFE [19]. Using this MnO2/Pd composite catalyst, the charging potential of the air electrode may nearly decrease to its theoretical value (3.7 V vs Li/Li+) and a stable specific capacity of 225 mA��h/g for the air electrode could be obtained during 20 charge-discharge cycles. In addition, another new Pd-coated manganese dioxide composite catalyst with a core-shell structure was synthesized by depositing Pd on the surface of ��-MnO2 nanorod particles, and exhibited a good electrocatalytic performance for the oxygen reduction in alkaline media [20]. The MnO2/Pd core-shell catalyst composed of MnO2 cores and less Pd shell may not only reduce the Pd dosage within the catalyst but also improve the electro-catalytic activity for the oxygen reduction reaction (ORR) by a synergetic effect between MnO2 and Pd [21]. Unfortunately, the electrocatalytic behavior of MnO2/Pd catalysts with a core-shell structure for rechargeable lithium-air batteries has attracted less attention until now.
In this work, a novel Pd-coated manganese dioxide composite catalyst (MnO2/Pd) with a core-shell structure was prepared for rechargeable lithium-air batteries. Hollow microspheres composed of ��-MnO2 nanoneedles were synthesized by a hydrothermal method, and Pd nanoparticles were precipitated on the surface of the as-prepared hollow ��-MnO2 microspheres by an electroless deposition method at room temperature. The electrocatalytic performance of the MnO2/Pd core-shell catalyst was investigated in an air electrode supported by carbon materials (Super P carbon).
2 Experimental
All chemical reagents used in the experiments were of analytical grade and used without further purification.
2.1 Preparation of hollow ��-MnO2 microspheres
The manganese dioxide (��-MnO2) microspheres with a hollow structure were synthesized by a hydrothermal method reported in Ref. [22]. KMnO4 (0.474 g), MnSO4��H2O (0.845 g), FeCl3��6H2O (0.054 g) and 1 mL of concentrated sulfuric acid��98%��were mixed in 15 mL of de-ionized water and stirred with a magnetic stirrer at room temperature for 10 min to form a homogeneous solution. Then the obtained solution was transferred into a Teflon (PTFE)-lined autoclave (20 mL) and heated at 150 ��C for 20 min. The obtained precipitates were filtered, washed with distilled water and dried at 60 ��C for 4 h in air.
2.2 Preparation of hollow ��-MnO2/Pd core-shell microspheres
The Pd-coated manganese dioxide microspheres with a hierarchical core-shell structure were prepared by an electroless deposition of Pd on the surface of the as-prepared hollow ��-MnO2 microspheres. 0.3 g ��-MnO2 microspheres dispersed in an ethanol/water solution (1:1, in volume ratio) were mixed with 100 mL of 3.1 mmol/L PdCl2 solution ultrasonically. Then the NaBH4 was added dropwise to the solution with a continuous magnetic stirring. After the reductive reaction performed at room temperature for 20 min, the obtained product was washed with de-ionized water and dried in a vacuum oven at 70 ��C for 12 h.
2.3 Characterization of hollow microsphere catalysts
The surface morphology of the as-prepared catalysts was investigated by a Hitachi S-4800 field emission scanning electron microscope (FE-SEM) and the components were analyzed by a HORIBA 7593-H energy-dispersive spectrometer. The X-ray diffraction (XRD) patterns of the as-prepared catalysts were recorded by a Rigaku D/max-2500 diffractometer with a Cu K�� radiation. The nitrogen adsorption-desorption measurement was conducted by a Quadrasorb SI surface area analyzer at 77 K. The specific surface area of the as-prepared catalysts was calculated by a Brunauer- Emmett-Teller (BET) method and experimental points were obtained at a relative pressure of 0.05-0.25.
2.4 Preparation of air electrode including hollow microsphere catalysts
The catalytic layers of air electrodes were composed of Super P carbon, the as-prepared hollow ��-MnO2/Pd core-shell catalyst, and polyvinylidene fluoride (PVDF) solutions in a mass ratio of 75:15:10. For comparision, the air electrode composed of Super P, the as-prepared ��-MnO2 catalyst and PVDF solutions with the same composition was also prepared. When preparing the air electrode, the catalytic layer was tiled on a foam nickel current collector, and then dried at 60 ��C in a vacuum oven for 24 h. The loading content of the catalytic layer in an air electrode is about 5 mg/cm2.
2.5 Electrochemical measurements of air electrode containing hollow microsphere catalysts
The electrochemical behavior of the as-prepared air electrodes was tested by a lithium-air coin battery which had four 3 mm-diameter holes on the side of shells of battery. This lithium-air coin battery was assembled from the as-prepared air electrode and a lithium foil (0.5 mm in thickness) used as the counter electrode in a 2032-type coin cell. The electrolyte was 1 mol/L LiClO4/PC+DEC (1:1 in volume ratio) and a Celgard 2300 membrane was used as the cell separator. The assembling process of the lithium-air coin battery was performed in an argon filled glove box (MIKROUNA Advanced 2440/750) with water and oxygen level less than 1��10-6.
To avoid some side effects produced by the humidity and CO2 in air, all electrochemical measurements of the lithium-air coin battery were conducted in a homemade cylinder which was filled with dry and pure oxygen gas at a pressure of 1��105 Pa. The charge-discharge behavior of the lithium-air coin battery was measured by a Solartron analytical system (1400/1470E) at different current densities in a voltage range of 2.0-4.5 V. The air electrode capacity was calculated according to the total mass of the air electrode (carbon + catalyst + binder).
3 Results and discussion
Figure 1 shows the XRD patterns of the as-prepared MnO2 and the as-prepared Pd-coated MnO2 catalysts. As shown in Fig. 1(a), it is seen that the XRD diffraction peaks of the as-prepared MnO2 catalyst are assigned to a pure ��-MnO2 tetragonal phase (Joint Committee on Powder Diffraction Standards (JCPDS) Card No. 44-0141). Compared with the XRD pattern of the as-prepared MnO2 catalyst (Fig. 1(a)), there are four new peaks located at about 40.1��, 46.7��, 68.1�� and 82.1�� in the XRD pattern of the Pd-coated MnO2 catalyst (Fig. 1(b)). These four peaks are well attributed to the lattice faces of Pd (111), Pd (200), Pd (220) and Pd (311) (JCPDS Card No. 65-2867), respectively. This indicates that the as-prepared ��-MnO2 catalyst is coated by Pd.
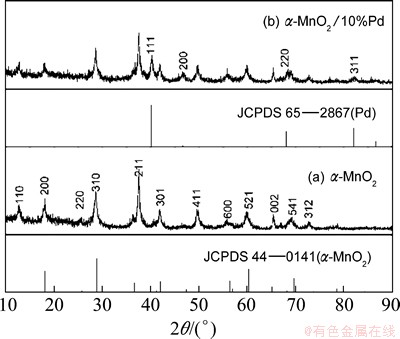
Fig. 1 XRD patterns of as-prepared catalysts
Figure 2 shows the curves of N2 adsorption- desorption isotherm measurements of the as-prepared ��-MnO2 catalyst. The N2 adsorption/desorption curves exhibit a typical IV shape, indicating that the as-prepared ��-MnO2 catalyst has a characteristic of the mesoporous [18]. The specific surface area of the as-prepared ��-MnO2 catalyst is estimated to be about 145 m2/g.
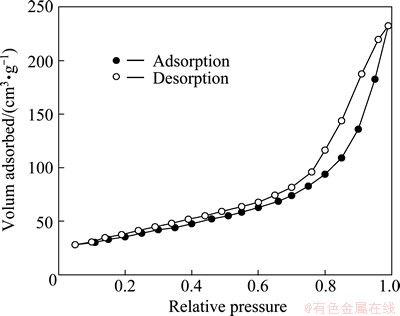
Fig. 2 N2 adsorption-desorption isotherms of as-prepared ��-MnO2 catalyst
Figure 3 shows the SEM and TEM images of the as-prepared ��-MnO2 catalyst. As shown in Fig. 3(a), the as-prepared ��-MnO2 catalyst has a typical microspherical morphology which is composed of many hollow microspheres with the oriented nanoneedles arranged on the surface. The diameter of these microspheres is 1-2 ��m. Furthermore, it is seen from the surface ruptures that the ��-MnO2 microspheres are hollow. The TEM image further indicates that the nanoneedles with a length of 100-200 nm form a hollow ��-MnO2 microsphere structure. This hierarchical microstructure results in a large specific surface area of the ��-MnO2 catalyst.

Fig. 3 SEM (a) and TEM (b) images of as-prepared ��-MnO2 catalyst

Fig. 4 SEM (a), TEM (b) and HRTEM (c) images of as-prepared ��-MnO2/Pd catalyst
Figure 4 shows the SEM, TEM and HRTEM images of the as-prepared Pd-coated ��-MnO2 catalyst. As shown in Fig. 4(a), the surface morphology of the composite catalyst is similar to that of the above-mentioned hollow ��-MnO2 catalyst in Fig. 3(a). Namely, the original morphology has been retained after the deposition of Pd. From Fig. 4(b), it is clear that Pd nanoparticles with a diameter of about 5 nm are distributed on the surface of ��-MnO2 nanoneedles evenly. Furthermore, no obvious agglomeration of Pd nanoparticles appears on the surface of the ��-MnO2 catalysts. In addition, the HRTEM image of the Pd-coated ��-MnO2 catalyst is shown in Fig. 4(c). The (110) lattice spacing of the as-prepared ��-MnO2 catalyst is about 0.67 nm and the (111) lattice spacing of Pd deposited on the surface of the ��-MnO2 catalyst is 0.24 nm, which match well with the standard lattice spacing of the ��-MnO2 (d110=0.6919 nm) and Pd (d111=0.2246 nm), respectively. These results also demonstrate that the composite MnO2/Pd catalysts with a core-shell structure have been prepared successfully by an electroless deposition of Pd on the surface of the ��-MnO2 catalyst.
Figure 5 shows the EDS analytical result of the as-prepared Pd-coated ��-MnO2 catalyst. It shows the existence of palladium, manganese and oxygen in the composite ��-MnO2/Pd catalyst. The mass fraction of Pd approximates 8.88 %, which is close to the feed ratio of 10 %.
The initial charge-discharge curves of the ��-MnO2/Super P air electrode and the ��-MnO2/Pd/Super P air electrode at different current densities are shown in Fig. 6. The specific discharge capacity of the ��-MnO2/ Pd/Super P air electrode is 1220 mA��h/g at a current density of 0.1 mA/cm2, while 1089.6 mA��h/g is obtained from the ��-MnO2/Super P air electrode at the same current density. Furthermore, the specific discharge capacities of the ��-MnO2/Pd/Super P air electrode are 921.4 and 419.3 mA��h/g at the current densities of 0.2 and 0.5 mA/cm2, respectively. Under the same conditions, 700.0 and 203.9 mA��h/g are obtained from the ��-MnO2/Super P air electrode, respectively. These results show that the specific discharge capacities of above two air electrodes decrease with the increase of current densities owing to the polarization of air electrodes. The reasons may be that both the blocking of insoluble discharge products within pores of the air electrode and the decrease of the oxygen concentration in the electrolyte become more and more serious with the increase of current densities [7,23].
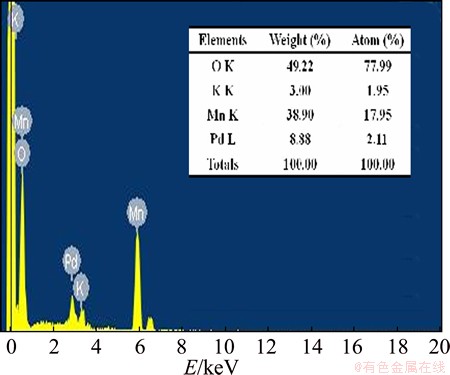
Fig. 5 EDS analysis result of as-prepared ��-MnO2/Pd catalyst
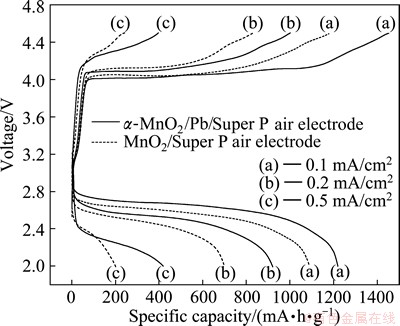
Fig. 6 Charge-discharge curves of both ��-MnO2/Pd/Super P air electrode and MnO2/Super P air electrode in 1��105 Pa O2 atmosphere between 2.0-4.5 V at different current densities
From Fig. 6, we could also find that every charge- discharge voltage difference of the ��-MnO2/Pd/Super P air electrode at different current densities is lower than that of the ��-MnO2/Super P air electrode at the same current density. This means that the composite ��-MnO2/Pd catalyst has higher electrocatalytic activity than the ��-MnO2 catalyst for the oxygen reduction reaction (ORR) and the oxygen evolution reaction (OER) [13], which is ascribed to the high catalytic activity of Pd nanoparticles anchored on the surface of the ��-MnO2 catalysts [19]. Furthermore, the polarization of both the ��-MnO2/Pd/Super P air electrode and the MnO2/Super P air electrode is significant in the initial stage of charge-discharge. This is due to the decomposition of the carbonate-based electrolyte [24].
Figure 7 shows the charge-discharge curves of the above two air electrodes at a current density of 0.1 mA/cm2 during different charge-discharge cycles. As shown in Fig. 7(a), the initial specific discharge capacity of the ��-MnO2/Super P air electrode is 1089.6 mA��h/g, and then steadily declines to 262.8 mA��h/g after 13 charge-discharge cycles. This indicates that the capacity retention rate of the ��-MnO2/Super P air electrode is only 24.1%. By comparison, the ��-MnO2/Pd/Super P air electrode exhibits a better charge-discharge cycling performance. The initial specific discharge capacity of the ��-MnO2/Pd/Super P air electrode is 1220 mA��h/g, and then steadily decreases to 567.6 mA��h/g after 13 charge-discharge cycles. Namely, its capacity retention rate is about 47.3%. These results show that the composite ��-MnO2/Pd catalyst not only improves the electrocatalytic activity of the oxygen reduction in the discharge process, but also facilitates decomposition of the discharge products in the charge process [13].
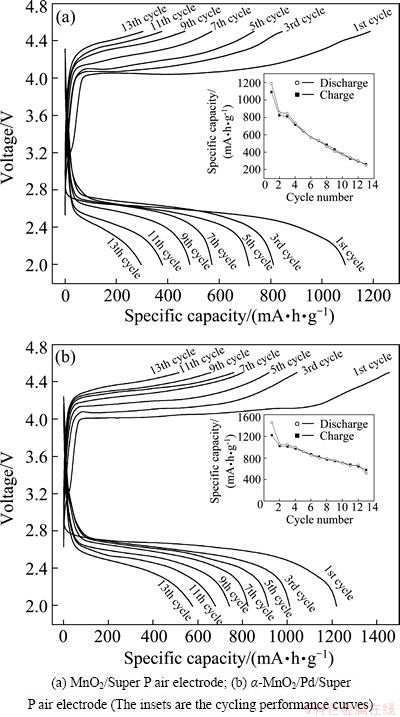
Fig. 7 Charge-discharge curves of air electrodes in 1��105 Pa O2 atmosphere between 2.0-4.5V at current density of 0.1 mA/cm2
4 Conclusions
A novel Pd-coated ��-MnO2 catalyst with a hierarchical core-shell structure was prepared by an electroless deposition method. The results of SEM and TEM confirm that the as-prepared composite MnO2/Pd catalyst has a microspherical structure with Pd nanoparticles deposited on the surface of the oriented ��-MnO2 nanoneedles. These ��-MnO2 nanoneedles form a hollow ��-MnO2 microsphere structure. The initial specific discharge capacity of the ��-MnO2/Pd/Super P air electrode is 1220 mA��h/g and the capacity retention rate is about 47.3% after 13 charge-discharge cycles. Compared with the ��-MnO2/Super P air electrode, both the energy conversion efficiency and the charge- discharge cycling performance are improved. Obviously, this kind of hollow ��-MnO2/Pd catalysts with a complicated core-shell structure is a promising catalyst for the rechargeable lithium-air battery.
References
[1] XIAO J, WANG D, XU W, WANG D, WILLIFORD R E. Optimization of air electrode for Li/air batteries [J]. Journal of the Electrochemical Society, 2010, 157: A487-A492.
[2] KRAYTSBERG A, EIN-ELI Y. Review on Li�Cair batteries- Opportunities, limitations and perspective [J]. Journal of Power Sources, 2011, 196: 886-893.
[3] GIRISHKUMAR G, MCCLOSKEY B, LUNTZ A C, SWANSON S, WILCKE W. Lithium-air battery promise and challenges [J]. The Journal of Physical Chemistry Letters, 2010, 1: 193-203.
[4] PADBURY R, ZHANG X. Lithium�Coxygen batteries-Limiting factors that affect performance [J]. Journal of Power Sources, 2011, 196: 4436-4444.
[5] ABRAHAM K M, JIANG Z. A Polymer electrolyte-based rechargeable lithium/oxygen battery [J]. Journal of the Electrochemical Society, 1996, 143: 1-5.
[6] OGASAWARA T, DEBART A, HOLZAPFEL M, NOVAK P, BRUCE P G. Rechargeable Li2O2 electrode for lithium batteries [J]. Journal of the American Chemical Society, 2006, 128: 1390-1393.
[7] READ J. Characterization of the lithium/oxygen organic electrolyte battery [J]. Journal of the Electrochemical Society, 2002, 149: A1190-A1195.
[8] PARK C K, PARK S B, LEE S Y, LEE H, JANG H, CHO W I. Electrochemical performances of lithium-air cell with carbon materials [J]. Bulletin of the Korean Chemical Society, 2010, 31: 3221-3224.
[9] XIAO J, HU J, WANG D, HU D, XU W, GRAFF G L, NIE Z, LIU J, ZHANG J G. Investigation of the rechargeability of Li�CO2 batteries in non-aqueous electrolyte [J]. Journal of Power Sources, 2011, 196: 5674-5678.
[10] DEBART A, PATERSON A J, BAO J, BRUCE P G. ��-MnO2 nanowires: A catalyst for the O2 electrode in rechargeable lithium batteries [J]. Angewandte Chemie International Edition, 2008, 47: 4521-4524.
[11] CHENG H, SCOTT K. Carbon-supported manganese oxide nanocatalysts for rechargeable lithium�Cair batteries [J]. Journal of Power Sources, 2010, 195: 1370-1374.
[12] LI J, WANG N, ZHAO Y, DING Y, GUAN L. MnO2 nanoflakes coated on multi-walled carbon nanotubes for rechargeable lithium- air batteries [J]. Electrochemistry Communications, 2011, 13: 698-700.
[13] LU Y C, XU Z, GASTEIGER H A, CHEN S, HAMAD- SCHIFFERLI K, SHAO-HORN Y. Platinum-gold nanoparticles a highly active bifunctional electrocatalyst for rechargeable lithium-air batteries [J]. Journal of the American Chemical Society, 2010, 132: 12170-12171.
[14] LI Y, WANG J, LI X, LIU J, GENG D, YANG J, LI R, SUN X. Nitrogen-doped carbon nanotubes as cathode for lithium�Cair batteries [J]. Electrochemistry Communications, 2011, 13: 668-672.
[15] LI Y, WANG J, LI X, GENG D, BANIS M N, LI R, SUN X. Nitrogen-doped graphene nanosheets as cathode materials with excellent electrocatalytic activity for high capacity lithium-oxygen batteries [J]. Electrochemistry Communications, 2012, 18: 12-15.
[16] WANG H, YANG Y, LIANG Y, ZHENG G, LI Y, CUI Y, DAI H. Rechargeable Li-O2 batteries with covalently coupled MnCo2O4- graphene hybrid as oxygen cathode catalyst [J]. Energy & Environmental Science, 2012, 5: 7931-7935.
[17] ZHANG G Q, HENDRICKSON M, PLICHTA E J, AU M, ZHENG J P. Preparation, characterization and electrochemical catalytic properties of hollandite Ag2Mn8O16 for Li-air batteries[J]. Journal of Electrochemical Society, 2012, 159: A310-A314.
[18] YANG Y, SHI M, ZHOU Q F, LI Y S, FU Z W. Platinum nanoparticle�Cgraphene hybrids synthesized by liquid phase pulsed laser ablation as cathode catalysts for Li-air batteries [J]. Electrochemistry Communications, 2012, 20: 11-14.
[19] THAPA A K, ISHIHARA T. Mesoporous ��-MnO2/Pd catalyst air electrode for rechargeable lithium�Cair battery [J]. Journal of Power Sources, 2011, 196: 7016-7020.
[20] XU M W, GAO G Y, ZHOU W J, ZHANG K F, LI H L. Novel Pd/��-MnO2 nanotubes composites as catalysts for methanol oxidation in alkaline solution [J]. Journal of Power Sources, 2008, 175: 217-220.
[21] SUN W, HSU A, CHEN R. Palladium-coated manganese dioxide catalysts for oxygen reduction reaction in alkaline media [J]. Journal of Power Sources, 2011, 196: 4491-4498.
[22] HE X, YANG M, NI P, LI Y, LIU Z H. Rapid synthesis of hollow structured MnO2 microspheres and their capacitance [J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2010, 363: 64-70.
[23] YANG X H, XIA Y Y. The effect of oxygen pressures on the electrochemical profile of lithium/oxygen battery [J]. Journal of Solid State Electrochemistry, 2010, 14: 109-114.
[24] CHEN Y H, FREUNBEGER S A, PENG Z Q, BARDE F, BRUCE P G. Li-O2 battery with a dimethylformamide electrolyte [J]. Journal of the American Chemical Society, 2012, 134: 7952-7957.
�� ��1,2���� ǿ1��ɣ ��2���� ��2�����˽�1,2��������3
1. ����ѧ ����ѧԺ����� 300072��
2. ����Դ�о��� ��ѧ��������Դ�ص�ʵ���ң���� 300384��
3. �Ͽ���ѧ �Ƚ���Դ���ϻ�ѧ�������ص�ʵ���ң���� 300071
ժ Ҫ������NaBH4��Һ��ԭPdCl2�ķ����ھ������״�״����Ħ�-MnO2�п����ϳ���Pd���Ʊ�һ�֦�-MnO2/Pd�˿����ϴ�����Ϊ��һ���ɳ��-������صĴ�����TEM��XRD��EDS�ȷ����ķ�������������ڴ���������Pd�������ȵطֲ��ڴ�״��-MnO2�п�����棬Pd�ڴ����е���������ԼΪ8.88%����ŵ���Խ�������������״�״�Ħ�-MnO2�п��������ȣ���-MnO2/Pd���ϴ�������˿����缫������ת��Ч�ʺͳ�ŵ�ѭ�����ܡ���Super P̼���Ϻ����Ʊ��Ħ�-MnO2/Pd���ϴ��������ɵĿ����缫��0.1 mA/cm2�����ܶ��µ��״ηŵ���������Դ�1220 mA��h/g����13�γ�ŵ�ѭ���������������Ϊ47.3%�������״�״��-MnO2/Pd�˿����ϴ�����һ����ǰ;�Ŀ�����ش�����
�ؼ��ʣ��-������أ����ϴ�������״�п��˿ǽṹ
(Edited by Xiang-qun LI)
Foundation item: Project (20973124) supported by the National Natural Science Foundation of China; Project (KLAEMC-OP201101) supported by the Open Project of Key Laboratory of Advanced Energy Materials Chemistry of Ministry of Education (Nankai University), China
Corresponding author: Qiang XU; Tel: +86-13702108410; E-mail: xuqiang@tju.edu.cn; Xing-jiang LIU; E-mail: xjliu@nklps.org
DOI: 10.1016/S1003-6326(14)63043-0

