��-�������������̷��ⶨ���ڽ��������е���ɢϵ��
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2014���12��
�������ߣ���ٻٻ �� Դ ������
����ҳ�룺4030 - 4037
�ؼ��ʣ��⣻��ɢϵ����Һ̬��������-����������������
Key words��hydrogen; diffusion coefficient; liquid metal; solid-gas eutectic; unidirectional solidification
ժ Ҫ�����ڹ�-�������������̼����͵���Ͻ�Ķ�������ԭ�������һ�ֲⶨ���ڽ�����������ɢϵ���ķ�������Cu-H2ϵΪ�����о������ѹ��������ȶȶ����ڽ����������۵㴦��ɢϵ����Ӱ�졣�����ʾ����ͬ���ղ�������������õ���ɢϵ������С���Ҳ��ø÷�����õ����ڽ���Cu�������¶���ص���ɢϵ�������������в���ʵ�鷽����õĽ���ǺϵýϺã�����֤�˸÷����Ŀ����ԡ���Cu-H2ϵ�⣬�����������ڽ���Mg��Si�Լ�Cu-34.6%Mn�Ͻ��۵��¶������е���ɢϵ��ֵ�Լ��¶���ص���ɢϵ�����̡�
Abstract: Based on the solid-gas eutectic unidirectional solidification technique and the principle of unidirectional solidification of single-phase alloy, a new method for evaluating the diffusion coefficient of hydrogen in liquid metals was proposed. Taking Cu-H2 system for example, the influences of argon partial pressure and superheat degree of melt on the diffusion coefficient of hydrogen in liquid metal were studied and the predicted values were similar to each other. The obtained temperature-dependent equation for diffusion coefficient of hydrogen in liquid copper is comparable with experimental data in literature, which validates the effectiveness of this method. The temperature-dependent equations for diffusion coefficient of hydrogen in liquid Mg, Si and Cu-34.6%Mn alloy were also evaluated by this method, along with the values at the melting point of each metal and alloy.
Trans. Nonferrous Met. Soc. China 24(2014) 4030-4037
Qian-qian YANG1, Yuan LIU1,2, Yan-xiang LI1,2
1. School of Materials Science and Engineering, Tsinghua University, Beijing 100084, China;
2. Key Laboratory for Advanced Materials Processing Technology of Ministry of Education, Tsinghua University, Beijing 100084, China
Received 16 December 2013; accepted 18 June 2014
Abstract: Based on the solid-gas eutectic unidirectional solidification technique and the principle of unidirectional solidification of single-phase alloy, a new method for evaluating the diffusion coefficient of hydrogen in liquid metals was proposed. Taking Cu-H2 system for example, the influences of argon partial pressure and superheat degree of melt on the diffusion coefficient of hydrogen in liquid metal were studied and the predicted values were similar to each other. The obtained temperature-dependent equation for diffusion coefficient of hydrogen in liquid copper is comparable with experimental data in literature, which validates the effectiveness of this method. The temperature-dependent equations for diffusion coefficient of hydrogen in liquid Mg, Si and Cu-34.6%Mn alloy were also evaluated by this method, along with the values at the melting point of each metal and alloy.
Key words: hydrogen; diffusion coefficient; liquid metal; solid-gas eutectic; unidirectional solidification
1 Introduction
Investigation of the diffusion process in liquid metals is vital for gaining an effective understanding of nucleation, crystal growth, and liquid dynamics. The diffusion coefficient in particular is essential to the study of microstructural evolution, and has been the subject of interest for a number of materials and metallurgical researchers over recent years [1-4]. What is more, the similarity in physical properties between liquid metals and semiconductors means that such research can potentially have broader implications.
Up to now, a number of different experimental methods have been developed to measure the diffusion coefficient of solute atoms in liquid metals [5,6], i.e., capillary-reservoir, diffusion couple, shear-cell, plane source, electrochemical concentration-cell, slow neutron scattering, nuclear magnetic resonance and electrochemical methods. Furthermore, a number of theoretical models have also been developed over the past several decades, such as the Einstein relation model [6], rigid sphere model [7], density fluctuation model [8], and a modified ��hole�� theory model based on the Arrhenius relation [9]. However, despite this progress, it is still more difficult to obtain the diffusion coefficient of solute atoms in liquid metals than in solid ones because of the high sensitivity to external convection effects [10]. Experimental data regarding the diffusion coefficient for a small number of liquid metals are quite scarce and the results obtained tend to be quite diverse due to variation in the experimental method [11, 12]. In addition, many theoretical models cannot be applied widely, due to the absence of data for some important physical parameters. For example, CAHOON [9] presented the diffusion coefficient for solute impurity in liquid metals:
 (1)
(1)
where Z2 is the effective relative valence calculated by the Engel-Brewer ground state valence and a is the screening potential factor which is related to both the solvent properties and Z2. CHHABRA and ROY [13] regarded that the ratio of diameters between the solute impurity and solvent atoms (dA and dB) exerted a tremendous influence on the diffusion coefficient of solute impurity. The corresponding equation is
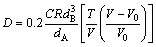 (2)
(2)
where V0 is the atomic volume of liquid metal with the infinite viscosity and C is the characteristic constant of liquid metal. The complicity and difficulty in acquiring those above parameters is a hindrance to the development of those theoretical models.
Compared with other atoms, the small atomic radius of hydrogen allows it to penetrate far more readily into metals and react with almost all other elements of the periodic table. The reaction exerts an impact on the microstructure and performance. Taking steel for example, the diffusion of hydrogen can accelerate the crack propagation which could result in the occurrence of hydrogen embrittlement (HE, a phenomenon which is notorious for significantly reducing the strength of steel) [14]. In other words, the appearance of HE is affected by the diffusion rate of hydrogen. Meanwhile, as for the technology of liquid state hydrogenation, the redistribution of alloy elements and the ascension of nucleation rate owing to the different diffusion coefficients of hydrogen in a and b phases could improve the microstructure of TC21 alloy [15]. In addition, the diffusion coefficient of hydrogen in liquid metal is the key parameter which influences the pore size in lotus-type porous structure [16]. Therefore, the study of hydrogen diffusion is quite important, particularly in the liquid metals within the casting industry. However, the high flammability of hydrogen and risks associated with high temperatures make it far more difficult to apply conventional means of measuring the diffusion coefficient of hydrogen.
The solid-gas eutectic unidirectional solidification technique [17-19] (or Gasar process) is a new method for fabricating porous materials with long cylindrical pores aligned in one direction. In this process, a melt saturated with hydrogen is solidified unidirectionally in a closed high-pressure chamber, which subsequently induces the formation of bubbles due to the difference in hydrogen solubility between the liquid and solid phases. By adjusting processing parameters such as the partial pressures of hydrogen and argon, or the superheat degree of melt, the initial hydrogen concentration and the required one for bubble nucleation of the melt will be changed. If the initial hydrogen concentration is smaller than the later one, the bubble-free layer will be formed. The diffusion coefficient of hydrogen in the liquid metal has an essential impact on the accumulation rate of hydrogen at the solid-liquid interface and finally determines the thickness of the initial bubble-free layer at the bottom of the ingot.
By measuring the thickness of the initial bubble-free layer and combining the principle of unidirectional solidification of single-phase alloy, a new method of evaluating the diffusion coefficient of hydrogen in liquid metals was proposed. The predicted result for Cu-H2 system is comparable with literature values, which verifies the viability of this method. The values at the melting point and temperature-dependent equations for diffusion coefficients of hydrogen in liquid Mg, Si and Cu-34.6%Mn alloy were also predicted by this method. This method is easy to be carried out and solves the difficult problem of getting the diffusion coefficient of hydrogen in high-melting metals. Meanwhile, the obtained values through this method are much valuable for investigating the structure evolution of casting process, especially for the solid-gas eutectic unidirectional solidification.
2 Experiment and calculation
A simple mold casting technique was utilized for unidirectional solidification. The apparatus used is depicted in Fig. 1(a), and consists of a vacuum system that incorporated both the melting part and solidification part. To fabricate the ingot, high-purity (99.99%) material was first melted under vacuum. A gas mixture of hydrogen and argon was then introduced into the chamber, and the melt was held at a constant superheat degree until the hydrogen was uniformly dissolved in the melt (usually 10-90 min which was decided by melting method). Finally, the melt was poured into an insulated mold with a copper chiller, and then unidirectionally solidified upwards. This resulted in the formation of unique regular porous structure, which was known as a lotus-type porous structure owing to its resemblance to the roots of the lotus plant. This processing technique is a variation on the so-called solid-gas eutectic reaction, in which the melt is solidified into a solid solution and entrapped gas phase (L S+H2) [19]. Lotus-type porous Cu, Mg, Si and Cu-34.6%Mn alloy were fabricated by this method, respectively.
S+H2) [19]. Lotus-type porous Cu, Mg, Si and Cu-34.6%Mn alloy were fabricated by this method, respectively.
The ingot produced was sectioned along its central axis into two parts using a spark-erosion wire-cutting machine. The height (H) was determined as the average distance between the initial nucleation bubbles and the bottom of the ingot, as shown in Figs. 1(b) and (c).
According to the Sievert��s law, the original hydrogen concentration (C0) in liquid metal can be expressed as
 (3)
(3)
where ��(T) is the solubility coefficient; T, Tm and ��T are the initial temperature, melting point and the superheat degree of the melt, respectively; A and B are the solubility constants of the corresponding metal; pH2 is the hydrogen partial pressure in the chamber.
At the beginning of unidirectional solidification, the value of C0 is less than the hydrogen concentration required for bubble nucleation. A single solid phase is therefore formed, and the excess hydrogen is expelled into the liquid phase. This leads to a continuous increase in the hydrogen concentration of the liquid phase ahead of the solid-liquid interface (CL(x��)), as shown in Fig. 2. When CL* is sufficiently increased, an abundant number of bubbles are nucleated and eventually grow into the elongated pores depicted in Fig. 1(c).

Fig. 1 Schematic of solid-gas eutectic unidirectional solidification apparatus (a), formation of initial bubble-free layer (b) and evolution of lotus-type porous structure (c)
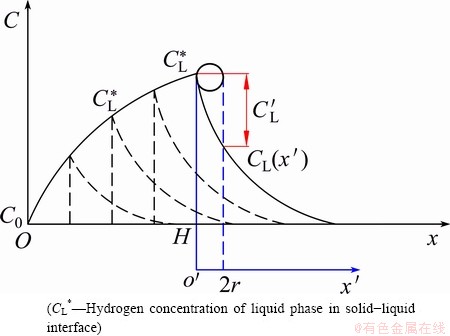
Fig. 2 Distribution of hydrogen in liquid ahead of solid-liquid interface during solidification of initial bubble-free layer
According to the principle of unidirectional solidification of single-phase alloy, the formation of the initial bubble-free layer corresponds to the initial growth stage. As the diffusion coefficient of hydrogen in liquid phase is generally three orders of that in solid [14], only the diffusion of hydrogen in the liquid phase needs to be considered. Therefore, regardless of the convection in the melt, CL* can be expressed as [14]
 (4)
(4)
where k0 is the distribution coefficient of hydrogen; v is the solidification velocity; DL is the diffusion coefficient of hydrogen in the liquid phase; x is the height of the solid phase. This demonstrates that there is a positive correlation between v and CL*; while a negative correlation exists between DL and CL*.
The investigation of bubble nucleation in Gasar solidification [20,21] revealed that heterogeneous nucleation of impurities (such as alumina, silicon oxide) in the pits and cracks was the most likely mechanism when CL* satisfied the following conditions:
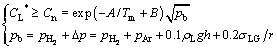 (5)
(5)
where pb is the gas pressure inside the bubble, from which the concentration of hydrogen for bubble nucleation (Cn) is determined; ��p is the additional pressure provided by the argon partial pressure (pAr) in the chamber, the hydrostatic pressure (ps) of the melt above the bubble and the capillary pressure (pc); ��L is the density; ��LG is the surface tension of the melt; g is the gravitational acceleration; h is the height of the melt above the bubble; r is the average radius of the initial nucleation bubbles.
Theoretically, bubbles will start to nucleate once CL* is equal to Cn and the corresponding value of x in Eq. (4) is exactly the height of the initial bubble-free layer (H). However, the solid-liquid interface is not simple as the gas bubbles grow into the melt with a convex shape, which is assumed to be spherical (radius of r) in this study. Consequently, the hydrogen concentration for bubble nucleation should be satisfied by CL��, which is an average value for the hydrogen concentration in the liquid bordered by a distance of 2r from the solid-liquid interface, as shown in Fig. 2.
The unidirectional solidification of single-phase alloy has previously been investigated by LI and LIU [22], who obtained a theoretical expression for the solute distribution in the liquid phase ahead of the solid-liquid interface during the initial growth stage:

 (6)
(6)
where x�� is the distance from the solid-liquid interface in the liquid phase. By substituting the height of the initial bubble-free layer (H) for x into Eq. (6), and integrating x�� in the range of 0 to 2r, CL�� can be obtained as follows:

 (7)
(7)
When CL�� is equal to Cn, the diffusion coefficient of hydrogen (DL) in the liquid phase can be calculated by solving Eqs. (3), (5), and (7). As the hydrogen concentration in the liquid phase attenuates exponentially with increasing distance from the interface, diffusion only needs to be considered within a short distance (x'��DL/v, which is called the characteristic distance). As the temperature of liquid phase changes slightly in this range, DL can be regarded as being equal to the value at the melting point of the metal in the liquid phase (i.e., DLm=DL).
According to the Arrhenius relation [9], both the self-diffusion and diffusion of solute impurities in the liquid metals can be given as
 (8)
(8)
where HD is the apparent activation energy or diffusion activation energy; Rg is the gas constant; and D0 is the pre-exponential factor, which can be calculated by substituting DLm into Eq. (8):
 (9)
(9)
LARSSON et al [23] have provided an empirical formula for calculating the value of HD for the self-diffusion of liquid metals and semiconductors:
 (10)
(10)
In the rigid sphere model, CHHABRA and ROY [13] believed that the diffusion coefficient of solute impurity in liquid metals was proportional to the self-diffusion coefficient of liquid metal itself and the proportionality coefficient was the ratio of atom diameters of metal and solute impurity. This means that HD only depends on the metal but nothing on the solute impurity with regards to both self-diffusion and the diffusion of solute impurity in liquid metals.
Thus, the temperature-dependent equations for the diffusion coefficient of hydrogen in liquid metals and semiconductors can be obtained by incorporating Eqs. (9) and (10) into Eq. (8):
 (11)
(11)
3 Results and discussion
In this section, a Cu-H2 system was chosen as the basis for a theoretical model, as lotus-type porous Cu had previously been fabricated by the solid-gas eutectic unidirectional solidification technique [17]. What��s more, several temperature-dependent equations for diffusion coefficient of hydrogen had already been reported which allowed for verification of the predicted result.

Fig. 3 Pressure conditions for formation of lotus-type porous structure and bubble-free layer with various superheat degrees of melt

Fig. 4 Longitudinal section views of porous Cu (a-c) for (a1-a3) respectively, Mg (d), Si (e) and Cu-34.6%Mn alloy (f)
Based on this Cu-H2 system, it was determined that in order to obtain an initial bubble-free layer in a lotus-type structure, the hydrogen concentration for bubble nucleation (Cn) must be greater than C0. This means that a certain amount of argon (pAr) is indispensable for the formation of bubble-free layer. However, a lotus-type structure cannot form if the amount of argon introduced is too great. The value of Cn must also satisfy the following expression [24]:
 (12)
(12)
According to Eqs. (3) and (5), Eq. (12) can be expressed by ignoring ps and pc, as both values are much smaller than pH2 and pAr:

 (13)
(13)
The pressure conditions required for the formation of the lotus-type structure and bubble-free layer are indicated by the shaded area shown in Fig. 3(a). With an increase in the superheat degree of the melt, both the maximum and minimum values for argon pressure are increased for a given hydrogen pressure. Three sets of parameters for the shaded area were therefore selected, as shown in Fig. 3. Longitudinal section views of the three ingots are shown in Fig. 4. The heights of the initial bubble-free layer (H) and the average radius of the initial nucleation bubbles (r) were obtained and listed in Table 1 for each sample.
According to the study by LIU [25], there is a relationship between the solidification velocity and pore diameter, which can be expressed as
 (14)
(14)
where ��LS is the interfacial energy between liquid and solid phases; �� is the porosity of the obtained ingot; TE (��Tm) is the eutectic temperature; ��Hm and ��S are the fusion enthalpy and density of the solid phase, respectively; F is a constant determined by the solid-gas system, melt temperature, and gas pressure in the chamber. By submitting the average radius of r and �� into Eq. (14), values of v are obtained, as shown in Table 1.
Using the equations outlined in Section 2, values for the diffusion coefficient of hydrogen in liquid Cu at the melting point were obtained under three different conditions, i.e., 4.4��10-3, 3.8��10-3 and 3.9��10-3 cm2/s.
Figure 4 shows that the largest value of H for the initial bubble-free layer exists when pAr is equal to 707 kPa and ��T equals 50 K. Reducing the pressure of argon allows the bubbles to nucleate at a lower concentration, while increasing the superheat degree of melt increases the initial concentration of hydrogen in the liquid phase, making both easier for bubbles to nucleate and reducing the distance of hydrogen enrichment required during solidification. The similarity in the diffusion coefficients obtained with different experimental parameters means that the error produced by this method is relatively small and acceptable. By submitting the average value of 4.0��10-3 cm2/s into Eq. (11), a temperature-dependent equation for the diffusion coefficient of hydrogen in liquid Cu is obtained:
 (15)
(15)
Figure 5 shows a comparison between the predicted results of DL,Cu and the literature values. From this, the data obtained by the traditional method are dispersed over a wide range due to the inevitable influence of convection and the inherent limitations of some methods. It has in fact been reported that variation in the published values of diffusion coefficients is between 50% and 100% for most metals [12]. Though the predicted results were somewhat greater than most of the literature values, it remained within an order of magnitude, and thus this method is considered reliable.
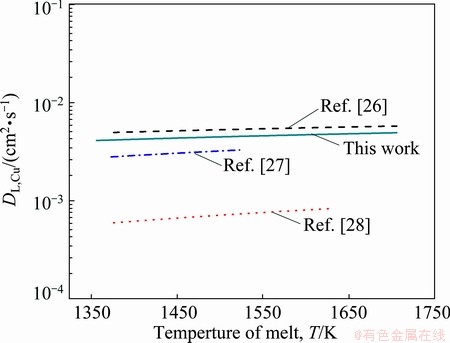
Fig. 5 Comparison between predicted results and literatures�� values of diffusion coefficients of hydrogen in liquid Cu
Lotus-type porous Mg, Si and Cu-34.6%Mn alloy were also fabricated under the conditions listed in Table 1. As with the Cu-H2 system, Figs. 3(b)-(d) and 4(d)-(f) show the determination of the experimental parameters based on the theoretical calculations and longitudinal section views, respectively. Finally, temperature- dependent equations for the diffusion coefficient of hydrogen in liquid Mg, Si and Cu-34.6%Mn alloy were obtained as follows:
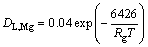 (16)
(16)
 (17)
(17)
 (18)
(18)
From this, the respective diffusion coefficients of hydrogen at the melting point for each metal are determined as1.75��10-2, 4.4��10-3 and 4.9��10-3 cm2/s. The physical property parameters used in the fabrication and calculation of the Cu-H2, Mg-H2, Si-H2 and Cu-34.6%Mn-H2 systems are shown in Table 2.
Table 1 Processing and obtained parameters of Cu-H2, Mg-H2, Si-H2 and Cu-34.6%Mn-H2 systems

Table 2 Physical property parameters of Cu-H2, Mg-H2, Si-H2 and Cu-34.6%Mn-H2 systems

4 Conclusions
1) A new method for evaluating the diffusion coefficient of hydrogen in liquid metals was proposed, by combining the solid-gas eutectic unidirectional solidification technique and the principle of unidirectional solidification of single-phase alloy.
2) For Cu-H2 system, the influence of argon pressure and superheat degree of melt on the diffusion coefficient of hydrogen in liquid phase is relatively small and acceptable. The obtained temperature-dependent equation for diffusion coefficient of hydrogen in liquid copper is comparable with experimental data in literature, thus validating the effectiveness of this method.
3) The temperature-dependent equations for diffusion coefficient of hydrogen in liquid Mg, Si and Cu-34.6%Mn alloy were evaluated, along with the values at the melting point of each metal and alloy.
References
[1] AKHTER J I, AHMED E, AHMAD M. Study of diffusion coefficients in liquid noble metals [J]. Materials Chemistry and Physics, 2005, 93(2-3): 504-507.
[2] KORKMAZ S D, KORKMAZ S. A study for structure and inter-diffusion coefficient of liquid K1-xC5x metal alloys [J]. Physics and Chemistry of Liquids, 2011, 49(6): 801-810.
[3] SUN Wei-hua, ZHANG Li-jun, WEI Ming, DU Yong, HUANG Bai-yun. Effect of liquid diffusion coefficients on microstructure evolution during solidification of Al356.1 alloy [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(12): 3722-3728.
[4] SEQUEIRA C A C, AMARAL L. Role of Kirkendall effect in diffusion processes in solids [J]. Transactions of Nonferrous Metals Society of China, 2014, 24(1): 1-11.
[5] MURAKAMI T, KOYAMA T. Application of electrochemical method to measure diffusion coefficient in liquid metal [J]. Journal of the Electrochemical Society, 2011, 158(8): F147-F153.
[6] IIDA T, GUTHRIE R I L. The physical properties of liquid metals [M]. XIAN Ai-ping, WANG Lian-wen. Beijing: Science Press, 2006: 219-249. (in Chinese)
[7] PROTOPAPAS P, ANDERSEN H C, PARLEE N A D. Theory of transport in liquid metals. I. Calculation of self-diffusion coefficients [J]. The Journal of Chemical Physics, 1973, 59(1): 15-25.
[8] SWALIN R A, LEAK V G. Diffusion of heterovalent solutes in liquid silver [J]. Acta Metallurgica, 1965, 13(5): 471-478.
[9] CAHOON J R. A modified ��Hole�� theory for solute impurity diffusion in liquid metals [J]. Metallurgical and Materials Transactions A, 1997, 28(3): 583-593.
[10] WAX J F, ALBAKI R, BRETONNET J L. Temperature dependence of the diffusion coefficient in liquid alkali metals [J]. Physical Review B, 2002, 65(1): 014301-1-9.
[11] LU H M, LI G, ZHU Y F, JIANG Q. Temperature dependence of self-diffusion coefficient in several liquid metals [J]. Journal of Non-crystalline Solids, 2006, 352(26-27): 2797-2800.
[12] LIU Yong, YANG Jian-gao. Cemented carbides of function alloy graded and other new structures [M]. Changsha: Central South University, 2010: 64-79. (in Chinese)
[13] CHHABRA R P, ROY A K. Diffusivity of oxygen and nitrogen in liquid metals: An explicit formulation [J]. Zeitschrift fur Metallkunde, 1988, 79(1): 64-67. (in Russian)
[14] LI Yan-xiang, WU Ai-ping. Principle of materials processing [M]. Beijing: Tsinghua University Press, 2005: 81-97. (in Chinese)
[15] WANG Shu-jie. Effect of liquid state hydrogenation on the microstructure and mechanical properties of Ti600 and Tc21 alloys [D]. Harbin: School of Materials Science and Engineering, Harbin Institute of Technology, 2008: 23-55. (in Chinese)
[16] LIU Yuan, LI Yan-xiang, WAN Jiang, ZHANG Hua-wei. Metal-gas eutectic growth during unidirectional solidification [J]. Metallurgical and Materials Transactions A, 2006, 37(9): 2871-2878.
[17] SHAPOVALOV V I. Method for manufacture of porous articles: USA, 5181549 [P]. 1993-01-26.
[18] JIANG G R, LI Y X, LIU Y. Influence of solidification mode on pore structure of directionally solidified porous Cu-Mn alloy [J]. Transactions of Nonferrous Metals Society of China, 2011, 21(1): 88-95.
[19] SHAPOVALOV V I, BOYKO L. Gasar��A new class of porous materials [J]. Advanced Engineering Materials, 2004, 6(6): 407-410.
[20] LIU Yuan, LI Yan-xiang. Theoretical analysis of bubble nucleation in GASAR materials [J]. Transactions of Nonferrous Metals Society of China, 2003, 13(4): 830-834.
[21] LIU Yuan, LI Yan-xiang, ZHANG Hua-wei. Fabrication of lotus-structured porous magnesium with gasar process [J]. Acta Metallurgica Sinica, 2004, 40(11): 1121-1126. (in Chinese)
[22] LI Yan-xiang, LIU Bai-cheng. Initial composition transient during crystal growth [J]. Acta Metallurgica Sinica B, 1988, 24(1): 12-16. (in Chinese)
[23] LARSSON S J, ROXBERGH C, LODDING A. Self-diffusion in liquid alkali metals [J]. Physics and Chemistry of Liquids, 1972, 3(3): 137-146.
[24] ZHANG Hua-wei. Theoretical and experimental study on unidirectional solidification of metal�Cgas eutectics [D]. Beijing: Department of Mechanical Engineering, Tsinghua University, 2006: 31-39. (in Chinese)
[25] LIU Yuan. Fundamental study on directional solidification of metal-gas eutectic for fabrication of lotus-structured porous metals [R]. Beijing: Department of Mechanical Engineering, Tsinghua University, 2003: 28-43. (in Chinese)
[26] SACRIS E M, PARLEE N A D. The diffusion of hydrogen in liquid Ni, Cu, Ag, and Sn [J]. Metallurgical Transactions, 1970, 1(12): 3377-3382.
[27] CHERNEGA D F, VASHCHENKO K I, IVANCHUK D F. Diffusion of H in molten Cu [J]. Izvest VUZ Tsvetnaya Met, 1973 (4): 120-122. (in Russian)
[28] SIGRIST P, FEICHTINGER H K, MARINCEK B. New stationary method for determination of gas-diffusion coefficients in metals and alloys [J]. Zeitschrift fuer Physikalische Chemie-Frankfurt, 1977, 107(2): 211-218. (in Russian)
[29] NAKAHATA T, NAKAJIMA H. Fabrication of lotus�Ctype porous silicon by unidirectional solidification in hydrogen [J]. Materials Science and Engineering A, 2004, 384(1-2): 373-376.
[30] ZENG X C, STROUD D. The solid-liquid interfacial tension of diamond-structure Si and Ge [J]. Journal of Physics. 1989, 1(9): 1779-1783.
[31] KOSTINA T K, BAUM B A, KUROCHKIN K T. Solubility of hydrogen in silicon [J]. Izvestiya Akademii Nauk SSSR, Neorganicheskie Materialy, 1970, 6(1): 117. (in Russian)
[32] JIANG Guang-rui. Study on hydrogen solubility in molten alloys and directional solidification of porous Cu-Mn alloy [D]. Beijing: Department of Mechanical Engineering, Tsinghua University, 2010: 69, 147. (in Chinese).
��ٻٻ1���� Դ1,2��������1,2
1. �廪��ѧ ����ѧԺ������ 100084��
2. �廪��ѧ �Ƚ���������������ص�ʵ���ң����� 100084
ժ Ҫ�����ڹ�-�������������̼����͵���Ͻ�Ķ�������ԭ�������һ�ֲⶨ���ڽ�����������ɢϵ���ķ�������Cu-H2ϵΪ�����о������ѹ��������ȶȶ����ڽ����������۵㴦��ɢϵ����Ӱ�졣�����ʾ����ͬ���ղ�������������õ���ɢϵ������С���Ҳ��ø÷�����õ����ڽ���Cu�������¶���ص���ɢϵ�������������в���ʵ�鷽����õĽ���ǺϵýϺã�����֤�˸÷����Ŀ����ԡ���Cu-H2ϵ�⣬�����������ڽ���Mg��Si�Լ�Cu-34.6%Mn�Ͻ��۵��¶������е���ɢϵ��ֵ�Լ��¶���ص���ɢϵ�����̡�
�ؼ��ʣ��⣻��ɢϵ����Һ̬��������-����������������
(Edited by Xiang-qun LI)
Foundation item: Project (51271096) supported by the National Natural Science Foundation of China; Project (NCET-12-0310) supported by Program for New Century Excellent Talents in University, China
Corresponding author: Yuan LIU; Tel: +86-10-62789328; E-mail: yuanliu@tsinghua.edu.cn
DOI: 10.1016/S1003-6326(14)63565-2

