��п����ֱ�ӽ�������ѧ����뾭��ģ��
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2017���10��
�������ߣ�Nima SADEGHI Javad MOGHADDAM Mehdi OJAGHI ILKHCHI
����ҳ�룺2272 - 2281
�ؼ��ʣ�����ѧ��ֱ�ӽ�������п��������ģ�ͣ�����
Key words��kinetics; direct leaching; sphalerite; shrinking core model (SCM); pilot plant
ժ Ҫ�������Թ�ģ��ʽ��Ӧ����ֱ�ӽ���ƶ����п�����о����������ѧ��Ϊ���㾲ѹ����������������������������Ŀ�ע�뵽��ֱ���õ�ֱ��8 m�Ĺ�ʽ��Ӧ���У������ӷ�Ӧ���ĵײ����롣�����˳�ʼ����Ũ�ȡ���Ӧ�¶ȡ�����ֱ������ʼ����пŨ�ȡ���Ũ�Ⱥ��������Է�Ӧ����ѧ��Ӱ�졣�����������п�����ֱ�ӽ�����ѭ������ģ�ͣ������ܻ�ѧ��Ӧ���ƣ�����ۻ��Ϊ49.7 kJ/mol�����ð뾭��ģ�������ù��̣��õ�������������Ũ�ȡ���п����Ũ�ȺͿ���ֱ���ķ�Ӧ�����ֱ�Ϊ0.982��0.189��-0.097��-0.992������SEM-EDS����п���������Ӧǰ��Ŀ������з��������ֵ�п�Ľ����ʵ���60%ʱ�����ڷ�Ӧ����������⣬�����������������䣬��Ӱ�������Һ���������ĽӴ���
Abstract: The direct leaching kinetics of an iron-poor zinc sulfide concentrate in the tubular reactor was examined. All tests were carried out in the pilot plant. To allow the execution of hydrostatic pressure condition, the slurry with ferrous sulfate and sulfuric acid solution was filled into a vertical tube (9 m in height) and air was blown from the bottom of the reactor. The effects of initial acid concentration, temperature, particle size, initial zinc sulfate concentration, pulp density and the concentration of Fe on the leaching kinetics were investigated. Results of the kinetic analysis indicate that direct leaching of zinc sulfide concentrate follows shrinking core model (SCM). This process was controlled by a chemical reaction with the apparent activation energy of 49.7 kJ/mol. Furthermore, a semi-empirical equation is obtained, showing that the order of the iron, sulfuric acid and zinc sulfate concentrations and particle radius are 0.982, 0.189, -0.097 and -0.992, respectively. Analysis of the unreacted and reacted sulfide particles by SEM-EDS shows that insensitive agitation in the reactor causes detachment of the sulfur layer from the particles surface in lower than 60% Zn conversion and lixiviant in the face with sphalerite particles.
Trans. Nonferrous Met. Soc. China 27(2017) 2272-2281
Nima SADEGHI1, Javad MOGHADDAM2, Mehdi OJAGHI ILKHCHI1
1. Faculty of Materials Engineering, Sahand University of Technology, Tabriz, Iran;
2. Materials and Metallurgical Department, University of Zanjan, Zanjan, Iran
Received 11 August 2016; accepted 30 September 2017
Abstract: The direct leaching kinetics of an iron-poor zinc sulfide concentrate in the tubular reactor was examined. All tests were carried out in the pilot plant. To allow the execution of hydrostatic pressure condition, the slurry with ferrous sulfate and sulfuric acid solution was filled into a vertical tube (9 m in height) and air was blown from the bottom of the reactor. The effects of initial acid concentration, temperature, particle size, initial zinc sulfate concentration, pulp density and the concentration of Fe on the leaching kinetics were investigated. Results of the kinetic analysis indicate that direct leaching of zinc sulfide concentrate follows shrinking core model (SCM). This process was controlled by a chemical reaction with the apparent activation energy of 49.7 kJ/mol. Furthermore, a semi-empirical equation is obtained, showing that the order of the iron, sulfuric acid and zinc sulfate concentrations and particle radius are 0.982, 0.189, -0.097 and -0.992, respectively. Analysis of the unreacted and reacted sulfide particles by SEM-EDS shows that insensitive agitation in the reactor causes detachment of the sulfur layer from the particles surface in lower than 60% Zn conversion and lixiviant in the face with sphalerite particles.
Key words: kinetics; direct leaching; sphalerite; shrinking core model (SCM); pilot plant
1 Introduction
Leaching of sphalerite was, usually, carried out under pressure and oxygen plays a key role in zinc dissolution process [1,2]. Also, atmospheric leaching of sphalerite has been studied by some researchers [3-8] and it has been shown that added ferric ions or other oxidizing agents (in solution) usually oxidize sphalerite in atmospheric media. From these methods, an integrated method has, also, been proposed where oxygen gas is used to sphalerite leaching in the atmospheric media [9-11]. In this case, ferrous ionic species are oxidized by oxygen blowing in the reactor. The generated ferric ions are reduced to ferrous ion by sphalerite:
Fe2(SO4)3(aq)+ ZnS(s)��ZnSO4(aq)+2FeSO4(aq)+S0(s) (1)
The ferrous ion (Fe2+) is re-oxidized in order to continue the leaching. Therefore, concentration of ferric ions, as an oxidant, could affect kinetic of zinc sulfide leaching. Oxygen blowing rate, particle size, temperature and acid concentration are other effective factors in direct leaching of sphalerite.
MARKUS et al [6] have investigated particle size variations during leaching process. The results confirmed the formation of the product layer on the surface particles. Moreover, other reports [1,8,12] confirmed shrinking core model (SCM) in atmospheric leaching that a sulfur layer is formed on solid particles. Three known control regimes in sulfide leaching kinetics are liquid film diffusion, solid product diffusion and chemical reaction controls. Several reports [8,13,14] have refused liquid film diffusion control, by determination of the processes activation energy (Ea). Furthermore, due to intense mixing in the reactor, sphalerite leaching may not be controlled by the liquid film diffusion.
On the other hand, a major feature of the kinetic system is the chemical reaction step and reacting mass transport coupled in series. In this case, the chemical reaction occurs in an interface between the original solid and the reaction product. If we assume that the zinc sulfide particles have a spherical geometry and the chemical reaction is the rate-controlling step, then it is expression of the shrinking core model to describe the dissolution kinetics of the process as follows:
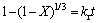 ,
,
 (2)
(2)
When the diffusion of lixiviant (such as Fe3+) through the elemental sulfur layer is the rate-controlling step, the following expression can be described as kinetic model:
 ,
,
 (3)
(3)
where X is the fraction reacted and kr and kd are the apparent rate constants for chemical reaction control and product diffusion in the product, respectively. Also, kc is the chemical reaction rate constant, CA is the concentration of the lixiviant in the solution, b is stoichiometric coefficient of the reactant in the leaching reaction, r0 is the initial radius of the solid particle, n is the order of reaction with respect to CA and D is the diffusion coefficient in the product layer. If the chemical reaction on the surface or diffusion through the product layer controls the leaching rate, there must be a linear relation among the left side of Eq. (2) or Eq. (3) and time.
Results of the investigation on determination of kinetic mechanism of sphalerite leaching were totally different. DUTRIZAC [14] and PECINA et al [8] indicated that the kinetics of sphalerite dissolution in ferric sulfate media is controlled by a chemical reaction on the surface of the particles. Also, XIE et al [1] mentioned that the interface chemical reaction is the controlling step in the pressure leaching.
On the other hand, some authors [7,15] have reported that the kinetics could be controlled by a non-reaction mechanism. An investigation [16] reported that both phenomenon of diffusion in product layer and the reaction between the particle surface and the product layer play a key role in controlling reaction rate. Generally, the kinetic model of zinc sulfide leaching is affected by different factors, such as agitation rate, and minerals composition in ore [17].
Even though all these studies provide valuable background on sphalerite dissolution kinetics, their results do not cover the dissolution kinetics during direct atmospheric leaching process. Moreover, studies about direct leaching of sphalerite [18,19] were carried out at bench scale, while the effect of various factors on direct leaching kinetics should be surveyed at the larger scale.
The aim of this work is to investigate effects of kinetic characteristics (such as kinetic control regime, activation energy of the dissolution and reaction order of different parameters) on direct leaching of zinc sulfide concentrate. All tests were performed in tubular reactor and pilot scale and then the collected data were analyzed by SCM. Influences of the particle size, acid concentration, zinc sulfate concentration, temperature, and solid to liquid (S/L) ratio were also studied on sphalerite direct saturation kinetics. The activation energy of the dissolution process was evaluated and semi-empirical rate equation was presented based on the experimental and calculated results.
2 Experimental
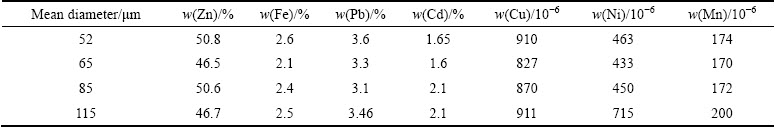
The zinc concentrate enriched through the flotation of Angouran mine sulfide ore in the Aria flotation plant (Zanjan, Iran) was used in this work. First, sulfide concentrate was ground in rod mill and then it was sieved to obtain the particle size distribution. Mean particle size and chemical compositions of the concentrate were determined by LPSA (laser particle size analyzer) and atomic absorption spectroscopy, respectively. The crystalline ferrous sulfate and sulfuric acid of industrial grade were used to make up all needed solutions. Analytical grade chemicals were used for solution analysis. Results of sulfide concentrate analysis can be seen in Table 1. The low content of iron is remarkable, which can influence zinc dissolution rate.
Table 1 Chemical composition of different fractions of complex concentrate
The contents of the metallic ions (Zn, Fe) in the sample solutions obtained from the leaching experiments were analyzed by atomic absorption spectroscopy (Varian, AA240). The ferrous and ferric ion concentrations were analyzed by titration of cerium (IV) sulfate with Phenanthroline indicator and EDTA with the salicylic acid indicator, respectively. The sulfuric acid concentration was determined by titration of sodium hydroxide and Methyl yellow indicator.
Leaching experiments were carried out in a tube reactor equipped with a thermometer, air inlet at bottom of tube, indirect steam coil, pump and a prime tank for making solution. It should be noted that for ideal agitation, slurry was circulated from bottom to top of the tube. Volume of the reactor, slurry height, rate of circulation of pulp and rate of air blowing were 0.44 m3, 9 m, 80 L/min and 180 L/min, respectively. First, the solution was prepared in the prime tank with acidity, determined iron concentration and desired temperature. After preparing slurry in the main tank, the slurry was pumped into the tubular reactor and the first sample was taken at that time.
3 Results and discussion
3.1 Direct leaching curve trends
Figure 1 presents zinc extraction for 0.2 mol/L Fe, 52 ��m mean diameters of particles and 30 g/L solid to liquid ratio at 0.25 and 0.5 mol/L H2SO4. There is a linear relationship between zinc extraction and time in initial period, but when zinc extraction is increased by more than 65%, the dissolution rate is reduced in both curves, obviously. BUBAN et al [20] observed similar results in direct leaching experiments, which were carried out in the sulfate solution. SOUZA et al [7] and WEISENER et al [21] attributed the decrease in zinc dissolution rate to the formation of an elemental sulfur layer. However, a different approach was used by DUTRIZAC and MACDONALD [22] and XU et al [23], and they reported that the produced quantity of elemental sulfur is too low so that the diffusion resistance is small. So, a comprehensive study should be carried out in direct leaching sphalerite to understand kinetics of the zinc dissolution and other product formation.
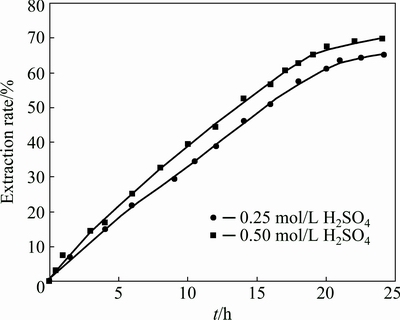
Fig. 1 Zn extraction rates for 0.2 mol/L Fe, 52 mm mean size of particles
3.2 Kinetics model
Two kinetic regimes were studied in leaching process. Equations (2) and (3) can be used to describe the leaching of zinc when only one step, chemical reaction [12,14,22,24] or diffusion through the reaction product [4,8], controls the entire process. However, some investigations [7,25] confirmed both equations. When the chemical reaction is first-order with respect to lixiviant (such as Fe3+) concentration, linear regression analysis is used for evaluation of each model (Eqs. (2) and (3)). The closer the correlation coefficient (R2) to 1, the better the regression line will be, which fits the data in this way.
The correlation coefficients for 0.2 and 0.4 mol/L Fe were calculated from above equations in Fig. 2. Findings of the study revealed that the values for correlation coefficient (R2) of chemical reaction control are closer to 1 than diffusion control regime. This result is in balance with findings observed in DUTRIZAC��s study where they determined that sphalerite leaching by ferric sulfate is controlled by chemical reaction.
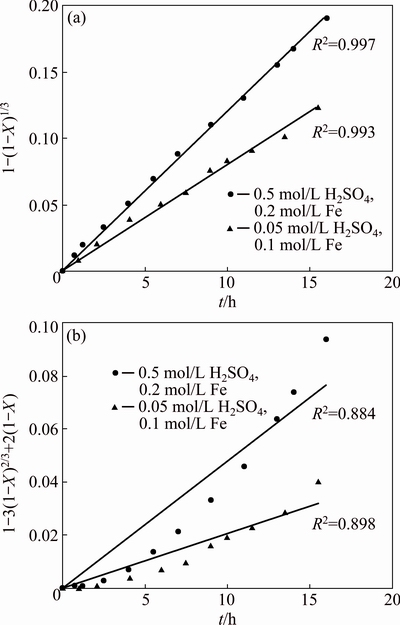
Fig. 2 Comparison of shrinkage core model for chemical reaction control (a) and diffusion control (b)
3.2.1 Influence of initial iron concentration
Direct leaching reaction involves ferric ions (Eq. (1)) directly, which is regenerated by ferrous ions and oxygen gas reaction, and it would be expected that the ferric sulfate concentration would be an important parameter in the process. According to the Fe3+ analysis, in constant sulfuric acid concentration, there is a direct relationship between Fe3+ and total iron concentration in the solution. Hence, the study of the iron concentration effect can be useful on the zinc sulfide dissolution rate.
For the reactions at 87 ��C, 0.15 g/L sulfuric acid and 30 g/L solid to liquid ratio, apparent rate constants (kr) were deduced from the slopes of the individual 1-(1-X)1/3 versus time straight lines, and these rate constants are displayed as a function of the iron concentration in Fig. 3. The plot from both ln kr and ln[Fe] values was used to determine the order of a reaction with respect to the Fe concentration. As it is seen in Fig. 4, the order of the iron concentration is 0.982, being close to 1 and similar to those values available for the oxidative leaching [26,27].
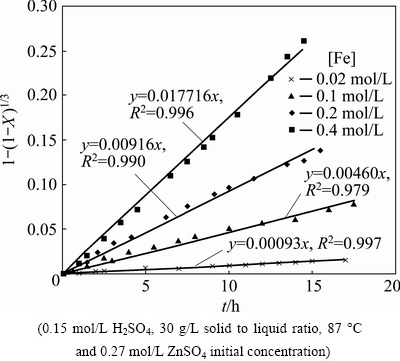
Fig. 3 Kinetic plot of surface chemical reaction for dissolution of zinc sulfide concentrate in different concentrations of ferrous sulfate
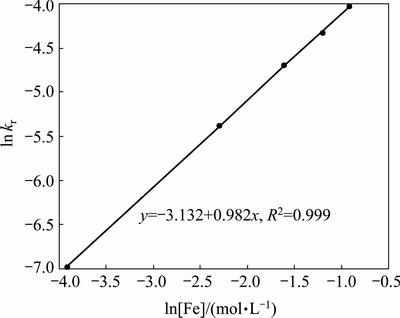
Fig. 4 ln kr vs ln[Fe] plot
On the other hand, the reaction order determined by AYDOGAN [28], DUTRIZAC [14], DUTRIZAC and MACDONALD [22] and SOUZA et al [7] is approximately 0.50. The main reason for such disagreement may be due to the difference in the ferric sulfate sources in these studies. In the previous studies [7,14], the ferric sulfate was added to the solution at the beginning of the reaction and Fe3+ concentration was decreased by ZnS oxidation, while the ferric ions are regenerated dynamically by Fe2+ oxidation in the tubular reactor.
3.2.2 Influence of H2SO4 concentration
The effect of H2SO4 concentration on the dissolution rate of 55 g/L of sphalerite was surveyed at 87 ��C in 0.2 mol/L Fe solution. The apparent rate constants (kr) were calculated from the straight line slopes of 1-(1-X) 1/3 versus time curves (Fig. 5(a)). The apparent rate constant is the function of H2SO4 concentration of the solution. As acid concentration increases, the zinc sulfide leaching rate increases, accordingly, from about kr=0.0078 at 0.05 mol/L H2SO4 to kr=0.0120 at 0.5 mol/L H2SO4. Moreover, the reaction order with respect to H2SO4 concentration was calculated by slope line of ln kr versus ln[H2SO4] (Fig. 5(b)). As can be seen, the reaction order of H2SO4 is 0.189.
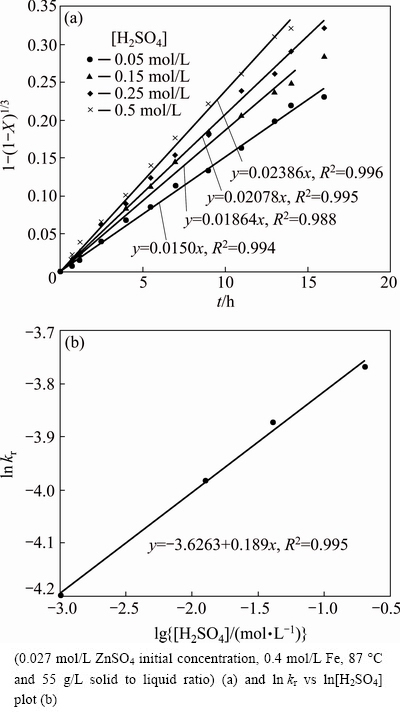
Fig. 5 Kinetic plot of surface chemical reaction for dissolution of zinc sulfide concentrate at different concentrations of H2SO4
CRUNDWELL [12] and SOUZA et al [7] have reported conflicting results for the reaction order with respect to sulfuric acid concentration. They mentioned that the reaction order was around 1, while DUTRIZAC [14] reported that the reaction rate increases gradually for more than 0.1 mol/L sulfuric acid concentrations. These observations were attributed to direct acid attack reaction increasing in zinc sulfide leaching process. However, LAMPINEN et al [19] reported that the sulfuric acid concentration has no significant influence on the reaction rate of the direct leaching of zinc, but they did not present any reaction order for H2SO4 concentration.
In addition to the acid attack reaction, the sulfuric acid has a desirable effect on Fe2+ oxidation reaction rate [29] that can increase zinc dissolution rate, consequently. Thus, the sulfuric acid concentration would rather increase zinc leaching and Fe2+ oxidation rates in the tubular reactor, simultaneously.
3.2.3 Influence of mean particle size
Different sizes of sphalerite were leached at 87 ��C in 0.4 mol/L FeSO4-0.25 mol/L H2SO4 to assess the effect of grinding on the dissolution rate of zinc, and the leaching kinetic curves under chemical control regime are presented in Fig. 6(a). The kinetic constant (kr) changes from 0.02412 to 0.1090 when the mean size of the particle increases from 52 to 115 mm. It is clear that the rate increases, significantly, as the particle size of sphalerite decreases. A decrease in particle size causes an increase in the solid��s total surface area. Also, the size of particles with lower iron content could have a greater impact on dissolution rate due to lower reactivity of sphalerite [7,30]. de LOS SANTOS et al [30] have reported that the presence of a catalytic surface of pyrite with sphalerite favors the electronic transfer of sphalerite to pyrite in the ferric sulfate solution. So, the catalytic and galvanic effects of pyrite decrease particle size effects in the zinc extraction of sphalerite.
lnkr versus lnr0 plot is presented in Fig. 6(b). As it can be seen, the slope of the straight line in the plot shows that the order of reaction with respect to the initial radius of the particle is close to -1. This value confirmed surface reaction control in SCM that kr varies with the inverse of the initial particle radius (kr��1/r0). The result is in agreement with the results obtained by DUTRIZAC [14] and XIE et al [1] .
3.2.4 Influence of temperature
Figure 7 illustrates the 1-(1-X)1/3 versus time curves for the dissolution of zinc when Angouran zinc sulfide concentrate (65 mm) was leached at various temperatures (70, 80, 87 and 95 ��C) in 0.4 mol/L FeSO4-0.15 mol/L H2SO4 solutions. The dissolution curves are essentially linear at 70-95 ��C and the rate constants (kr) were calculated from slopes of the straight lines. The reaction rate dependence on the temperature follows the Arrhenius equation [31].
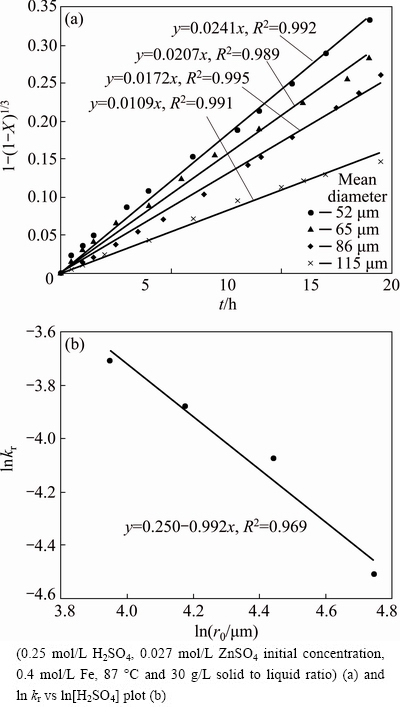
Fig. 6 Kinetic plot of surface chemical reaction for dissolution of zinc sulfide concentrate with different mean particle sizes
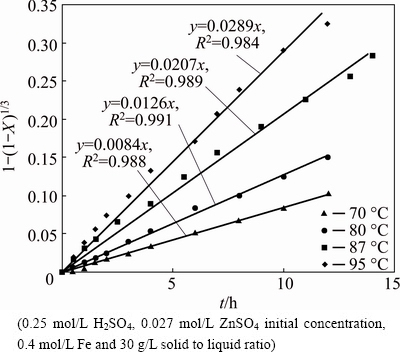
Fig. 7 Kinetic plot of surface chemical reaction for dissolution of zinc sulfide concentrate at different temperatures
By plotting the natural logarithm of the rate constant (ln k) against the reciprocal of the reaction temperature (1/T), the Arrhenius plot is shown in Fig. 8. The slope of this plot (-E/R) is -5.978 and the apparent activation energy for sphalerite direct leaching is obtained as 49.70 kJ/mol by multiplying gas constant. Similar results were reported by other authors [1,22,25], who calculated in SCM with chemical control regime.
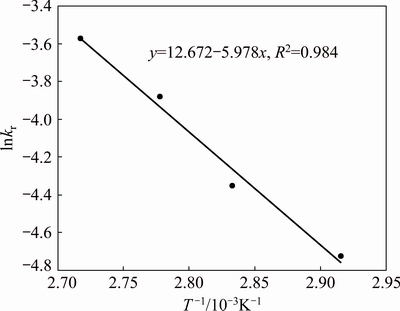
Fig. 8 Arrhenius plot of reaction rate against reciprocal temperature
3.2.5 Influence of ZnSO4 initial concentration and pulp density
In any commercial leaching process, the electrowinning output could be restored to the leaching reactors, which may lead to higher initial concentration of zinc in the solution. Increasing ZnSO4 initial concentration in the solution could affect Fe2+ re-oxidation and zinc dissolution, simultaneously [14,19]. Accordingly, a series of tests were done at 87 ��C where sphalerite was leached using 0.4 mol/L FeSO4-0.15 mol/L H2SO4 solutions, to which different initial concentrations of ZnSO4 were added. As can be seen in Fig. 9, the kinetic constant (kr) was calculated from straight line slope of 1-(1-X)1/3 versus time which was achieved at 0.02403, 0.01902, 0.01650 for 0.027, 0.5 and 1 mol/L initial zinc concentration, respectively. It is clear that ZnSO4 species has a damaging effect on the dissolution rate of direct leaching and this should be considered in the design of industrial processes.
Also, to survey the effect of the pulp density on the rate of sphalerite dissolution, rate constants were calculated from kinetic curves and the calculated rate constants are displayed in Fig. 10. Various masses (30, 55 and 80 g/L) of the sphalerite were leached at 87 ��C in 0.4 mol/L FeSO4-0.15 mol/L H2SO4 solution. The kinetic constant (kr) was reduced by increasing solid to liquid ratio, significantly. Based on Eq. (1), change in pulp density changes reactant (Fe3+ and ZnS) ratio which influences reaction rate. Also, increasing pulp density affects the fluid flow through solid particles and mass transfer of lixiviant around the particle is decreased, accordingly [32].
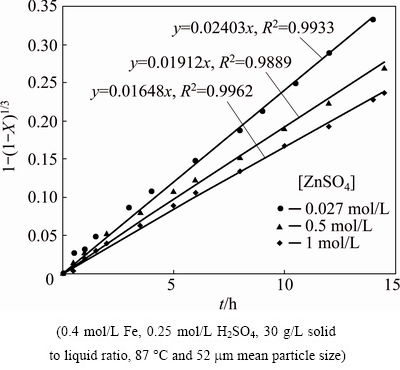
Fig. 9 Kinetic plot of surface chemical reaction for dissolution of zinc sulfide concentrate in different ZnSO4 initial concentrations
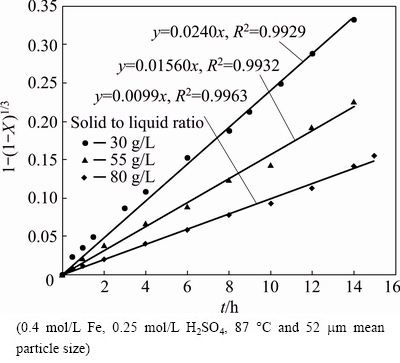
Fig. 10 Kinetic plot of surface chemical reaction for dissolution of zinc sulfide concentrate in different solid to liquid ratios
The reaction orders related to zinc concentration and solid to liquid ratio are determined in Figs. 11(a) and (b), respectively. In Fig. 11(a), the order of the initial zinc sulfate concentration is 0.097 that shows increase in the initial concentration of zinc sulfate changes the apparent rate constant, slightly. The order of pulp density is calculated to be 0.886, which implies significant impact of the factor on dissolution rate.
3.2.6 Mathematical modeling of sphalerite direct leaching
The apparent rate constant (kr) for the surface chemical reaction was calculated from slopes of the straight lines in the previous sections. The slope of the line in each plot presents the calculated order of dissolution rate with respect to the related parameter for a chemical reaction controlled process. Accordingly, the orders of the reaction related to iron concentration, sulfuric acid concentration, zinc sulfate concentration and initial radius of the particle are 0.982, 0.189, 0.097 and 0.992, respectively. Also, the apparent activation energy is obtained to be 49.70 kJ/mol. Hence, a mathematical model for direct leaching of zinc sulfide concentrate is as follows:
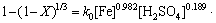
 (7)
(7)
According to the value of activation energy and the inverse relationship between kr and initial particle radius, chemical reaction was stabilized as control regime. Furthermore, plotting the rate constant )kr) values versus the right side expressions of Eq. (7) gives a k0 value of 29.131, 17.016, 12.204 for 30, 55 and 80 g/L of solid to liquid ratio, respectively.
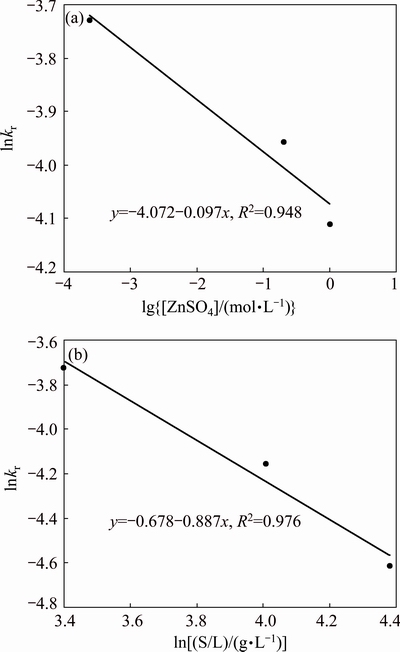
Fig. 11 ln kr vs ln[ZnSO4] plot (a) and ln kr vs ln[S/L] plot (b)
To test the accordance between the experimental values and the calculated values from the empirical equation, a plot of these values was drawn. As can be seen in Fig. 12, a good coincidence was achieved between the calculated and the experimental values.
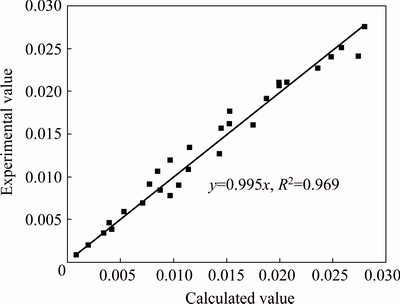
Fig. 12 Comparison of experimental and calculated data of sphalerite concentrate dissolution in direct leaching of zinc sulfide concentrate
3.3 Morphology of leaching residues
The morphology of the leaching residual was examined at different time intervals of the process by SEM-EDS (Fig. 13). The particle��s precipitation after 10%, 35% and 60% Zn conversion is presented in Fig. 13.
As can be seen in Fig. 13(a), the particle surface is approximately clear; however, the sulfur is dispersed on the particle. The sulfur particle is dispersed on the sphalerite surfaces in Fig. 13(b). In Fig. 13(c), the sulfur layer covers parts of the particles but the leach liquor is faced with the sphalerite particles. The morphology of the leach residue implies that major amount (higher than 60%) of zinc sulfide is dissolved in the solution without sulfur interference, whereas some studies [7,21] reported that sulfur covers particle surface after 35%-40% zinc extraction. The Fe3+ concentration analyses show that only 10% of iron concentration is oxidized to Fe3+ and the ferric ions attacks to the mineral surface, slowly. A thin sulfur layer is formed on the surface by the reaction of Fe3+ attacks. It seems that extreme turbulence in the present reactor causes the break-down of the thin sulfur layer on sphalerite surface and dispersion in the system.
SANTOS et al [33] considered that leaching residues showed no change in the sphalerite composition after the leaching by the electron microprobe analysis. In this regard, the elemental analysis result of this micrograph is presented in Table 2. According to Table 2, the content of sulfur increased on residue surface, while the zinc content was decreased in the zinc dissolution reaction progress. Furthermore, the XRD pattern of leaching residue is demonstrated in Fig. 14. The sulfur peaks are observed in the pattern, obviously, which confirms that the sulfide layer is formed on the surface.

Fig. 13 SEM photographs of zinc concentrate and leach residues
Table 2 EDS analysis result of residue
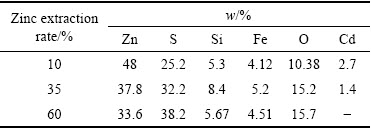
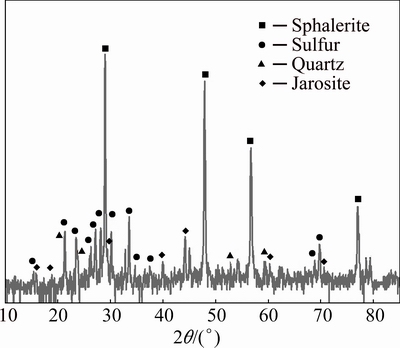
Fig. 14 XRD pattern of leaching residue
The direct leaching process has been designed in more than one stage to overcome sulfide barrier layer, as a possible problem [10,34,35]. The sulfur is removed from residue and the sulfur-free residue is recycled back into the leaching process. Present processes could reduce sulfide barrier layer and increase leaching rate in the first stage. So, the mass of residue for subsequent operations is reduced, obviously.
4 Conclusions
The kinetics of direct leaching of zinc sulfide concentrate was investigated in the tubular reactor (9 m in height) and pilot scale. Results of the present study reveals that the values of correlation coefficient (R2) for chemical reaction control process are closer to 1 than those for diffusion control. The dissolution rate increases when the iron concentration, in proportion to the amount of re-oxidized ferrous concentration, is increased and kinetic constant and iron concentration have a linear relationship with each other. As presented by chemical reaction control regime in SCM, the reaction rate is inversely proportional to the particle size. Also, the rate is increased with increasing temperature and the apparent activation energy is 49 kJ/mol. The high value (>40 kJ/mol) of the apparent activation energy and the inverse relationship between the kinetic apparent rate constant (kr) and particle size stabilize the chemical reaction as control step of leaching. SEM�CEDX analyses on residue confirm the shrinking core model, so that elemental sulfur covers some parts of particle surface.
Acknowledgments
The authors gratefully acknowledge the Zanjan Zinc Khalessazan Industries Company (ZZKICO) for the financial and technical support of this work.
References
[1] XIE Ke-qiang, YANG Xian-wan, WANG Ji-kun, YAN Jiang-feng, QING feng-shen. Kinetic study on pressure leaching of high iron sphalerite concentrate [J]. Transactions of Nonferrous Metals Society of China, 2007, 17(1): 187-194.
[2] DREISINGER D B, PETERS E. The oxidation of ferrous sulphate by molecular oxygen under zinc pressure-leach conditions [J]. Hydrometallurgy, 1989, 22(1-2): 101-119.
[3] DUTRIZAC J E. The leaching of sulphide minerals in chloride media [J]. Hydrometallurgy, 1992, 29(1): 1-45.
[4] BABU M N, SAHU K K, PANDEY B D. Zinc recovery from sphalerite concentrate by direct oxidative leaching with ammonium, sodium and potassium persulphates [J]. Hydrometallurgy, 2002, 64(2): 119-129.
[5] HAGHSHENAS D F, ALAMDARI E K, BONAKDARPOUR B, DARVISHI D, NASERNEJAD B. Kinetics of sphalerite bioleaching by Acidithiobacillus ferrooxidans [J]. Hydrometallurgy, 2009, 99(3): 202-208.
[6] MARKUS H, FUGLEBERG S, VALTAKARI D, SALMI T, MURZIN D Y, LAHTINEN M, Kinetic modelling of a solid-liquid reaction: Reduction of ferric iron to ferrous iron with zinc sulphide [J]. Chemical Engineering Science, 2004, 59(4): 919-930.
[7] SOUZA A D D, PINA P D S,  V A, SILVA C A D, SIQUEIRA P D F. The leaching kinetics of a zinc sulphide concentrate in acid ferric sulphate [J]. Hydrometallurgy, 2007, 89(1): 72-81.
V A, SILVA C A D, SIQUEIRA P D F. The leaching kinetics of a zinc sulphide concentrate in acid ferric sulphate [J]. Hydrometallurgy, 2007, 89(1): 72-81.
[8] PECINA T, FRANCO T, CASTILLO P, ORRANTIA E. Leaching of a zinc concentrate in H2SO4 solutions containing H2O2 and complexing agents [J]. Minerals Engineering, 2008, 21(1): 23-30.
[9] SVENS K, KERSTIENS B, RUNKEL M. Recent experiences with modern zinc processing technology [J]. Erzmetall, 2003, 56(2): 94-103.
[10] HAAKANA T, SAXTON B, LEHTINEN L, TAKALA H, LAHTINEN M, SVENS K, RUONALA M, GONGMING X. Outotec direct leaching application in China [J]. Journal of the Southern African Institute of Mining and Metallurgy, 2008, 108(5): 245-251.
[11] SVENS K, SVENS K R. Consulting incorporation. direct leaching alternatives for zinc concentrates [C]//Honorary Symposium on Hydrometallurgy, Electrometallurgy and Materials Characterization. Hoboken, PA: TMS, 2012: 191-206.
[12] CRUNDWELL F K. Kinetics and mechanism of the oxidative dissolution of a zinc sulphide concentrate in ferric sulphate solutions [J]. Hydrometallurgy, 1987, 19(2): 227-242.
[13] ESPIARI S, RASHCHI F, SADRNEZHAAD S K. Hydrometallurgical treatment of tailings with high zinc content [J]. Hydrometallurgy, 2006, 82(1): 54-62.
[14] DUTRIZAC J E. The dissolution of sphalerite in ferric sulfate media [J]. Metallurgical and Materials Transactions B, 2006, 37(2): 161-171.
[15] SAHU S K, SAHU K K, PANDEY B D. Leaching of zinc sulfide concentrate from the Ganesh-Himal Deposit of Nepal [J]. Metallurgical and Materials Transactions B, 2006, 37(4): 541-549.
[16] HASANI M, KOLEINI S M J, KHODADADI A. Kinetics of sphalerite leaching by sodium nitrate in sulfuric acid [J]. Journal of Mining and Environment, 2015, 7(1): 1-12.
[17] KOLEINI S M J, SABERIAN K, ABDOLAHI M, MEHRPOUYA H. Extraction of indium from zinc sulphide concentrates [J]. Asian Journal of Chemistry, 2009, 21(7): 5611-5620.
[18] HAAKANA T, LAHTINEN M, TAKALA H, RUONALA M, TURUNEN I. Development and modelling of a novel reactor for direct leaching of zinc sulphide concentrates [J]. Chemical Engineering Science, 2007, 62(18): 5648-5654.
[19] LAMPINEN M, LAARI A, TURUNEN I. Kinetic model for direct leaching of zinc sulfide concentrates at high slurry and solute concentration [J]. Hydrometallurgy, 2015, 153: 160-169.
[20] BUBAN K R, COLLINS M J, MASTERS I M, TRYTTEN L C. Comparison of direct pressure leaching with atmospheric leaching of zinc concentrates [C]//Lead-Zinc 2000. Pittsburgh, PA: TMS, 2000: 727-738.
[21] WEISENER C G, SMART R, GERSON A R. Kinetics and mechanisms of the leaching of low Fe sphalerite [J]. Geochimica et Cosmochimica Acta, 2003, 67(5): 823-830.
[22] DUTRIZAC J E, MACDONALD R J C. The dissolution of sphalerite in ferric chloride solutions [J]. Metallurgical Transactions B, 1978, 9(4): 543-551.
[23] XU Zhi-feng, JIANG Qing-zheng ,WANG Cheng-yan. Atmospheric oxygen-rich direct leaching behavior of zinc sulphide concentrate [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(12): 3770-3787.
[24] JIN Zuo-mei, WARREN G W, HENEIN H. Reaction kinetics of the ferric chloride leaching of sphalerite an experimental study [J]. Metallurgical Transactions B, 1984, 15(1): 5-12.
[25] BOBECK G E, SU H. The kinetics of dissolution of sphalerite in ferric chloride solution [J]. Metallurgical Transactions B, 1985, 16(3): 413-424.
[26] JIAN S, LING G A O, JIANQUN L I N, HONGBIN W U, JIANQIANG L I N. Kinetics and modeling of chemical leaching of sphalerite concentrate using ferric iron in a redox-controlled reactor [J]. Chinese Journal of Chemical Engineering, 2013, 21(8): 933-936.
[27] da SILVA G. Relative importance of diffusion and reaction control during the bacterial and ferric sulphate leaching of zinc sulphide [J]. Hydrometallurgy, 2004, 73(3): 313-324.
[28] AYDOGAN S. Dissolution kinetics of sphalerite with hydrogen peroxide in sulphuric acid medium [J]. Chemical Engineering Journal, 2006, 123(3): 65-70.
[29]  J, SALMI T, TURUNEN I, LUOMA M. Kinetics of oxidation of ferrous sulfate with molecular oxygen [J]. Chemical Engineering Science, 1999, 54(19): 4223-4232.
J, SALMI T, TURUNEN I, LUOMA M. Kinetics of oxidation of ferrous sulfate with molecular oxygen [J]. Chemical Engineering Science, 1999, 54(19): 4223-4232.
[30] ESTRADA-de LOS SANTOS F, RIVERA-SANTILLAN R E, TALAVERA-ORTEGA M, BAUTISTA F. Catalytic and galvanic effects of pyrite on ferric leaching of sphalerite [J]. Hydrometallurgy, 2016, 163: 167-175.
[31] LEVENSPIEL O. Chemical reaction engineering [M]. 3rd edition. Wiley, 1999.
[32] APPA H, DEGLON D A, MEYER C J. Numerical modelling of mass transfer in an autoclave [J]. Hydrometallurgy, 2014, 147: 234-240.
[33] SANTOS  M C, MACHADO R M, CORREIA M J N, REIS M T A, ISMAEL M R C. CARVALHO J M R. Ferric sulphate/ chloride leaching of zinc and minor elements from a sphalerite concentrate [J]. Minerals Engineering, 2010, 23(8): 606-615.
M C, MACHADO R M, CORREIA M J N, REIS M T A, ISMAEL M R C. CARVALHO J M R. Ferric sulphate/ chloride leaching of zinc and minor elements from a sphalerite concentrate [J]. Minerals Engineering, 2010, 23(8): 606-615.
[34] HALFYARD J E, HAWBOLDT K. Separation of elemental sulfur from hydrometallurgical residue: A review [J]. Hydrometallurgy, 2011, 109(1): 80-89.
[35] LI Hai-long, WU Xian-ying, WANG Ming-xia, WANG Jun, WU Shao-kang, YAO Xiao-long, LI Li-qing. Separation of elemental sulfur from zinc concentrate direct leaching residue by vacuum distillation [J]. Separation and Purification Technology, 2015, 138: 41-46.
Nima SADEGHI1, Javad MOGHADDAM2, Mehdi OJAGHI ILKHCHI1
1. Faculty of Materials Engineering, Sahand University of Technology, Tabriz, Iran;
2. Materials and Metallurgical Department, University of Zanjan, Zanjan, Iran
ժ Ҫ�������Թ�ģ��ʽ��Ӧ����ֱ�ӽ���ƶ����п�����о����������ѧ��Ϊ���㾲ѹ����������������������������Ŀ�ע�뵽��ֱ���õ�ֱ��8 m�Ĺ�ʽ��Ӧ���У������ӷ�Ӧ���ĵײ����롣�����˳�ʼ����Ũ�ȡ���Ӧ�¶ȡ�����ֱ������ʼ����пŨ�ȡ���Ũ�Ⱥ��������Է�Ӧ����ѧ��Ӱ�졣�����������п�����ֱ�ӽ�����ѭ������ģ�ͣ������ܻ�ѧ��Ӧ���ƣ�����ۻ��Ϊ49.7 kJ/mol�����ð뾭��ģ�������ù��̣��õ�������������Ũ�ȡ���п����Ũ�ȺͿ���ֱ���ķ�Ӧ�����ֱ�Ϊ0.982��0.189��-0.097��-0.992������SEM-EDS����п���������Ӧǰ��Ŀ������з��������ֵ�п�Ľ����ʵ���60%ʱ�����ڷ�Ӧ����������⣬�����������������䣬��Ӱ�������Һ���������ĽӴ���
�ؼ��ʣ�����ѧ��ֱ�ӽ�������п��������ģ�ͣ�����
(Edited by Bing YANG)
Corresponding author: Javad MOGHADDAM; Tel: +98-243-3054364; Fax: +98-243-2383400; E-mail: moghaddam@znu.ac.ir, hastyir@yahoo.com
DOI: 10.1016/S1003-6326(17)60253-X

