
Evolution of intermetallic phases of Al-Zn-Mg-Cu alloy during heat treatment
FAN Xi-gang(樊喜刚)1, JIANG Da-ming(蒋大鸣)1, MENG Qing-chang(孟庆昌)1,
LI Nian-kui (李念奎)2, SUN Zhao-xia(孙兆霞)2
1. Analysis and Measurement Center, Harbin Institute of Technology, Harbin 150001, China;
2. Northeast Light Alloy Co Ltd, Harbin 150060, China
Received 28 July 2006; accepted 15 September 2006
Abstract: Al-Zn-Mg-Cu alloy is a favorable choice for aerospace applications requiring good combination of strength and toughness, which is greatly influenced by the coarse intermetallic particles. The evolution of intermetallic particles in an Al-Zn-Mg-Cu alloy during heat treatment was studied by field emission gun scanning electron microscopy (FEG-SEM) and X-ray diffractometry(XRD). The results show that there are lamellar eutectic structure (α(Al)+Mg(Zn,Al,Cu)2) and Al7Cu2Fe particles in the solidified structure. The Al7Cu2Fe particles are embedded in the eutectic structure. The content of eutectic structure decreases with the increase of holding time and disappears after 24 h. The size and morphology of Al7Cu2Fe particles exhibit no change during the heat treatment. It is found that the Al2CuMg phase is formed during the treatment at 460 ℃. A transformation process from the primary eutectic phase Mg(Zn,Al,Cu)2 to Al2CuMg is observed, and the transformation mechanism and kinetics are analyzed. The Al2CuMg constituents form in the primary Mg(Zn,Al,Cu)2 phase, and grow along the eutectic microstructure.
Key words: Al-Zn-Mg-Cu alloy; eutectic structure; intermetallic particles; phase transformation
1 Introduction
The Al-Zn-Mg-(Cu) alloys are widely used in the aeronautics industry for their specific strength. The copper, as one of the principal alloying elements, improves considerably the age-hardening potential and resistance to the corrosion cracking of the alloys[1-2]. However, coarse intermetallic particles (0.5-10 μm) such as Al7Cu2Fe and S (Al2CuMg) phases are present in the alloys, which consume a great amount of copper and lead to lower toughness[3-4]. The nature and detrimental effect of these coarse particles have been studied by HAHN et al[5-8].
The coarse Al7Cu2Fe particles generally formed during ingot solidification, and exhibited small change during subsequent heat treatment in the Al-Zn-Mg-Cu alloys. In the Al-Cu-Mg and 7055 (Al-8Zn-2Mg-2Cu) alloys, the coarse S phase was present in the as-cast materials, and dissolved into the matrix during the subsequent heat treatment with the changing of its composition[9-11]. The implication is that the S phase formed during the ingot solidification and was metastable during the heat treatment. The formation of the S phase during the solidification and the dissolving process in subsequent heat treatment were studied[12-13]. Despite detailed studies of the type of primary eutectic structure in the as cast and as homogenized alloys, less attention was focused on the evolution processing of eutectic structures during homogenization, and the transformation process of primary eutectic structures to residual coarse constitutes is not clear entirely.
In a previous study[14], it is found that the S phase are not present in the as cast structure in a 7150 alloy (Al-6Zn-2Mg-2Cu), and it is formed during the homogenization. In the present study, the nucleation and transformation process of the S phase during the homogenization were studied, and the evolution of newly formed S phase was analyzed.
2 Experimental
The 7150 alloy was prepared via semi-continuous casting method. The composition of the alloy is shown in Table 1. The as-cast ingots were 200 mm in diameter. Specimens were collected at the region of 50 mm from ingot surface. As-cast specimens were homogenized at 460 ℃ for various times then quenched into the water.
The microstructure was analyzed by optical microscopy and scanning electron microscopy(SEM). The specimens were ground and polished. For optical structure observation and examination, specimens were etched in Keller’s reagent. SEM observation was carried out on the S-4700 microscope. The specimens for SEM observation were polished before the test. The as-cast and homogenized specimens were analyzed by energy dispersive spectroscopy (EDS), X-ray diffractometry (XRD) and differential scanning calorimetry (DSC).
Table 1 Chemical composition of 7150 alloy (mass fraction, %)

3 Results and discussion
3.1 Evolution of primary particles
The primary particles in the as cast alloy are mainly Mg(Zn, Al, Cu)2 phase, including a small amount of Al7Cu2Fe phase. The Al7Cu2Fe phase is formed during solidification, and exhibits no change during the subsequent homogenization. The lamellar eutectic structures in the as-cast alloy are shown in Fig.1(a). The bright phase is Mg(Zn, Al, Cu)2, which is divided by the α(Al) (black area). The Mg(Zn, Al, Cu)2 is dissolved into the matrix during homogenization. From Fig. 1(b), it can be seen that the continuous Mg(Zn, Al, Cu)2 phase evolves into isolated particles and decreases in content after homogenization at 460 ℃ for 24 h. The residual particles in Fig.1(b) are S phase formed during the homogenization, which is supported by the XRD and DSC results as below. The size of the S is larger than that of the primary Mg(Zn, Al, Cu)2. The evolution of area fraction of the coarse particles during homogenization is shown in Fig.2. The fraction of coarse particles in the as cast alloy is about 5.6%, and it decreases quickly in the initial 12 h homogenization.
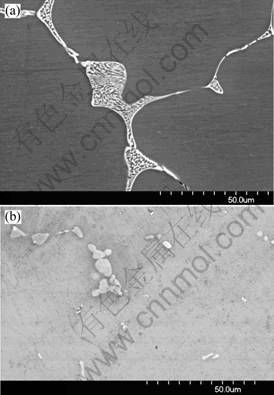
Fig.1 SEM images of evolution of primary particles: (a) As-cast material; (b) Homogenized at 460 ℃ for 24 h
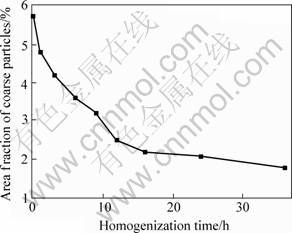
Fig.2 Evolution of area fraction of coarse particles during homogenization
The XRD analysis results for the phase evolution are shown in Fig. 3. From Fig.3, it can be seen that the Mg(Zn, Al, Cu)2 is present in the as-cast alloy, and the peaks about the S can not be observed. The peaks about the S can be observed after homogenization for 12 h, indicating the S is formed during the heat treatment. At the same time, the peaks about the Mg(Zn, Al, Cu)2 become weaker gradually, resulting from the decreasing of its fraction. It is easy to identify the existence of Al2CuMg from the DSC results, as shown in Fig.4. The peaks A and B are attributed to the melting of Mg(Zn, Al, Cu)2 and S, respectively. It is obviously that the S is not present in the as-cast alloy and formed during the homogenization. The Mg(Zn, Al, Cu)2 decreases gradually and disappears in the alloy after homogeniza-
tion for 24 h.
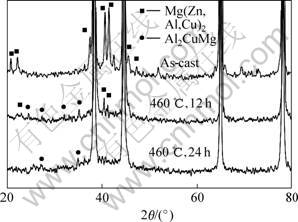
Fig.3 XRD patterns of phase evolution
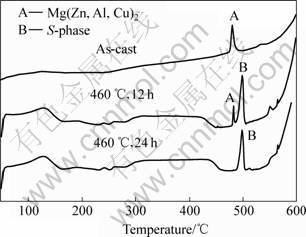
Fig.4 DSC results of phase evolution
3.2 Eutectoid transformation from Mg(Zn, Al, Cu)2 to S-phase
From Fig.5(b), it is found that the Mg(Zn, Al, Cu)2 is brighter than the S phase. So it is easy to analyze the evolution of Mg(Zn, Al, Cu)2 and the nucleation of S during the homogenization. The S phase nucleates at the boundary between the primary Mg(Zn, Al, Cu)2 phase and the matrix. This is due to the high free energy on the interface. After nucleation, the S phase grows into the bulk of the Mg(Zn, Al, Cu)2 particle. The newly formed S is conjunct with the primary Mg(Zn, Al, Cu)2 phase. It seems that the S is one part of the primary phase, suggesting that it is transformed from Mg(Zn, Al, Cu)2. From the phase diagram, the S(Al2CuMg) is present at 460 ℃ for 7150 alloy, but the Mg(Zn, Al, Cu)2 is not stable. Therefore, increasing the holding time at 460 ℃, the Mg(Zn, Al, Cu)2 particles resolve into the matrix, and some of these particles transform into the S phase.
The lamellar Mg(Zn, Al, Cu)2 phase resolves into the matrix and transforms into isolate particles during the homogenization, as shown in Fig.5. There is little Mg(Zn, Al, Cu)2 phase (bright phase) present in the alloy after homogenization for 12 h. At the same time, some of the Mg(Zn, Al, Cu)2 phase transform into the S. The fraction of the transformed S phase particles increases quickly with heating time(shown in Figs.3-4). The morphology of the newly formed S is consistent with that of primary Mg(Zn, Al, Cu)2 particles. With the growth of the S particles, they are exhibited in elliptic morphology. The number of the S particles is increased with the increase of the holding time within 12 h. After 12 h, the number is decreased gradually, and the size is increased with the homogenization, suggesting the coalescence of the S particles. Furthermore, the fraction of the S is decreased after 12 h(shown in Fig.2). This indicates that some of S particles dissolve into the matrix during the homogenization resulted from the diffusion of copper from the interdendritic regions to the inner regions of dendrite.
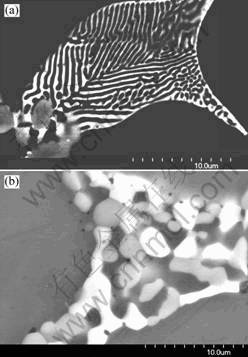
Fig. 5 Evolution of newly formed S-phase particles: (a) As-cast; (b) Homogenization for 12 h
In the as-cast alloy, the solute distributes heterogeneously within the grain. More solute is present in the primary particles and the α(Al) in interdendritic regions. During the homogenization, the solute diffuses into the dendrite and the primary Mg(Zn, Al, Cu)2 phase dissolves into the matrix. The compositions of the second phases are listed in Table 2. As reported in Refs.[4,8], the primary Mg(Zn, Al, Cu)2 phase is solid solution with extended composition ranges containing four elements. The copper and magnesium are rich in the primary Mg(Zn, Al, Cu)2, which will supply sufficient solute for nucleation of the S. From Table 2, it can be seen that the S phase contains lower zinc content compared with the primary phase, which indicates that the zinc diffuses out of the primary Mg(Zn, Al, Cu)2 phase during the homogenization and results in the formation of Al2CuMg.
Table 2 Compositions of phases present in alloy(mole fraction, %)

According to the observations above, the phase transformation of the primary particles can be expressed as:
Mg(Zn, Al, Cu)2→S(Al2CuMg)
The phase transformation is controlled by the diffusion of zinc. The reaction is close to the eutectoid phase transformation from Alm(Fe, Mn) to Al3(Fe,Mn) in AA5182 alloy, which is controlled by the diffusion of aluminum[15]. It should be pointed out that this reaction included the diffusion of other elements such as copper, magnesium and aluminium. The S phase also contains four elements. The diffusion of the solute within the grain during the homogenization is analyzed by EDS. The results are shown in Fig.6. The diffusion rate of zinc is faster than that of copper. After 1 h, the zinc distributes nearly homogeneously. A great amount of S particles have formed as shown in Fig.5(b), indicating the great influence of the diffusion of zinc on the phase transformation. The copper content changes greatly with the increase of holding time, which results from the diffusion of the copper from the interdendritic regions to the center regions of dendrite. This explains the decrease of the fraction of newly formed S phase during the homogenization.
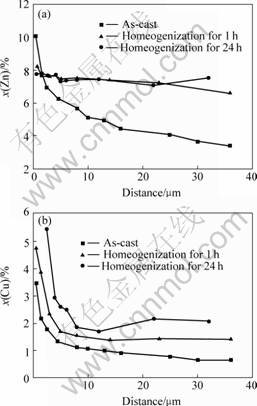
Fig.6 Evolution of solute distribution during homogenization:(a) Zinc content; (b) Copper content
4 Conclusions
1) A phase transformation of the primary particle from Mg(Zn, Al, Cu)2 to S occurs during the homogenization at 460 ℃.
2)The newly formed S nucleates at the boundaries between the primary particles and the matrix. After nucleation, the S phase grows along the primary Mg(Zn, Al, Cu)2 particles. The coalescence of S occurs with the decrease of its fraction during the homogenization.
3)The transformation process from Mg(Zn, Al, Cu)2 to S is controlled by the diffusion of solute, and the diffusion of zinc has a great influence on this process.
Acknowledgements
Authors are also grateful to the Northeast Light Alloy Co. Ltd. for production of materials.
References
[1] DESCHAMPS A, BRECHET Y, LIVET F. Influence of copper addition on precipitation kinetics and hardening in Al-Zn-Mg alloy[J]. Materials Science and Technology, 1999, 15(9): 993-1000.
[2] CHINH N Q, LENDVAI J, PING D H, HONO K. The effect of Cu on mechanical and precipitation properties of Al-Zn-Mg alloys[J]. Journal of Alloys and Compounds, 2004, 378: 52-60.
[3] SALAMCI E, COCHRANE R F. Investigation of secondary phase particles in spray deposited 7000 series aluminium alloys[J]. Materials Science and Technology, 2002, 18(12): 1445-1452.
[4] LI X M, STARINK M J. Effect of compositional variations on characteristics of coarse intermetallic particles in overaged 7000 aluminium alloys[J]. Materials Science and Technology, 2001, 17: 1324-1328.
[5] HAHN G T, ROSENFIELD A R. Metallurgical factors affecting fracture toughness of aluminium alloys[J]. Metallurgical Transa-
ctions A, 1975, 6A: 653-668.
[6] THOMPSON D S. Metallurgical factors affecting high strength aluminium alloy production[J]. Metallurgical Transactions A, 1975, 6A: 671-683.
[7] HASHEMI H R, ASHOORI H. Microstructure and tensile properties of squeeze cast Al-Zn-Mg-Cu alloy[J]. Materials Science and Technology, 2001, 17: 639-643.
[8] XIE F Y, YAN X Y, DING L, ZHANG F, CHEN S L, CHU M, CHANG Y A. A study of microstructure and microsegregation of aluminum 7050 alloy[J]. Materials Science and Engineering A, 2003, A335: 144-153.
[9] MUKHOPADHYAY A K. On the nature of the second phase particles present in an as-cast Al-Cu-Mg-Ag alloy[J]. Scripta Materialia, 1999, 41(6): 667-672.
[10] MONDAL C, MUKHOPADHYAY A K. On the nature of T(Al2Mg3Zn3) and S(Al2CuMg) phase present in as-cast and annealed 7055 aluminum alloy[J]. Materials Science and Engineering A, 2005, A391: 367-376.
[11] CHEN K H, LIU H W, ZHANG Z, LI S. The improvement of constituent dissolution and mechanical properties of 7055 aluminum alloy by stepped heat treatments[J]. Journal of Materials Processing Technology, 2003, 142:190-196.
[12] MUKHOPADHYAY A K, RAO R, RAMA V V. Characterization of S(Al2CuMg) phase particles present in as-cast and annealed Al-Cu-Mg(-Li)-Ag alloys[J]. Materials Science and Engineering A, 1999, 268: 8-14.
[13] KIM Y C, KIM D H, KIM W T, HAN Y S, LEE H I. Effect of silver addition on formation of secondary phases in squeeze cast Al-Cu-Mg alloys[J]. Materials Science and Technology, 2001, 17(2): 215-222.
[14] FAN X G, JIANG D M, MENG Q C, ZHONG L. The microstructural evolution of an Al-Zn-Mg-Cu alloy during homogenization[J]. Materials Letters, 2006, 60: 1475-1479.
[15] LI Y J, ARNBERG L. A eutectoid phase transformation for the primary intermetallic particle from Alm(Fe, Mn) to Al3(Fe, Mn) in AA 5182 alloy[J]. Acta Materialia, 2004, 52(10): 2945-2952.
(Edited by YANG You-ping)
Foundation item: Project(2004AA5BG018) supported by the Science and Technology Development Fund of Harbin, China
Corresponding author: FAN Xi-gang; Tel: +86-451-86417617; E-mail: xigangfan@163.com