Recovery of Zn, Pb, Fe and Si from a low-grade mining ore by sulfidation roasting-beneficiation-leaching processes
��Դ�ڿ������ϴ�ѧѧ��(Ӣ�İ�)2020���1��
�������ߣ�֣���� ������ ��Խ ������ �ӽ� ������
����ҳ�룺37 - 51
Key words��low-grade mining ore; comprehensive recovery; sulfidation roasting; flotation; magnetic separation; leaching
Abstract: To recover Zn, Pb, Fe and Si from a low-grade mining ore in the Lanping basin, Yunnan Province, China, a novel technology using the roasting with pyrite and carbon followed by beneficiation and hydrochloric acid leaching was proposed. Firstly, several factors such as pyrite dosage, roasting temperature, carbon powder dosage, holding time and particle size affecting on the flotation performance of Zn (Pb) and magnetic separation performance of Fe were simultaneously examined and the optimum process parameters were determined. A flotation concentrate, containing 17.46% Zn and 3.93% Pb, was obtained, and the Zn and Pb recoveries were 86.04% and 69.08%, respectively. The obtained flotation tailing was concentrated by a low-intensity magnetic separator. The grade of iron increased from 5.45% to 43.45% and the recovery of iron reached 64.87%. Hydrochloric acid leaching was then carried out for the magnetic separation tailing and a raw quartz concentrate containing 81.05% SiO2 was obtained. To further interpret the sulfidation mechanism of smithsonite, surface morphology and component of the sample before and after reactions were characterized by XRD and EPMA-EDS. The aim was to achieve the comprehensive utilization of the low-grade mining ore.
Cite this article as: LAN Zhuo-yue, LAI Zhen-ning, ZHENG Yong-xing, LV Jin-fang, PANG Jie, NING Ji-lai. Recovery of Zn, Pb, Fe and Si from a low-grade mining ore by sulfidation roasting-beneficiation-leaching processes [J]. Journal of Central South University, 2020, 27(1): 37-51. DOI: https://doi.org/10.1007/s11771-020-4276-3.

J. Cent. South Univ. (2020) 27: 37-51
DOI: https://doi.org/10.1007/s11771-020-4276-3
LAN Zhuo-yue(��Խ)1, 2, LAI Zhen-ning(������)2, ZHENG Yong-xing(֣����)1,LV Jin-fang(������)2, PANG Jie(�ӽ�)2, NING Ji-lai(������)1
1. State Key Laboratory of Complex Nonferrous Metal Resources Clean Utilization, Kunming University of Science and Technology, Kunming 650093, China;
2. Faculty of Land Resource Engineering, Kunming University of Science and Technology, Kunming 650093, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2020
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2020
Abstract: To recover Zn, Pb, Fe and Si from a low-grade mining ore in the Lanping basin, Yunnan Province, China, a novel technology using the roasting with pyrite and carbon followed by beneficiation and hydrochloric acid leaching was proposed. Firstly, several factors such as pyrite dosage, roasting temperature, carbon powder dosage, holding time and particle size affecting on the flotation performance of Zn (Pb) and magnetic separation performance of Fe were simultaneously examined and the optimum process parameters were determined. A flotation concentrate, containing 17.46% Zn and 3.93% Pb, was obtained, and the Zn and Pb recoveries were 86.04% and 69.08%, respectively. The obtained flotation tailing was concentrated by a low-intensity magnetic separator. The grade of iron increased from 5.45% to 43.45% and the recovery of iron reached 64.87%. Hydrochloric acid leaching was then carried out for the magnetic separation tailing and a raw quartz concentrate containing 81.05% SiO2 was obtained. To further interpret the sulfidation mechanism of smithsonite, surface morphology and component of the sample before and after reactions were characterized by XRD and EPMA-EDS. The aim was to achieve the comprehensive utilization of the low-grade mining ore.
Key words: low-grade mining ore; comprehensive recovery; sulfidation roasting; flotation; magnetic separation; leaching
Cite this article as: LAN Zhuo-yue, LAI Zhen-ning, ZHENG Yong-xing, LV Jin-fang, PANG Jie, NING Ji-lai. Recovery of Zn, Pb, Fe and Si from a low-grade mining ore by sulfidation roasting-beneficiation-leaching processes [J]. Journal of Central South University, 2020, 27(1): 37-51. DOI: https://doi.org/10.1007/s11771-020-4276-3.
1 Introduction
The Jinding Pb-Zn deposit, which locates at the Lanping basin in Yunnan Province, is the largest Pb-Zn deposit in China, and probably the youngest sediment-hosted super giant Zn-Pb deposit in the world [1, 2]. With the continuous exploitation of sulfide ores and high-grade oxide ores, large quantities of low-grade mining ores have been produced. Statistically, there are about 32 million tons of ores stockpiled in the open air [3]. They potentially endanger the environment, as the metals they contain may be released to the environment in hazardous form. Smithsonite is the most valuable mineral component in the mining ore, in which the average grade of zinc was about 6% [4]. This needs processing the ores to obtain a marketable product. For this type of raw material, flotation is the most commonly used method for the Zn recovery.
Both of the cationic and anionic collectors are used for the flotation of zinc oxide mineral. In the available literature, it was found that the flotation with amine as collectors and Na2S as modifier was of wide use for the zinc recovery [5, 6]. But for the Lanping zinc oxide ore characterized by the high content of slime and valuable metals distributed in fine size fraction, significant amounts of Na2S had to be added ascribed to the non-selective adsorption on the slime, resulting in the ineffectiveness of flotation [7, 8]. De-sliming prior to the flotation was also proved to be an effective method, but the valuable metals distributed in the fine fraction lost in this process [9]. By contrast, the anionic collector, mainly fatty acids, seemed to be potentially feasible in the flotation, but the gangue and iron bearing mineral also reported the concentrate due to the poor selectivity, making the following separation more complicated [10]. More seriously, no matter what methods were adopted, large quantities of tailings, containing heavy metals, were still discharged in the process, exhibiting potential environmental and safe risks for the local residents.
Generally, sulfidation-xanthate flotation method is less sensitive to the slime. After treating with Na2S at a moderate dosage, the hydrophilicity of the mineral surface decreases because of the presence of chemisorbed sulfide ion and in the present case, the zinc oxide minerals should be effectively collected by the xanthate [11]. Factually, their sulfidation are extremely difficult to be achieved, resulting in the ineffective mineral processing. To improve the surface sulfidation of zinc oxide minerals, WU et al [12] found that the surface solubility of the smithsonite in the presence of ammonium ions significantly decreased, corresponding to its flotation recovery increased by 25%. However, this was only carried out for a single smithsonite and its effectiveness for the zinc oxide ore with a complex composition was not illustrated. Other methods such as mechanical- chemical and hydrothermal sulfidation were also reported [13, 14], but their further application seemed to be difficult due to the slow transformation process and poor recovery of zinc.
Recently, sulfidation roasting has received considerable attentions to improve the sulfidation of zinc oxide minerals. LI et al [15] investigated the sulfidation roasting of a low-grade zinc oxide ore containing 7.57% Zn with elemental sulfur and the sulfidation extent of Zn was 95%. A flotation concentrate containing 38.9% Zn was obtained from the treated ore and the recovery of Zn was 88.2%. ZHENG et al [16] adopted a method of temperature gradient for transforming the zinc carbonate into the zinc sulfide and a zinc sulfidation extent of 97.29% was obtained. HAN et al [17] found that the addition of iron oxides during the roasting of ZnO with elemental sulfur could not only improve the sulfidation of ZnO, but also promote the formation and growth of ZnS crystals. However, these studies about sulfidation roasting have been mainly restricted to the investigation of process optimization, especially for the improvement of the sulfidation extent. They required introducing significant amounts of sulfur, but most of which were lost due to the nature of easier volatilization and then discharged in the form of SO2, resulting in the environmental pollution. Factually, the flotation performances of oxide minerals depend on whether a stable sulfide film can be formed at the mineral surface to the greatest extent. Therefore, it seemed unreasonable to improve their flotation performances by increasing the sulfidation extents. In addition, the recovery of other elements such as Fe and Si was also not considered.
In our knowledge, little work has been reported on the comprehensive utilization of a low-grade mining ores by the roasting with pyrite and carbon followed by the benefication and leaching processes. Roasting with pyrite and carbon powder was firstly carried out, resulting in hydrophobicity improvement of Zn-Pb oxide minerals and a distinct improvement of magnetic susceptibility of ferric oxide mineral. After flotation with the conventional collector (xanthate), a Zn-Pb bulk concentrate and a tailing, mainly containing Fe, Si and Ca, were obtained. Then, the tailing was concentrated by a low-intensity magnetic separator and a magnetic concentrate was produced. To further recover Si from the magnetic separation tailing, hydrochloric acid leaching was finally performed to remove the calcite and a raw quartz concentrate was obtained. The hydrophobic transformation was directly characterized by the flotation (not sulfidation extent) to optimize the roasting conditions. The phase, morphology and surface changes were detected by XRD and EPMA-EDS analyses to interpret the roasting mechanisms. The goal of this paper was to find a feasible way to realize the full economic value of the low-grade mining ore and nearly no tailings discharged.
2 Experimental
2.1 Materials
The representative sample was provided from the Jinding lead-zinc mine, whose chemical composition is shown in Table 1, which reports a mining ore with low-grades of zinc, lead and iron. Phase compositions of zinc, lead and iron in the ore are shown in Table 2, which reveals the sample with a high oxidization extent. In addition, the percentage of iron sulfide accounting for 32.08% indicates that there were parts of natural sulfidation agents. Figure 1 shows the XRD pattern of the sample. It was known that the main zinc mineral was smithsonite and the gangue was mainly quartz and calcite. The lead bearing minerals were not detected due to its low content. According to Table 2 and other similar reports [15], the lead bearing mineral was mainly cerussite. In the available literature [15, 18], the Al2O3 and MgO mainly occurred as kaolin (Al2(Si2O5)(OH)4) and dolomite (CaMg(CO3)2), respectively.
Table 1 Chemical composition of mining ore (mass fraction, %)
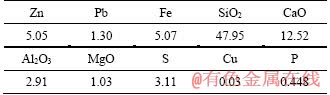
Table 2 Phase composition of zinc, lead and iron in mining ore (mass fraction, %)
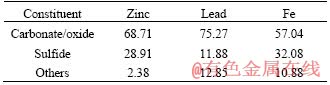
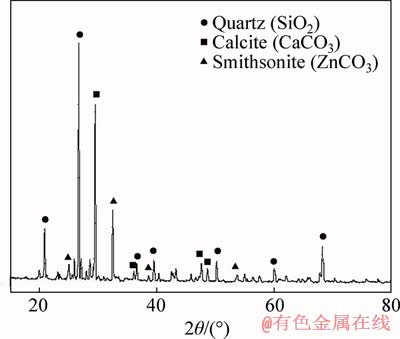
Figure 1 XRD pattern of mining ore
Carbon powder containing 55% C, 21% ash, 24% volatiles was used as the reducing agent. Pyrite containing 46% Fe, 47% S, 5% SiO2 and 2% Al2O3 was used as the sulfidation agent. Copper sulphate, sodium hexametaphosphate, ammonium dibutyl dithiophosphate and butyl xanthate were used in the flotation tests. Hydrochloric acid was used as the leaching agent.
2.2 Theoretical fundamental
2.2.1 Roasting with pyrite and carbon
The related reactions during the roasting of low-grade mining ore with the pyrite and carbon are listed as follows [16, 19-22]:
FeS2��FeS+1/2S2(g) (1)
ZnCO3��ZnO+CO2(g) (2)
PbCO3��PbO+CO2(g) (3)
3/2S2(g)+2ZnO��2ZnS+SO2(g) (4)
3/2S2(g)+2PbO��2PbS+SO2(g) (5)
S2(g)+2ZnO+C��2ZnS+CO2(g) (6)
S2(g)+2PbO+C��2PbS+CO2(g) (7)
3Fe2O3+C��2Fe3O4+CO(g) (8)
6Fe2O3+C��4Fe3O4+CO2(g) (9)
C+CO2(g)��2CO(g) (10)
3Fe2O3+CO(g)��2Fe3O4+CO2(g) (11)
The pyrite is firstly decomposed into ferrous sulfide and sulfur vapor (Eq. (1)). Meantime, the lead and zinc carbonates are dissociated into their respective oxides (Eqs. (2)-(3)) in the temperature range of 500-800 ��C. Then, the generated sulfur vapor will react at the surfaces of the formed oxides, resulting in the formation of sulfide films (Eqs. (4)- (5)). When the carbon is introduced into the system, the sulfidation reactions at the surface will be enhanced [20]. On the one hand, the sulfur dosage decreases from 1.5 mole to 1.0 mole when the lead or zinc oxide is fixed at 2 mole (Eqs. (6)-(7)). On the other hand, the amount of SO2 gas will decrease, which is favorable for the environment.
The reduction reactions of iron oxides simultaneously occur during the surface sulfidation of Zn-Pb oxides, as presented in Eqs. (8)-(11). Generally, the addition of carbon can not only improve the sulfidation roasting, but also transform the iron oxides into magnetite in the thermodynamics.
2.2.2 Upgrading quartz by hydrochloric acid leaching
In the magnetic separation tailing, the main minerals are quartz and calcite. The quartz can be further upgraded by the selective leaching using the hydrochloric acid as leaching agent. The related reactions are listed as follows [23, 24]:
CaCO3+2HCl��CaCl2+CO2(g)+H2O (12)
FeO+2HCl��FeCl2+H2O (13)
The quartz will not be dissolved in the process and then remained in the residue, which can be used as a raw quartz concentrate. The calcite will be completely dissolved and then enter the leaching liquor, which can be used to prepare the anhydrous CaCl2.
2.3 Experimental method
The general flow sheet for processing the low-grade mining ore is shown in Figure 2. Roasting of the ore was conducted in a muffle furnace. The prepared powder sample was firstly mixed with the carbon powder and pyrite at a desired weight ratio and then was loaded into an alundum crucible equipped with a cover. Finally, the crucible was placed in the furnace and heated. After the holding time of 30-90 min, the sample was cooled and then used for the flotation tests.
The roasting conditions were mainly optimized by the flotation tests, as shown in Figure 2 (Dotted part). In addition, several parameters, such as carbon powder dosage and temperature, were simultaneously examined by the magnetic separation for the flotation tailing. When the optimal roasting conditions were determined, the close-circuit flotation tests were performed and the Zn-Pb sulfides concentrate was obtained. The flotation tailing was used for the magnetic separation and a magnetic concentrate was produced. The magnetic tailing was further upgraded and a raw quartz concentrate was obtained.
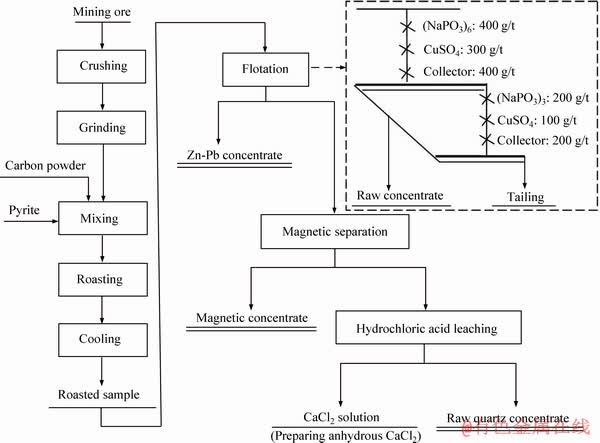
Figure 2 General flow sheet for processing the mining ore
2.4 Property changes and analytical techniques
The obtained samples were examined on a X-ray powder diffractometer (Cu K��, ��=1.5406  ). The transformation of morphology and composition were detected by the scanning electron microscope (JEOL. Ltd., JSM-6360LV) and energy spectrometer (EDAX. Ltd., EDX-GENESIS 60S). The EPMA (JXA-8230, 20 kV) line scanning was carried out to compare the surface differences between the natural sphalerite and the treated smithsonite. The leaching performance of magnetic separation tailing was further characterized by examining the content of CaO in the residue.
). The transformation of morphology and composition were detected by the scanning electron microscope (JEOL. Ltd., JSM-6360LV) and energy spectrometer (EDAX. Ltd., EDX-GENESIS 60S). The EPMA (JXA-8230, 20 kV) line scanning was carried out to compare the surface differences between the natural sphalerite and the treated smithsonite. The leaching performance of magnetic separation tailing was further characterized by examining the content of CaO in the residue.
3 Results and discussion
3.1 Tests for roasting with pyrite and carbon powder
3.1.1 Effect of pyrite dosage
The surfaces of smithsonite and cerussite can be transformed into sulfides at high temperatures in the presence of pyrite. So it was initially investigated under these conditions: particle size of -74 ��m (85%), carbon powder dosage of 3%, roasting temperature of 700 ��C, holding time of 60 min and the results are shown in Figures 3(a) and (b). It was seen that part of zinc and lead oxide minerals were recovered when there was no addition of the pyrite. This can be ascribed to the sulfidation caused by the iron sulfides in the raw sample (Table 2). The recoveries of lead and zinc increased from 59.88% to 73.95% and from 62.15% to 77.95%, respectively, when the pyrite dosage increased from 0% to 3%, whereas the grades of lead and zinc decreased from 4.33% to 3.74% and from 22.01% to 16.27%, respectively. With the further increase in the pyrite dosage, their recoveries slightly fluctuated and their grades further decreased. This can be explained that the generated iron sulfides reported the flotation concentrate. Therefore, the optimal pyrite dosage was determined to be 3%. In addition, it was found that the flotation performance of lead oxide mineral seemed to be similar to that of the zinc oxide mineral. Accordingly, the flotation performance of zinc oxide mineral was only examined in the following tests.
According to the ideal reactions (Eqs. (6) and (7)) and the component of lead and zinc (Tables 1 and 2), pyrite dosage of 7% was approximately added to completely transform the lead and zinc oxides into the sulfides. However, the practical pyrite dosage was lower than the theoretical value. It was deduced that the sulfidation extents of lead and zinc should not be high. In other words, the sulfidation reactions only occurred at the outlayer of the oxides. In the present case, the treated lead and zinc oxides were well recovered by the conventional flotation. On the contrary, their flotation performances became bad, especially for their grades, with the increase in the pyrite dosage. In the available literature, the increase of sulfidation agent dosage would improve the sulfidation extents of lead and zinc oxides [15, 16, 25]. Therefore, it was concluded that the flotation responses were not an absolutely positive correlation with their sulfidation extents.
3.1.2 Effect of roasting temperature
Figures 3(c) and (d) present the flotation performance of zinc and magnetic separation performance of iron with respect to the roasting temperature, respectively. From Figure 3(c), it was seen that the zinc recovery increased when the temperature increased from 600 ��C to 700 ��C, which could be ascribed to the fact that the higher the roasting temperature was, the more sulfur vapor was generated, resulting in more sulfides formed at the surface of zinc oxide mineral. The recovery of zinc nearly kept constant when the temperature increased to 750 ��C. With the further increase in the temperature, the recovery of zinc sharply decreased. This could be explained as follows [20, 25]:
SiO2+2ZnO��Zn2SiO4 (14)
Fe2O3+ZnO��ZnFe2O4 (15)
Compared with the ZnO, the zinc silicates and zinc ferrites are difficult to be sulfurized, making the formation of sulfides difficult [20]. In addition, the grade of zinc decreased with the increase in the temperature. This result was ascribed to the aggregation of particles enhanced with the increase in the temperature, resulting in the gangues also reporting the concentrate.
Figure 3(d) presents the magnetic separation performances of iron bearing mineral with respect to the roasting temperature. The lower recovery of iron at temperature lower than 700 ��C may be attributed to the inadequate reduction of iron oxides (Eqs. (8)-(11)). The bad separation performances at temperature higher than 750 ��C may be ascribed to the aggregation of particles and even the excessive reduction of iron oxides to ferrous oxide [26]. Combining with the zinc flotation performances, the optimal roasting temperature was determined to be 700 ��C.
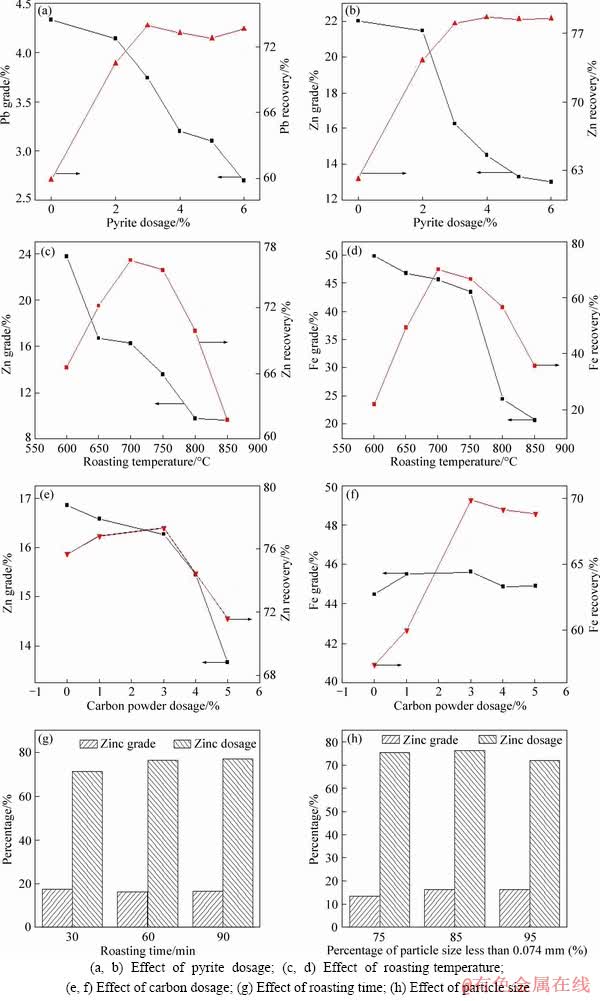
Figure 3 Factors affecting beneficiation performances:
3.1.3 Effect of carbon powder dosage
Figures 3(e) and 3(f) exhibit the effects of carbon powder dosage on the flotation performance of zinc and magnetic separation performance of iron, respectively. It was seen that the recovery of zinc slightly increased and the iron recovery obviously increased when the carbon powder dosage increased from 0% to 3%. The flotation performance of zinc became bad and the magnetic separation performance of iron had little changes with the increase in the carbon dosage. The bad flotation performances of zinc may be ascribed to the non-selective adsorption of collector at the surface of the carbon powder [27, 28]. Accordingly, the optimal carbon powder dosage was determined to be 3%.
3.1.4 Effect of holding time and particle size
Figure 3(g) shows the relationship between the flotation performance of zinc and holding time. It was seen that the zinc recovery increased from 71.44% to 77.95% when the roasting time increased from 30 min to 60 min. With the further increase in the roasting time, there was little change. On the other hand, the grade of zinc nearly kept constant in the time range. Therefore, the optimal roasting time was determined to be 60 min.
The effect of particle size on the flotation performance of zinc is presented in Figure 3(h). It was known that the zinc grade increased and zinc recovery nearly kept constant when the percentage of particle size of -74 ��m increased from 75% to 85%. With the further increase in the percentage, the flotation performance of zinc became bad, which may be ascribed to large quantities of the generated slimes, resulting in the non-selective adsorption of flotation reagents [29, 30]. Therefore, the optimal percentage of particle size of -74 ��m was determined to be 85%.
3.2 Tests for close-circuit flotation and magnetic separation
3.2.1 Close-circuit flotation tests
The close-circuit flotation tests for the roasted material were carried out by a bulk flotation process, as shown in Figure 4. The flotation results are shown in Table 3. It was known that a flotation concentrate containing 17.46% Zn and 3.93% Pb was obtained and the zinc and lead recoveries were 86.04% and 69.08%, respectively. In addition,Table 4 also listed the mainly chemical compositions of the flotation concentrate and tailing.
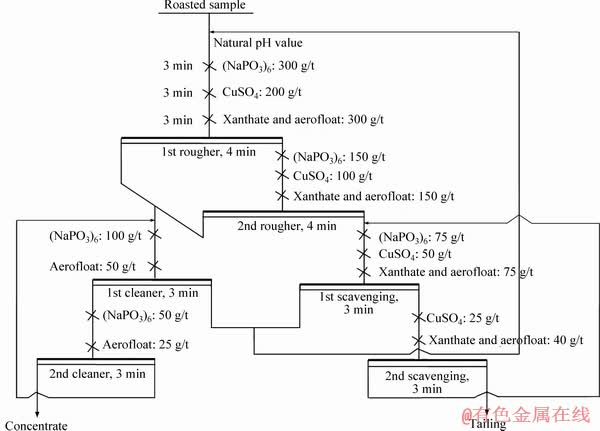
Figure 4 Flow chart of close-circuit flotation test
Table 3 Results of close-circuit flotation for roasted sample (%)
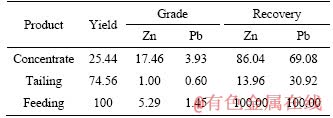
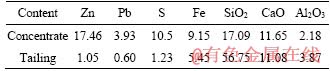
Table 4 Main chemical composition of flotation concentrate and tailing (%)
These results confirmed that the Zn-Pb oxides after the surface modification were well recovered by the conventional flotation.
3.2.2 Magnetic separation for flotation tailing
Figure 5 shows the effect of magnetic induction on the magnetic separation performances. It was known that the Fe grade decreased from 49.85% to 43.45%, whereas the Fe recovery increased from 46.24% to 64.87% for the flotation tailing when the magnetic induction increased to 0.27 T. With the further increase in the magnetic induction, both the grade and recovery of iron slightly fluctuated. So the optimal magnetic induction was determined to be 0.27 T. XRD patterns of the magnetic separation products obtained under the optimal condition are shown in Figure 6. It was seen that there were peaks of quartz, indicating that the quartz reported the magnetic concentrate due to the aggregation of particles at high temperatures. This explained the magnetic concentrate with a low-grade of iron. In addition, it was known that the main components of tailing were quartz and calcite. The latter can be removed by the hydrochloric acid leaching to upgrade the quartz.
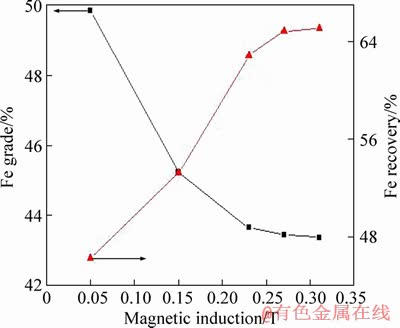
Figure 5 Effect of magnetic induction on magnetic separation performance
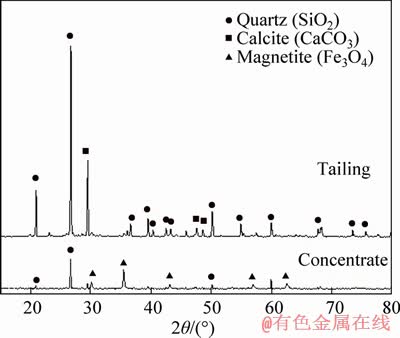
Figure 6 XRD patterns of magnetic separation products
3.3 Hydrochloric acid leaching tests for magnetic separation tailing
The hydrochloric acid leaching tests for the magnetic separation tailing were carried out and the results are shown in Figure 7. The effect of hydrochloric acid concentration on the leaching performance of CaO was firstly examined under these conditions: liquid/solid ratio of 3, stirring rate of 250 r/min, leaching time of 60 min, as shown in Figure 7(a). It was seen that the content of CaO in the residue decreased from 3.67% to 0.66% when the initial concentration increased from 10% to 20%. With the further increase in the initial concentration, the content of CaO was nearly constant. Therefore, the optimal initial concentration was determined to be 20%.
The effect of liquid/solid ratio on its leaching performance is exhibited in Figure 7(b). It was known that the content of CaO slightly decreased when the liquid/solid ratio increased from 1 to 3. This result illustrated that the increase of the volume of leaching solution made the reaction between minerals and leaching solution sufficiently occur, resulting in the leaching rate accelerated by the efficiency of mass transfer [31]. The content of CaO in the residue slightly increased with the increase in the liquid/solid ratio. Therefore, the optimal liquid/solid ratio was determined to be 3.
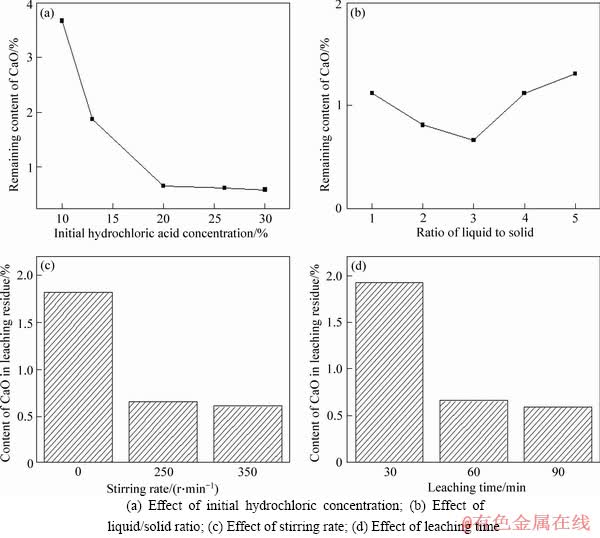
Figure 7 Factors influencing leaching performances:
Figure 7(c) presents the decrease of CaO content in the residue in contrast with no stirring. The further increase in the stirring rate seemed not to affect the content. So, the optimal stirring rate was determined to be 250 r/min. The effect of the time on its leaching performance was shown in Figure 7(d). It was seen that the leaching time had an obvious effect on the leaching performance of CaO within 60 min. With the further increase in the leaching time, its leaching performance had little change. Therefore, the optimal leaching time was determined to be 60 min.
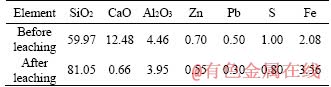
In addition, Table 5 also lists the main composition of the magnetic separation tailing before and after leaching. It was seen that the content of SiO2 increased from 59.97% to 81.05% after the hydrochloric acid leaching. This product can be used as a flux in the pyrometallurgical process. On the other hand, the content of CaO decreased from 12.48% to 0.66% after the leaching, but the contents of Al2O3 and Fe had little changes, indicating that the leaching liquid was mainly composed of CaCl2, which will be used as a kind of raw material for producing the anhydrous CaCl2. Therefore, the full economic value of the low-grade mining ore was achieved after adopting this novel method, as well as no tailings discharged.
Table 5 Main composition of magnetic separation tailing before and after leaching/%
3.4 Properties changes during roasting
3.4.1 XRD analyses
The XRD patterns of the mining ores treated under different conditions are shown in Figure 8. From Figures 8(a) and (b), it was seen that the peak of smithsonite disappeared after the roasting in contrast with the raw sample (Figure 1). When the pyrite was introduced into the system (Figure 8(c)), the peaks of PbS and ZnS occurred, indicating that the outlayers of the obtained zinc and lead oxides were transformed into their sulfides. According to Figure 3, these treated Zn-Pb oxides were well recovered by the conventional flotation. When the pyrite and carbon powder were simultaneously introduced (Figure 8(d)), there was no obvious change for the sample. Combining with Figure 6, the hematite and limonite were reduced to magnetite in the presence of carbon, but the peak of the magnetite was not observed (Figure 8(d)).
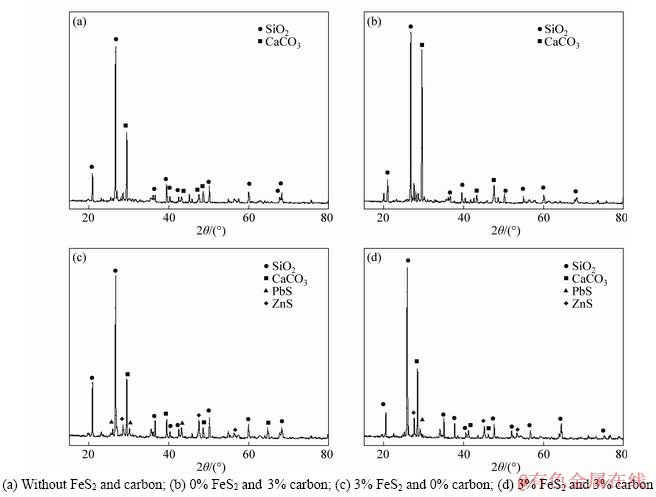
Figure 8 XRD patterns of mining ores treated under different conditions:
3.4.2 EPMA-EDS analyses
Figure 9 shows the BSE images and EDS spectra of the roasted products at different temperatures. From Figures 9(a), (c) and (e), it was seen that the Zn-Pb oxide particles were covered with the sulfide films and these particles were nearly liberated from other minerals such as magnetite, calcite and quartz when the roasting temperature was fixed at 600 ��C. The mineral particles began to aggregate when the roasting temperature increased to 700 ��C. It was especially noticed that large quantities of fine grains at a size fraction of 1-2 ��m deposited at the surface of larger particle. With the further increase in the temperature (Figure 9(e)), the aggregation of mineral particles was enhanced and part of particles started to form solid solution. These results explained the effect of the roasting temperature on the zinc flotation performance (Figure 3(c)) and iron magnetic separation performance (Figure 3(d)). From Figures 9(b), (d) and (f), it was known that the surfaces of zinc oxides were not only covered with sulfides, but also with other gangue such as silicon, iron and calcium bearing minerals, although the roasting temperature was fixed at 600 ��C. This could be accounted by the fact that significant amounts of slimes were easily produced during the grinding of the mining ore and the generated slimes attracted with the target mineral due to the interaction force between the particles [32, 33].
3.4.3 EPMA analyses
To further investigate the artificial film formed at the surface of zinc oxide mineral, EPMA analyses were carried out and the results are shown in Figure 10 and Table 6. From Figure 10(a), it was observed that the natural smithsonite occurred as irregular lump with smooth dissociation surfaces. Figure 10(b) shows that the zinc concentration at the surface of smithsonite slightly fluctuated and there was nearly no sulfur signal. When the raw sample was roasted at 700 ��C without any additive, the smithsonite exhibited rough surfaces (Figure 10(c)). From Figure 10(d), it was deduced that these formed surfaces were attributed to the thermal decomposition of the smithsonite and the uneven adsorption of sulfur vapor at the surface. Combining with Table 2, it was known that the sulfur was originated from the iron sulfide in the raw sample. This result further confirmed that part of the Zn-Pb oxide minerals were recovered when there was no addition of the pyrite (Figure 3(a)). With the introduction of the pyrite (3%) and carbon (3%), the surfaces of zinc oxide mineral became smooth (Figure 10(e)) and the zinc concentration fluctuated in the narrow range (Figure 10(f)), indicating that the morphology and component of the artificial films were very similar to that of the natural sphalerite (Figures 10(g) and (h)) [34]. This finding explained that the zinc oxide mineral presented a perfect flotation performance under this condition (Figure 3(a)).
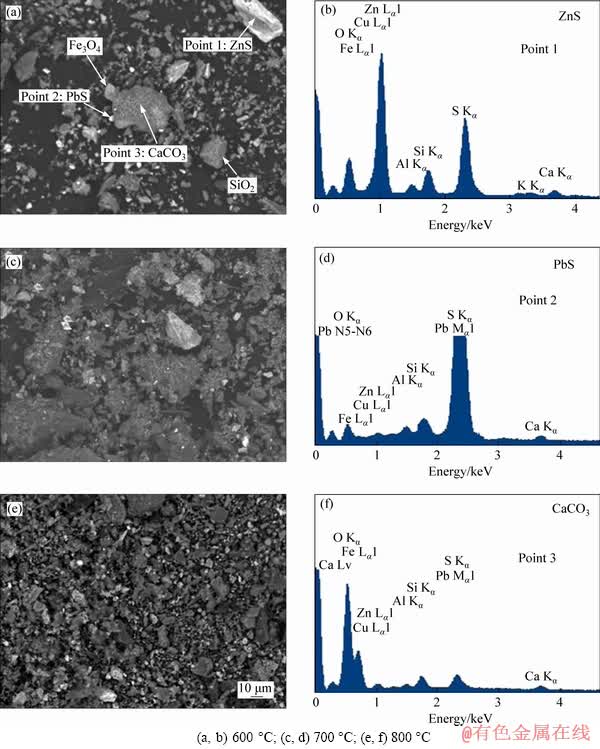
Figure 9 BSE images (a, c, e) and EDS spectra (b, d, f) of roasted products at different temperatures:
The statistical values of the concentration of elemental Zn and S at the surfaces of the obtained samples are listed in Table 6. It was seen that the average concentration of elemental sulfur at the sample surface increased from 0.05% to 8.43% when the raw sample was heated at 700 ��C without any additive. The average concentration of elemental sulfur increased to 24.80%, which was near to the concentration of sulfur (24.13%) at the surface of natural sphalerite when the pyrite (3%) was added. On the other hand, the minimum and maximum concentrations of the elemental zinc and sulfur also suggested that these elements were not evenly distributed at the surface of zinc oxide mineral.
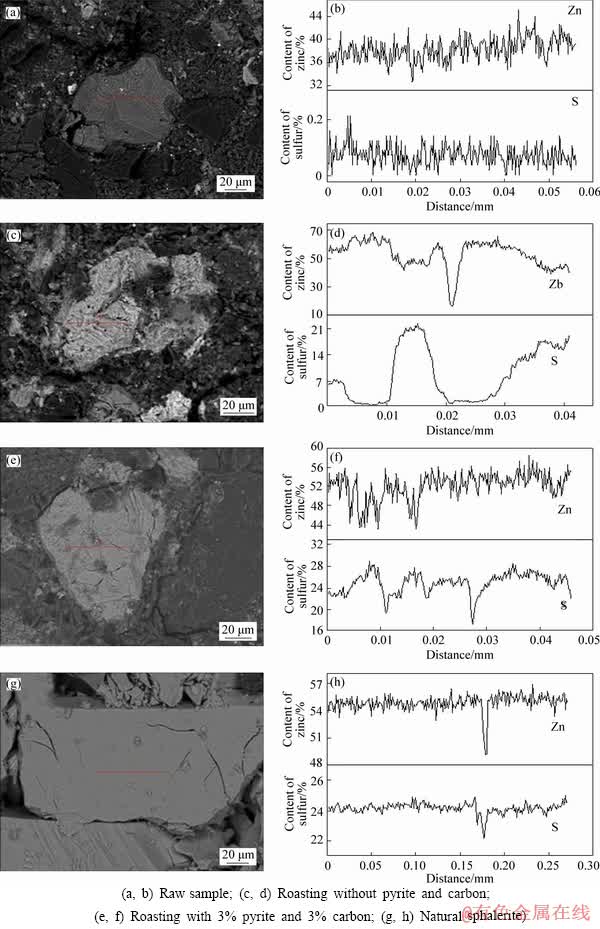
Figure 10 EPMA line scan spectra of single zinc sulfide particles obtained under different conditions:
Table 6 Concentration of elemental Zn and S at surface of obtained particle
4 Conclusions
1) The Zn, Pb, Fe and Si in the mining ore were recovered using the sulfidation roasting followed by the benefication and leaching processes and no tailing was discharged in the whole process.
2) The optimum roasting parameters were determined to be pyrite dosage of 3%, temperature of 700 ��C, carbon powder dosage of 3%, roasting time of 60 min and particle size of <74 ��m (85%). Under these conditions, a flotation concentrate containing 17.46% Zn and 3.93% Pb was obtained and the zinc and lead recoveries were 86.04% and 69.08%, respectively. In addition, it was concluded that the flotation responses were not an absolutely positive correlation with their sulfidation extents. Magnetic separation was carried out for the flotation tailing. The grade of iron increased from 5.45% to 43.45% and the recovery of iron reached 64.87%.
3) The optimum leaching conditions were determined to be initial hydrochloric acid concentration of 20%, liquid/solid ratio of 3, stirring rate of 250 r/min and leaching time of 60 min. Under the optimal leaching conditions, the content of SiO2 in the magnetic separation tailing increased from 59.97% to 81.05% and this leaching residue can be used as a flux in the pyrometallurgical process.
4) The outlayer of the zinc oxide mineral was transformed into zinc sulfide after the surface modification. The generated surface was very similar to that of the natural sphalerite, corresponding to a perfect flotation performance of the zinc oxide mineral.
References
[1] ZAW K, PETERS S G, CROMIE P, BURRETT C, HOU Zeng-qian. Nature, diversity of deposit types and metallogenic relations of South China [J]. Ore Geology Reviews, 2007, 31(1): 3-47. DOI: 10.1016/j.oregeorev.2005. 10.006.
[2] TANG Yong-yong, BI Xian-wu, FAYEK M, HU Rui-zhong, WU Li-yan, ZOU Zhi-chao, FENG Cai-xia, WANG Xin-song. Microscale sulfur isotopic compositions of sulfide minerals from the Jinding Zn�CPb deposit, Yunnan Province, Southwest China [J]. Gondwana Research, 2014, 26(2): 594-607. DOI: 10.1016/j.gr.2013.07.021. http://news.hexun. com/2015-06-10/176628271.html.
[3] China United Business Daily. Lead zinc mine in Asia: The world-class technical problem of development has been solved [EB/OL] [2015-06-10]. http://news.hexun.com/2015- 06-10/176628271.html.
[4] ZHANG Jin-rang, WEN Han-jie, QIU Yu-zhuo, ZHANG Yu-xu, LI Chao. Ages of sediment-hosted Himalayan Pb�CZn�CCu�CAg polymetallic deposits in the Lanping basin, China: Re�COs geochronology of molybdenite and Sm�CNd dating of calcite [J]. Journal of Asian Earth Sciences, 2013, 73: 284-295. DOI: 10.1016/j.jseaes.2013.04.041.
[5] EJTEMAEI M, IRANNAJAD M, GHARABAGHI M. Influence of important factors on flotation of zinc oxide mineral using cationic, anionic and mixed (cationic/anionic) collectors [J]. Minerals Engineering, 2011, 24(13): 1402-1408. DOI: 10.1016/j.mineng.2011.05.018.
[6] KIERSZNICKI T, MAJEWSKI J, MZYK J. 5-alkylsalicylal- doximes as collectors in flotation of sphalerite, smithsonite and dolomite in a Hallimond tube [J]. International Journal of Mineral Processing, 1981, 7(4): 311-318. DOI: 10.1016/ 0301-7516(81)90026-0.
[7] WILLS B A, FINCH FRSC J A, PENG F. Chapter 12- Froth Flotation [M]// Wills Mineral Processing Technology, 2016: 265-380. DOI: 10.1016/B978-0-08-097053-0.00012-1.
[8] LAN Zhuo-yue, LI De-fei, LIU Quan-jun, TONG Xiong. Study on flotation of lead-zinc oxide ore from Yunnan [J]. Advanced Materials Research, 2013, 807-809: 2317-2322. DOI: 10.4028/www.scientific.net/AMR.807-809.2317.
[9] HOSSEINI S H, FORSSBERG E. Studies on selective flotation of smithsonite from silicate minerals using mercaptans and one stage desliming [J]. Mineral Processing & Extractive Metallurgy, 2011, 120(2): 79-84. DOI: 10.1179/ 1743285510Y.0000000001.
[10] HOSSEINI S H, FORSSBERG E. Adsorption studies of smithsonite flotation using dodecylamine and oleic acid [J]. Mining, Metallurgy & Exploration, 2006, 23(2): 87-96. DOI: 10.1007/BF03403341.
[11] ONAL G, BULUT G, GUL A, KANGAL O, PEREK K T, ARSLAN F. Flotation of Aladag oxide lead�Czinc ores [J]. Minerals Engineering, 2005, 18(2): 279-282. DOI: 10.1016/j. mineng.2004.10.018.
[12] WU Dan-dan, MA Wen-hui, WEN Shu-ming, BAI Shao-jun, DENG Jiu-shuai��YIN Qiong. Contribution of ammonium ions to sulfidation-flotation of smithsonite [J]. Journal of the Taiwan Institute of Chemical Engineers, 2017, 78: 20-26. DOI: 10.1016/j.jtice.2017.05.015.
[13] WANG Jun, LU Jin-feng, ZHANG Qi-wu, SATIO F. Mechanochemical sulfidization of nonferrous metal oxides by grinding with sulfur and iron [J]. Industrial & Engineering Chemistry Research, 2003, 42(23): 5813-5818. DOI: 10.1021/ie030046b.
[14] LIANG Yan-jie, CHAI Li-yuan, MIN Xiao-bo, TANG Chong-jian, ZHANG Hai-jing, KE Yong, XIE Xian-de. Hydrothermal sulfidation and floatation treatment of heavy-metal-containing sludge for recovery and stabilization [J]. Journal of Hazardous Materials, 2012, 217-218: 307-314. DOI: 10.1016/j.jhazmat.2012.03.025.
[15] LI Yong, WANG Ji-kun, WEI Chang, LIU Chun-xia, JIANG Ji-bo, WANG Fan. Sulfidation roasting of low grade lead�Czinc oxide ore with elemental sulfur [J]. Minerals Engineering, 2010, 23(7): 563-566. DOI: 10.1016/j.mineng. 2010.01.004.
[16] ZHENG Yong-xing, LIU Wei, QIN Wen-qing, JIAO Fen, HAN Jun-wei, YANG Kang, LUO Hong-lin. Sulfidation roasting of lead and zinc carbonate with sulphur by temperature gradient method [J]. Journal of Central South University, 2015, 22(5): 1635-1642. DOI: 10.1007/s11771- 015-2681-9.
[17] HAN Jun-wei, LIU Wei, ZHANG Tian-fu, XUE Kai, LI Wen-hua, JIAO Fen, QIN Wen-qing. Mechanism study on the sulfidation of ZnO with sulfur and iron oxide at high temperature [J]. Scientific Reports, 2017, 7: 42536. DOI: 10.1038/srep42536.
[18] XUE Chun-ji, ZENG Rong, LIU Shu-wen, CHI Guo-xiang, QING Hai-ruo, CHEN Yu-chuan, YANG Jian-min, WANG Deng-hong. Geologic, fluid inclusion and isotopic characteristics of the Jinding Zn�CPb deposit, western Yunnan, South China: A review [J]. Ore Geology Reviews, 2007, 31(1): 337-359. DOI: 10.1016/j.oregeorev.2005.04.007.
[19] ZHENG Yong-xing, LV Jin-fang, LIU Wei, QIN Wen-qing, WEN Shu-ming. An innovative technology for recovery of zinc, lead and silver from zinc leaching residue [J]. Physicochemical Problems of Mineral Processing, 2016, 52(2): 943-954. DOI: 10.5277/ppmp160233.
[20] ZHENG Yong-xing, LIU Wei, QIN Wen-qing, LUO Hong-lin, HAN Jun-wei. Mineralogical reconstruction of lead smelter slag for zinc recovery [J]. Separation Science and Technology, 2014, 49(5): 783-791. DOI: 10.1080/ 01496395.2013.863342.
[21] LV Jin-fang, ZHANG Han-ping, TONG Xiong, FAN Chun-lin, YANG Wen-tao, ZHENG Yong-xing. Innovative methodology for recovering titanium and chromium from a raw ilmenite concentrate by magnetic separation after modifying magnetic properties [J]. Journal of Hazardous Materials, 2017, 325: 251-260. DOI: 10.1016/j.jhazmat. 2016.11.075.
[22] LI Chao, SUN Heng-hui, BAI Jing, LI Long-tu. Innovative methodology for comprehensive utilization of iron ore tailings: Part 1. The recovery of iron from iron ore tailings using magnetic separation after magnetizing roasting [J]. Journal of Hazardous Materials, 2010, 174(1-3): 71-77. DOI: 10.1016/j.jhazmat.2009.09.018.
[23] BIYOUNE M G, ATBIR A, BARI H, HASSNAOUI L, MONGACH E, KHADIR A, BOUKBIR L, BELLAJROU R, ELHADEK M. Remineralization of permeate water by calcite bed in the Daoura��s plant (south of Morocco) [J]. The European Physical Journal Special Topics, 2017, 226(5): 931-941. DOI: 10.1140/epjst/e2016-60181-6.
[24] GIREESH V S, VINOD V P, KRISHNAN NAIR S, NINAN G. Catalytic leaching of ilmenite using hydrochloric acid: A kinetic approach [J]. International Journal of Mineral Processing, 2015, 134: 36-40. DOI: 10.1016/j.minpro. 2014.11.004.
[25] ZHENG Y X, LIU W, QIN W Q, HAN J W, YANG K, WANG D W. Improvement for sulphidation roasting and its application to treat lead smelter slag and zinc recovery [J]. Canadian Metallurgical Quarterly, 2015, 54(1): 92-100. DOI: 10.1179/1879139514Y.0000000155.
[26] HAN Jun-wei, LIU Wei, QIN Wen-qing, PENG Bing, YANG Kang, ZHENG Yong-xing. Recovery of zinc and iron from high iron-bearing zinc calcine by selective reduction roasting [J]. Journal of Industrial & Engineering Chemistry, 2015, 22(2): 272-279. DOI: 10.1016/j.jiec.2014.07.020.
[27] LIU Han-qiao, WEI Guo-xia, ZHANG Rui. Removal of carbon constituents from hospital solid waste incinerator fly ash by column flotation [J]. Waste Management, 2013, 33 (1): 168-174. DOI: 10.1016/j.wasman.2012.08.019.
[28] WANG Li, SUN Wei, ZHANG Qing-peng. Recovery of vanadium and carbon from low-grade stone coal by flotation [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(11): 3767-3773. DOI: 10.1016/s1003-6326(15) 64020-1.
[29] KIENKO L A, VORONOVA O V. Selective flotation of fine-ingrained carbonate-fluorite ore in pulp of increased dispersion uniformity [J]. Journal of Mining Science, 2014, 50(1): 176-181. DOI: 10.1134/s1062739114010244.
[30] MEHDILO A, IRANNAJAD M, ZAREI H. Smithsonite flotation from zinc oxide ore using alkyl amine acetate collectors [J]. Separation Science and Technology, 2014, 49(3): 445-457. DOI: 10.1080/01496395.2013.838966.
[31] HUANG Jin-xiu, CHEN Meng-jun, CHEN Hai-yan, CHEN Shu, SUN Quan. Leaching behavior of copper from waste printed circuit boards with Bronsted acidic ionic liquid [J]. Waste Management, 2014, 34(2): 483-488. DOI: 10.1016/j. wasman.2013.10.027.
[32] ZHOU Fang, WANG Lou-xiang, XU Zheng-he, RUAN Yao-yang, ZHANG Zhen-yue, CHI Ruan. Role of reactive oily bubble in apatite flotation [J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2017, 513: 11-19. DOI: 10.1016/j.colsurfa.2016.11.024.
[33] GUI Xia-hui, XING Yao-wen, RONG Guo-qiang, CAO Yi-jun, LIU Jiong-tian. Interaction forces between coal and kaolinite particles measured by atomic force microscopy [J]. Powder Technology, 2016, 301: 349-355. DOI: 10.1016/ j.powtec.2016.06.026.
[34] LV Jin-fang, TONG Xiong, ZHENG Yong-xing, XIE Xian, WANG Cong-bing. Study on the surface sulfidization behavior of smithsonite at high temperature [J]. Applied Surface Science, 2018, 437: 13-18. DOI: 10.1016/j.apsusc. 2017.12.163.
(Edited by HE Yun-bin)
���ĵ���
��������-ѡ��-�������ո�Ч���յ�Ʒλ�ɿ��ʯ�е�п��Ǧ������
ժҪ��Ϊ�˴���ƺ������Ʒλ�ɿ��ʯ�л���п��Ǧ��������м�Ԫ�أ����һ���µı���-ѡ��-�������գ����չ����������˻������̼�ۡ��о��˻����������������¶ȡ�̼������������ʱ��Ϳ������ȵ����ض�Ǧп��ѡЧ��������ѡ������Ϊ��Ӱ�죬ȷ������ѹ��ղ������õ��˺�п17.46% �ͺ�Ǧ 3.93%�ĸ�ѡ��������п��Ǧ�Ļ����ʷֱ�Ϊ86.04%��69.08%����ѡβ���������ѡ��ҵ����ú���43.45%��������Ϊ64.87%������������ѡβ����������������ö������躬��Ϊ81.05%��ʯӢɰ������XRD��SEM-EDS��EPMA�Ȳ����ֶΣ�����п��Ӧǰ���������ò����ɽ��з�������һ����ʾ����п������������о���Ϊ��ʵ�ֵ�Ʒλ�ɿ��ʯ���ۺ����á�
�ؼ��ʣ���Ʒλ�ɿ��ʯ���ۺϻ��գ����գ���ѡ����ѡ������
Foundation item: Project(51604131) supported by the National Natural Science Foundation of China; Project(2017FB084) supported by the Yunnan Province Applied Basic Research Project, China; Project(2018T20150055) supported by the Testing and Analyzing Funds of Kunming University of Science and Technology, China
Received date: 2018-09-10; Accepted date: 2019-03-15
Corresponding author: ZHENG Yong-xing, PhD, Associate Professor; Tel: +86-13669710518; E-mail: yongxingzheng2017@126.com; ORCID: 0000-0003-1032-6146; LV Jin-fang, PhD, Lecturer; E-mail: jflv2017@126.com; ORCID: 0000-0001- 7890-3101

