
Preparation and electrochemical performance of
hollow-spherical polypyrrole/V2O5 composite
WANG Yuan-hong1, LIU Heng1, ZHU Ding1, GUO Zai-ping2, LIU Hua-kun2, DOU Shi-xue2
1. College of Materials Science and Engineering, Sichuan University, Chengdu 610065, China;
2. Institute for Superconducting and Electronic Materials, University of Wollongong, NSW 2522, Australia
Received 6 July 2010; accepted 6 December 2010
Abstract: In order to improve the lower practical capacity and bad cyclability of crystalline V2O5 (c-V2O5), the vanadium oxide (V2O5) and polypyrrole (PPy) hybrid with hollow-spherical(HS) structure was studied. HS nanocomposite comprised of conductive polypyrrole and vanadium pentoxide (PPy/V2O5) was synthesized by polymerization of pyrrole monomer (Py) in the hollow-microspherical V2O5 host. This novel hybrid was characterized by X-ray diffraction (XRD), scanning electron microscopy (SEM), transmission electron microscopy (TEM) and tested as the cathode material for lithium-ion batteries (LIB) by galvanostatic cell cycling and electrochemical impedance spectroscopy (EIS). The hollow-spherical polypyrrole/vanadium oxide (HS-PPy/V2O5 ) composites, in which PPy molecules are intercalated between the layers of V2O5, exhibit slight reduced capacity and substantially improve cyclability and electrochemical activity compared with the pure HS-V2O5.
Key words: V2O5; hollow microspheres; cathode; lithium-ion batteries
1 Introduction
The transition metal oxide V2O5 has attracted great attention as cathode material for lithium-ion batteries (LIB) due to its high theoretical capacity and energy density [1]. However, many limitations such as poor cycling behavior and weak power performance are yet to be solved [2-3]. In general, two functionalization routes are widely used to optimize the overall properties of the V2O5 cathode. One route is the insertion of metallic cations or conductive polymer in the interlamellar space of the oxide, forming the so-called “bronze vanadium oxide” or the organic-inorganic hybrid, respectively. Taking the latter for instance, the composites usually have advantages of both organic and inorganic components and exhibit good synergistic effect [4-5], so the modified materials are expected to obtain higher electrical conductivity and more stable layered-structure, and thus, better electrochemical properties [6-7]. The other route is the production of V2O5 in nanometric scale. In this kind of material, the diffusion path is shorter than that in a macroscopic material and the kinetic of intercalation is faster due to the high area to mass ratio [8-9].
Recent studies showed that the hollow spheres of metal oxide integrate the small size effects of ultrafine particles, such as surface effect, volume effect and quantum effect. The spherical shell material has some features, such as ability to hold large amounts of guest molecules or large-sized object, low density, large specific surface, and strong surface penetrability [10-11]. Therefore, when the hollow spheres are used as the cathode materials of lithium-ion batteries, they can present good electrochemical properties.
The objective of this work is to combine the two routes mentioned above to prepare a nanostructual polymer/V2O5 hybrid with particular morphology. Recently, the synthesis of the hollow-microspherical V2O5 (HS-V2O5) crystal was reported [12]. The objective of this work is to study the influence of the PPy/V2O5 hybrid and check the viability of this previous insertion step to improve the electrochemical behaviour. The PPy is selected as the conductive polymer to modify this product to obtain a novel hollow-microspherical hybrid nanocomposite. The as-prepared nanocomposite is tested as the cathode material for LIB and the effects of PPy on the electrochemical properties of the HS-V2O5 are discussed.
2 Experimental
HS-PPy/V2O5 nanocomposite was synthesized by injection and polymerization of Py in the V2O5 host. Briefly, 0.20 g hollow microspheres of c-V2O5 (preparation described elsewhere [12]), 0.728 g ferric chloride (used as oxidant) and 0.32 g C7H7NaO3S (used as dopant) were added in 20 mL distilled water. After stirring the suspension in ice bath for a moment, different amount (0.01, 0.02, 0.03 and 0.04 mL) of pyrrole monomer was injected into the reaction system under the protection of Ar atmosphere. The subsequent polymerization process lasted about 4 h and the formed precipitate was collected by centrifugation, washed several times with ethanol and then heated at 80 ℃ under vaccum to obtain the final product.
The mass of PPy in the whole composite was quantified by the conversion rate (n) of PPy during the polymerization process, which was determined by the following equation:
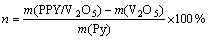 (1)
(1)
where the m(PPy/V2O5), m(V2O5) and m(Py) represent the mass of the as-synthesized composite, the host V2O5 and the injected pyrrole monomer, respectively. In this work, the conversion rate of PPy varied from 18.2% to 19.5% with the injection dosage of Py (0.01-0.04 mL).
The XRD patterns were measured by a Philips X’pert diffractometer using a graphite monochromator and Cu Kα radiation. The scanning range was 10°-60°. The SEM and TEM images of the samples were taken with JEOL JSM-5900LV scanning electron microscope and JEOL JEM-100CX transmission electron microscopy, respectively.
For the electrochemical study, the working electrodes were prepared by mixing the active material with acetylene black (conducting additive) and PVDF (binder) in a mass ratio of 16:3:1, followed by pressing the mixture onto Al foils. Such pallets were vacuum-dried at 80 ℃ for 12 h before entering the argon-?lled glove box in which the gas-tight coin cells were assembled. The metallic Li, membrane (Celgard 2400) and 1 mol/L LiPF6/EC/DEC (1:1 by volume) were used as the counter and reference electrodes, separator and electrolyte, respectively. Galvanostatic charge/ discharge tests were performed over the potential region of 4.0-2.0 V at a constant current density of 80 mA/g using Neware BTS-610. The EIS of each electrode was conducted on an advanced electrochemical system PARSTAT2273, with an AC voltage amplitude of 5 mV over the frequency range of 10 mHz-100 kHz.
3 Results and discussion
3.1 X-ray diffraction study
The XRD patterns of the hollow-spherical vanadium oxide (HS-V2O5) before and after modification with PPy (V(Py)=0.03 mL) are presented in Fig. 1. The pristine V2O5 crystallizes in an orthorhombic structure (PDF 41-1426) with its most intense peak located at 20.369°, which corresponds to the (001) diffraction and is related to the typical layered-structure. The PPy/V2O5 composite displays a set of diffraction peaks which approximate to those of the unmodified V2O5, indicating that the layered-structure is preserved in the composite material. The crystallite sizes estimated from the Scherrer equation for pure and modified V2O5 are 100 and 110 nm, respectively.
Figure 1 also confirms the intercalation of organic polymer into layers of V2O5. The (001) reflection shifts to lower angle of 20.369° and the corresponding interlayer expands from 4.3573 to 4.3792 ? calculated by Jade 5.0 software, which are in accordance with the previous reports for the organic polymer insertion [13-14] and suggest that a parallel polymer chain lies between the V2O5 slabs. Thus, a more stable structure during the reversible insertion/extraction of Li+ can be expected because the PPy molecules can serve as supports to prevent the layers from collapsing. The increment in the d(001) in our experiment is much lower than that reported by BOYANO et al [14]. However, the different V2O5 matrixes used in each research should be taken into account. Comparing with the amorphous V2O5 xerogel used by BOYANO et al [14], the highly crystallized V2O5 has more strong bonding energy and better thermodynamics stability, therefore, the relatively lower change in the structure upon modifying is reasonable.

Fig. 1 XRD patterns of hollow microspheres of V2O5 (a) and PPy/V2O5 (b)
3.2 SEM and TEM study
Representative SEM and TEM images for pure V2O5 and PPy/V2O5 hybrid are displayed in Fig. 2 and Fig. 3, respectively. Figs. 2(a) and (b) clearly show the microspherical morphologies of V2O5. The modification with PPy seems not to affect this morphology strongly as can be seen in Figs. 2(c) and (d). The microspheres of both samples are accumulated by many crystal grains with an average size of about 100 nm, which coincides with the XRD conclusion. These fine grains may provide highly developed surface area for charge-transfer reaction and short paths for Li+ diffusion during the electrochemical reaction. The inner cavities are noticeable in Fig. 3, which may produce a buffer effect on the volume change and relieve the pulverization during the electrochemical cycling. After intercalating the PPy, the wall of the microspheres becomes thicker, corresponding to the interlayer expansion analyzed in XRD as well.
3.3 Electrochemical tests
Galvanostatic cycling tests were carried out at a current density of 80 mA・h/g, which corresponds to the C/7 rate, assuming that the full capacity of V2O5 is 560 mA・h/g. The charge/discharge curves for HS-V2O5 and HS-PPy/V2O5 hybrid electrodes (V(Pr)=0.03 mL) are compared in Fig. 4. At the beginning of the cycle, both samples exhibit three typical plateaus, corresponding to the coexistence of α+ε, ε+δ and δ+γ phases, respectively [4], which means that the modification with PPy causes negligible impact on the phase compositions of V2O5 during the charging/discharging process. A slight decrease in potential is observed for HS-PPy/V2O5 whose plateaus appear at ~3.4, 3.3 and 2.4 V analogous to other V2O5-based hybrids [15-16]. The decreased voltage seems to be independent of cathodic polarization but associated with a partial reduction of vanadium during the polymerization of the organic molecules [17]. The presence of lower valence state of vanadium may lead to a decrease in the amount of inserted Li+ and a farther decrease in the discharge capacity. The initial capacity of pure oxide achieves 324.3 mA・h/g but declines steeply as the retention after the 50th cycle is only 32.7%. On the other hand, despite of lower maximum capacity 236.4 mA・h/g, the PPy/oxide claims significantly improved cyclability with 89.1% capacity retention at the same cycling condition (in Fig. 5).

Fig. 2 SEM images of hollow microspheres of V2O5 (a, b) and PPy/V2O5(c, d)

Fig. 3 TEM images of hollow microspheres of V2O5 (a) and PPy/V2O5(b)
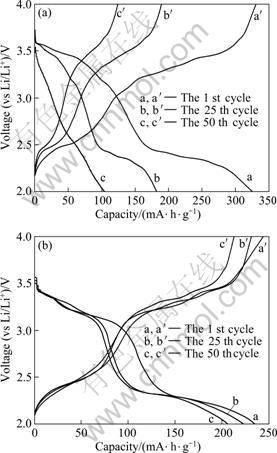
Fig. 4 Voltage―specific capacity curves of hollow microspheres of V2O5 (a) and PPy/V2O5 composite (b) at the 1st, 25th and 50th cycle charge/discharge at discharge rate of C/7
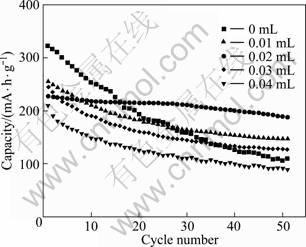
Fig. 5 Cycle behaviour of hollow microspheres of V2O5 and PPy/V2O5 produced by different dosages of pyrrole
It is important to note that there is so much difference in the character of discharge curves of two samples. The 50th discharge curve of pristine HS-V2O5 is sloping without any plateau, suggesting the inexistence of multiphase region in terms of thermodynamics. As for HS-PPy/V2O5 hybrid electrode, three distinct plateaus on the last discharge curve imply the maintenance of reversible phase transitions which depend on the amount of Li+ inserted into the host and on the relevant change in the crystal structure. Therefore, the enhanced cycling behavior can be partly attributed to the better reversibility of insertion/extraction of Li+, which probably results from the more stable structure supported by the PPy molecules as analyzed by XRD.
In addition, Fig. 5 shows that the capacity retention of PPy/c-V2O5 does not increase monotonously with the content of PPy but attains maximum when the injected amount of Py is 0.02 mL. Further research is needed to offer a convincing explanation for this phenomenon.
The Nyquist plots obtained for HS-PPy/V2O5 composite electrode compared with HS-V2O5 electrode in the full-charged state are shown in Fig. 6. As can be seen, two distinct regions are observed for both samples [18]: 1) a semicircle at high frequencies related to a charge-transfer process; 2) a Warburg linear region at intermediate frequency values, associated with a mass transfer process. It is obvious that the charge-transfer resistance (RCT) of PPy/c-V2O5 composite electrode is much smaller compared with that of V2O5 electrode, implying the larger exchange current density (J0) of surface electrochemical reaction in PPy/V2O5 composite electrode. This enhanced electrochemical activity can be ascribed to the presence of high conductive PPy, and also be beneficial for the overall electrochemical properties, especial the cyclability.
Fig. 6 Electrochemical impedance spectroscopic traces of hollow microspheres of V2O5 (a) and PPy/V2O5(b)
As mentioned earlier, Nyquist diagrams show a semicircle and a region at low frequencies characterized by a straight line with a phase angle of 45°. Starting from the equation bellow [19]:
 (2)
(2)
and using the slope of the impedance modulus |Z| as a function of the inverse of square root of angular frequency (ω-1/2), the apparent coefficient diffusion was determined for the three electrodes. Figure 7 shows the linear variation of the modulus of impedance versus the inverse of the ω-1/2. The values of diffusion coefficients were calculated from the slope. It can be seen from Fig. 7 that the slope of PPy/V2O5 electrode is smaller than that of HS-V2O5. It means that the diffusion coefficient of PPy/V2O5 electrode is greater than that of HS-V2O5. Thus, the insertion of PPy helps to open lamellar spacing of V2O5 and increases the dispersion speed of ions in lamella, which plays an important role in improving electrochemical properties of composite material, corresponding to the interlayer expansion analyzed in XRD as well.
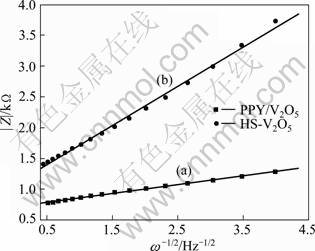
Fig. 7 Impedance modulus |Z| as function of square root of angular frequency (ω-1/2) for PPy/V2O5 (a) and HS-V2O5 (b) at different applied potentials
4 Conclusions
1) PPy/V2O5 hollow microspherical hybrid is prepared by injection and polymerization method. The conductive polymer lies between the V2O5 lamellas and does not strongly affect the morphology of V2O5 host.
2) Comparing with the bare HS-V2O5 electrode, this novel hybrid cathode exhibits slightly reduced capacity and substantially improved cyclability. The lower discharge capacity can be ascribed to the partial reduction of vanadium during the polymerization of polymer, and the greatly enhanced cycling performance is due to the better reversibility of insertion/extraction of Li+ and the better electrochemical activity, which presumably result from the more stable structure and the higher conductivity of HS-PPy/V2O5 composite, respectively.
References
[1] PASSERINI S, RESSLER J J, LE D B, OWENS B B, SMYRL W H. High rate electrodes of V2O5 aerogel [J]. Journal of Electroanalytical Chemistry, 1999, 44(13): 2209-2217.
[2] COUSTIER F, PASSERINI S, SMYRL W H. Dip-coated silver-doped V2O5 xerogels as host materials for lithium intercalation [J]. Solid State Ionics, 1995, 7(4): 780-785.
[3] BADDOUR R, PEREIRA-RAMOS J P, MESSINA R, PERICHON J. A thermodynamic, structural and kinetic study of the electrochemical lithium intercalation into the xerogel V2O5・1.6 H2O in a propylene [J]. Electroanalytical Chemistry, 1991, 314: 8l-101.
[4] FRITZ H, ROBERTO M T, DANIEL A B, JOSE E P S, SUSANA I C T. Electrochemical and Raman studies on a hybrid organic-inorganic nanocomposite of vanadium oxide and a sulfonated polyaniline [J]. Electrochimica Acta, 2001, 46: 3555-3562.
[5] HARRELD J, WONG H P, DAVE B C, DUNN B, NAZAR L F. Synthesis and properties of polypyrrole-vanadium oxide hybrid aerogels [J]. Journal of Non-Crystalline Solids, 1998, 225: 319-324.
[6] SUSUMU K, SHINGO M, HIDEMASA T. Charge-discharge properties of chemically prepared composites of V2O5 and polypyrrole as positive electrode materials in rechargeable Li batteries [J]. Electrochimica Acta, 2000, 46(1): 91-97.
[7] HUGUENIN F, GIROTTO E M, TORRESI R M, BUTTRY D A. Transport properties of V2O5/polypyrrole nanocomposite prepared by a sol-gel alkoxide route [J]. Journal of Electroanalytical Chemistry, 2000, 147(7): 2437-2444.
[8] SPAHR M E, NOV?K P, HAAS O, NESPER R, Electrochemical insertion of lithium, sodium, and magnesium in molybdenum(VI) oxide [J]. Journal of Power Sources, 1995, 54(2): 346-351.
[9] CEPAK V M, HULTEEN J C, CHE G, JIRAGE K B, LAKSHMI B B, FISHER R R, MARTIN C R, YONEYAMA H. Chemical strategies for template syntheses of composite micro- and nanostructures [J]. Chemistry of Materials, 1997, 9(5): 1065.
[10] HUANG H, REMSEN E E. Nanocages derived from shell cross-linked micelle template [J]. Advanced Materials, 1999, 121(15): 3085-3806.
[11] MANDAL T K, FLEMING M S, WALT D R. Production of hollow polymeric microspheres by surface-confined living radical polymerization on silica templates[J]. Chemistry of Materials, 2000, 12(11): 3481-3487.
[12] ZHU D, LIU H, LV L, YAO Y D, YANG W Z. Hollow microspheres of V2O5 and Cu-doped V2O5 as cathode materials for lithium-ion batteries[J]. Scripta Materialia, 2008, 59(6): 642-645.
[13] G?OMEZ-ROMERO P, TORRES-G?OMEZ G. Conducting organic polymers with electroactive dopants. Synthesis and electrochemical properties of hexacyanoferrate-doped polypyrrole [J]. Synthetic Metals, 1998, 98(2): 95-102.
[14] BOYANO I, BENGOECHEA M, de MEATZA I, MIGUEL O, CANTERO I, OCHOTECO E, RODR?GUEZ J, LIRA-CANT?M, G?MEZ-ROMERO P. Improvement in the Ppy/V2O5 hybrid as a cathode material for Li ion batteries using PSA as an organic additive[J]. Journal of Power Sources, 2007, 166(2): 471-477
[15] HITZKY E R. Conducting polymers intercalated in layered solids [J]. Advanced Materials, 1993, 5(5): 334-340.
[16] ZHU Ding, LIU Heng, YAO Ya-dong, YANG Wei-zhong. Fabrication and characterization of nanostructural vanadium pentoxide hollow microspheres [J]. Journal of Inorganic Materials, 2008, 23(1): 43-48.
[17] GOWARD G R, LEROUX F, NAZAR L F. Poly(pyrrole) and poly(thiophene)/vanadium oxide interleaved nanocomposites: Positive electrodes for lithium batteries[J]. Electrochem Acta, 1998, 43(10-11): 1307-1313.
[18] HO C, RAISTRICK I D, HUGGINS R A. Lithium ion conduction in Li5A1O4, Li5GaO4 and Li6ZnO4[J]. Electrochem Acta, 1980, 11(8): 343-350.
空心聚吡咯/V2O5复合材料的制备及其电化学性能
王远洪1, 刘 恒1, 朱 丁1, 郭再萍2, 刘华坤2, 窦诗学2
1. 四川大学 材料科学与工程学院,成都 610065;
2. Institute for Superconducting and Electronic Materials, University of Wollongong, NSW 2522, Australia
摘 要:为改善晶态V2O5 (c-V2O5)正极材料实际容量较低、循环性能较差等问题,制备了空心球聚吡咯/ V2O5复合材料。利用导电吡咯单体(Py)在中空型V2O5层间发生原位氧化聚合反应制备聚吡咯(PPy)/中空型V2O5复合材料(HS-PPy/V2O5)。采用X射线衍射仪(XRD)、扫描电镜(SEM)、透射电镜(TEM)对样品进行表征,采用恒流充放电测试和电化学阻抗(EIS)测试样品的电化学性能。结果表明,Py单体已插入中空型V2O5层间,与纯中空型V2O5相比,制备的HS-PPY/V2O5复合材料比容量虽然有所减小,但是循环稳定性有较大的提高。
关键词:V2O5;空心微球;正极;锂离子电池
(Edited by LI Xiang-qun)
Foundation item: Project (50574063) supported by the National Natural Science Foundation of China
Corresponding author: LIU Heng; Tel: +86-28-85410272; Fax: +86-28-85413003; E-mail: h_liu@scu.edu.cn
DOI: 10.1016/S1003-6326(11)60857-1