J. Cent. South Univ. Technol. (2010) 17: 674-682
DOI: 10.1007/s11771-010-0539-8
Effect of Nb on plasticity and oxidation behavior of TiAlNb intermetallic compound by density functional theory
LI Yan-feng(李燕峰)1, 2, XU Hui(徐慧)1, 2, SONG Zhao-quan(宋招权)1, MA Song-shan(马松山)2
1. School of Materials Science and Engineering, Central South University, Changsha 410083, China;
2. School of Physical Science and Technology, Central South University, Changsha 410083, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2010
Abstract: Based on the pseudo potential plane-wave method of density functional theory (DFT), Ti1-xNbxAl (x=0, 0.062 5, 0.083 3, 0.125, 0.250) crystals’ geometry structure, elastic constants, electronic structure and Mulliken populations were calculated, and the effects of doping on the geometric structure, electronic structure and bond strength were systematically analyzed. The results show that the influence of Nb on the geometric structure is little in terms of the plasticity, and with the increase of Nb content, the covalent bond strength remarkably reduces, and Ti-Al, Nb-M (M=Ti, Al) and other hybrid bonds enhance; meanwhile, the peak district increases and the pseudo-energy gap first decreases and then increases, the overall band structure narrows, the covalent bond and direction of bonds reduce. The population analysis also shows that the results are consistent with the electronic structure analysis. The density of states of TiAlNb shows that Nb doping can enhance the activity of Al and benefit the form of Al2O3 film. All the calculations reveal that the room temperature plasticity and the antioxidation properties of the compounds can be improved with the Nb content of 8.33%-12.5% (mole fraction).
Key words: TiAl; density functional theory; Nb doping; plasticity; oxidation behavior
1 Introduction
TiAl-based alloy, especially the TiAl intermetallic compound, is considered to be one of the most promising heat-resistant structural materials in the space of light aircraft engines and automotive components because of its low density, high specific strength and elastic modulus, and good creep resistance [1-2]. However, the weak high temperature oxidation resistance and room temperature plasticity of the compound are the major drawbacks for its wider application.
Previous studies show that alloying is an effective method to improve the oxidation resistance and room temperature plasticity of TiAl [3]. DONCHEV et al [4] found that TiAl alloy’s high-temperature oxidation resistance can be enhanced by handling of halogen (F, Cl, Br and I). JIANG et al [5] reported that Si and Nb can improve its high temperature oxidation, too. TEM observations [6] of the oxidation of Nb-ion-implanted TiAl indicated that the implantation of Nb is effective to generate very ?ne Al2O3 scales grains at 1 200 K, which improves the oxidation resistance of TiAl. Through the research on the influence of the content of the doping atom, LIN et al [7] found that a high content of Nb can make TiAl compound have a good high temperature oxidation resistance. Studies showed that Mn [8], V [9], Cr+Nb [10] can also improve the TiAl alloy’s low-temperature plasticity. However, some of the researched elements to enhance ductility can reduce the oxidation, such as Y, Cr and Mn [11]. And research on the comprehensive impacts on the elements is still lacking [12].
Many papers based on the theoretical calculation of TiAl and TiAlNb have been published [13-16].WU et al [13] employed the first-principle method to study the effect of Nb on the bonding characteristics of TiAl intermetallic compound with the emphasis on the electronic interactions between Ti (Al) and O atoms. Phase interface in multiphase Ti3Al-based alloys [14] was analyzed by improved Thomas-Fermi-Dirac (TFD) theory, which calculated and analyzed the preliminary interpretation of the phase interface electronic structure and bonding of the micro-impact toughness of alloy from the level of electronic structure. DANG et al [15] studied the site substitution behavior of the 4d transition elements and the alloying effect of Nb and Mo by using the first-principle discrete variational (DV) and Dmol3 method, and found that Nb and Mo gave rise to strong interaction and charge transfer with the neighboring host atoms of TiAl, resulting in a strong solution strengthening effect.
LI and LIU [16] calculated electronic structure of Ti3Al with Nb doped. The result indicated that Ti3Al-based alloys with the addition of macro-alloyed Nb cause a partly disordered α2 phase, incorporating the toughening phase while weakening the Ti-Ti covalent bond, which increases α (uniform deforming factor) and Gc (cleave energy) and thus the embrittlement of Ti3Al-based alloys as well as TiAl, is basically improved [17].
All these researches are based on a specified content of Nb doped and give an ordinary conclusion about the electronic band and structure, the influence of the Nb content is neglected, except that LIU et al [18] studied the effect of large range Be addition (0-20.83% Be, mole fraction) on the lattice distortion and dislocation mobility in TiAl by DFT calculations. Particularly, the systematic research of plasticity and oxidation mechanism by Nb doping with changing content on the TiAl intermetallic compound is still lacking [19].
In this work, Ti1-xNbxAl (x=0, 0.062 5, 0.083 3, 0.125, 0.250) crystal structures are relaxed, and their elastic moduli and electronic structures are calculated. The change of the TiAlNb ternary intermetallic compounds’ bonding by doping Nb is discussed, and the influences on the high-temperature oxidation resistance and plasticity are systematically analyzed.
2 Calculations
2.1 Models and parameters
γ-TiAl belongs to the Berthollide type intermetallic compounds, which can keep in an ordered state at any temperature by solid state. It consists of Ti and Al atoms that alternate in (002) surface, and its lattice constants in [100] and [010] direction are different from those in [001] direction, for its structure belongs to face-centered cubic crystal. The crystal’s parameters, which belong to space group 123 (P4/MMM) [20], are as follows: a= 4.046 5×10-10 m, b=4.046 5×10-10 m, c=4.095 0×10-10 m, and α=β=γ=90?. The crystal structure is shown in Fig.1 (the white balls represent Ti, the gray balls represent Al, and the black balls represent Nb).
Theoretical and experimental results show that Nb in γ-TiAl tends to occupy the Ti site [21-22] and generate a new ternary intermetallic compound [23] easily. So in this work, the supercells of Ti1-xNbxAl are built (x=0, 0.062 5, 0.083 3, 0.125, 0.250) and the cell structures are shown in Fig.1.
All calculations are based on pseudo-potential plane wave method of density functional theory (DFT). BFGS (Broyden-Fcetcher-Goldfarb-Shanne) algorithm is used to relax the crystal structures, and the exchange- correlation contribution is described within the generalized gradient approximation (GGA) proposed by PERDEW et al (PW91) [24]. The Vanderbilt ultra-soft pseudo-potential is used to describe the ion-solid interaction with the valence electrons. The valence electron of Al is 3s23p1, while that of Ti and Nb is 4s23d2 and 4d45s1, respectively. Kohn-shan orbits are expanded with plane wave and the cut-off energy (Ecut) is set to 280 eV, the self-consistent calculation accuracy is set to 1.0×10-5 eV/atom, and the K-point in Brillouin zone is set to 5×5×5 by using Monkhorst-Pack form.
2.2 Geometric parameters and elastic constants
The lattice constants and mechanical constants of Ti1-xNbxAl after optimization are shown in Table 1. From Table 1, it can be seen that the average volume of cell increases with the increase of Nb content, but the axial ratio (c/a) tends to decrease.
Because the anisotropy caused by axial ratio is related to the dislocation motion in crystal, the decrease of the lattice axial ratio can improve the plasticity of TiAl [8]. As shown in Fig.2(a), c/a decreases overall with the increase of Nb content. It is known that lower c/a can reduce the anisotropy of crystals, which is helpful for the plastic deformation. So, it seems that Nb is beneficial to improving the plasticity of the alloys. However, in a certain range, the average volume of cell becomes larger by the increase of Nb content (Fig.2(b)). γ-TiAl phase has 60%-70% of the metal bond and 30%-40% covalent bond [25]. The larger cell volume will reduce the bond strength of fracture strength and the plastic deformation will be reduced. Thus, the geometry changed by Nb doping is not the main reason for TiAlNb’s plasticity.
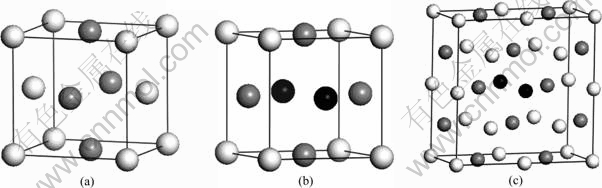
Fig.1 Crystal structures of compounds: (a) TiAl; (b) TiAl2Nb; (c) Ti7Al8Nb
Table 1 Geometric parameters (a, c), bulk modules (B) and elastic modules (E) of crystal after optimization


Fig.2 Curves of parameters at different Nb contents: (a) Axis ratio (c/a); (b) Average cell volume; (c) Bulk modulus; (d) Elastic modulus along each axis
Figs.2(c) and (d) show the curves of bulk modulus B and elastic modulus with each axis (Ex, Ey, Ez). The bulk modulus B significantly increases with the increase of Nb doping content, which indicates that the strength of the system increases, as well as Ez; but Ex and Ey show concussion-typed changes. It can be seen that when the Nb content is below 6.25% (mole fraction), Ex and Ey are significantly decrease, while Ez increases, which means that the mechanical anisotropy can be reduced. When the Nb content is about 1.72%, the three basic components of elastic modulus are equal. With the increase of Nb content, the gap between the elastic modulus of three components becomes larger and the maximum appears in 6.25% of Nb content, then the three components tend to converge. When the Nb doping content is above 15%, Ex and Ey show a decreasing trend, while Ez continues to increase, which causes the significantly promoted anisotropy. Overall, its mechanics anisotropy shows oscillating changes with the increase of Nb content.
Thus, the doping of Nb improves the strength of the compound and changes the bond strength in different directions, which directly influences the dislocation and pinning. Geometric analysis shows that, when the Nb content is 1.72% or 8.33%-12.5%, the ternary intermetallic compounds tend to be isotropic and have better plasticity, and the other proportion of Nb content will result in the opposite effect.
3 Results and discussion
3.1 Density of states (DOS) of TiAlNb
The metal bond can enhance the ductility and plasticity, while the covalent bond can produce inhibition of plasticity because of its fixed direction and great strength of bond, so the changes of the system’s covalent with the Nb joining are particularly analyzed. Figs.3-7 show the DOS of Ti1-xNbxAl. Because the DOS distribution near the Fermi level determines the properties, attention is especially paid to the DOS in the vicinity of the Fermi level, which is set to zero. The Fermi level corresponds to the vertical dash-dot line.
Fig.3 shows the total DOS (TDOS) and partial DOS (PDOS) for TiAl compound. The TDOS has a strong symmetry by the Fermi level: in the vicinity of the Fermi level, the DOS is mainly provided by the Ti 3d orbital, with a little Al 3p orbital, such as the peaks at -0.5 eV and 2.0 eV, which consist of Ti 3d-Ti 3d bonds; when it is slightly away from the Fermi level, it is mainly provided by the hybrid bonds consisting of Ti 3d and Al 3p, such as the peaks at -2.5 eV and 3.8 eV; and in the farther area (<-2.5 eV or >5 eV) of the Fermi level, the DOS is mainly provided by the Al 3p orbital, with a little hybrid bonds by Ti 3d and Al 3p electrons.
Figs.4-7 show the TDOS for Ti1-xNbxAl (x= 0.062 5, 0.083 3, 0.125) and PDOS for s-, p- and d- electrons of them. The above analysis is similar to the DOS for Ti1-xNbxAl. In the vicinity of the Fermi level, it is noted that the DOS is mainly provided by the Ti 3d and Nb 4d orbital, the bonds change from Ti 3d-Ti 3d to Ti 3d-Ti 3d and Ti 3d-Nb 4d hybrid bonds, and the Fermi level located in the left peak of the pseudo-energy gap and the Fermi value is reduced gradually with the increase of Nb content. When it is slightly away from the Fermi level, the bonds change from Ti 3d-Al 3p to Ti 3d-Al 3p and Al 3p-Nb 4d hybrid bonds. And the hybrid bonds of Al 3p-Nb 4d increase in the low energy-level area of the Fermi level.
The present calculation shows that the Nb doping significantly inhibits the Al―Al covalent bond in the high energy-level area (>5 eV) and the energy broadening becomes narrower distinctly than that of the compound without Nb doping.
Particularly, for Al atom(Fig.3(c)), the DOS is mainly provided by the Al 3p orbital in the vicinity of the Fermi level of TiAl cell, then, s- and p- orbital co-exist in the low energy-level area (≤-2.5 eV) and high energy- level area (>2.5 eV). After Nb is doped (e.g. Fig.4(c)), the PDOS of Al is obviously changed that s- and p- electrons are substantially reduced in the high energy level area.

Fig.3 Total and partial DOS for TiAl: (a) DOS of TiAl; (b) DOS of Ti in TiAl; (c) DOS of Al in TiAl

Fig.4 Total and partial DOS for Ti7Al8Nb: (a) DOS of Ti7Al8Nb; (b) DOS of Ti in Ti7Al8Nb; (c) DOS of Al in Ti7Al8Nb; (d) DOS of Nb in Ti7Al8Nb
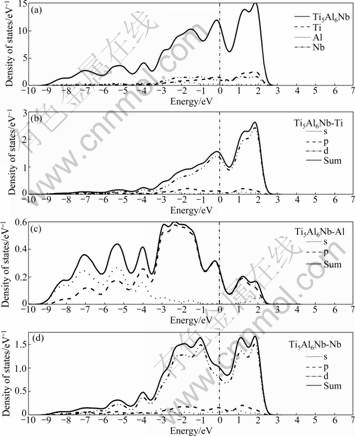
Fig.5 Total and partial DOS for Ti5Al6Nb: (a) DOS of Ti5Al6Nb; (b) DOS of Ti in Ti5Al6Nb; (c) DOS of Al in Ti5Al6Nb; (d) DOS of Nb in Ti5Al6Nb
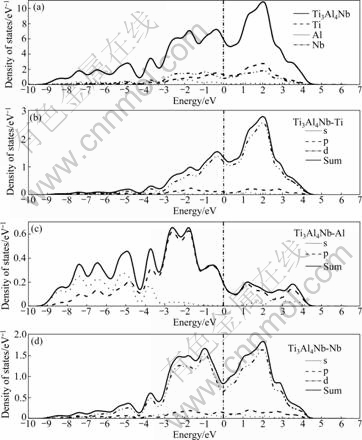
Fig.6 Total and partial DOS for Ti3Al4Nb: (a) DOS of Ti3Al4Nb; (b) DOS of Ti in Ti3Al4Nb; (c) DOS of Al in Ti3Al4Nb; (d) DOS of Nb in Ti3Al4Nb
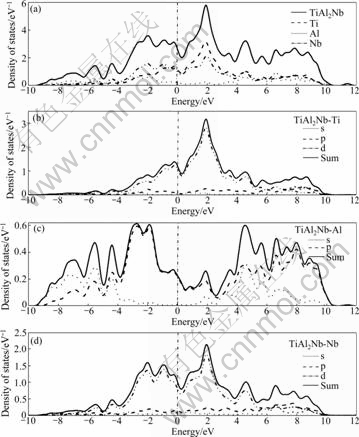
Fig.7 Total and partial DOS for TiAl2Nb: (a) DOS of TiAl2Nb; (b) DOS of Ti in TiAl2Nb; (c) DOS of Al in TiAl2Nb; (d) DOS of Nb in TiAl2Nb
3.2 Band structure of TiAlNb
Figs.8-12 show the band structures of Ti1-xNbxAl, which offer an intuitive observation of the band change.
It is noted that the difference between the upper part and the lower part of Fermi level is not significant without Nb doing (Fig.8) and a great content of Nb doping (Fig.12), reveals a clearly parabolic-type curve, which typically indicates that the system has covalent bond components and exhibits strong covalent. Meanwhile, in the other doping conditions, such as Figs.9-11, the energy bands are continuous and intensive across the Fermi level, which shows a strong metallic nature.
It can also be seen that, the energy band with Nb doping is clearly narrower than that without Nb doping, much smaller, which indicates that Nb doping increases in particular the fluctuations of conduction band (CB) are the effective mass of the electrons, reduces the non-local degree, and weakens the expansion of atomic orbital, and the covalent degree.

Fig.8 Energy band structure for TiAl

Fig.9 Energy band structure for Ti7Al8Nb
From the DOS calculation we know that the conduction band is mainly composed of Al―Al bond. Thereby, it can be concluded that the Nb doping inhibits the p-valence electrons of Al atom.

Fig.10 Energy band structure for Ti5Al6Nb
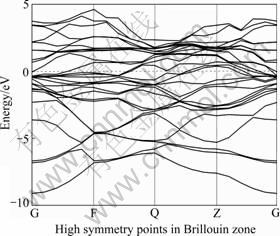
Fig.11 Energy band structure for Ti3Al4Nb
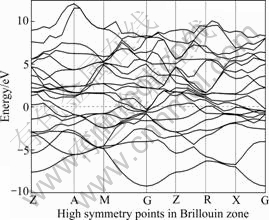
Fig.12 Energy band structure for TiAl2Nb
3.3 Impact on property
According to the calculations, it is learned that with the increase of Nb content, the hybrid bond becomes more complex. Despite the decrease in the peak near the Fermi level, the peak area increases, so as to enhance the bonding strength. With a little Nb doping, Nb in the site of Ti inhibits the Al―Al covalent bond, so the pseudo-energy gap is reduced, which demonstrates the reduction of the system’s covalent (Fig.4). And then, when the Nb content reaches a certain level (e.g. more than 25%, see Fig.7) as a main component, the Al―Al covalent bond can be strengthened to enhance the binding energy and the ordered energy of the system [26], which may promote the system’s covalent.
Therefore, it is believed that in the initial period of doping, Nb plays a role in inhibiting Al―Al covalent bond. The hybrid bond (Ti 3d-Nb 4d and Al 3p-Nb 4d) enhances the strength of the system and reduces the anisotropy of the bonding in order to improve the plasticity of the system. When the Nb content is increased to 25% or more, the bonding of system changes enormously by the formation of a new phase and Nb begins to play a role in enhancing the Al―Al covalent bond, so the covalent of the system improves, and the plasticity is deteriorated.
On the other hand, since the free energies of the formation of TiO2 and Al2O3 are very close, it is always easy to generate a mixture of TiO2 and Al2O3 on the surface, which destroys the dense oxide layer by Al2O3 [27]. Thus, the key factor to improve the oxidation behavior of the ternary intermetallic compound is to enhance the activity of Al and inhibit oxidation of Ti.
By comparing electronic structure of Al before and after doping (Fig.3(c) and Fig.4(c)), it is noted that Al s- electrons are scattered in the energy levels of -2.5- -10.0 eV and 2.5-10.0 eV before Nb is doped. After Nb is added, the peak below the Fermi level moves closer to the Fermi surface, respectively, from -2.5 eV, -5.5 eV and -7.0 eV to -1.5--2.0 eV, -4.5 eV and -5.0--3.0 eV, and forms several peak districts. The changes indicate that Nb doping enhances the density of s- and p- electrons of Al in the vicinity of the Fermi level, which increases their interactions and is helpful for the interaction of Al and O atoms, as well as the formation of Al2O3-oxide-film.
Fig.3(b) and Fig.4(b) show the electronic structures of Ti. The peak decreases significantly in the vicinity of the Fermi level, and the value of the Fermi level EF becomes slightly lower with the Nb doping, which indicates that the electrons in the vicinity of Fermi level are less and the reaction activity is slowed. But, there is no evidence that the change of Nb content can take great changes for the antioxidation properties of the compounds. The conclusion is consistent with the experimental and theoretical analysis [12-13].
3.4 Mulliken populations
The calculated Mulliken populations of the systems are given in Table 2, which can be used to analyze the bonding strength.
When Nb is not added, the Al―Al covalent bond is the strongest bond in TiAl compounds, and the Ti―Al hybrid bond is the weakest one, in which the bond strength of Al―Al and Ti―Ti is about 437.9% and 272.4% of that of Ti―Al hybrid bond. So, the bonds create a greater anisotropy, which is the reason for its poor room temperature plasticity. With Nb doped in the site of Ti, the Ti―Ti bond and the Al―Al covalent bond are conspicuously reduced, which is easy to activate the dislocation and reduce the stacking fault energy to induce the twinned crystal; meanwhile, the hybrid bond including Al―Ti strength is enhanced, so does the strength of the system.
In this work, the average level of bonding strength is described by the data of standard deviation. The present calculation shows that the standard deviation is the smallest when the Nb content is 6.25% to 12.5%, which is in agreement with the conclusion by DOS and bonding analysis.
4 Conclusions
(1) Mechanical parameter shows that the Nb content is an element that impacts the anisotropy. When the Nb content is about 1.72% or 8.33%, the three basic components of elastic modulus are close or even equal. Geometric analysis also indicates that the anisotropy and plasticity of the ternary compounds may be better when the Nb content is 8.33%-12.5%.
(2) Nb doping in the site of Ti causes a strong
hybrid bond. When Nb is doped, the main bonds in the vicinity of the Fermi level change from Ti―Ti to Ti―Ti and Ti 3d-Nb 4d hybrid bond; the main bonds in the slightly away from the Fermi level change from Ti 3d- Al 3p to Ti 3d-Al 3p and Al 3p-Nb 4d hybrid bonds; Al 3p-Nb4 hybrid bonds increase greatly in the low energy-level district and Al―Al covalent bond in high energy-level district is inhibited. All these changes of bonds reduce the anisotropy of the TiAlNb ternary compounds. Mulliken populations show that Ti―Ti and Al―Al band strength declines and Ti―Al hybrid bond strength increases. And the calculated standard deviation of the bond strength is very small when the content of Nb doping is 6.25%-12.5%.
Table 2 Mulliken population of systems

(3) An analysis of the DOS and PDOS for TiAlNb reveals that, Nb not only reduces the electron density of Ti atom near Fermi energy level, but also enhances the interactions between Al (s) electrons and O (p) electrons. When the Nb content is 8.33%-12.5%, TiAlNb ternary compounds have better plasticity and oxidation behavior than TiAl compounds. However, the phase transformation by high temperature and high Nb content need further research.
References
[1] DEY S R, SUWAS S, FUNDENBERGER J J, RAY R K. Evolution of crystallographic texture and microstructure in the orthorhombic phase of a two-phase alloy Ti-22Al-25Nb [J]. Intermetallics, 2009, 17(8): 622-633.
[2] KRUMLT, OBRTLIKK, PETRENECM, POLAKJ. Low-cycle fatigue properties of TiAl alloy with high Nb content [J]. International Journal of Materials Research, 2009, 100(3): 349-352.
[3] WU Xin-hua. Review of alloy and process development of TiAl alloys [J]. Intermetallics, 2006, 14(10): 1114-1122.
[4] DONCHEV A, RICHTER E, SCHUTZE M. Improvement of the oxidation behaviour of TiAI-alloys by treatment with halogens [J]. Intermetallics, 2006, 14(10): 1168-1174.
[5] JIANG Hui-ren, WANG Zhong-lei, MA Wen-shuai. Effects of Nb and Si on high temperature oxidation of TiAl [J]. Trans Nonferrous Met Soc China, 2008, 18: 512-517.
[6] SHIGEJI T, ZHU Y C, KAZUHISA F, NOBUYA I. TEM observations of the initial oxidation stages of Nb-ion-implanted TiAl [J]. Oxidation of Metals, 2002, 58(3/4): 375-390.
[7] LIN Jun-pin, XU Xiang-jun, WANG Yan-li, SONG Xi-ping, CHEN Guo-liang. High temperature deformation behaviors of a high Nb containing TiAl alloy [J]. Intermetallics, 2007, 15(5): 668-674.
[8] KONG Fan-tao, CHEN Zi-yong, TIAN Jing, CHEN Yu-yong. Methods of improving room temperature ductility of TiAl based alloys [J]. Rare Metal Materials and Engineering, 2003, 32(2): 81-86. (in Chinese)
[9] JIN Guang-xi, QIAO Li-jie, GAO Ke-zuo, MU Cun-long, QIAO Ben-jian, ZUO Wu-yang. Effect of Mn and V on hot corrosion of TiAl alloy [J]. Acta Metallurgica Sinica, 2004, 40(2): 179-184.
[10] XIAO Dai-hong, HUANG Bai-yun. Superplastic behavior and microstructure evolution of as-cast Ti-47Al-8Cr-2Nb alloy at lower temperature [J]. The Chinese Journal of Nonferrous Metals, 2008, 18(10): 1749-1755. (in Chinese)
[11] WANG Fu-hui, TANG Zhao-lin. Oxidation and protection of TiAl intermetallics [J]. Chinese Journal of Materials Research, 1998, 12(4): 337-344. (in Chinese)
[12] JUNG H G, KIM K Y. Effect of ternary elements on the oxidation behavior of aluminized TiAl alloys [J]. Oxidation of Metals, 2002, 58: 197-201.
[13] WU Hong-li, ZHANG Wei, GONG Sheng-kai. Effect of Nb on the bonding characteristics of TiAl intermetallic compounds: A first-principle study [J]. Acta Chimica Sinica, 2008, 66(14): 1669-1675. (in Chinese)
[14] QU Hua, LIU Wei-dong, ZHANG Kun, LIU Zhi-lin. Study on the valence electron structure of 0001α//110β interface in Ti3Al-based alloys [J]. Rare Metal Materials and Engineering, 2005, 34(10): 1569-1573.
[15] DANG Hong-li, WANG Chong-yu, YU Tao. First-principles investigation on alloying effect of Nb and Mo in γ-TiAl [J]. Chin Phys Soc, 2007, 56(5): 2838-2844.
[16] LI Wen, LIU Gui-fu, KONG Xiao-hua. Valence electron structures and alloying behavior of Ti-aluminides [J]. Acta Armamentarii, 1999, 20(2): 151-155. (in Chinese)
[17] XIE Hua, CHEN Wen-zhe, QIAN Kuang-wu. Influence of manganese and niobium on electronic structure and brittleness of TiAl [J]. Rare Metals, 2004, 28(2): 350-353.
[18] LIU Yong-li, LI Hong, ZHANG Lin, WANG Shao-qing, YE Heng-qiang. First-principles study of Be content on the structural and mechanical properties of TiAl alloy [J]. Journal of Alloys and Compounds, 2009, 475: 401-407.
[19] ZHANG Lan-zhi, WANG Bao-yi, WANG Dan-ni, WEILong, LINJun-pin, WANGWen-jun. Study of doping effect of Nb in TiAl alloy by position annihilation spectrum [J]. Acta Metall Sin, 2007, 43(3): 269-272.
[20] NOVOSELOVA T, MALINOV S, SHA W, ZHECHEVA A. High-temperature synchrotron X-ray diffraction study of phases in a gamma TiAl alloy [J]. Materials Science and Engineering A, 2004, 371: 103-112.
[21] SCABAROZI T H, GENNAOUI C, ROCHE J, FLEMMING T, WITTENBERGER K, HANN P, ADAMSON B, ROSENFIELD A, BARSOUM M W, HETTINGER J D, LOFLAND S E. Combinatorial investigation of (Ti1-xNbx)2Al [J]. Applied Physics Letters, 2009, 95(10): 101907-3.
[22] TALA′AT A K, YONG Y, CHRISTIAN K, TORBEN B, LIU Z G. Investigation of the ordering and atomic site occupancies of Nb-doped TiAl/Ti3Al intermetallics [J]. Surf Interface Anal, 2007, 39: 257-261.
[23] CHEN Guo-liang, WANG Jin-guo, LIN Jun-pin. A new intermetallic compound in TiAl+Nb composition area of the Ti-Al-Nb ternary system [J]. Intermetallics, 2005, 13(3): 329-333.
[24] PERDEW J P, CHEVARY J A, VOSKO S H, JACKSON K A, PEDERSON M R, SINGH D J, FIOLHAIS C. Atoms, molecules, solids, and surfaces: Applications of the generalized gradient approximation for exchange and correlation [J]. Phys Rev B, 1992, 46: 6671-6687.
[25] XING Fei, LIU Yan-ping, HAN Pei-de, LIU Xiao-ping. First-principles study on diffusion mechanisms of Nb and C atoms on TiAl (010) surface [J]. New Technology and New Process, 2008, 11: 86-89. (in Chinese)
[26] ZHANG Wei, LIU Yong, HUANG Jin-song, LIU Bin, HE Yao-hui. Research progress and prospects for refractory TiAl alloy with high Nb content [J]. Rare Metals Letters, 2007, 26(8): 1-6. (in Chinese)
[27] SHEN Y, DING X F, WANG F G, TAN Y, YANG J M. High temperature oxidation behavior of Ti2Al2Nb ternary alloys [J]. Journal of Materials Sciences, 2004, 39: 6583-6588.
Foundation item: Project(07JJ3102) supported by Hunan Provincial Natural Science Foundation, China; Project(k0902132-11) supported by Changsha Municipal Science and Technology, China
Received date: 2009-11-27; Accepted date: 2010-02-25
Corresponding author: XU Hui, PhD, Professor; Tel: +86-731-88879207; E-mail: xuekb3@mail.csu.edu.cn
(Edited by YANG You-ping)