
Flotation separation of Xixia andalusite ore
ZHOU Ling-chu, ZHANG Yi-min
College of Resources and Environmental Engineering,
Wuhan University of Science and Technology, Wuhan 430081, China
Received 10 June 2010; accepted 29 October 2010
Abstract: The flotation behavior of Xixia andalusite was studied with different collectors. The tests were conducted with a pure andalusite sample in micro-flotation cell and then with an ore sample in a mechanical flotation cell. The surface charge was measured on pure andalusite sample and the isoelectric point (IEP) was found to be pH 5.2. Andalusite is floated with an amine collector above IEP where mineral surface is negatively charged. In contrast, an alkyl sulfonate collector (sodium dodecyl benzene sulfonate) is effective in the pH range where andalusite surface is positively charged. Furthermore, andalusite is floated with oleate in neutral and mildly alkaline pH values. 55.3% Al2O3 is produced with 75.6% andalusite recovery when using the alkyl benzene sulfonate collector. Starch is found to be an effective depressant for gangue minerals in alkyl benzene sulfonate flotation. Moreover, slimes are found to have an adverse effect on the flotation of andalusite. Therefore, pretreatment by desliming and pre-float (to remove carbonaceous species) prior to andalusite flotation is necessary.
Key words: andalusite; flotation; collectors; desliming; pre-float; isoelectric point (IEP)
1 Introduction
Although three anhydrous aluminum silicate minerals, andalusite, kyanite and sillimanite, are of the same chemical composition, Al2O3・SiO2, they have different crystal structures and different physical properties. Andalusite, with orthorhombic crystals, is one of the most important minerals used in the production of advanced refractory material [1-2].
Andalusite ores usually contain 5%-15% andalusite. Other minerals contained in the ores are mainly quartz, biotite and garnet [3-4]. The ores must be beneficiated in order to be used for the refractory material industry. In general, the andalusite concentrate contains not less than 54% Al2O3 and not more than 42% SiO2, 1.5% Fe2O3, 2% TiO2, 0.1% CaO and 0.1% MgO [5]. Currently, the beneficiation of andalusite ores is commercially realized by gravity concentration, magnetic separation and froth flotation [6-9].
Flotation is the most commonly used method for fine-grained andalusite beneficiation. There are many reagents for andalusite flotation. For example, at pH 4, oleic acid, linoleic acid and linolenic acid can all be used as andalusite collectors [9]. Research has shown that when a small amount of biotite is contained in an andalusite ore, other anionic collectors, such as naphthenic acid or its salt, can be used to float andalusite in acidic media [3-4]. A French company, DAMREC, reported in 1987 that, at pH ≤3.5 and solids 15%-30%, alkyl sulfonate can be used as a collector for andalusite flotation. An andalusite concentrate of the highest standard, the KF class, assaying 60% Al2O3 and 0.6% Fe2O3, was produced. This method was patented in Europe in 1989 (Patent No. 238479). After dissolving the aluminum ions from the surface with dilute acid, a cationic collector, dodecylamine acetate, is a very good collector for andalusite at pH 7 [3-4].
The Xixia andalusite mine, located in the Henan province of China, is a large sedimentary metamorphic deposit. It is of a simple ore type. Most of the deposits are porphyritic biotite and quartz schist. The flowsheet of the operating consists of selective grinding, scrub desliming, heavy liquid pre-concentration and dry high-intensity magnetic separation. A coarse concentrate is produced with 55%-58% Al2O3 and 27.9% recovery (concentrate mass recovery of 2.7%). Fine andalusite particles lost in tails must be recovered in order to increase andalusite recovery. The objective of this research is to understand the flotation behavior of andalusite and then to develop a flowsheet to recover lost fine andalusite particles by means of flotation.
2 Experimental
In this research work, the flotation behavior of andalusite was investigated. The ore sample was collected from Yangnaigou zone of Xixia mine, Henan province, China. In the first stage of the study, micro-flotation tests were conducted on pure andalusite sample obtained by hand-sorting. In the second stage, batch and continuous flotation tests were conducted on the ore sample. The results of chemical analysis of test samples are given in Table 1.
According to the chemical analysis, the pure mineral sample contained 96.2% andalusite. Mineralogical examinations on the ore sample showed that quartz, biotite and garnet were the main gangue minerals. Other impurities were muscovite, staurolite, almandine, plagioclase, chlorite, and carbonaceous materials. A minute amount of titanium indicates that this ore could be suitable for the manufacture of refractory materials.
Micro-flotation tests were conducted using an XFG micro-flotation cell (40 mL). The mineral sample was dry ground using an agate mortar and pestle and then dry screened. The fraction in -0.104+0.04 mm was used for the micro-flotation tests. 5 g sample was used in each test. Flotation properties of andalusite were determined with respect to pH using different types of collectors. Surface charge of pure andalusite was measured against pH using a JS94J enhanced micro-electrophoresis meter.
Small-scale batch flotation tests on the ore sample were performed in a 0.5 L XFD mechanical flotation cell. The mass of the ore sample used in each test was 250 g. Dodecylamine (DDA), sodium dodecylbenzene sulfonate (SDBS) and sodium oleate (NaOl) were used as the flotation collectors. Continuous flotation tests on the ore sample were performed in a series of 0.5-1.0 L lab-used miniature continuous mechanical flotation cells. The equipment had a capacity of 3 kg/h and was run in 24 h continuous operation. The flotation conditions were similar to those used in the batch flotation tests.
3 Results and discussion
3.1 Micro-flotation of pure mineral sample
Previous micro-flotation tests were conducted with each of the three collectors at varying dosages. Then the optimum dosage for each collector was chosen to test the effect of flotation pH. The results of pH tests are shown in Fig. 1 which also presents the optimum dosage for each collector, i.e. 10 mg/g for DDA, 1 mg/g for SDBS and 2 mg/g for NaOl.
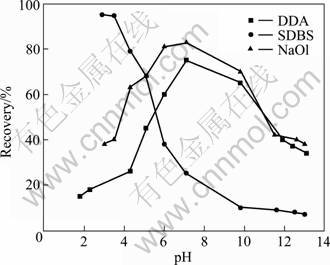
Fig. 1 Results of andalusite flotation using 10 mg/g DDA, 1 mg/g SDBS and 2 mg/g NaOl
It is found that DDA is a cationic collector, giving relatively better flotation results in alkaline medium; SDBS is an anionic collector with more effective in acidic pH, especially pH 3-4; NaOl, another anionic collector, floats andalusite the best in the range of pH 6-9.
The zeta potential of andalusite is given in Fig. 2 as a function of pH. It is seen that the IEP of andalusite is around pH 5.2.
3.2 Ore flotation
Since SDBS had shown more favorable flotation performance than DDA and NaOl in micro-flotation of pure mineral sample, only SDBS was used in the ore flotation tests. In the micro-flotation tests, SDBS of analytical grade was used. In the ore flotation tests, industrial petroleum sulfonate was used. The main component of the petroleum sulfonate is SDBS.
Table 1 Chemical analysis of pure andalusite and andalusite ore sample
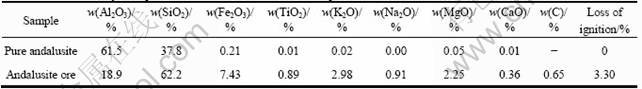
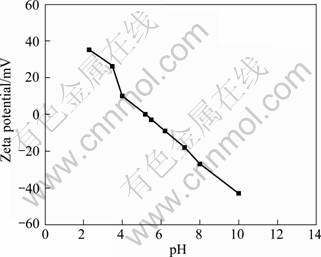
Fig. 2 Zeta potential of andalusite as function of pH
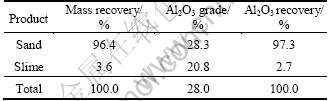
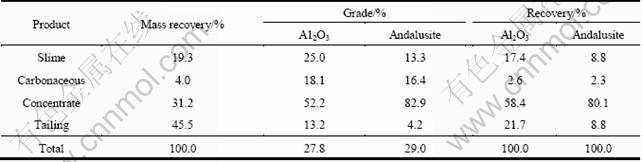
Previous scoping tests indicate that for the Xixia andalusite ore, desliming and pre-float (to remove carbonaceous species) before andalusite flotation were necessary, and the best flotation results were obtained in acidic pH range. Therefore the sample was ground to be less than 0.076 mm using a steel ball mill and deslimed before flotation. The material balance of slimes and the sand is presented in Table 2.
Table 2 Material balance of slimes and sand
Batch flotation tests were carried out with petroleum sulfonate at pH 3.5 since micro-flotation tests had showed that the best flotation results using SDBS was achieved at pH 3-4. Batch flotation tests showed that the optimum dosage for petroleum sulfonate at pH 3.5 was 3 200 g/t. The results can be seen in Table 3. All tests conducted later were carried out under these conditions.
Modifiers such as sodium silicate (Na2SiO3) and starch were tested to enhance separation. Sodium silicate was first tested as a depressant for gangue minerals but proved to be ineffective. Then the starch was tested. The results can be seen in Table 4. The test results show that starch was good depressant for the selective flotation of andalusite in this ore when petroleum sulfonate was used as collector.
Following simple one-stage flotation tests, additional flotation tests were conducted. The flowsheets are opened-circuit flowsheet with multi-stage cleaning. The results are presented in Tables 5-7.
Table 5 indicates the importance of adding an appropriate amount of H2SO4 during the cleaner stages in order to maintain the slurry pH at about 3.5. This ensured high concentrate Al2O3 grade and recovery. Addition of both starch and sulphuric acid to the cleaner stages did not further improve flotation results. Addition of starch alone without acid gave poor Al2O3 recovery due to its loss to the middling. Therefore, addition of starch in the cleaner stages was redundant.
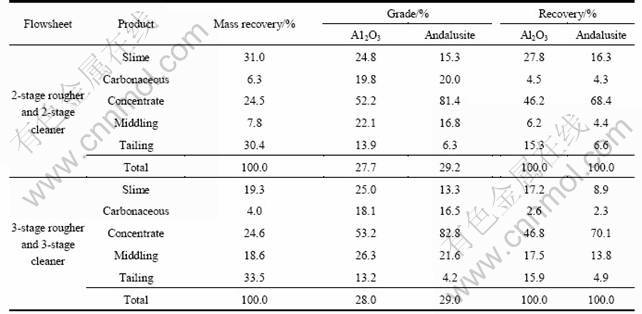
Based on the above test results, continuous flotation tests were also conducted. It was determined that the flowsheet should consist of three stages of rougher followed by three stages of cleaner; petroleum sulfonate should be used as the collector; starch should be used as the depressant; the flotation pH should be between 3 and 4 in both rougher and cleaner. This flowsheet is a closed-circuit flowsheet with 3-stage rougher and 3-stage cleaner. In a closed-circuit where the middling (the cleaner tailing) is recirculated to respective preceding stages. The results are presented in Table 8.
Table 3 Flotation results at pH 3.5 with various petroleum sulfonate dosages
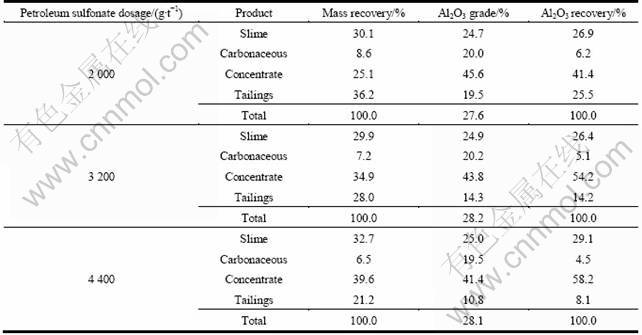
Table 4 Effect of starch dosage (3 200 g/t petroleum sulfonate, pH 3.5)
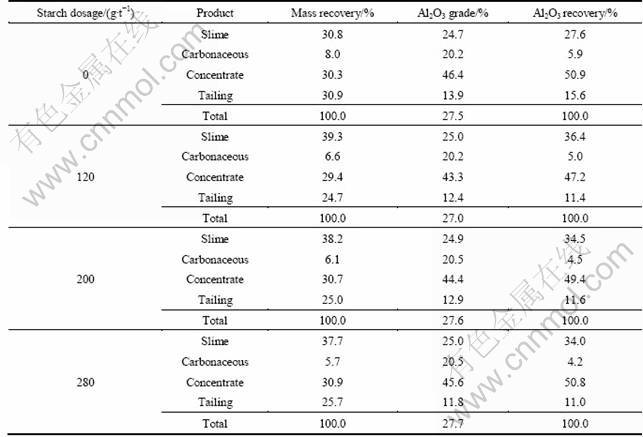
Table 5 Results of cleaner flotation with various reagent dosages
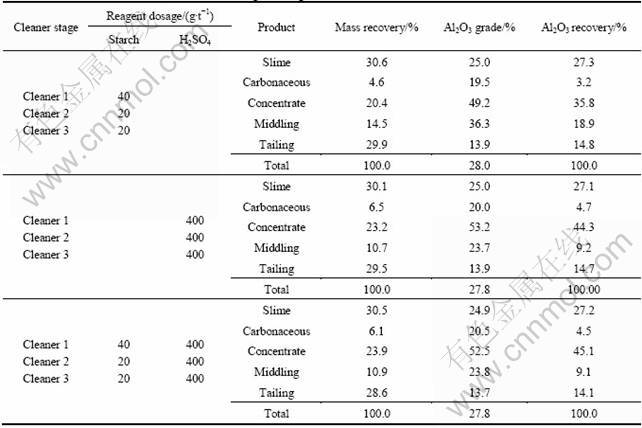
Table 6 Effect of cleaner stages on concentrate grade and recovery
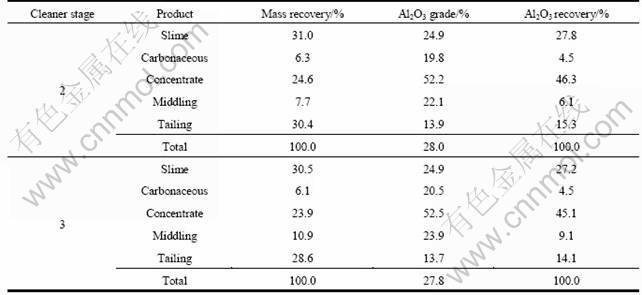
Table 7 Effect of rougher and cleaner stages on flotation results
3.3 Discussion
In this investigation, tests were carried out on both a pure andalusite mineral sample and an andalusite ore sample to outline the flotation separation of andalusite.
The adsorption of many collectors to oxide and silicate surfaces is through electrostatic interaction [10-12]. In this case, the IEP of the minerals is the key [13-14]. Zeta potential values of andalusite with respect to pH show that the IEP of this mineral is around pH 5.2 (Fig. 2). Below the IEP, andalusite surface is positively charged; thus, negatively charged alkyl sulfonate ions may adsorb in the pH range (Fig. 1). Above the IEP, the andalusite surfaces are negatively charged and alkyl sulfonate ions are repelled from the surface. On the other hand, ammonium (RNH3+) ions, which are positively charged, are adsorbed on the negatively charged surface (Fig. 1). Therefore, andalusite presents much better flotation behaviour with the sulfonate collectors below IEP and with the ammonium collectors above IEP. The flotation behaviour of andalusite presented in Fig. 1 suggests that electrostatic interaction occurs between collector and mineral surface. Studies on the zeta potential of quartz and biotite prove that the IEP of quartz and biotite are pH 2.1 and pH 3.8, respectively [15]. In this investigation, the IEP of andalusite was found to be around 5.2; therefore, it is safe to presume that, in the pH range of 3-4, the quartz surfaces were negatively charged and the biotite surfaces carried little charges, while the andalusite surfaces were positively charged. Therefore selective adsorption of petroleum sulfonate and thus separation could be obtained in the pH range. This explains why the andalusite recovery decreased sharply when pH was increased to above 5, as shown in Fig. 1.
Table 8 Continuous flotation results of andalusite ore with 3-stage rougher and 3-stage cleaner
Although oleate is an anionic collector, it floats andalusite above IEP (Fig. 1). This indicates that chemical adsorption rather than electrostatic interaction occurs between the oleic ions and andalusite surface. Flotation experiments conducted with oleate at different pH values show that andalusite floats well in a pH range where formation of Al(OH)2+ or Al(OH)2+ complexes is favored. On the other hand, recovery decreases as pH increases beyond 9 because of the formation of Al(OH)3(s) or Al(OH)4- [16-17]. The floatability was the best around pH 7.5 that was almost natural.
Amine is tested in micro-flotation of the pure mineral but not in batch flotation of the ore sample. This is because that, in the pH range where amine effectively floats andalusite, the floatabilities of quartz or biotite is similar to that of andalusite, and separation of andalusite from the gangue minerals is not anticipated. Quartz and biotite are negatively charged even in acidic pH values; therefore, ammonium cations can easily adsorb on to their surfaces [13, 18-19].
As the floatability of garnet is similar to that of andalusite and a small amount of biotite may be entrained, the concentrate (Table 8) has a high Fe2O3 grade, around 5.25%. Accordingly, a high-intensity magnetic separation is needed to remove the iron and titanium from the flotation concentrate. After the magnetic separation, the Al2O3 content rises to 55.3% that is translated to 85.9% andalusite, with a recovery of 75.6%. The results of chemical analysis of the final concentrate are given in Table 9. This concentrate meets the market specifications.
Table 9 Chemical analysis of final andalusite concentrate (mass fraction, %)

4 Conclusions
1) The IEP of andalusite is found to be around pH 5.2. DDA, a cationic collector, is therefore effective in alkaline pH region; on the other hand, SDBS, an anionic collector, is effective in acidic pH range. However, NaOl acts more effectively between pH 6 and 9.
2) Because of the presence of slimes and carbonaceous species, desliming and pre-float are necessary prior to andalusite flotation. The experiment results show that effective separation of andalusite from quartz and biotite could be achieved in pH ≤3.5 with SDBS or petroleum sulfonate as the collector and starch as the depressant.
3) The process consisting of desliming, pre-float, andalusite flotation at pH ≤3.5 with starch as the depressant and SDBS as the collector, followed by high-intensity magnetic separation, produces an andalusite concentrate of 55.3% A12O3 equivalent to 85.9% andalusite at an andalusite recovery of 75.6%.
Acknowledgements
The authors of this paper are grateful to Dr. DAI Zong-fu, Dr. SONG Shao-xian for reviewing the manuscript and making fruitful suggestions in finalizing the paper.
References
[1] ZHANG Hui-min, WANG Li-jin, JIA Dian-zen. Application and progress of researches on andalusite material field [J]. Journal of Xinjiang University: Natural Science Edition, 2006, 23(1): 53-57. (in Chinese)
[2] BURT J B, ROSS N L, ROSS J A. Equations of state and structures of andalusite to 9.8 GPa and sillimanite to 8.5 GPa [J]. American Mineralogist, 2006, 91: 319-326.
[3] XIA Shao-zhu, FENG Qi-gui, HOU Ruo-zhou, ZHANG Ye, LI Yan-hua. Andalusite family mineral resources and beneficiation [J]. Metal Mine, 1994, 212(2): 37-44. (in Chinese)
[4] XIA Shao-zhu, FENG Qi-gui, HOU Rou-zhou, ZHANG Ye. Andalusite family mineral resources and beneficiation [J]. Metal Mine, 1994, 213(3): 36-42. (in Chinese)
[5] JIN Qing-guo, LI Jiang, LIU Yan-jun. Status of deposits, production and application of China’s andalusite [J]. Naihuo Cailiao, 2002, 36(5): 284-287. (in Chinese)
[6] YANG Da-bing, ZHANG Yi-min, YANG Shi-yong, CHEN Tie-jun. Heavy medium separation study of coarse andalusite [J]. Mining and Metallurgical Engineering, 2003, 23(2): 33-35. (in Chinese)
[7] FAN Shao-liang, LI Yan-hua. Study on the technology of andalusite ore of Zhang county, Gansu province [J]. Metal Mine, 1999, 282(12): 37-39. (in Chinese)
[8] LI Xue-xia, LI Biao. Research and practice of DMG electromagnetic pulsating high gradient magnetic separator in iron removal from andalusite [J]. Metal Mine, 2001, 297(3): 39-41. (in Chinese)
[9] LI Xiao-jing, YUAN Chu-xiong, YUAN Ji-zu. Studies on the flotation behaviour of andalusite and the mechanism of its interaction with flotation collectors [J]. Journal of Wuhan University of Technology, 1993, 15(2): 63-68. (in Chinese)
[10] BULUT G, YURTSEVER C. Flotation behavior of bitlis andalusite ore [J]. Miner Process, 2004, 73: 29-36.
[11] WENG Da, ZHOU Ling-chu. Selection and functions of floatation agent in the flotation of andalusite [J]. Journal of Wuhan Yejin University of Science and Technology, 1998, 21(1): 1-4. (in Chinese)
[12] WENG Da, ZHOU Ling-chu. Exploration into the collecting mechanism of alkyl sulphonate during flotation of andalusite [J]. Journal of Wuhan Yejin University of Science and Technology, 1998, 21(2): 134-137. (in Chinese)
[13] GAUDIN A M, FUERSTENAU D W. Quartz flotation with cationic collectors [J]. Transactions of AIME, 1955, 202: 958-962.
[14] VIJAYA K T V, PRABHAKARA S, BHASKAR R G. Adsorption of oleic acid at sillimanite/water interface [J]. Journal of Colloid and Interface Science, 2002, 247(2): 275-281.
[15] PARKS G A. Aqueous surface chemistry of oxides and complex oxide minerals isoelectric point and zero point of charge [C]//GOULD R F. Equilibrium Concepts in Naturel Water Systems. Advances in Chemistry Series. Washington, DC: American Chemical Society, 1967: 121-160.
[16] FUERSTENAU M C, PALMER B R. Anionic flotation of oxides and silicates [C]//FUERSTENAU M C. Flotation A. M. Gaudin Memorial. New York: American Institute of Mining, Metallurgical, and Petroleum Engineers, 1976: 148-197.
[17] ZHANG Guo-fan, FENG Qi-ming, LU Yi-ping, OU Le-ming. Mechanism on diaspore and kaolinite collected by sodium oleate [J]. The Chinese Journal of Nonferrous Metals, 2001, 11(2): 298-301. (in Chinese)
[18] BLEIER E, GODDARD E D, KULKARNI R D. The structural effects of amine collectors on the flotation of quartz [C]//FUERSTENAU M C. Flotation A M GAUDIN Memorial. New York: American Institute of Mining, Metallurgical, and Petroleum Engineers, 1976, 1: 117-147.
[19] JIANG Hao, HU Yue-hua, QIN Wen-qing, WANG Yu-hua, WANG Dian-zuo. Mechanism of flotation for diaspore and aluminium-silicate minerals with alkyl-amine collectors [J]. The Chinese Journal of Nonferrous Metals, 2001, 11(4): 688-692. (in Chinese)
西峡红柱石矿石的浮选分离
周灵初,张一敏
武汉科技大学 资源与环境工程学院,武汉 430081
摘 要:研究应用不同捕收剂时红柱石的浮选行为。采用西峡红柱石纯矿物进行微型浮选试验,利用红柱石矿石进行小型浮选试验,测定纯红柱石表面的电荷。结果表明,红柱石的等电点为pH 5.2。在pH 值高于等电点时,矿物表面带负电荷,可用胺类捕收剂浮选红柱石。相反,红柱石表面带正电荷,用烷基磺酸盐捕收剂(十二烷基苯磺酸钠)浮选红柱石比较有效。在中性和弱碱性pH 范围时用油酸盐可以浮选红柱石。用烷基磺酸盐捕收剂时可以获得含55.3% Al2O3,红柱石回收率为75.6%的红柱石精矿。在用烷基磺酸盐捕收剂时,淀粉是脉石矿物的有效抑制剂。此外,试验发现矿泥对红柱石浮选有不利影响。因此,浮选前必须脱碳和脱泥。
关键词:红柱石;浮选;捕收剂;脱泥;预浮选;等电点
(Edited by LI Xiang-qun)
Corresponding author: ZHOU Ling-chu; Tel: +86-27-86483758; E-mail: chaulc@yahoo.cn
DOI: 10.1016/S1003-6326(11)60871-6