文章编号:1004-0609(2015)-07-1969-09
含硼铁精矿还原过程中硼矿物的热力学行为
余建文,韩跃新,高 鹏
(东北大学 资源与土木工程学院,沈阳 110819)
摘 要:依据广泛应用的同族化合物线性递变规律,通过拟合钙、镁化合物生成反应标准吉布斯自由能之间的线性关系,由不同温度条件下Ca3(BO3)2和Ca2B2O5生成反应的标准吉布斯自由能求得相应温度条件下Mg3(BO3)2和Mg2B2O5生成反应的标准吉布斯自由能,进而基于化学反应的标准吉布斯自由能与温度之间的近似线性关系,推导Mg3(BO3)2和Mg2B2O5生成反应标准吉布斯自由能,并通过试验进行了验证与探讨。热力学计算及XRD物相分析结果表明:在还原焙烧过程中,含硼铁精矿中的硼镁石(Mg2[B2O4(OH)](OH))分解为遂安石(Mg2B2O5),遂安石进一步与蛇纹石的分解产物镁橄榄石或顽火辉石反应最终转变成小藤石(Mg3(BO3)2)。
关键词:含硼铁精矿;同族线性规律;还原焙烧;物相演变
中图分类号:TF704.7 文献标志码:A
Thermodynamic behavior of boron minerals in iron concentrate during direct reduction
YU Jian-wen, HAN Yue-xin, GAO Peng
(College of Resources and Civil Engineering, Northeastern University, Shenyang 110819, China)
Abstract: According to the homologous linear rule applied widely in the chemistry area, the linear equation between the formation reaction Gibbs free energy of calcium and magnesium composite compounds was fitted out. The formation reaction Gibbs free energies of Mg3(BO3)2 and Mg2B2O5 are obtained from the formation reaction of Ca3(BO3)2 and Ca2B2O5 at different temperatures. Then, the thermodynamic calculation models for the formation reaction of Mg3(BO3)2 and Mg2B2O5 were deduced based on the approximate linear rule between the reaction Gibbs free energy and temperature, and it was verified by the laboratory tests. The results of thermodynamic calculation and XRD phase identification indicate that ascharite in the iron concentrate will be transformed into suanite firstly, and then into kotoite by reacting with the serpentine decomposition product of serpentine during the roasting process.
Key words: boron-bearing iron concentrate; homologous linear rule; reduction roasting; phase evolution
我国硼矿资源主要有硼镁石矿和硼铁矿,由于可直接利用的硼镁石矿已近枯竭,开发复杂的硼铁矿资源已迫在眉睫[1-2]。辽宁翁泉沟硼铁矿石是我国特大 型硼资源基地,已探明储量2.8亿t,其中B2O3储量为2184万t,占全国B2O3总储量的58%左右,铁储量近亿吨,是硼、铁共生的特大型沉积变质再造型硼矿床[3-5]。硼铁矿中矿物种类多,结构复杂,硼和铁均属贫矿,矿物属细粒不均匀嵌布、共生关系密切,选矿方法仅能初步分离硼和铁得到含硼铁精矿及硼精 矿[6-9]。其中含硼铁精矿中含B2O3 4%~6%(质量分数),占原矿硼总量的30%(质量分数),这部分硼资源若不能回收为硼化工业可以利用的原料,将造成硼资源的巨大流失。
针对含硼铁精矿中硼的回收,国外鲜有相关文献报道。国内早在20世纪80年代中期,王裕民等[1]在选择性还原理论基础上提出了含硼铁精矿的火法分离工艺流程,因其简单、高效而极具发展前景。近年来,随着直接还原技术的不断发展与演变,科研工作者围绕直接还原技术,相继提出了含硼铁精矿高炉法、直接还原-电炉熔分、含碳球团还原熔分等综合利用方案[10-14]。以上工艺的基本原理是通过选择性还原将含硼铁精矿中铁氧化物还原为金属铁,而难以还原的硼氧化物则残留在渣中,金属铁及脉石在高温条件下发生熔化,利用铁水和熔渣在密度、表面张力等方面的差异,实现铁与富硼渣的分离。显然,在上述工艺过程中,硼氧化物以液态形式存在于熔渣中,从而不可避免地造成硼的挥发损失和活性低等问题。针对上述工艺中的缺陷,付小佼等[15]提出了采用含硼铁精矿与煤粉按一定配比进行还原-磁选工艺,该工艺虽然能够避免硼的挥发问题,但由于还原煤粉与含硼铁精矿混合工艺的限制,煤灰最终会进入硼精粉中而降低硼的品位。
含硼铁精矿尚无成熟的加工工艺和技术,当前研究以火法为主,许多研究者对火法工艺过程中铁氧化物的还原过程进行了大量的研究工作,但含硼铁精矿中硼氧化物焙烧过程中的物相转化与演变规律及其与脉石矿物之间的热力学行为的研究还鲜有报道。因 此,本文作者依据化学领域广泛应用的同族化合物线性递变规律,通过拟合钙、镁化合物生成反应吉布斯自由能之间的线性关系式,推导了Mg3(BO3)2和Mg2B2O5生成反应ΔGΘ-T关系式,结合XRD物相分析,揭示含硼铁精矿中硼氧化物焙烧过程中的物相转化与演变规律及其与脉石矿物之间的热力学行为,为含硼铁精矿火法高效利用提供理论依据。
1 实验
1.1 含硼铁精矿
采用阶段磨矿阶段磁选-分级分离工艺回收得到的低品位铁精矿,其化学成分如表1所列,物相分析如图1所示。
由表1及图1中可以看出,铁精矿的主要成分为Fe3O4、Mg3[Si2O5](OH)4和Mg2[B2O4(OH)](OH),其他杂质成分较少。由于铁精矿品位较低,铁含量为55.55%,未能达到标准铁精矿的品位(>60%),同时,铁精矿中B2O3含量为4.22%。这主要是由于磁铁矿与硼镁石、蛇纹石嵌布关系密切。
表1 某企业含硼铁精矿成分
Table 1 Composition of boron-bearing iron concentrate from enterprise (mass fraction, %)

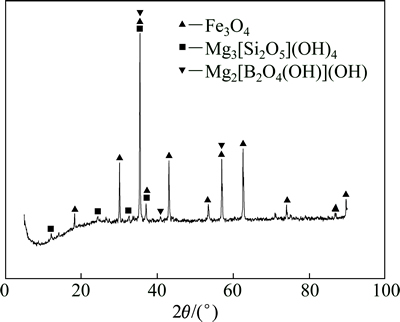
图1 含硼铁精矿的XRD谱
Fig. 1 XRD pattern of boron-bearing iron concentrate
采用矿物解离分析仪(MLA)研究矿物的解离和嵌布特征,其结果如图2所示。结果表明:磁铁矿多以单体矿物形式产出,形状和粒径不一,其单体解离度为89.59%,主要和蛇纹石连生;硼镁石多以板状、柱状产出,粒径一般为10~20 μm,与其他矿物关系密切,约有64%的硼镁石以连生体形式产出,较常见的情况有:硼镁石与蛇纹石或磁铁矿连生,被大颗粒磁铁矿包裹或被蛇纹石包裹。

图2 含硼铁精矿的嵌布特征
Fig. 2 Dissemination characteristics of boron-bearing iron concentrate (Mag-Magnetite; Srp-Serpentine; Asc-Ascharite)
1.2 还原剂
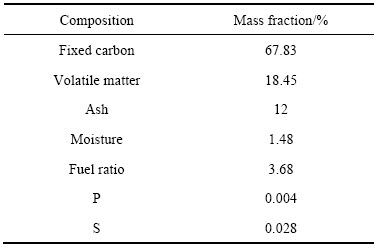
本试验使用烟煤为还原剂,取样破碎至粒度低于2 mm以备用。其工业分析及化学成分分析结果如表2所列。
由表2可知,试验用煤粉固定碳和挥发分含量高,分别为67.83%和18.45%,同时,煤的灰分及有害元素S、P含量相对较低,可知本次试验所用烟煤为低灰分、低硫磷的优质还原剂。
表2 煤工业分析及化学成分
Table 2 Industry and chemical analysis of coal
1.3 实验方法
本实验主要模拟隧道窑煤基还原焙烧过程。先将铁精矿与适量的水混匀,在5 MPa的压力下制备成d15 mm×20 mm柱团,柱团经干燥后供后续焙烧实验使用。含硼铁精矿还原焙烧实验在程控高温箱式电阻炉中进行。首先,将柱团外配过量煤粉装入刚玉坩埚中(20 g含硼铁精矿外配15 g煤粉),待炉膛温度达到预设温度(分别为1323、1373、1423及1473 K),焙烧一定时间(150 min)后,将焙烧好的物料取出水淬冷却,磨细后磁选,磁性部分为铁粉,非磁性部分为硼精粉,并对其进行XRD物相分析,工艺流程如图3所示。
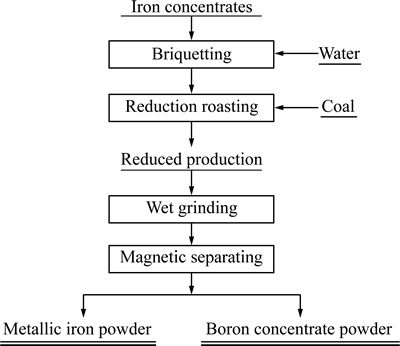
图3 含硼铁精矿处理的工艺流程
Fig. 3 Treatment technological process of boron-bearing iron concentrate
2 结果与讨论
2.1 硼镁复合化合物标准反应吉布斯自由能的估算
限于现有的热力学数据手册中尚无Mg3(BO3)2和Mg2B2O5两种复合化合物的标准反应吉布斯自由能函数的数据,仅有硼钙复合化合物热力学计算模型见诸文献。本研究中基于化学领域广泛采用的同族复合化合物标准反应吉布斯自由能的线性规律[16-18]来估算所缺的标准反应吉布斯自由能值。
根据从热力学数据手册中查得的简单氧化物和单质在1123、1223、1323、1423及1523 K时的热力学数据[19],经计算求得了估算时所用的热力学数据,如表3所列。
根据表3中的热力学数据,将不同温度条件下各种钙盐和与其对应的镁盐的标准反应吉布斯自由能(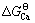 和
和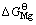 )对应作图(如图4所示),得到的拟合直线方程如下:
)对应作图(如图4所示),得到的拟合直线方程如下:
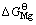 (1123 K)=0.869
(1123 K)=0.869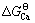 (1123 K)+30.12 (1)
(1123 K)+30.12 (1)
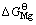 (1223 K)=0.866
(1223 K)=0.866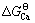 (1223 K)+33.32 (2)
(1223 K)+33.32 (2)
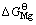 (1323 K)=0.863
(1323 K)=0.863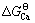 (1323 K)+36.48 (3)
(1323 K)+36.48 (3)
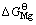 (1423 K)=0.861
(1423 K)=0.861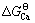 (1423 K)+39.60 (4)
(1423 K)+39.60 (4)
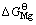 (1523 K)=0.857
(1523 K)=0.857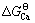 (1523 K)+42.66 (5)
(1523 K)+42.66 (5)
3MgO(s)+B2O3(l)=Mg3B2O6(s) (6)
2MgO(s)+B2O3(l)=Mg2B2O5(s) (7)
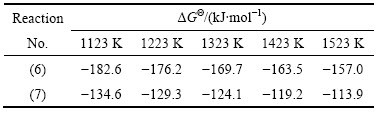
其线性相关系数R分别为0.99499、0.99499、0.99499、0.99448和0.99398,可见该方法的可靠性还是比较高的。由不同温度条件下已知的ΔGΘ(Ca3B2O6)和ΔGΘ(Ca2B2O5)值,根据相应的拟合直线方程(1)、(2)、(3)、(4)和(5)求得不同温度条件下ΔGΘ(Mg3B2O6)及ΔGΘ(Mg2B2O5)的值,其结果如表4所列。
根据表4中的热力学数据,将不同温度和与其对应的ΔGΘ(Mg3B2O6)和ΔGΘ(Mg2B2O5)的值作图(见图5),得到的拟合直线方程如下:
ΔGΘ(Mg3B2O6, 1123~1523 K)=-254.340+0.0639T (8)
ΔGΘ(Mg2B2O5, 1123~1523 K)=-192.355+0.0515T (9)
其线性相关系数R分别为1和0.99950,可知其可信度较高。同时,根据能量最低原理可知,Mg3B2O6较Mg2B2O5更稳定,Mg2B2O5有转变为Mg3B2O6的趋势。
2.2 硼矿物焙烧热力学
文献[20-21]中指出,在固态条件下,只要温度控制在1806 K(1533 ℃)以下,以固体碳为还原剂,铁的氧化物可以还原为金属铁,而硼镁石中的硼仍以氧化物状态存在,最后经磁选实现硼铁分离。因此,在本次实验过程中不考虑硼氧化物还原而挥发的问题。
含硼铁精矿在焙烧过程中,除了铁氧化物将按Fe2O3→ Fe3O4→ FeO→Fe顺序逐级还原外,其他主要矿物硼镁石、蛇纹石则发生分解反应,反应式如下所示[22-26]:
T<873 K时,
Mg2[B2O4(OH)](OH)(s)=Mg2B2O5(s)+H2O(g) (10)
T>1173 K时,
2Mg3Si2O5(OH)4(s)=2Mg2SiO4(s)+Mg2(Si2O6)(s)+4H2O(g) (11)
表3 估算不同温度条件下硼镁复合化合物标准反应吉布斯自由能所用参数
Table 3 Parameters for estimating standard reaction Gibbs free energy of B-Mg composite compounds at different temperatures
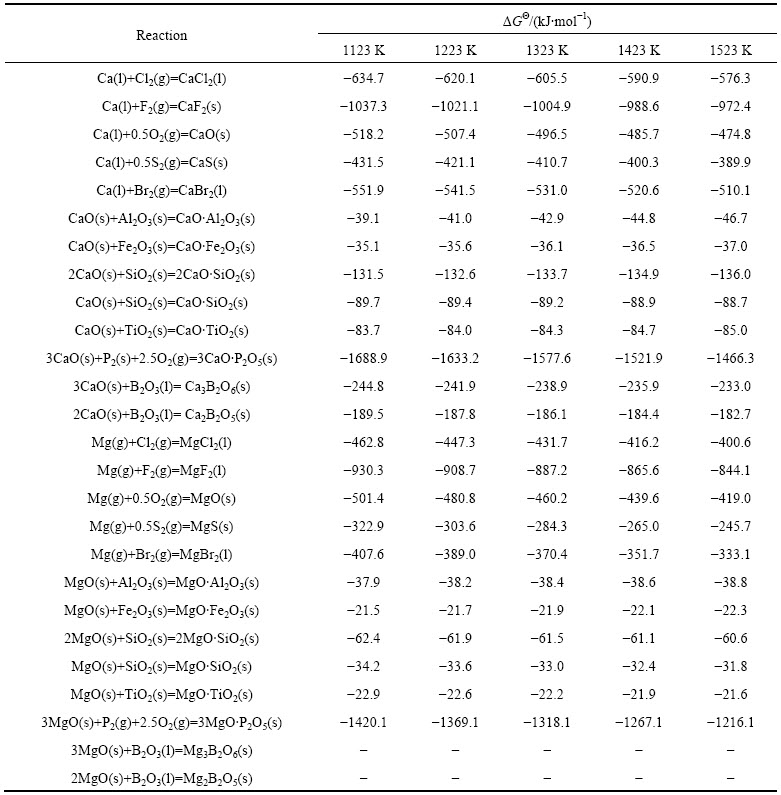
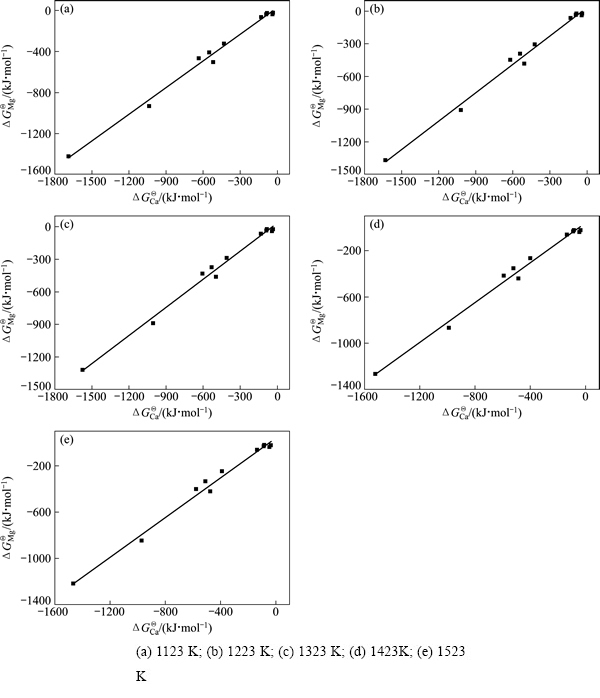
图4 不同温度条件下镁盐与钙盐的ΔGΘ间的同系线性关系
Fig. 4 Linear relationship of standard reaction Gibbs free energy of Ca and Mg salts at different temperatures
表4 不同温度条件下估算的硼镁复合化合物标准反应吉布斯自由能
Table 4 Standard reaction Gibbs free energy of B-Mg composite compounds at different temperatures
即硼镁石在焙烧过程会分解形成遂安石 (Mg2B2O5),蛇纹石在受热过程中会最终转变为镁橄榄石(Mg2SiO4)和顽火辉石(Mg2(Si2O6))。此外,由热力学数据手册有
2MgO(s)+SiO2(s)=Mg2SiO4(s)
ΔG12Θ=-67.2+0.00431T (12)
2MgO(s)+2SiO2(s)=Mg2(Si2O6)(s)
ΔG13Θ=-82.2+0.0122T (13)
根据盖斯定律,通过耦合反应(6)、(7)和(12)可得反应(14):
2Mg2B2O5(s)+Mg2SiO4(s)=2Mg3(BO3)2(s)+SiO2(s)
ΔG14Θ=-56.77+0.0205T (14)
同样地,耦合反应(6)、(7)和(13)可得反应(15):
2Mg2B2O5(s)+Mg2(Si2O6(s))=2Mg3(BO3)2(s)+2SiO2(s)
ΔG15Θ=-41.77+0.0126T (15)
可能发生的反应还有
3Mg2B2O5(s)=2Mg3(BO3)2(s)+B2O3(l)
ΔG16Θ=68.385-0.0267T (16)
在焙烧过程中,硼氧化物可能发生的化学反应(14)、(15)及(16)的热力学分析如图6所示。
由图6可知,在本次实验1323~1473 K的温度范围内,含硼铁精矿中硼镁石的分解产物遂安石与蛇纹石的分解产物镁橄榄石和顽火辉石的反应均能自发进行,而遂安石进一步分解为小藤石和三氧化二硼在热力学上则难以进行。
2.3 硼矿物焙烧物相演变规律
不同焙烧温度下所得硼精粉产物的物相变化,如图7所示。由图7分析可知,随着焙烧温度的变化,硼精粉的物相组成也发生变化。当焙烧温度为1323~1373 K时,硼精粉的XRD谱中均出现了遂安石(Mg2B2O5)和镁橄榄石(Mg2SiO4)及石英(SiO2)的特征衍射峰(见图7(a)和(b)),说明含硼铁精矿中的硼镁石和蛇纹石在焙烧过程中已经发生了分解反应,分别生成遂安石和镁橄榄石(见反应(10)和(11)),遂安石虽然也开始反应(如反应(14)和(15)所示),但由于在1323~1373 K温度下,遂安石的反应(固-固反应)速度较慢,反应不充分,故仍有Mg2B2O5的衍射峰。随着温度从1373 K升高到1423 K,矿石中的遂安石与镁氧化物反应更充分,Mg2B2O5的衍射峰消失,而Mg3(BO3)2的衍射峰强度显著增强,表明遂安石反应充分,已全部转化为Mg3(BO3)2含硼相。随着反应温度继续升高到1473 K,硼精粉的物相组成则无变化。
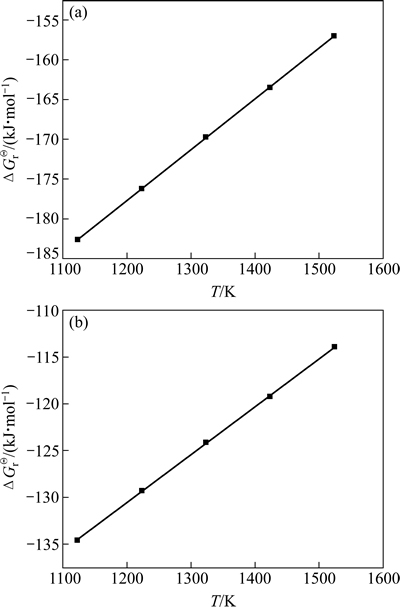
图5 反应(6)和(7)标准吉布斯自由能与温度之间的关系
Fig. 5 Relationship between standard Gibbs free energy of reactions (6) (a) and (7) (b) and temperature
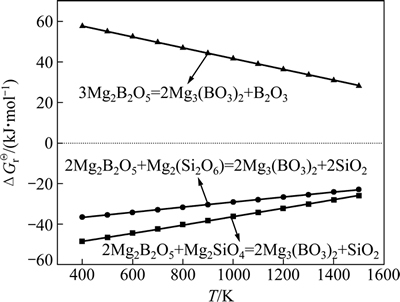
图6 硼氧化物复分解反应中ΔGTΘ和T的关系
Fig. 6 Relationship between ΔGTΘ and T for boron-bearing minerals double decomposition reaction
综上所述,含硼铁精矿在还原焙烧过程中,硼矿物将按硼镁石(Mg2[B2O4(OH)](OH))→遂安石(Mg2B2O5)→小藤石(Mg3(BO3)2)顺序依次发生物相转变。
3 结论
1) 该含硼磁铁精矿中含Fe 55.55%、B2O3 4.22% (质量分数),铁主要以磁铁矿的形式存在,硼主要以硼镁石的形式存在,主要脉石矿物为蛇纹石。其中磁铁矿的单体解离度89.59%,主要和蛇纹石连生;硼镁石的连生度高,约有64%的硼镁石以连生体形式产出,主要与蛇纹石、磁铁矿连生。
2) 依据化学中广泛采用的同族线性规律,通过拟合钙、镁复合化合物标准反应吉布斯自由能之间的线性关系,推导了所缺的Mg3(BO3)2和Mg2B2O5生成反应吉布斯自由能与温度之间的线性关系式,并且可信度较高。
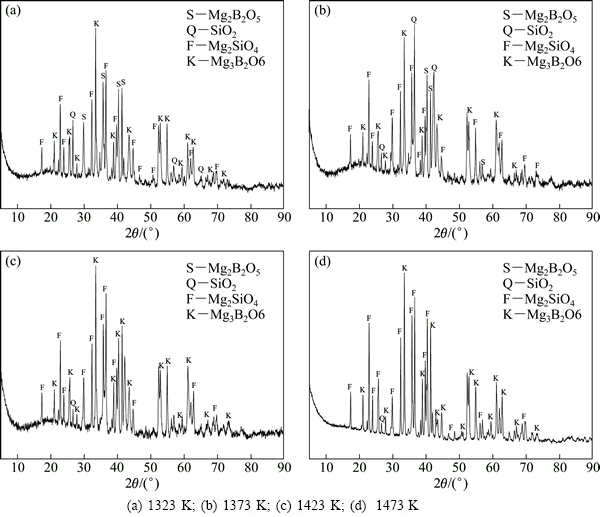
图7 不同焙烧温度下硼精粉的物相演变
Fig. 7 Phase evolution of boron-bearing concentrate obtained at different temperatures
3) Mg3B2O6较Mg2B2O5稳定,Mg2B2O5有转变为Mg3B2O6的趋势。含硼铁精矿在还原焙烧过程中,硼矿物将按硼镁石(Mg2[B2O4(OH)](OH))→遂安石(Mg2B2O5)→小藤石(Mg3(BO3)2)顺序依次发生物相转变,并最终以小藤石相稳定存在。
REFERENCES
[1] 王裕民, 刘素兰, 张显鹏, 崔传孟, 樊占国.硼铁矿资源综合利用[J]. 矿业快报, 2000(10): 58-61.
WANG Yu-min, LIU Su-lan, ZHANG Xian-peng, CUI Chuai-meng, FAN Zhan-guo. The comprehensive utilization of ludwigite resources[J]. Express Information of Mining Industry, 2000(10): 58-61.
[2] 刘 然, 薛向欣, 姜 涛, 张淑会, 黄大威. 硼铁矿综合利用概况与展望[J].矿产综合利用, 2006(2): 33-37.
LIU Ran, XUE Xiang-xin, JIANG Tao, ZHANG Shu-hui, HUANG Da-wei. Comprehensive utilization of ludwigite and its prospect[J]. Multipurpose Utilization of Mineral Resources, 2006(2): 33-37.
[3] 曹 钊, 曹永丹, 桂 富. 硼铁矿资源开发利用研究现状及进展[J]. 矿产综合利用, 2013(2): 17-19.
CAO Zhao, CAO Yong-dan, GUI Fu. Research status and progress of the exploitation and utilization of paigeite ore[J]. Multipurpose Utilization of Mineral Resources, 2013(2): 17-19.
[4] 李艳军, 韩跃新. 辽宁凤城硼铁矿资源的开发与利用[J]. 金属矿山, 2006(7): 8-11.
LI Yan-jun, HAN Yue-xin. Exploitation and utilization of boron-iron ore resource in Fengcheng Liaoning[J]. Metal Mine, 2007(6): 8-11.
[5] 李艳军, 高 太, 韩跃新. 硼铁矿工艺矿物学研究[J]. 有色矿冶, 2006, 22(6): 14-16.
LI Yan-jun, GAO Tai, HAN Yue-xin. Research on processing mineralogy of paigeite[J]. Non-ferrous Mining and Metallurgy, 2006, 22(6): 14-16.
[6] 赵庆杰, 何长清, 王常任, 连相泉. 硼铁矿磁选分离综合利用新工艺[J]. 东北大学学报(自然科学版), 1996, 17(6): 588-592.
ZHAO Qing-jie, HE Chang-qing, WANG Chang-ren, LIAN Xiang-quan. New process of multipurpose utilization of ludwigite[J]. Journal of Northeastern University (Natural Science), 1996, 17(6): 558-592.
[7] 张金华, 王 耀. 硼铁矿综合利用选矿试验研究[J].金属矿山, 2009(11): 212-216.
ZHANG Jin-hua, WANG Yao. Dressing test research of comprehensive utilizing ludwigite[J]. Metal Mine, 2009(11): 212-216.
[8] 连相泉, 王常任. 辽宁凤城地区硼铁矿石适宜选矿工艺[J]. 东北大学学报(自然科学版), 1997, 18(3): 238-241.
LIAN Xiang-quan, WANG Chang-ren. Investigation on suitable mineral separation technology for paigeite in Fengcheng, Liaoning[J]. Journal of Northeastern University (Natural Science), 1997, 18(3): 238-241.
[9] 李艳军, 韩跃新. 硼铁矿选矿分离研究新进展[J]. 金属矿山, 2005(8): 161-163.
LI Yan-jun, HAN Yue-xin. New process of paigeite concentration and separation research[J]. Metal Mine, 2005(8): 161-163.
[10] LIU S L, CUI C M, ZHANG X P. Pyrometallurgical separation of boron from iron in ludwigite ore[J]. ISIJ International, 1998, 38(10):1077-1079.
[11] 张显鹏, 郎建峰, 崔传孟, 刘素兰. 低品位硼铁矿在高炉冶炼过程中的综合利用[J]. 钢铁, 1995, 30(12): 9-11.
ZHANG Xian-peng, LANG Jian-feng, CUI Chuan-meng, LIU Su-lan. Comprehensive utilization of low grade ludwigite ore with blast furnace smelting[J]. Iron and Steel, 1995, 30(12): 9-11.
[12] 李壮年, 储满生, 王兆才, 柳政根. 凤城含硼铁精矿硼铁分离新工艺[C]//第七届(2009)中国钢铁年会论文集. 北京: 冶金工业出版社, 2009: 432-437.
LI Zhuang-nian, CHU Man-sheng, WANG Zhao-cai, LIU Zheng-gen. New technology of boron and iron separation from Fengcheng boron-bearing iron concentrate[C]//Seventh Annual Conference of Chinese steel. Beijing: Metallurgical Industry Press, 2009: 432-437.
[13] WANG G, WANG J S, DING Y G, MA S, XUE Q G. New separation method of boron and iron from ludwigite based on carbon bearing pellet reduction and melting technology[J]. ISIJ International, 2012, 52(1): 45-51.
[14] DING Y G, WANG J S, WANG G, MA S, XUE Q G. Comprehensive utilization of paigeite ore using iron nugget making process[J]. Journal of Iron and Steel Research, International, 2012, 19(6): 9-13.
[15] 付小佼, 于洪翔, 柳政根, 储满生. 含硼铁精矿选择性还原-选分新工艺的实验研究[J]. 东北大学学报(自然科学版), 2013, 34(7): 966-970.
FU Xiao-jiao, YU Hong-xiang, LIU Zheng-gen, CHU Man-sheng. Experimental Study on new process for selective reduction and separation of boron-bearing iron concentrate[J]. Journal of Northeastern University (Natural Science), 2013, 34(7): 966-970.
[16] 温元凯, 邵 俊. 离子极化导论[M]. 合肥: 安徽教育出版社, 1985.
WEN Yuan-kai, SHAO Jun. Ion polarization introduction[M]. Hefei: Anhui Education Press, 1985.
[17] 蒋明谦. 同族元素与化合物性能递变的规律性[J]. 吉林大学自然科学学报, 1979(4): 81-103.
JIANG Ming-qian. The regularity in the gradation of properties of the congeners of the elements and their compounds[J]. Acta Scientiarum Naturalium Universitatis Jilinensis, 1979(4): 81-103.
[18] 王 广, 王静松, 薛庆国, 丁银贵. MgO-B2O3二元系熔渣质量作用浓度计算模型[J]. 中国有色金属学报, 2011, 21(12): 3143-3148.
WANG Guang, WANG Jing-song, XUE Qing-guo, DING Yin-gui. Mass action concentration calculation model for MgO-B2O3 slag melt[J]. The Chinese Journal of Nonferrous Metals, 2011, 21(12): 3143-3148.
[19] 梁英教, 车荫昌, 刘晓霞, 李乃军. 无机物热力学数据手册[M]. 沈阳: 东北大学出版社, 1993.
LIANG Ying-jiao, CHE Yin-chang, LIU Xiao-xia, LI Nai-jun. Thermodynamic data notebook of inorganics[M]. Shenyang: Northeastern University Press, 1993.
[20] 蔡海涛, 张建良. 低品位硼铁矿硼铁分离的热力学研究[J]. 钢铁, 2008, 43(11): 31-36.
CAI Hai-tao, ZHANG Jian-liang. Thermodynamic study on boron and iron separation from low grade paigeite[J]. Iron and Steel, 2008, 43(11): 31-36.
[21] 赵庆杰. 硼铁矿选择性还原分离铁和硼[J]. 东北工学院学报, 1990, 11(2): 122-126.
ZHAO Qing-jie. Separation iron from boric iron ore by selective reduction[J]. Journal of Northeast University of Technology, 1990, 11(2): 122-126.
[22] 都兴红, 刘 慧, 牛亚慧, 娄太平, 付念新. 含镍蛇纹石中氧化镍的碳热还原过程[J]. 中国稀土学报, 2012, 30(专辑): 781-786.
DU Xing-hong, LIU Hui, NIU Ya-hui, LOU Tai-ping, FU Nian-xin. Carbonthermic reduction process of NiO in serpentine containing nickel[J]. Journal of Chinese Society of Rare Earths, 2012, 30(Spec. Issue): 781-786.
[23] 张保森, 徐滨士, 许 一, 巴志新, 王章忠. 纳米氧化镧对超细蛇纹石微粉热相变行为的影响[J]. 材料热处理学报, 2013, 34(12): 18-23.
ZHANG Bao-sen, XU Bin-shi, XU Yi, BA Zhi-xin, WANG Zhang-zhong. Effect of nano-scale lanthanum on phase transformation behavior of ultrafine serpentine powders[J]. Transactions of Materials and Heat Treatment, 2013, 34(12): 18-23.
[24] 崔传孟, 刘素兰, 张显鹏. 硼铁矿氧化焙烧试验研究[J]. 矿冶, 1995, 4(4): 78-81.
CUI Chuan-meng, LIU Su-lan, ZHANG Xian-peng. Research on oxidizing roasting of ferroludwigite ore[J]. Mining and Metallurgy, 1995, 4(4): 78-81.
[25] 刘 然, 薛向欣, 黄大成. 硼铁矿氧化焙烧失重及机理探讨[J]. 钢铁, 2007, 42(9): 9-11.
LIU Ran, XUE Xiang-xin, HUANG Da-wei. Study on mass loss of ludwigite in process of oxidizing roasting and its mechanism[J]. Iron and Steel, 2007, 42(9): 9-11.
[26] 赵庆杰, 何长清, 高明辉. 硼铁矿综合利用-硼精矿活化及含硼铁精矿改善烧结球团性能的机理[J]. 华东冶金学院学报, 1997, 14(3): 262-266.
ZHAO Qing-jie, HE Chang-qing, GAO Ming-hui. Reasonable utiligation of boronic magnetite ore[J]. Journal of East China University of Metallurgy, 1997, 14(3): 262-266.
(编辑 李艳红)
基金项目:国家自然科学基金资助项目(51204033);教育部新教师专项科研基金资助项目(20120042120051)
收稿日期:2014-11-17;修订日期:2015-04-01
通信作者:韩跃新,教授,博士;电话:024-83680602;E-mail:dongdafulong@mail.neu.edu.cn