˫(�ȼ�)�����̼������ͭ����ѡ���������ƻ���
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2017���4��
�������ߣ���־�� ��ΰ ������ ����� ���Ȼ� ��Ծ�� �Լƻ�
����ҳ�룺883 - 890
�ؼ��ʣ�ͭ����˫(�ȼ�)�����̼���ƣ����Ƽ�����ѡ���룻��������
Key words��copper-molybdenum sulfide ore; disodium bis(carboxymethyl) trithiocarbonate; depressant; flotation separation; physical adsorption
ժ Ҫ��С�����л����Ƽ�˫(�ȼ�)�����̼����(DBT)��һ��ͭ�����ѡ�������Ƽ�������λ�������������������DBT�ڻ�ͭ����������ǿ�ڻ����X���߹��������һ��֤��DBT�����ڻ�ͭ����棬���Ƽ�DBT���ڻ�ͭ��������������������Ϊ����ͭ���Ͼ���ѡ����������������DBT��һ��DZ�ڵĻ����Ѻ���ͭ��������Ƽ���
Abstract: This work focuses on the organic depressant, disodium bis(carboxymethyl) trithiocarbonate (DBT), as a selective depressant in copper-molybdenum sulfide flotation separation. Micro-flotation, Zeta potential, FTIR and XPS measurements were carried out to investigate the selective depression mechanism of DBT on chalcopyrite. Zeta potential and FTIR measurements revealed that DBT had higher affinity for chalcopyrite than molybdenite and the XPS results of chalcopyrite before and after treatment with DBT further proved that DBT adsorbed on chalcopyrite surface. The investigation indicates that the mechanism of DBT adsorbing on chalcopyrite is mainly physical adsorption. Locked circuit experiments were carried out and the results showed that DBT could be considered as a cleaner option in commercial Cu-Mo flotation separation circuits.
Trans. Nonferrous Met. Soc. China 27(2017) 883-890
Zhi-gang YIN, Wei SUN, Yue-hua HU, Qing-jun GUAN, Chen-hu ZHANG, Yue-sheng GAO, Ji-hua ZHAI
School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China
Received 19 February 2016; accepted 28 June 2016
Abstract: This work focuses on the organic depressant, disodium bis(carboxymethyl) trithiocarbonate (DBT), as a selective depressant in copper-molybdenum sulfide flotation separation. Micro-flotation, Zeta potential, FTIR and XPS measurements were carried out to investigate the selective depression mechanism of DBT on chalcopyrite. Zeta potential and FTIR measurements revealed that DBT had higher affinity for chalcopyrite than molybdenite and the XPS results of chalcopyrite before and after treatment with DBT further proved that DBT adsorbed on chalcopyrite surface. The investigation indicates that the mechanism of DBT adsorbing on chalcopyrite is mainly physical adsorption. Locked circuit experiments were carried out and the results showed that DBT could be considered as a cleaner option in commercial Cu-Mo flotation separation circuits.
Key words: copper-molybdenum sulfide ore; disodium bis(carboxymethyl) trithiocarbonate; depressant; flotation separation; physical adsorption
1 Introduction
Molybdenite (MoS2) is the most important molybdenum source and large amount of molybdenites are associated with copper sulfide ore as trace mineral [1]. Since molybdenite is a valuable mineral, high recovery and efficient separation with copper sulfide are more important [2]. The selective separation of sulfide minerals by froth flotation frequently relies on the use of various chemical additives that affect the floatability of individual ore components [3]. In the case of copper-molybdenum separation, the use of sodium hydro-sulfide (NaHS), sodium sulfide, Noke��s reagent (thiophosphorus or thioarsenic compounds), or even cyanides, is widely practiced for the selective depression of chalcopyrite which is the main copper sulfide mineral [4-6]. However, given the toxic nature of most of these chalcopyrite depressants, there have been significant efforts to determine the effect of non-toxic or low-toxic organic compounds on molybdenite and chalcopyrite flotation recovery [7-9], in an effort to find a benign reagent that will have a little impact on environment.
Alkyl trithiocarbonates are of importance in synthetic chemistry, biochemistry and industry [10,11]. They are especially used as pesticides in agriculture [12], reversible addition fragmentation chain transfer agent in the free radical polymerization reactions [13], lubricating additive [14], pharmaceuticals [15] and intermediate in organic synthesis [16]. Alkyl trithiocarbonates have also been explored in the minerals industry, for example, trithiocarbonate could be used as collector in the flotation of platinum group-minerals (PGMs) [17], auriferous pyrite [18], pyrite and copper [19,20].
Although much work has been done on the copper sulfide ore depressants for copper-molybdenum separation, there is still a lack of understanding on the flotation characteristics of copper-molybdenum sulfides when disodium bis (carboxymethyl) trithiocarbonate (DBT) is used as the depressant.
In this work, disodium bis(carboxymethyl) trithiocarbonate (DBT) was explored to evaluate its potential as a selective depressant in copper- molybdenum flotation separation. Micro-flotation tests, Zeta potential, FTIR and XPS measurements were carried out to investigate the mechanism. The flotation of pure chalcopyrite was depressed strongly by DBT while the floatability of molybdenite only slightly reduced in the tested pH range, and the optimal differential flotation of molybdenite and chalcopyrite was achieved under weakly alkaline condition. The results of FTIR spectroscopic, Zeta potential and XPS measurement indicate that the mechanism of DBT adsorbing on chalcopyrite is mainly as physical adsorption. Furthermore, DBT also tested in copper-molybdenum sulfide separation by flotation using copper- molybdenum rough concentrate to determine its possibility to be an effective selective depressant at bench scale testwork.
2 Experimental
2.1 Materials and reagents
Molybdenite and chalcopyrite obtained from Henan province in China, were crushed in a porcelain mortar and sieved. The fraction containing particles of 38-74 ��m fraction was used for flotation. The sample was treated with dilute nitric acid (0.1 mol/L) to eliminate oxide layers, and was treated with dilute sodium hydroxide (0.1 mol/L) to remove any hydrophobic impurities or residual reagents. After each leaching step, the residues were removed by washing the sample with distilled water until neutral. The sample was dried in vacuum desiccators and kept in a plastic bag and stored in a freezer to minimize oxidation. The rough concentrate, which was obtained from China Molybdenum, Co., Ltd., with 0.95% Cu and 7.23% Mo collected from a day shift, was used for the flotation separation of copper-molybdenum sulfides in the locked circuit flotation test. The mineralogical analysis results showed that both copper and molybdenum mainly existed in the form of chalcopyrite and molybdenite respectively and the gangue minerals were primarily quartz, biotite, muscovite, clay, fluorspar, chlorite, calcite, and so on.
The preparation of DBT was carried out according to Ref. [21] and the molecular structure was presented in Fig. 1. Dodecane and methyl isobutyl carbinol (Guoyao, China) were used as collector and frother at contents of 10��10-6 and 5��10-6, respectively, and sodium hydroxide and hydrochloric acid were used to adjust pH. Double distilled water was used in all tests.
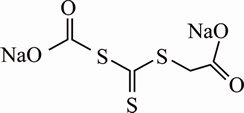
Fig. 1 Molecular structure of disodium bis (carboxymethyl) trithiocarbonate (DBT)
2.2 Flotation tests
A flotation machine of XFG-1600 type (mechanical agitation) with the volume of 40 mL was used in micro-flotation tests. The impeller speed was fixed at 1902 r/min. The mineral suspension was prepared by adding 2.0 g of single mineral to 40 mL of solutions in single mineral flotation tests. The mineral surfaces were cleaned by using ultrasound cleaner. The pH of the mineral suspension was adjusted to the desired operating value by adding HCl or NaOH stock solutions. The general reagent addition scheme involved depressant, collector and then frother addition with each stage having a 2 min conditioning period prior to the next reagent addition. Flotation concentrates were then collected for a total of 4 min. The floated and unfloated particles were collected, filtered and dried. In single mineral flotation, the recovery was calculated based on solid mass distributions between the two products. In order to assess the accuracy of flotation tests, the errors of the recovery were found to be within 2.0% after at least three tests for each condition, and the average values were reported.
Testworks were carried out on copper-molybdenum rougher concentrate in a series of flotation cell in the cleaner flotation stage to examine the potential of using DBT as a selective depressant in copper-molybdenum separation(XFD-0.5L, XFGCII Model Hanging Cell Flotation Machine).
2.3 Zeta potential experiments
Zeta potential experiments were conducted using the Malvern Zeta Sizer Nano Series. Zeta potential measurements for each mineral were carried out in 0.01 mol/L NaCl electrolyte solution. A freshly ground 0.05 g mineral sample (100% passing 5 ��m sieve) was dispersed in 100 mL of electrolyte solution and mixed for 5 min. The coarse mineral particles were allowed to settle for 0.5 h and the supernatant containing the fine particles were transferred into a folded capillary cell, ensuring that there were no bubbles in the cell. The results presented here were the average of five independent measurements.
2.4 FTIR spectroscopic measurements
1 g of molybdenite or chalcopyrite with particle size less than 38 ��m was ground to about 2 ��m in an agate mortar, and placed into a 250 mL beaker with 100 mL of 0.05 mol/L reagent. After the pH of the suspension was adjusted to desired value using NaOH or HCl, the suspension was conditioned for 20 min. The conditioned mineral solids were separated from the solution by centrifugation. The solid sample was washed four times with distilled water of the same pH as in the reaction treatment then dried under vacuum at room temperature for 24 h. The Fourier transform infrared (FTIR) spectra of DBT, untreated and treated minerals were collected by a Bruker Alpha (Thermo, USA) FTIR spectrophotometer.
2.5 XPS measurement
In order to investigate the chemical composition of surface films, the chalcopyrite samples before and after treatment with DBT solution at pH 8.0 were analyzed using X-ray photoelectron spectroscopy. The XPS spectra of mineral particles were recorded with a K-alpha 1063 (Thermo Scientific Co., USA) spectrometer with Al K�� as sputtering source at 12 kV and 6 mA, with pressure in the analytical chamber at 1.0��10�C12 Pa. All binding energies were referenced to the neutral C 1s peak at 285.0 eV to compensate for the surface-charging effects. XPS Peak 4.1 software was used to fit the XPS peaks.
3 Results and discussion
3.1 Flotation
The plots of the floatability of molybdenite and chalcopyrite as a function of pH (4-12) with and without depressant are shown in Fig. 2. As shown in Fig. 2, both molybdenite and chalcopyrite show high floatability with a high recovery of more than 80% and 60%, respectively, in the pH range in the absence of DBT. These recoveries tendency of molybdenite and chalcopyrite are in agreement with the reports in Refs. [7,22], and the high recovery of chalcopyrite might be attributed to the formation of sulfur elements as discussed in the following XPS analysis section. However, when 50 mg/L DBT was added, the floatability of both chalcopyrite and molybdenite dropped slowly. At pH 12, the flotation recovery of chalcopyrite dropped from about 75% in the absence of DBT, to about 39% in the presence of 50 mg/L DBT. For molybdenite, the recovery dropped from 85% in the absence of DBT, to about 74% in the presence of 50 mg/L DBT at pH 12. As shown in Fig. 2, the maximum recovery difference between chalcopyrite and molybdenite occurred in the pH range of 8-12. These results prompted the implementation of a series of tests with the objective of confirming the influence of depressant concentration on the floatability of molybdenite and chalcopyrite. Therefore, testwork was conducted by fixing pH at 8 and varying DBT concentration, as this value is close to neutral and economical in Cu-Mo commercial flotation separation.
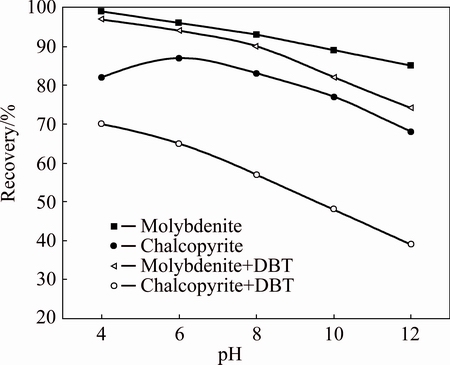
Fig. 2 Flotation of single minerals of chalcopyrite and molybdentie as function of pH by kerosene (10 mg/L) and MIBC (4 mg/L) with or without DBT (50 mg/L)
The effect of DBT concentration on molybdenite and chalcopyrite flotation is presented in Fig. 3. As shown in Fig. 3, the recovery of chalcopyrite decreased from 83% to 9% and that of molybdenite decreased from 93% to 88% as the DBT concentration increased from 0 to 150 mg/L. A window with 80% separability was obtained using DBT at a concentration of 150 mg/L. Furthermore, the result also showed that the recovery difference between molybdenite and chalcopyrite was enlarged gradually as the depressant concentration was increased. Therefore, it is possible to use DBT as a selective depressant in copper-molybdenum separation.
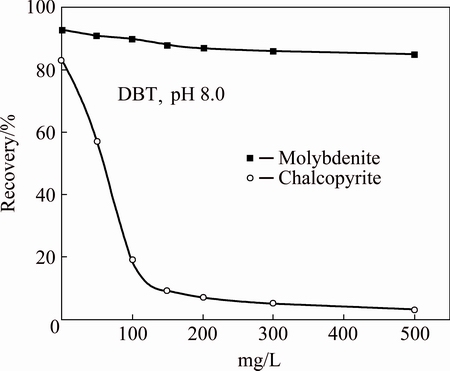
Fig. 3 Floatability of molybdenite and chalcopyrite at different concentrations
3.2 Zeta potential
The Zeta potential of chalcopyrite was first determined as a function of pH, open to the atmosphere, at a fixed ionic strength of 0.01 mol/L NaCl in the absence of DBT, and these results are presented in Fig. 4. The figure shows that the Zeta potential of chalcopyrite depends on the pH: it is positive at low pH values, goes through Zero and becomes negative as the pH increases. The isoelectric point (IEP) (pH 4.8) of chalcopyrite sample used in this investigation is close to the value reported by other researchers [23,24]. As shown in Fig. 4, the DBT makes the Zeta potential of chalcopyrite more negative. Because the molecular structure of this organic compound contains trithiocarbonate and carboxylic acid functional groups, the more negative Zeta potential of chalcopyrite in the presence of DBT may be associated to the adsorption of DBT on the chalcopyrite surface.
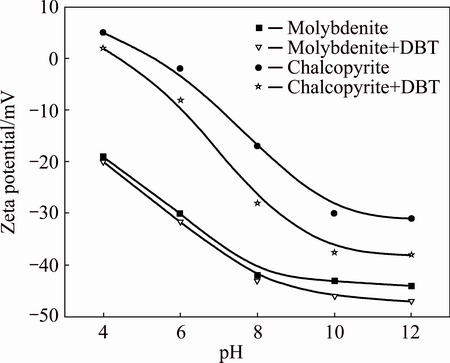
Fig. 4 Zeta potential of minerals as function of pH in absence and presence of DBT (50 mg/L)
The Zeta potential curves for molybdenite and molybdenite treated with DBT (Fig. 4) showed that the surfaces were electronegative throughout almost the entire pH range, with isoelectric points located at pH values less than 4. It became clear that beginning at pH 6, there was an accentuated increase in the electronegativity of the molybdenite surface, which was potentially due to the oxidation and hydrolysis of MoO3 to 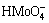 and
and 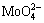 , which increased the electronegativity of the surface [22]. When molybdenite was treated with DBT, Fig. 4 clearly indicates that its adsorption onto the molybdenite surface also affected the Zeta potential, thus promoting a displacement of the curve to the region of lower electronegativity. But, the presence of DBT had little influence on the Zeta potential of the molybdenite and promoted only a small increase in its electronegativity. The negative Zeta potential of molybdenite in the presence of DBT suggests that the DBT molecules also adsorbed at molybdenum surface sites.
, which increased the electronegativity of the surface [22]. When molybdenite was treated with DBT, Fig. 4 clearly indicates that its adsorption onto the molybdenite surface also affected the Zeta potential, thus promoting a displacement of the curve to the region of lower electronegativity. But, the presence of DBT had little influence on the Zeta potential of the molybdenite and promoted only a small increase in its electronegativity. The negative Zeta potential of molybdenite in the presence of DBT suggests that the DBT molecules also adsorbed at molybdenum surface sites.
The Zeta potentials of chalcopyrite decreased significantly whereas Zeta potentials of molybdenite decreased to a minor extent only, which indicates a higher adsorbed amount of DBT on chalcopyrite surface than on molybdenite surface. The strongest depression of chalcopyrite, as measured by flotation, appears to coincide with the strongest decrease in Zeta potential at pH 6-12. It should be noticed that DBT transformed into its acid form which is present as yellow precipitation in solution under acid condition (pH<5). The difference of the Zeta potential of DBT adsorbed chalcopyrite or molybdenite in alkaline and acid conditions might be explained by the transformation of chemical formula of DBT in different pH conditions (salt-acid).
3.3 FTIR study
Fourier transform infrared (FTIR) spectroscopic measurements were taken to delineate the mechanism by which reagents adsorb on mineral surfaces. The spectrum of DBT only shows the 500-2000 cm-1 region, since this area includes most of the main adsorption bands attributable to the depressant. In the spectrum of DBT, the main bands at 1734.17, 1397.10, 1349.90, 1207.22, 1047.64, 874.41 and 663.90 cm-1 were due to C=O stretching, CH2 scissoring, CH2 wagging, C��O stretching, C=S stretching, C��C stretching and C��S stretching, respectively. The FTIR spectra of chalcopyrite before and after treatment with DBT are presented in Fig. 5. As shown in Fig. 5(a), the results of DBT adsorbed on chalcopyrite after treating with DBT, the new bands appeared near 1051.90, 1204.81, 1347.91, 1394.23, 872.14 cm-1 were due to C=S stretching, C��O stretching, CH2 wagging, CH2 scissoring, C��C stretching, respectively, which indicated that the adsorption of DBT on chalcopyrite is mainly as physical adsorption because of lower shift value of IR bands on spectra of surface of the chalcopyrite compared with DBT spectrum. The FTIR spectra of molybdenite before and after treatment with DBT are presented in Fig. 5(b). It can be seen that the characteristic bands of DBT did not present on the surface of treated molybdenite surface. The coverage of DBT on molybdenite surface might be removed by the sample preparation procedure. The solid sample was washed four times with distilled water of the same pH as in the reaction treatment, which indicated that the adsorbed DBT molecules on molybdenite surface might be removed. Comparing the results of flotation and Zeta potential, we can clearly conclude that the interaction between DBT and molybdenite probably is physical adsorption.
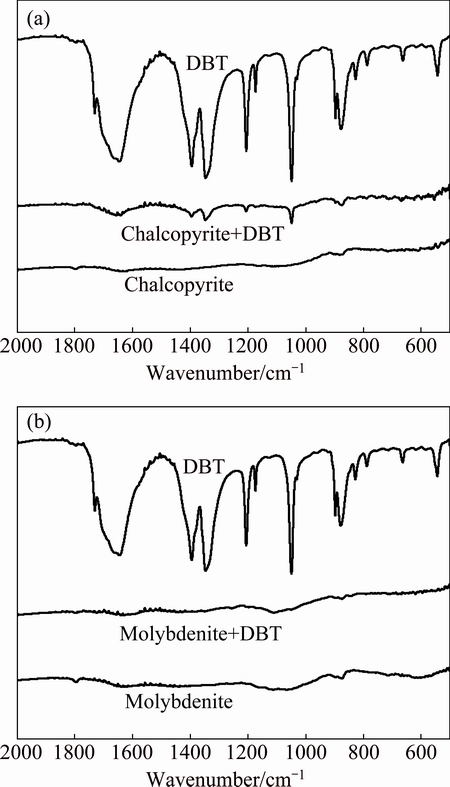
Fig. 5 FTIR spectra of DBT and depressant treated minerals at pH 8.0
3.4 XPS analyses of chalcopyrite treated with depressant
The FTIR spectra of molybdenite before and after treatment with DBT at pH 8.0 indicated that the interaction between DBT and molybdenite might be physical adsorption. However, chalcopyrite adsorbed more DBT than molybdenite. Therefore, the XPS spectra of chalcopyrite before and after treatment with DBT at pH 8.0 were selected and further investigated through XPS analysis in this section.
The C 1s spectrum (Fig. 6) of the baseline chalcopyrite without reaction with DBT depressant has main component at 284.81 eV. It should be noted that at least part of the intensity at 286.18 eV may result from carbon contamination. After interaction with DBT, the broad contribution appearing at 288.61 eV likely represents C atoms of depressant, probably the C bonded to O/S(C=S/C=O), which were in close to the carbon atoms of dixanthogen [25], thiourea[26], xanthate (CS2) [27] and the C bonded to N, S and O in IPETC [28]. The broad peak at 286.22 eV probably assigned to the C atoms of methylene group (��CH2��) or the carbon contamination presented in chalcopyrite (286.18 eV).
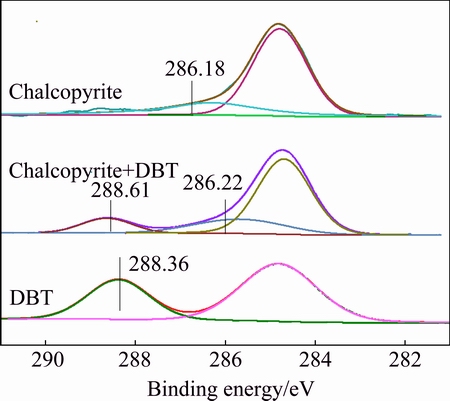
Fig. 6 C 1s spectra of chalcopyrite with and without DBT treatment at pH 8.0
The S 2p spectrum of the initial chalcopyrite is shown in Fig. 7. It has been reported that the S 2p3/2 peak originating from fully coordinated sulfur atoms in chalcopyrite��s crystal structure, i.e. monosulfide (S2-), centers at 161.4 eV [29,30] and elsewhere it has been reported to be at 161.5 eV [31]. These results agree well with those of our experiments(S 2p3/2 peaks, centered at 161.38 eV). On the other hand, the position of the S 2p3/2 peak for disulfide species (S22-) are reported to be 162.48 eV [32] and 162.5 eV [30], and these again agree well with the position of the second S 2p3/2 peak in our data(S 2p3/2 peaks, centered at 162.54 eV). In addition to the two S 2p doublets, Fig. 8 presents another peak positioned at 163.73 eV(Sn2-/S0), which is similar to previous XPS studies on electrochemically oxidized chalcopyrite [33]. The fitted S 2p3/2 peak at 169.22 eV is attributed to sulphate (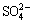 ), which is in good agreement with the report [34]. The chalcopyrite surface is mildly oxidized [35].
), which is in good agreement with the report [34]. The chalcopyrite surface is mildly oxidized [35].
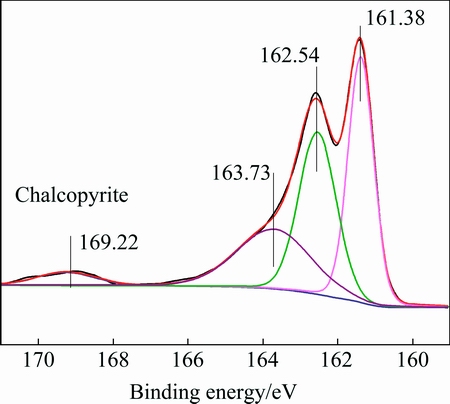
Fig. 7 S 2p spectrum of chalcopyrite at pH 8.0
In Fig. 8, the S 2p spectrum of DBT can be fitted by three doublets with the position of the S 2p3/2 splitting peaks at 161.14 and 162.59 eV and about 163.85 eV. The average FWHM values for these three components were about 1.32, 1.23, and 1.4 eV, respectively. The S 2p spectrums obtained from the chalcopyrite conditioned in DBT solution at pH 8 indicated the existence of several electron binding stages (Fig. 8). The S 2p line can be fitted by four doublets with the position of the S 2p3/2 splitting peaks at 161.48, 162.62, 164.07 and about 165.27 eV. The two former doublets have positions characteristic of the sample of chalcopyrite [29,30-32], while the excess of sulfur (latter doublets) might be assigned to S atoms in adsorbed DBT, in line with growing content of carbon (Fig. 6). Compared to the S 2p binding energy of DBT and the excess of sulfur presented on DBT treated chalcopyrite surface. The sulphate peak (169.23 eV) is clearly present in the spectra of original chalcopyrite at pH 8; however, this species vanished with the addition of depressant, which indicates that the oxidation of chalcopyrite is hindered with the addition of DBT which means a layer of DBT molecules might be covered on chalcopyrite surface. The intensity of the peak at 161.48 and 162.62 eV further proved that the extra sulfur atoms on chalcopyrite surface were donated by DBT.
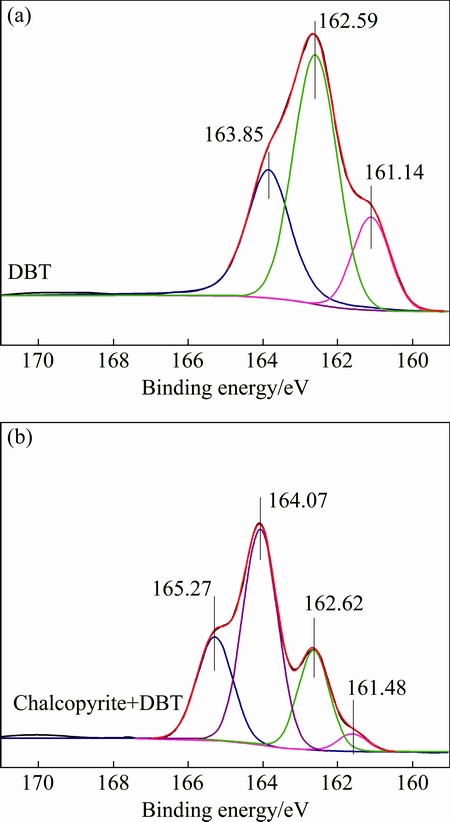
Fig. 8 S 2p spectra of DBT (a) and DBT-treated chalcopyrite (b) at pH 8.0
The Cu 2p XPS studies are shown in Fig. 9. The results suggested that binding energies for Cu 2p3/2 and Cu 2p1/2 peaks of original chalcopyrite are 932.47 and 952.29 eV, respectively. These results are in good agreement with those reported by GHAHREMANINEZHAD et al [30] and YANG et al [33]. After treatment with DBT, the binding energies for Cu 2p3/2 and Cu 2p1/2 peaks of chalcopyrite were shifted to lower binding energies (932.13 and 952.01 eV), which indicate that the differences might be attributed to the electron donation of sulfur atoms in depressant molecule. From the S 2p and Cu 2p spectra, it is possible to draw a conclusion that the sulfur atoms (electron donor) might interact with the copper ions (electron acceptor) under the tested conditions since the excess S 2p spectra obtained from chalcopyrite samples before and after DBT conditioning were shifted to higher binding energy, meanwhile, the Cu 2p spectra were shifted to lower binding energy.
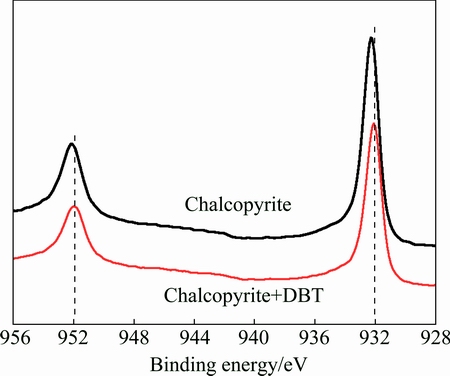
Fig. 9 Cu 2p spectra of chalcopyrite with and without DBT treatment at pH 8.0
3.5 Flotation separation of copper-molybdenum sulfides
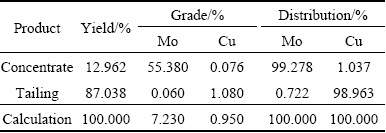
The pure mineral flotation tests using chalcopyrite and molybdenite only showed a marginal effect of depressant in copper-molybdenum sulfide separation [7]. However, since the pure mineral flotation tests could not demonstrate the competitive adsorption of DBT on the two minerals, testworks were carried out on copper-molybdenum bulk concentrate to evaluate the potential to use DBT as a selective depressant in copper-molybdenum separation. In the locked-circuit tests, it is not completed until the mass balance of concentrate plus tailing equal to feed was achieved. In this investigation, the average values of three tests were reported. The depressant used in bench scale tests were prepared according to literature without separation and purification [21]. The bulk concentrate, which was obtained from China Molybdenum Co., Ltd., with 0.95% Cu and 7.23% Mo collected from a day shift, was used in the locked-circuit flotation test. The results of locked-circuit were summarized in Table 1.
Table 1 Results of locked circuit test
4 Conclusions
1) The flotation performance and Zeta potentials of chalcopyrite and molybdenite indicated that a higher amount of DBT adsorbed on chalcopyrite surface than on molybdenite surface. The IR spectra and XPS results of chalcopyrite before and after treatment with DBT indicated that the mechanism of DBT adsorbing on chalcopyrite is mainly as physical adsorption.
2) Locked-circuit experiments were carried out and satisfied recovery and grade of molybdenite concentrate can be achieved (Table 1). Clearly, DBT will be a very selective flotation depressant in the differential separation of molybdenite from chalcopyrite, which could provide a viable alternative to sodium cyanide, sodium sulfide and sodium hydro-sulfide that are routinely used in commercial Cu-Mo flotation separation circuits.
Acknowledgements
The authors are grateful for the financial support from Jinduicheng Molybdenum Group Co., Ltd., (JDCMMC); The bench scale experiments of copper- molybdenum separation were conducted at China Molybdenum Co., Ltd.
References
[1] HIRAJIMA T, MORI M, ICHIKAWA O, SASAKI K, MIKI H, FARAHAT M, SAWADA M. Selective flotation of chalcopyrite and molybdenite with plasma pre-treatment [J]. Minerals Engineering, 2014, 66-68: 102-111.
[2] TRIFFETT B, VELOO C, ADAIR B J I, BRADSHAW D. An investigation of the factors affecting the recovery of molybdenite in the Kennecott Utah copper bulk flotation circuit [J]. Minerals Engineering, 2008, 21: 832-840.
[3] ANSARI A, PAWLIK M. Floatability of chalcopyrite and molybdenite in the presence of ligosulfonates.
[4] YIN Wan-zhong, ZHANG Li-rong, XIE Feng. Flotation of Xinhua molybdenite using sodium sulfide as modifier [J]. Transactions of Nonferrous Metals Society of China, 2010, 20: 702-706.
[5] ABDOLLAHI H, MANNAFI Z, SHAFAEI S Z. Romoval of copper from molybdenite concentrate by mesophilic and extreme thermophilic microorganisms [J]. International Journal of Mining Science and Technology, 2013, 23: 827-834.
[6]  J M, MAHAMUD M M, DZIOBA R. Tannins: the organic depressants alternative in selective flotation of sulfides [J]. Journal of Cleaner Production, 2014, 84: 723-726.
J M, MAHAMUD M M, DZIOBA R. Tannins: the organic depressants alternative in selective flotation of sulfides [J]. Journal of Cleaner Production, 2014, 84: 723-726.
[7] LI Ming-yang, WEI De-zhou, LIU Qi, LIU, Wen-bao, ZHENG, Ji-min, SUN Hong-jie. Flotation separation of copper-molybdenum sulfides using chitosan as a selective depressant [J]. Minerals Engineering, 2015, 83: 217-222.
[8] LI Ming-yang, WEI De-zhou, SHEN Yan-bai, LIU Wen-gang, CAO Shu-ling, LIANG Guang-quan. Selective depression effect in flotation separation of copper-molybdenum sulfides using 2,3-disulfanylbutanedioic acid [J]. Transactions of Nonferrous Metals Society of China, 2015, 25: 3126-3132.
[9] CHEN Jian-hua, LAN Li-hong, LIAO Xing-jin. Depression effect of pseudo glycolythiourea acid in flotation separation of copper- molybdenum [J]. Transactions of Nonferrous Metals Society of China, 2013, 23: 824-831.
[10] URBANIAK K, MLOSTON G, GULEA M, MASSON S, LINDEN A, HEIMGARTNER H. Thio- and dithioesters as dipolarophiles in reactions with thiocarbonyl ylides [J]. European Journal of Organic Chemistry, 2005, 8: 1604-1612.
[11] DEHMEL F, CIOSSEK T, MAIER T, WEINBRENNER S, SCHMIDT B, ZOCHE M, BECKERS T. Trithiocarbonates- exploration of a new head group for HDAC inhibitors [J]. Bioorganic and Medicinal Chemistry Letters, 2007, 17: 4746-4752.
[12] HAMM P C, GODFREY K L. Method of destroying vegetation with trithiocarbonates, US patent, US70427857A [P]. 1961-07-25.
[13] POSTMA A, DAVIS T P, LI Guo-xin, MOAD G, O, SHEA M S. Synthesis of well-defined polystyrene with primary amine end groups through the use of phthalimido-functional RAFT agents [J]. Macromolecules, 2006, 39: 5307-5318.
[14] ALI M F, ABBAS S A. Review of methods for the demetallization of residual fuel oils [J]. Fuel Process Technology, 2006, 87: 573-584.
[15] DEHMEL F, WEINBRENNER S, JULIUS H, CIOSSEK T, MAIER T, STENGEL T, FETTIS K, BURKHARDT C, WIELAND H, BECKERS T. Trithiocarbonates as a novel class of HDAC inhibitors: SAR studies, isoenzyme selectivity, and pharmacological profiles [J]. Journal of Medicinal Chemistry, 2008, 51: 3985-4001.
[16] OLIVA A, MOLINARI A, SANCHEZ L. New application of dimethyl trithiocarbonate-methyl thio carbonylation of 2,4-pentanedion and some beta-oxoesters [J]. Synthetic Communnications, 1998, 28: 3381-3386.
[17] VOS C F, DAVIDTZ J C, MILLER J D. Trithiocarbonates for PGM flotation [C]//International platinum conference platinum surges ahead. Sun city: The southern African Institute of Mining and Metallurgy, 2006, 169-174.
[18] MAKANZA A T, VERMANNK M K G, DAVIDTZ J C. The flotation of auriferous pyrite with a mixture of collectors [J]. International Journal of Mineral Processing, 2008, 86: 85-93.
[19] VENTER J A, VERMAAK, M K G. Mechanisms of trithiocarbonate adsorption: A flotation perspective [J]. Minerals Engineering, 2008, 21: 1044-1049.
[20] DU P R, MILLER J D, DAVIDTZ J C. Preliminary examination of electrochemical and spectroscopic features of trithiocarbonate collectors for sulfide mineral flotation [J]. Transactions of Nonferrous Metals Society of China, 2000, 10: 12-18.
[21] OLEXANDRA R, ROMAN L. Synthesis and anticancer activity in vitro of some 2-thioxo-4-thiazolidone derivatives [J]. Farmacia, 2007, 55(6): 640-648.
[22] BRAGA P F A, CHAVES A P, LUZ A B, FRANCA S C A. The use of dextrin in purification by flotation of molybdenite concentrate [J]. International Journal of Mineral Processing, 2014, 127: 23-27.
[23] MITCHELL T K, NGUYEN A V, EVANS G M. Heterocoagulation of chalcopyrite and pyrite minerals in flotation separation [J]. Advances in Colloid and Interface science. 2005, 114-115: 227-237.
[24] LORENZE R B, MAURICIO E, EDUARDO V, PABLO H, et al. Adsorption of biosolids and their main components on chalcopyrite, molybdenite and pyrite: Zeta potential and FTIR spectroscopy studies [J]. Minerals Engineering, 2015, 78: 128-135.
[25] SZARGAN R, KARTHE S. XPS studies of xanthate adsorption on pyrite [J]. Applied surface science, 1992, 55: 227-232.
[26] SRINIVASAN V, WALTON R A. X-ray photoelectron spectra of inorganic molecules. XX. Observation concerning the sulfur 2p binding energies in metal complexes of thiourea [J]. Inorganica Chimica Acta, 1977, 25: 185-186.
[27] DENG Mei-jiao, KARPUZOV D, LIU Qing-xia, XU Zheng-he. Cryo-XPS study of xanthate adsorption on pyrite [J]. Surface and Interface Analysis, 2013, 45(4): 805-810.
[28] BUCKLEY A N, HOPE G A, LEE K C, PETROVIC E A, WOODS R. Adsorption of O-isopropyl-N-ethyl thionocarbamate on Cu sulfide ore minerals [J]. Minerals Engineering, 2014, 69: 120-132.
[29] MIELCZARSKI J A, CASES J M, ALNOT M, EHRHARDT J J. XPS characterization of chalcopyrite, tetrahedrite, and tennanite surface products after different conditiong. 1. Aqueous solution at pH 10 [J]. Langmuir, 1996, 12: 2519-2530.
[30] GHAHREMANINEZHAD A, DIXON D G, ASSELIN E. Electrochemical and XPS analysis of chalcopyrite(CuFeS2) dissolution in sulfuric acid solution [J]. Electrochimica Acta, 2013, 87: 97-112.
[31] MCCARRON J J, WALKER G W, BUCKLEY A N. An X-ray photoclectron spectroscopic investigation of chalcopyrite and pyrite surfaces after condition in sodium sulfide solutions [J]. International Journal of Mineral Processing, 1990, 30(1-2): 1-16.
[32] KLAUBER C, PARKER A, BRONSWIJK W V, WATLING H. Sulphur speciation of leached chalcopyrite surfaces as determined by X-ray photoelectron spectroscopy [J]. International Journal of Mineral Processing, 2001, 62: 65-94.
[33] YANG Yi, HARMER S, CHEN Miao. Synchrotro-based XPS and NEXAFS study of surface chemical species during electrochemical oxidation of chalcopyrite [J]. Hyrdometallurgy, 2015, 156: 89-98.
[34] PARKER A, KLAUBER C, KOUGIANOS A, BRONSWIJK W V. An X-ray photoelectron spectroscopy study of the mechanism of oxidative dissolution of chalcopyrite [J]. Hyrdometallurgy, 2003, 71: 265-276.
[35] YANG Yi, HARMER S, CHEN Miao. Synchrotro X-ray photoelectron spectroscopic study of the chalcopyrite leached by moderate thermophiles and mesophiles [J]. Minerals Engineering, 2014, 69: 185-195.
��־�գ��� ΰ��������������������Ȼ�����Ծ�����Լƻ�
���ϴ�ѧ ��Դ�ӹ������﹤��ѧԺ����ɳ 410083
ժ Ҫ��С�����л����Ƽ�˫(�ȼ�)�����̼����(DBT)��һ��ͭ�����ѡ�������Ƽ�������λ�������������������DBT�ڻ�ͭ����������ǿ�ڻ����X���߹��������һ��֤��DBT�����ڻ�ͭ����棬���Ƽ�DBT���ڻ�ͭ��������������������Ϊ����ͭ���Ͼ���ѡ����������������DBT��һ��DZ�ڵĻ����Ѻ���ͭ��������Ƽ���
�ؼ��ʣ�ͭ����˫(�ȼ�)�����̼���ƣ����Ƽ�����ѡ���룻��������
(Edited by Yun-bin HE)
Foundation item: Project (2016zzts109) supported by the Innovation Driven Plan of Central South University, China; Project (B14034) supported by the National 111 Project, China
Corresponding author: Wei SUN; Tel: +86-731-88830482; E-mail: sunmenghu@csu.edu.cn
DOI: 10.1016/S1003-6326(17)60100-6

