���������廯ѧ��CH4/CO2ת���о���չ
��Դ�ڿ������ϴ�ѧѧ��(��Ȼ��ѧ��)2021���1��
�������ߣ����� ������ ��ʱ�� ���� ���� ���Ĺ�
����ҳ�룺70 - 86
�ؼ��ʣ���ѧ����CH4/CO2ת��������������
Key words��chemical looping; CH4/CO2 conversion; Fe-based oxygen carriers
ժ Ҫ���ӹ����Ż���������ѡ�����Ż��Լ���Ӧ����ƽǶȶԻ�ѧ��CH4/CO2ת���о���չ����������̽�֡�Ŀǰ����Ķ��ֻ�ѧ��CH4/CO2ת�����տ��Թ��ΪCH4��CO2��ͬһ��Ӧ���Լ�CH4��CO2�ڲ�ͬ��Ӧ��2�ࡣ����ѧ���������������������ʺ��ڻ�ѧ��CH4/CO2ת���������������������������������������ܼ��鷴Ӧ�ԡ���ԭ��ȡ�����ѡ���ԡ�ѭ���ȶ��ԣ��Ժ��о��ص��ǻ��Ըߡ��ȶ��Ժá��ʺϴ��ģ�Ʊ������������壬CH4/CO2���������������ķ�Ӧ������Ҫ�����о�����������Ӧ���㷺���ڻ�ѧ�����̣�ȼ�Ϸ�Ӧ���������ʵ�ִ��ģ��ҵ���Ĺؼ��������о�������������������廯ѧ��CH4/CO2ת�������������õ�ǰ����
Abstract: The advances of chemical looping technology for CH4/CO2 conversion in term of process optimization, selection and development of oxygen carriers as well as reactor design were reviewed. The chemical looping CH4/CO2 conversion processes include CH4/CO2 reacting in the same reactor and different reactors. Thermodynamic calculation results prove that Fe-based oxygen carriers are promising in the field of chemical looping CH4/CO2 conversion. Different supports and promoters can improve reactivity to CH4, reduction extend, product selectivity and stability for Fe-based oxygen carrier. One of the key issues in the future is the development of Fe-based oxygen carriers that have high reactivity, excellent stability and are suitable for large-scale production. The reaction mechanism of CH4/CO2 on Fe-based oxygen carriers requires further research. Fluidized bed reactors have been employed in chemical looping processes widely, and the design of fuel reactor is crucial for industrialization of chemical looping CH4/CO2 conversion. These existing results show Fe-based chemical looping technology for CH4/CO2 conversion is promising.

DOI�� 10.11817/j.issn.1672-7207.2021.01.007
������1������2����ʱ��2������1������1�����Ĺ�2
(1. ��ʯ���Ͼ����̹�˾������ �Ͼ���211100��
2. ���ϴ�ѧ ��Դ�뻷��ѧԺ����Դ��ת��������̲�ؽ������ص�ʵ���ң����� �Ͼ���210096)
ժҪ���ӹ����Ż���������ѡ�����Ż��Լ���Ӧ����ƽǶȶԻ�ѧ��CH4/CO2ת���о���չ����������̽�֡�Ŀǰ����Ķ��ֻ�ѧ��CH4/CO2ת�����տ��Թ��ΪCH4��CO2��ͬһ��Ӧ���Լ�CH4��CO2�ڲ�ͬ��Ӧ��2�ࡣ����ѧ���������������������ʺ��ڻ�ѧ��CH4/CO2ת���������������������������������������ܼ��鷴Ӧ�ԡ���ԭ��ȡ�����ѡ���ԡ�ѭ���ȶ��ԣ��Ժ��о��ص��ǻ��Ըߡ��ȶ��Ժá��ʺϴ��ģ�Ʊ������������壬CH4/CO2���������������ķ�Ӧ������Ҫ�����о�����������Ӧ���㷺���ڻ�ѧ�����̣�ȼ�Ϸ�Ӧ���������ʵ�ִ��ģ��ҵ���Ĺؼ��������о�������������������廯ѧ��CH4/CO2ת�������������õ�ǰ����
�ؼ��ʣ���ѧ����CH4/CO2ת��������������
��ͼ����ţ�X743 ���ױ�־�룺A ���ſ�ѧ(��Դ����)��ʶ��(OSID)
���±�ţ�1672-7207��2021��01-0070-16
WANG��Genbao1, HU��Jun2, CHEN��Shiyi2, ZHANG��Lei1, LI��Meng1, XIANG��Wenguo2
(1. Sinopec Nanjing Engineering & Construction Incorporation, Nanjing 211100, China;
2. Key Laboratory of Energy Thermal Conversion and Control of Ministry of Education, School of Energy and Environment, Southeast University, Nanjing 210096, China)
Abstract: The advances of chemical looping technology for CH4/CO2 conversion in term of process optimization, selection and development of oxygen carriers as well as reactor design were reviewed. The chemical looping CH4/CO2 conversion processes include CH4/CO2 reacting in the same reactor and different reactors. Thermodynamic calculation results prove that Fe-based oxygen carriers are promising in the field of chemical looping CH4/CO2 conversion. Different supports and promoters can improve reactivity to CH4, reduction extend, product selectivity and stability for Fe-based oxygen carrier. One of the key issues in the future is the development of Fe-based oxygen carriers that have high reactivity, excellent stability and are suitable for large-scale production. The reaction mechanism of CH4/CO2 on Fe-based oxygen carriers requires further research. Fluidized bed reactors have been employed in chemical looping processes widely, and the design of fuel reactor is crucial for industrialization of chemical looping CH4/CO2 conversion. These existing results show Fe-based chemical looping technology for CH4/CO2 conversion is promising.
Key words: chemical looping; CH4/CO2 conversion; Fe-based oxygen carriers
��Ȼ�������ú��ʯ����һ�����Ļ�ʯ��Դ������Ҫ�ɷ��Ǽ��顣��Ȼ��ֱ��ȼ�����þ���Ч��ͣ������Ȼ��ת�������õ��㷺��ע������������ת�������Ǽ���������������(steam methane reforming��SMR)�����ϳ������ٽ��ϳ��������Ʊ������������״����ϳ��͵Ȼ�ѧƷ[1-2]����ͳ��SMR����ʹ�ø����������������й�ʽ��Ӧ��������ȼ��ȼ���ṩ��Ӧ�ȣ�������ˮ������Ӧ��ͨ��ˮ���任�����롢�����Ⱥ������ջ������H2��CO���ʵ����ȵĺϳ���[3]����ͳ��SMR���մ��ڵ����Ա���Ҫ�У������뷴Ӧ����Ҫ��ߡ��������̸��ӡ�ȼ��ȼ�չ���CO2�ŷ�[4]����ȼ��ȼ�մ����ŷ�CO2����ȫ�������ů�������صı����£�����ʵ��CO2��ԭ�����ò��Ʊ��ϳ����ļ������������(dry reforming methane, DRM)����˹㷺�Ĺ�ע��Ȼ����DRM���վ��빤ҵ�����������ս����Ҫ����������Ĺ����������CH4/CO2��Ӧ�����д��������̼����ˮ���任��Ӧ�Լ������´��������ս�ᵼ�´�����Ӧ���ܿ��ٽ��ͣ��÷�Ӧ��ǿ���ȷ�Ӧ�����ø�Ч������ʵ��������Ӧ�����Ѷȵ�[5-6]����ˣ�ؽ��һ����Ч�ߡ����̼��ŷŵ͵�CH4/CO2ת�����ա���ѧ����һ�����͵Ļ�ѧת������Դ���ü������ü�����һ����Ӧ�ֽ�ɶ���ӷ�Ӧ������ѭ�����������д������ʺ�������ʵ���˲�����Է��롣1983�꣬RICHTER��[7]����˻���ѭ�������������ȼ�ռ������ü���ʹ��ȼ�Ϸ�Ӧ���Ϳ�����Ӧ����ȼ����ȼ�Ϸ�Ӧ���л�ԭ�����壬����ԭ�������ڿ�����Ӧ���б�����������������ȼ�շ�Ӧ�ֳ���2���������ӷ�Ӧ��������ȼ�չ��̵IJ������ԣ�ʵ����CO2��������N2���Է��룬��������ִ���ѧ�������ij���[8]������������ѧ��������Ϊ�������̼�Ч�ʸ��ܵ�Խ��Խ��Ĺ�ע�������˶��ּ�����Ŀǰ��Ҫ�л�ѧ��ȼ�ա���ѧ����������ѧ����������ѧ�����⡢��ѧ���ư�����ѧ�����������[9-11]����ѧ������Ҳ�������õ�ȼ����Ӧ�ԣ�����������ú�������ʵȹ���ȼ�ϡ��͵�Һ��ȼ���Լ���Ȼ������������������ȼ�ϵ�ת����չʾ�����õ�ǰ��[12]����ѧ��������������������������ϣ�ʵ��CH4�ĸ�Ч���ת����CO2�Ļ�ԭ�����á��������ߴӹ����Ż���������ѡ�����Ż�����Ӧ�����Լ���Ӧ����ƽǶ������˻�������������Ļ�ѧ����CH4/CO2ת����ϵ��о���չ��Ϊ��ѧ��CH4/CO2ת�����õ�δ����չ�ṩ�ο���
1 ��ѧ��CH4/CO2ת������
��ͳ��DRM�����У��������л�ԭԤ����֮��ͨ��CH4/CO2��������ɺϳ�����ʹ��ȼ��ȼ���ⲿ���ȡ�Ŀǰ�����˶��ֻ�ѧ������������������������գ����Է�ΪCH4��CO2�ڲ�ͬ��Ӧ���ڷ�Ӧ�Ĺ����Լ�CH4��CO2��ͬһ��Ӧ���ڷ�Ӧ�Ĺ��ա�
1.1��CH4��CO2�ڲ�ͬ��Ӧ���ڷ�Ӧ�Ĺ���
CH4��CO2�ڲ�ͬ��Ӧ���ڷ�Ӧ�Ĺ���ԭ��ͼ��ͼ1��ʾ�������黯ѧ��ȼ�չ����е��������ɿ�����ΪCO2���Ϳ���ʵ����CH4ת����ͬʱ��ԭCO2����ȼ�Ϸ�Ӧ���У��������������������ѧ���ʵIJ�ͬ��CH4�ᷢ��3�ַ�Ӧ����ȫ���������������Լ��ѽ⡣NAJERA��[12]��CO2���õĽǶȳ��������ѧ�����������(chemical looping dry reforming��CLDR)��CLDR��CH4��ѧ��ȼ����ʹ�ÿ�������������ı�Ϊʹ��CO2���������壬�����ط�������̶�����Ӧ����ʹ��������������˲��ԣ���������Ӧ�����CO2ת���ʴ���90%��MORE��[13]�Ʊ���FexNi1-x-CeO2�����壬ͨ������������ɷ���ȼ�Ϸ�Ӧ���л���˸ߺϳ���ѡ���ԣ���������Ӧ����ʹ��CO2���������壬1 000 ���£�Fe0.88Ni0.12-CeO2������CH4��������ѡ���Գ���90%��CO��H2���ʵ�����ԼΪ0.5��������CO2ת����ԼΪ90%��ʵ���˺ϳ����Ʊ���CO2��ԭ���ã�LOFBERG��[14-16]ͨ������CeO2������֤���˸ù��յĿ����ԣ�GE��[17]ʹ��V2O3������Ҳ��֤�˸ù��յĿ����ԡ�LIM��[18]����˻�ѧ�������ѽ���������(chemical looping reforming-decomposition��CLRD)��ȼ�Ϸ�Ӧ���з��������ѽ����ⷴӦ���ѽ����ɵ�̼�ͱ���ԭ�����������������Ӧ����CO2��Ӧ����CO���ù����ܹ�ʵ��CO��H2�ķ����Ʊ���SUN��[19]��NiyFe3-yO4-Ca2FexAl2-xO5����������Ʊ�����������ֱ�Ϊ96.3%��95.2%��H2��CO���ù��������������塢�����������Լ����������������������˴����о�[20-22]����CH4��CO2ͨ�벻ͬ�ķ�Ӧ����CH4�ѽ�����Ļ�̼������CO2�����α����ĵ��������˻�̼���´���ʧ������⣬CO2��H2�����ڲ�ͬ�ķ�Ӧ����Ҳ��������ˮ���任��Ӧ��
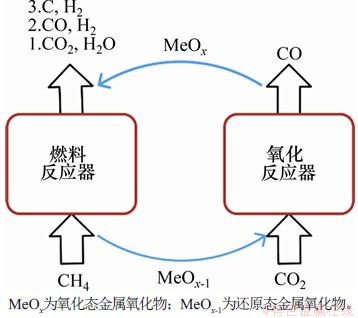
ͼ1��CH4��CO2�ڲ�ͬ��Ӧ���ڷ�Ӧ�Ĺ���ԭ��ͼ
Fig. 1��Schematic diagrams of chemical looping processes that CH4 and CO2 appear in different reactors
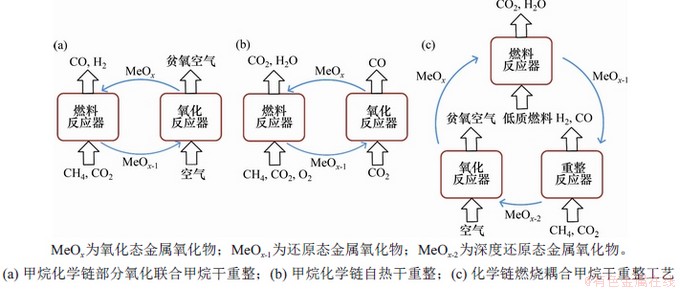
ͼ2��CH4��CO2��ͬһ��Ӧ���ڷ�Ӧ�Ĺ���ԭ��ͼ
Fig. 2��Schematic diagrams of chemical looping processes that CH4 and CO2 appear in the same reactor
�����������Ӧ���������Ĺ�Ӧ��ʽҲ�DZ��뿼�ǵ����⡣WARREN��[23]ʹ��̫���ܹ�Ӧ��ѧ����������������������������̫����ת��Ч�ʿ��Դﵽ10.06%����̫����ת��Ϊ��ѧ�ܴ��档BHAVSAR��[24]��CO2������Ӧ�������ӿ���������Ӧ������ǿ���ȵļ����������Ӧת��Ϊ���ȷ�Ӧ��ʵ�������ȣ����ڲ����˶����Ŀ���������Ӧ�������Ա���ʹ�øߺ��ܵĿշ�װ�ã��������Ч�ʣ����ұ���O2��ȼ�ϵ�ֱ�ӽӴ��������˱�ը���ա����ǣ����о��������������ȫ����������ᵼ�ºϳ�����ѡ���Խ���[25]��
��CH4��CO2�����ڲ�ͬ��Ӧ���У�����Ӧ�������ѡ����Ҫ��ϸߣ�ͨ���Ż������������������������ȷ�ʽʵ�ָ߲���ѡ������Ŀǰ�о����ص㡣
1.2��CH4��CO2��ͬһ��Ӧ���ڷ�Ӧ�Ĺ���
ͼ2��ʾΪ����CH4��CO2��ͬһ��Ӧ���ڷ�Ӧ�Ĺ���ԭ��ͼ��KANG��[26-27]���һ�ּ��黯ѧ�������������ϼ��������(chemical looping partial oxidation of methane with dry reforming of methane, CLPD)��ȡ�ϳ����ķ�������ͼ2(a)��ʾ���÷�����ȼ�Ϸ�Ӧ����Ӧ����ΪCO2��CH4������壬CO2��CH4�����ʵ����ȵ���1��CO2��CH4���ʵ����Ƚϵ͵ķ�Ӧ��ͬʱ���������������Ӧ�����鲿��������Ӧ�ͼ�����ȫ������Ӧ��������Ӧ���з�Ӧ��Ϊ�����������������������岢Ϊȼ�Ϸ�Ӧ���ṩ��������Ce����Fe2O3/Al2O3�������ϵ�ʵ����������CO2��CH4���ʵ�����Ϊ0.28ʱ��CH4ת���ʿɴ�93.11%��H2��CO���ʵ�����Ϊ2.04��KATHE��[28]Ҳʹ�����Ѹ��������彫����������뻯ѧ����ϣ�ʵ���ģ������ʾ��1 035 ����ͨ��CO2��CH4���ʵ�����Ϊ0.3�ķ�Ӧ����CH4ת����Ϊ94.1%������ϳ�����H2��CO���ʵ�����Ϊ1.41��GALVITA��[29-32]��CO2ת�������ȵĽǶȳ�������˼��黯ѧ�����ȸ���������(catalyst-assisted chemical looping auto-thermal dry reforming��CCAR)����ͼ2(b)��ʾ���÷�����ȼ�Ϸ�Ӧ����������Ӧ����ɣ�ʹ�þ��д�������˫���������壬ȼ�Ϸ�Ӧ����ͨ��ķ�Ӧ��ΪCH4��CO2��O2�����������CH4��CO2��Ni�������淢��������Ӧ�õ��ϳ��������ֺϳ����봿��ȼ���ṩ������ʣ��ϳ�������ʹ��������Ȼ�ԭ��������Ӧ������Ȼ�ԭ����������CO2��Ӧ����CO���ù��տ�����ʵ�����ȵ�ͬʱת�������CO2��SPALLINA��[33]����ѧ��ȼ�չ���������������������ϣ���ͼ2(c)��ʾ����ȼ�Ϸ�Ӧ����ʹ�õ���ȼ��������������澧���������ĵ��������������������д���������Ӧ����������������Ӧ����CH4/CO2�����������������Ӧ���������������о������Ľ�һ�����������������ɵĺϳ������Խ�һ������H2��CH3OH������ģ��������ʹ��NiO������ʱ������Ӧ����CH4ת���ʿɴ�92%������ʵ��H2��CH3OH�Ʊ��Ľ���CO2�ŷš�ZHU��[34]��������Ӧ�������ˮ����������Ӧ����ˮ��������������H2��������Ӧ���в����ĺϳ�����Ͽ�������H2/CO�ȿɵ��ĺϳ������ù��յĿ�����ʹ��Fe2O3/Al2O3�����������֤ʵ�������廹ԭ�̶�Ϊ33%ʱ��ͨ��CH4��CO2���ʵ�����Ϊ1�ķ�Ӧ����CH4ת���ʺͺϳ������ʷֱ���Դﵽ98.32%��3.84��
CH4��CO2�ڲ�ͬ��Ӧ���ڷ�Ӧʱ���������������������壻��CH4��CO2��ͬһ��Ӧ���ڷ�Ӧʱ�������������ڹ����������������ͬʱ��������������Ӧ�������ǰһ�ָ����ӡ�CH4��CO2��ͬһ��Ӧ���ڷ�Ӧ�����У�Ŀ�������ҪΪ�ϳ�������CH4��CO2�ڲ�ͬ��Ӧ���ڷ�Ӧ��Ŀ�������Զ���������ʵ�ָ�ѡ���Զ��������Լ���Ӧ���̿��Ƶ�Ҫ��ߡ�CH4��CO2��ͬһ��Ӧ���з�Ӧ�����Լ���CH4�ڹ��ɽ���������ѽ��̼���⡣������ԣ�CH4��CO2��ͬһ��Ӧ���ڷ�Ӧ�����ϳ������վ��и��õĹ�ҵ��ǰ����
2 ��ѧ��CH4/CO2ת���������о�
�������ڶ����Ӧ����ѭ��ʹ�ã��ǻ�ѧ�������Ĺؼ�[35]����ѧ����������������Ҫ�߱��������У�������������������Ӧ�����Լ����ȶ��ԣ���Դ�㷺�۸�������������Ѻã�ʹ����������Ӧ��ʱ��Ҫ���������������Ŀ�ĥ�������Լ���������[36]���о��������ڻ�ѧ��CH4/CO2ת����������ʱ������Ҫ���Ǽ��鷴Ӧ��ѡ���ԣ�CO2��ת��������
2.1�������������ѧ����
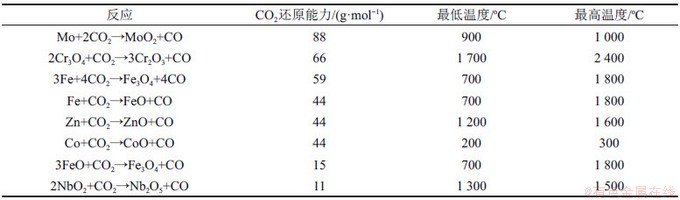
���õ�������ΪFe��Co��Ni��Cu��Mn���������600~1 200 ����CH4�볣��������ķ�Ӧ����ѧ������ͼ3��ʾ��lg KԽ��˵��������������CH4��ӦԽ���ף�MnO2/Mn2O3��Mn2O3/Mn3O4��Co3O4/CoO��CuO/Cu2O��Fe2O3/Fe3O4��NiO/Ni����������CH4������Fe2O3/Fe3O4��Mn2O3/Mn3O4��CuO/Cu2O��NiO/Ni������ʹCH4��ȫ����[37-38]��ZENG��[39]Ҳ������ѧ�Ƕ�ʹ������ѹpO2���¶ȵĹ�ϵ���������ʺ���ȫ�����Ͳ��������������壬�������úϳ�����Ũ���뾧����/ȼ�ϱ�������أ�FeO��Fe3O4��WO2��MoO2�ʺ���H2O��CO2�Ļ�ԭ�����1��ʾ��NAJERA��[12]��CO2�������ɽ������������������ѧɸѡ�������������������ɽ�������CO2��ԭ�������������������ڽϿ����¶ȷ�Χ�ھ��нϸߵ�ת���ʡ�����������������������۸��������Դ�㷺�������Ѻã��������ǰ����
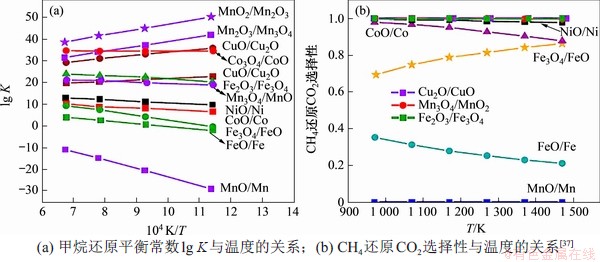
ͼ3����ͬ����������ѧ������
Fig. 3��Thermodynamic results of different oxides
��1����ͬ������CO2��ԭ����(����¶�ָCO2ת���ʳ���20%����ʼ�¶ȣ�����¶�ָ�۵�)
Table 1��CO2 reduction capacity of different oxygen carriers (minimum temperature is the starting temperature of CO2 conversion over 20%��maximum temperature is the melting temperature)
����ѧ����Ҳ֤���������������CH4/CO2�����Ŀ����ԣ�KATHE��[40]ʹ��Aspen������RGibbsģ�飬�Ա�����900 ���101.3 kPa��ʹ��O2���Ӽ����������Լ�ʹ��Fe2O3��Fe2O3��TiO2�������о����������������IJ���ֲ����������ʹ�þ�����������ʱ���ڽϴ��[O]/CH4��Χ�ڣ�1 mol CH4���Ի��2 mol���ϵĺϳ����������[41]Ҳ��CH4/CO2������벻ͬ��������ķ�Ӧ����������ѧ�������ڲ�ͬ�����������CH4��CO2��������Чת�������������ﻹԭ��FeO��Fe�Ļ����ʱ������ת���������98%��KANG��[26]��������CH4/CO2��������������ﷴӦʱCO2��CH4���ʵ����ȶԲ���ֲ���Ӱ�죬�������ͨ������CO2��CH4���ʵ����Ⱥ�[O]/CH4���Ի��H2��CO���ʵ�����Ϊ2�ĺϳ�����
2.2������������ĸ���
���������ڻ�ԭ�������ж��ּ�̬��Fe2O3��Fe3O4��FeO��Fe����ԭCO2��Ӧ�ʹ�CH4/CO2������Ӧ����Ҫ��������ﵽ��Ȼ�ԭ��״̬��FeO��Fe�������¶Ƚϵ�[42]����������������ڸ����³�ʱ�䷴Ӧ�ᵼ���ս��ʧ����ǿ����ܹ�Ӧ���ڻ�ѧ��CH4/CO2ת�����̵��������������Ҫ�˷������⡣��Σ���������������ȣ��������������ķ�Ӧ���Խϲ�¼���ת���ʵ�[35]����Ȼ�ԭ��Ҫ�ϳ��ķ�Ӧʱ�䣬�Լ����������Ӧ�Ĵ�����Ҳ��������ˣ�����������ͨ�����ɻ��Գɷ֡������Լ�������ɡ�
�����������ķ�Ӧ���ԡ�����ѡ���ԡ��ȶ��ԡ�����̼�����Ծ��нϴ��Ӱ��[43-46]��Ŀǰ����ѧ�����������������峣�õ�������Al2O3[47-49]��MgAl2O4[50-53]��SiO2[24]��TiO2[54-55]�Լ�ZrO2[56-57]�ȣ���Щ���岻�������ӵ��Ӵ���������������������Ҫ��֧�š���ɢ���Գɷֵ����á�ʹ��Al2O3��TiO2�Լ�SiO2Ϊ����ʱ����ԭ�����л�����FeAl2O4��FeTiO3�Լ�Fe2SiO4�ȶ��Գɷ֣��������������ͣ�Ӱ�����������һ����Ӧ����[38]��MgAl2O4�������õ����ȶ��Ժͱ����ݣ����һ�ѧ�����ȶ��������������ﷴӦ���ɶ��Բ��DHARANIPRAGADA��[51]�Ʊ���һϵ��Fe2O3����������10%~100%��Fe2O3/MgAl2O4����������CO2��ԭ����������Ʒ�ж�������MgFeAlOx�⾧ʯ�࣬MgFe3+AlOx���Ա���ԭ��MgFe2+AlOx������Fe2O3���ս��MgFeAlOx��������ԭ�����е��ս����أ�Fe2O3�����͵���Ʒ���ֳ���Fe2O3�����ߵ���Ʒ���õ��ȶ��ԣ�������������Ϊ10%��������CO���������BUELENS��[58]����������Ϊ10%��30%�Լ�50%��Fe2O3/MgAl2O4�������������1 000��H2��ԭCO2����ʵ�飬�������10Fe2O3-MgAl2O4���ȶ�����ã��������������١�DE VOS��[53]�Ա���Fe2O3/MgAl2O4��Fe2O3/Al2O3������ļ��黯ѧ���������ԣ�Fe2O3/MgAl2O4��������ֳ����õ��ȶ��ԣ�������ѭ�������б��ֽϺõĻ�еǿ�ȡ�CeO2�Լ����ѿ��������ϳ���ѡ���Ժã������������ͣ��۸��Ҿ������õ����ӵ��Ӵ����������������������������ʱ�����������������Ļ��ԡ��ı�����ѡ���ԡ�CeO2/Fe2O3�ڻ�ѧ������ˮ���������о������õ�CH4��Ӧ�Ժ���������[59-60]��ZHU��[61]��Ce1-xFexO2(x=0��0.1��0.2��0.3��0.4��0.5��0.6��1.0)���������ڻ�ѧ������ˮ���������о���������������±�������õľ���������CH4��ȫ��������CO2����������CO����Ҫ���Fe���������ർ��COѡ���Խ��ͣ�850 ����Ce0.5Fe0.5O2������������õ��ȶ��ԡ�GALVITA��[62]����ͬCeO2��������CeO2/Fe2O3���������ڻ�ѧ��CO2ת����CeO2������������70%ʱ�۲ⲻ��Fe2O3�࣬��500 �濪ʼ��Fe��һ��������Fe3O4��CeO2/Fe2O3������CO���������DZ�Fe2O3������Ĵ����ڶ��ѭ���б����ȶ���DHARANIPRAGADA��[42]��ϸ�о���CeO2/Fe2O3�������ڻ�ѧ��CO2ת�������е�ʧ��������ս��Ǹ�Fe������ʧ�����Ҫԭ��CHO��[63]����ʹ�����ӵ��ӵ�����Ϊ���������������������õļ��鲿��������Ӧ���ԣ�����ĵ����Ժ�������ԭ�ȶ��Զ�����������Ҫ��Ӱ�졣���ѿ��CH4��������ѡ���Ժõ����������ͣ���Fe2O3�����������Dz�������ѡ���ԲNEAL��[64]�����ѿ���Fe2O3����Ʊ���Fe2O3@La0.8Sr0.2FeO3-���˿�������������CH4�����������ϳ������ʱȸ��ѿ�������ߡ�
�����ļ�����Ը����������ȶ��ԣ��������������Fe2O3/Al2O3��������Ը�����������ȶ���[65]��Si����Fe2O3/Al2O3�������������Si-Al-O�ṹ������������ȶ�[66]�������ļ���Ҳ���Ը���������ķ�Ӧ���ԣ�HAFIZI��[67]ʹ��Ca����Fe2O3/Al2O3�����壬��700 ������CH4��ת���ʴﵽ100%�����ұȽ���Ce������Ca���������壬������������Ӱ��������Ļ��ԡ��ȶ����Լ�����̼���ܣ�ʹ��5%��Ce��Ca�����Ϳ������������ķ�Ӧ����[68]��IMTIAZ��[69-70]ʹ��Cu���Ե����������壬Cu0���Դٽ�FeO��ԭ��Fe0������Cu�����ڱ������������������Ŀ���̼���ܣ�QIN��[71]ʹ��1%��Cu�������������壬��700 ��ʱCH4ת������δ�����������470%��ϡ��������La��Ce��Y��Ҳ�����ڸ������������壬KANG��[72]����Y���Ե�Fe2O3/Al2O3������CH4ת���ʴ�98%��ͬʱCOѡ���Կɴ�98%����Ҫ����Ϊ������Y3Fe2Al3O12���ࡣQIN��[73-74]ʹ������La�������������壬550~780 �淶Χ�ڷ�Ӧ���ʶ�����δ���������壬����������������巴Ӧ���Ե�ͬʱά������������TANG��[75]��La/Ce���ε�Fe2O3/Al2O3���������ڻ�ѧ�������������La��Ce�������������������������Ŀ��ս����������Ʋ�����̼���γɡ�Ni����������������������CH4ת�����о���Ϊ�㷺������Ni��CH4���õĴ����ԣ�Ni���Ե�����������ͨ���ֿ��Գ�Ϊ˫���������壬����������������Ni��������������CH4�ķ�Ӧ��������������[76]�������ĵ���Ni/Fe�ȿ��Ըı�CH4��Ӧ��ѡ����[77]��CO2�����Խ���������Ni0[13]��Ni���Ե�����������Ҳ������CH4��CO2ͬʱת������CLPD�����У�Ni���Ե�Fe2O3/Al2O3���������CH4ת���ʱ�Fe2O3/Al2O3������ĸ�[27]����CCAR�����У�����Ni��Դ�����������������CH4��CO2��O2���ʵ�����Ϊ1.0:1.0:0.5����������Ȼ�ԭ[29]��
Fe2O3Ҳ�������������������ɾ��и��ѿ�ṹ�ĸ������������LaFeO3���ƶ�����ǿ��CH4��Ӧ����������������ѡ���Ժ�[78]�����㷺������CH4��ѧ���������̡�DAI��[79]ͨ����ͬ�¶��µ�����ʵ�鷢��LaFeO3��CH4��Ӧ�����뷴Ӧ�¶ȹ�ϵ�ϴ���(>800 ��)�ºϳ������ʸߡ�MIHAI��[78,80]�о�����CH4��ԭLaFeO3����Fe3+���ԽϿ�ת��ΪFe2+�����ǽ�һ����ԭ�Ƚ����ѣ����ҽϴ��LaFeO3�����б���Fe��O���������±�����������ӣ������ںϳ��������ɡ�A��Bλ���������LaFeO3��Ӧ���Եij��÷���[81-82]��ZHAO��[83]�Ʊ���La1.6Sr0.4FeCoO6����������CH4��ѧ��ˮ�������������ֽ�����ЭͬЧӦ������Ч���CH4��Ӧ�ԡ�MAITI��[84]ͨ���ܶȷ������ۼ����ʵ���о���La(1-x)SrxFe(1-y)CoyO(3-��)��������λ�γ�����CO2���������ܣ��������CO2���������ܿ��Ժ�������CO2ת��������ʵ����Ҳ����CO2������Чת���������������������Ҳ�ܵ��㷺��ע��SUN��[85]�Ʊ���һϵ��FeCa�������������Ca2Fe2O5���������һ����ԭ��Fe0�����Ҿ���������CH4��Ӧ�ԡ�����Ca2Fe2O5������������Ȼ�ԭ����Fe0�����㷺���ڻ�ѧ���������[86-88]��SHAH��[89-90]��������ѧ�������ģ�⣬����Ca2Fe2O5�������ڼ��黯ѧ�������ƺϳ�������Ч�ʸ��ڴ�ͳ����������������CuO��Ca2Fe2O5�������������CH4�ڽϵ��¶���ת��Ϊ�ϳ�����MILLER��[91]��ʹ���ܶȷ�������(DFT)�о�CH4��CaFe2O4��Ӧ����ʱ����H2��CO����ѧ����Ca2Fe2O5��Ӧ�Ŀ����Խϵͣ����Ca2Fe2O5�������õĺϳ���ѡ���ԡ�Fe2O3�������γɾ��м⾧ʯ�ṹ�ĸ��������HUANG��[92-93]�Ƚ���NiFe2O4��NiO-Fe2O3��NiO�Լ�Fe2O3�Ļ�ѧ��������������ܣ�4������������������Ӹߵ�������Ϊ��NiO��NiFe2O4��NiO-Fe2O3��Fe2O3������ԭ�����Ӹߵ�������ΪNiFe2O4��NiO-Fe2O3��Fe2O3��NiO��NiFe2O4��Fe��NiЭͬ���õ���NiFe2O4������ԭ����ǿ����ȻNiFe2O4��ѭ������������ȫ������������10��ѭ�������о��нϺõ��ȶ��ԡ�ZENG��[94-95]�Ʊ���CoFeAlOx�⾧ʯ�������壬ͨ�����ƻ�ԭ��ȿ�����Ч�����������ȶ��ԣ���ͨ�����벻ͬ����Ni��Cu��Coʵ���˼⾧ʯ�����������Ȼ�ԭ���������νṹ����CH4�����ԣ�ZHU��[96-97]�Ʊ���LaFe3Al9O19���������ڼ��黯ѧ�������������к��ʾ����ߴ�ĺ�����������������CH4��Ӧ�Ժͺϳ���ѡ���Ժã����������Ȼ�ԭ��û�в����ṹ�仯����ˣ�CO2��ԭ����ǿ��ѭ���ȶ���
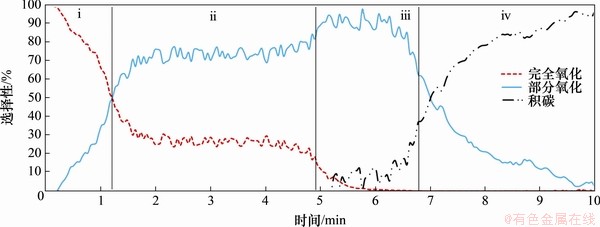
ͼ4��CH4��ԭFe2O3���Ľ�[102]
Fig.4��Four reaction regions of Fe2O3 reduced by CH4[102]
��Ȼ����ʯ�۸�����������Ѻã�������������ȿ�ʯ��CH4��ѧ��ȼ���о������õķ�Ӧ�Ժ��ȶ��ԣ�����ȵͼۺ�������Ҳ������CH4��ѧ��ȼ��[98]����ʯ�����ڲ����������Խϲ����CH4�Ʊ��ϳ������о����٣�SUN��[99]ʹ��Ni���Եij��������CH4/CO2��ѧ�������Ʊ��ϳ�����Ni������߳�����ķ�Ӧ���ԣ�CH4��Ni���Գ��������ͬʱ���������������������Ӧ�����Ʊ��ϳ�����
����ֵĿռ�ֲ�������������Ҳ��Ӱ�죬HU��[100]�Ʊ���Fe2O3/ZrO2@ZrO2�˿������������ڻ�ѧ��CO2��ԭ��������������ȶ��Զ�����ߡ�NEAL��[64]�Ʊ���Fe2O3@La0.8Sr0.2FeO3-�������壬�Dz�ĸ��ѿ�����ѡ���Ժã��˲�Fe2O3�������ߣ���Ч�ؽ�������ߵ����ơ�MORE��[101]��Fe2O3��NiO������Ϻ����ڻ�ѧ�������������CH4��Ni�������ɵ�C��H2��ԭFe2O3�������м���������Ч�ؽ���ͬ���ܵ��������������HU��[30]��CCAR�����в��ò�ͬ�Ŀռ�ֲ��������ʾ�ռ�ֲ��Բ���ֲ��Լ�CH4ת������Ӱ�졣
CH4��ԭFe2O3�����з����ķ�Ӧ����[102]����ͼ4��ʾ������Ϊ��ȫ�����������������ѽ�[49]��SHAFIEFARHOOD��[103]ͨ�����������о��˸����εķ�Ӧ�����������ʾ�������������˷�Ӧ�κͷ�Ӧ������MONAZAM��[104]�����ط�Ӧ���м�����CH4��ԭFe2O3�Ķ���ѧ�����������ԭ���̵�������ԭ������MIHAI��[78]���CH4��ԭ������Ĺ�������������Լ���������λ�����йأ�SHEN��[25]Ҳ����ʹ�ò�ͬ����ʱ���������ĺϳ���ѡ���Բ�ͬ�������ܶȷ������۵ļ�����Ի���۷�Ӧ������HUANG��[105]��ϸ������CH4��Fe2O3��Ӧ���̣������ʾCH4���ѽ��Լ�������Ǩ���Ƿ�Ӧ���̵����ʿ��Ʋ��衣CHENG��[106]������Fe2O3��������λ��CH4��Ӧ���̵�Ӱ�죬��������������λ������CH4���ѽ⣬�ٽ���Ӧ�Ľ��С�BENNET��[107]�Ƚ���Fe2O3�����Fe3O4�����CH4�ѽ���̣�CH4�ķ�Ӧ������������ı���ṹ�йء�
Ŀǰ�����㷺���ܵ�CH4/CO2�ڴ�������������Ļ�����ͼ5��ʾ����Ϊ4��[108]��CH4��������ֽ⡢CO2��������ֽ⡢���������γ��Լ��м������������Ѹ����ֵ��о����ڽ������淴Ӧ�����ڽ��������������о����٣�CH4/CO2�������������ķ�Ӧ������Ϊ�ټ���Ҳ��Ҫ��һ�����о���
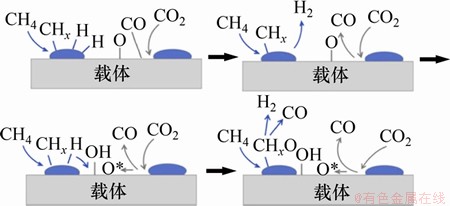
ͼ5�������������Ӧ����[108]
Fig. 5��Reaction steps for dry reforming of methane[108]
Ŀǰ���������������屻��������������ĸ��ԣ���ҪĿ��������鷴Ӧ�ԡ���ԭ��ȡ�����ѡ���ԡ�ѭ���ȶ��ԣ����Ǹ��Լ��������������Ӱ�������������ȷ���������徭���Ե�̽��Ҳ���٣�����Ҫ����������������������Լ��ʺϴ��ģ�������Ʊ�������չ�о���CH4����������(Fe2O3��Fe3O4��FeO��Fe)����ķ�Ӧ��������������Ե����ݣ�CH4/CO2�������������ķ�Ӧ���������CH4��CO2ת���ʵĹؼ�������Ҫ��������о���
3 ��ѧ��CH4/CO2ת���з�Ӧ�������
��Ӧ��Ҳ��ʵ�ֻ�ѧ�������Ĺؼ���Ŀǰ���ڻ�ѧ�����̵ķ�Ӧ���й̶��������������ƶ�����Ӧ��������CH4/CO2ת����ʾ����Ŀ�����ࡣ
�̶�����Ӧ���ṹ���������������־�ֹ����ͼ6��ʾ��SPALLINA��[33]ʹ�ù̶���֤ʵ�˻�ѧ��ȼ��������������ϵĿ����ԣ�ʵ��������CH4/CO2ת���������ֵ�ӽ������ҿ���ʵ������ƽ�⡣
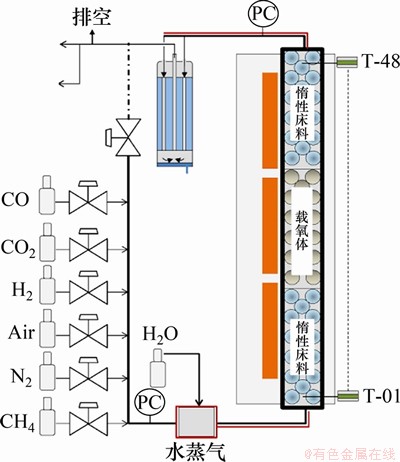
ͼ6���̶�����Ӧ��ʾ��ͼ[33]
Fig. 6��Schematic diagrams of fixed bed reactor[33]
��������Ӧ���ǻ�ѧ�������й㷺ʹ�õķ�Ӧ����Ŀǰ���ģ������������ʱ���ѳ���11 000 h[109]�����д�������ڻ�ѧ��ȼ�յ�˫����Ӧ����������Ӧ����̬���ӱ������١���������Ӧ���д�����Ҫ�й��ݴ����綯���Լ�ѭ���������õ����̽Ӵ������ڷ�Ӧ��������������Ӧ������Ҫ�����뷴Ӧ�ﵽ�Ż�ƥ�䣬�����������Ӧ����ȼ�ϻ�ԭ��Ӧ���еķ�Ӧ���Ӹ��ӣ����ȼ�Ϸ�Ӧ����ȼ��ת���ʺ�ѡ�����Ǵ��ģ����������о����ص�[110]���༶��ѧ����Ӧ���У�ȼ�Ϸ�Ӧ�������̽Ӵ���ʽ��˳�����������ڲ���ѡ���Է��棬�����Ӵ�����������CO2��H2O��˳���Ӵ�������CO��H2[4]�������ȼ��ת���ʷ��棬�������̽Ӵ�ʱ������Ҫ���������ֵ��͵���������Ӧ����Ʒ���[111-117]��ͼ7~11��ʾ��������ҵ��ѧ25 kWth CLCװ��(ͼ7)��ʹ��˫�����ݴ�ȼ�Ϸ�Ӧ��[111]�����н����ʾ������Ʒ���������Ч���ȼ��ת���ʡ����пƼ���ѧ5 kWth CLCװ��(ͼ8)��ʹ���²����ݴ��ϲ��Ķ���[112]���������ڹ��ݴ���ֻ�ԭ�����ϲ��Ķ���������������������������Ӧ�������ϴ�ѧSHEN��[113-114]������綯��ȼ�Ϸ�Ӧ��(ͼ9)���綯����Ӧ���п����Ӵ���֡�ͣ��ʱ�䳤���ڹ���ȼ�ϻ�ѧ��ת�������������ơ�ѭ����������Ӧ�������̽Ӵ��ȽϺò��ҿ��Խ�δ��ȫ��Ӧ�Ĺ����ٴη�Ӧ��������������Ļ�ԭ����ˣ����CLCװ��ʹ��ѭ��������ȼ�Ϸ�Ӧ��[115-116]�����ķʩ���ع�ҵ��ѧ1 MWth CLCװ��(ͼ10)�����ϴ�ѧXUE��[117]�����������������ѧ��������˸���������ȼ�Ϸ�Ӧ������ͼ11��ʾ����Ӧ���ϲ�Ϊ����������״̬���Ŷ�ǿ�ң����ʺ�Fe2O3ת��Fe3O4�Ŀ��ٷ�Ӧ���̣�����Ӧ���²��Ĺ��ݴ�״̬�����ٽ������ʺ�Fe3O4ת��ΪFeO��Fe�����ٷ�Ӧ���̣��ø����������ܹ��ڽ�Fe2O3��ԭ��FeO��ͬʱ��ʵ�ֽ�ȼ�����ļ�����ȫת��ΪCO2��H2O��
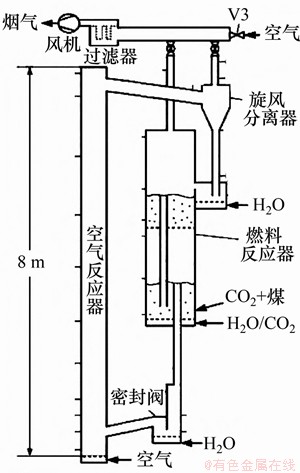
ͼ7��������ҵ��ѧ25 kWth CLCװ��(ȼ�Ϸ�Ӧ����˫�����ݴ�)[111]
Fig. 7��25 kWth CLC facility in Hamburg University and Technology (fuel reactor: two-stage bubbling fluidized bed)[111]
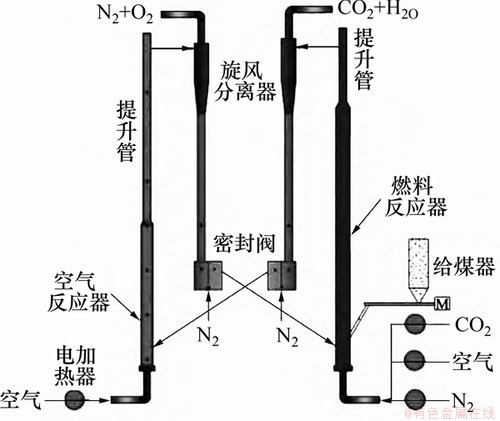
ͼ8�����пƼ���ѧ5 kWth CLCװ��(ȼ�Ϸ�Ӧ�����²����ݴ����ϲ��Ķ���)[112]
Fig. 8��5 kWth CLC facility in Huazhong University of Technology (fuel reactor: the bottom bubbling bed and the upper turbulent bed)[112]
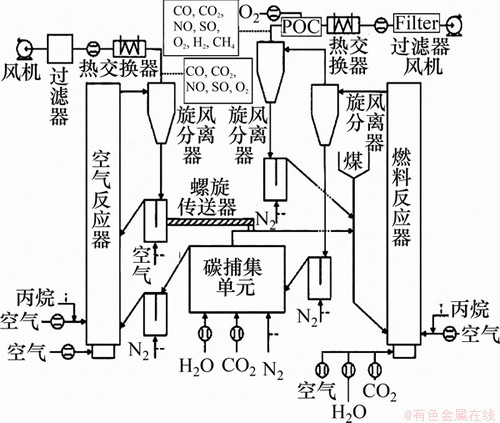
ͼ9�����ϴ�ѧ10 kWth CLCװ��(ȼ�Ϸ�Ӧ�����綯��)[114]
Fig. 9��10 kWth CLC facility in Southeast University (fuel reactor: spout fluidized bed)[114]

ͼ10����ķʩ���ع�ҵ��ѧ1 MWth CLCװ��(ȼ�Ϸ�Ӧ����ѭ��������)[115]
Fig .10��1 MWth CLC facility in Darmstadt University and Technology (fuel reactor: circulating bed)[115]
FAN��[118-119]����˻����ƶ�����ȼ�Ϸ�Ӧ������ͼ12��ʾ���ƶ���ȼ�Ϸ�Ӧ�����Կ������̽Ӵ�ʱ�䣬�Ӷ����ת���ʺͲ���ѡ���ԡ�KATHE��[28]���ƶ�����Ӧ���л������������������CH4/CO2ת��ʵ�飬1 039 ��ʱ��CO2��CH4���ʵ�����Ϊ0.3��Fe2O3��CH4���ʵ�����Ϊ0.83�����£�CH4ת����Ϊ92.3%��H2��CO���ʵ�����Ϊ1.40��
�̶�����Ӧ���У��������еǿ�ȵ�Ҫ��ͣ����ҹ̶���������ʵ�ּ�ѹ[120]���ڻ�ѧ��CH4/CO2ת�����������ɼ�ѹ�ϳ��������ڻ�ѧƷ�ĺϳɣ����ǹ̶�����Ӧ�������������������ƶ���ȼ�Ϸ�Ӧ�����Կ������̽Ӵ�ʱ�䣬�Ӷ����ת���ʺͲ���ѡ���ԣ��ƶ������������ڹ�������ѭ�����ʵͣ���������С����������Ӧ�������̽Ӵ��ã�����ѭ���������������������нϺõĹ�ҵӦ��ǰ�������Ƿ�Ӧ���������뷴Ӧ���Ż�ƥ��Ƚϸ��ӡ����������������Ļ�ѧ��ȼ�չ��̲�ͬ����������������Ļ�ѧ��CH4/CO2ת�����ն���Ҫ������ﵽ��Ȼ�ԭ��ʵ����Ȼ�ԭ���Ѷȸ������ȼ�Ϸ�Ӧ���������ʵ�ֻ�ѧ��CH4/CO2ת���Ĺؼ���
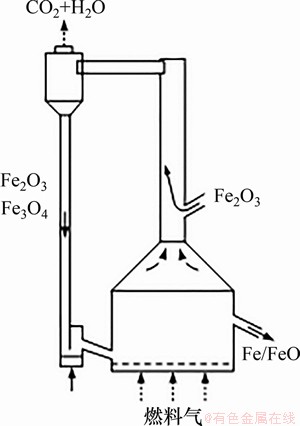
ͼ11������������ȼ�Ϸ�Ӧ��ԭ��ͼ[117]
Fig. 11��Schematic diagrams of compact fluidized bed fuel reactor[117]
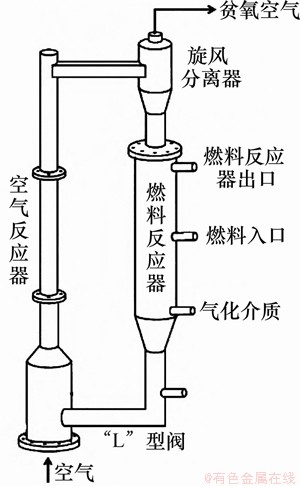
ͼ12��������������ѧ25 kWth CLCװ��[119](ȼ�Ϸ�Ӧ�����ƶ���)
Fig. 12��25 kWth CLC facility in Ohio State University[119] (fuel reactor: moving bed)
4 ����
1) ��ѧ��CH4/CO2ת�����տ��Կ˷���ͳ������������յ�ȱ�㣬���Ը���Ŀ�����ѡ���ʺϵ����̣�CH4��CO2��ͬһ��ͬ��Ӧ����ͨ�붼����ʵ��CH4��CO2��Чת������һ�ֱȽ���ǰ����CH4/CO2ת�����ա�
2) ����������۸��������Դ�㷺�������Ѻã����Ա�CH4��CH4/CO2�������ԭ�������ڽϿ��¶ȷ�Χ�ڻ�ԭCO2����һ���ʺϻ�ѧ��CH4/CO2ת�����յ������塣����������ĸ��Է�ʽ��Ҫ�ǵ������塢���������Գɷ��Լ��ռ�ֲ�����Ӧ���Ժá��۸�������ʺϴ��ģ�Ʊ����������������о��Ĺؼ���CH4��ԭ����������ķ�Ӧ�������Լ�CH4/CO2�������������������ĸ������������������������ϻ�������ָ����������Ƶ����ݣ�Ŀǰ������������Ҫ��һ���о���
3) ��ѧ��CH4/CO2ת���Ĵ��ģʾ����ĿĿǰ�����٣���ѧ�������з�Ӧ���й̶������ƶ�������������Ӧ������ѧ��CH4/CO2ת�����յ�ʵ����Ҫ����ܹ�ʵ��������������Ȼ�ԭ��ȼ�ϸ�Чת����ȼ�Ϸ�Ӧ����CH4��CO2��Ҫת�����ڴ��ģ��Ӧװ�ý�һ��֤ʵ��
�ο����ף�
[1] ��Сƽ, ����, ����, ��. ���黯ѧ�������ƺϳ������о���չ[J]. ����ˮ, 2017, 39(4): 198-202.
SU Xiaoping, WANG Li, YANG Wu, et al. Chemical looping reforming of methane for syngas production[J]. Ground Water, 2017, 39(4): 198-202.
[2] ������, ����, ������, ��. ���黯ѧ�������ƺϳ�������������о���չ[J]. ��Ȼ������(C1��ѧ�뻯��), 2019, 44(3): 106-116.
JIN Nannan, ZHANG Li, ZHU Yanyan, et al. Research progress in oxygen carriers for syngas production via chemical looping reforming of methane[J]. Natural Gas Chemical Industry, 2019, 44(3): 106-116.
[3] LUO Ming, YI Yang, WANG Shuzhong, et al. Review of hydrogen production using chemical-looping technology[J]. Renewable and Sustainable Energy Reviews, 2018, 81: 3186-3214.
[4] ����, ������. ��ѧ������ֱ�����⼼����չ[J]. ����ѧ��, 2015, 66(8): 2854-2862.
ZENG Liang��GONG Jinlong. Advances in chemical looping reforming for direct hydrogen production[J]. CIESC Journal, 2015, 66(8): 2854-2862.
[5] USMAN M, WAN DAUD W M A, ABBAS H F. Dry reforming of methane: influence of process parameters: a review[J]. Renewable and Sustainable Energy Reviews, 2015, 45: 710-744.
[6] YABE T, SEKINE Y. Methane conversion using carbon dioxide as an oxidizing agent: a review[J]. Fuel Processing Technology, 2018, 181: 187-198.
[7] RICHTER H J, KNOCHE K F. Reversibility of combustion processes[J]. ACS Symposium Series, 1983, 235: 71-85.
[8] ʷ���, ��˼��, Ǯ��. ��ѧ��������ú̿����Ч�����е��о���չ[J]. ����ѧ��, 2018, 69(12): 4931-4946.
SHI Xiaofei, YANG Siyu, QIAN Yu. Chemical looping technology for clean and highly efficient coal processes[J]. CIESC Journal, 2018, 69(12): 4931-4946.
[9] BHAVSAR S, NAJERA M, SOLUNKE R, et al. Chemical looping: to combustion and beyond[J]. Catalysis Today, 2014, 228: 96-105.
[10] ��һ��, �´�׳, �ž���, ��. ��ѧ��С����ת���о���չ[J]. �й���ѧ: ��ѧ, 2020, 50(3): 337-365.
DUAN Yifei, CHEN Cunzhuang, ZHANG Junshe, et al. Progress in chemical looping-based transformations of small molecules[J]. Sci Sin Chim, 2020, 50(3): 337-365.
[11] ZHU Xing, IMTIAZ Q, DONAT F, et al. Chemical looping beyond combustion: a perspective[J]. Energy & Environmental Science, 2020, 13(3): 772-804.
[12] NAJERA M, SOLUNKE R, GARDNER T, et al. Carbon capture and utilization via chemical looping dry reforming[J]. Chemical Engineering Research and Design, 2011, 89(9): 1533-1543.
[13] MORE A, BHAVSAR S, VESER G. Iron-nickel alloys for carbon dioxide activation by chemical looping dry reforming of methane[J]. Energy Technology, 2016, 4(10): 1147-1157.
[14] LOFBERG A, GUERRERO-CABALLERO J, KANE T, et al. Ni/CeO2 based catalysts as oxygen vectors for the chemical looping dry reforming of methane for syngas production[J]. Applied Catalysis B: Environmental, 2017, 212: 159-174.
[15] GUERRERO-CABALLERO J, KANE T, HAIDAR N, et al. Ni, Co, Fe supported on Ceria and Zr doped Ceria as oxygen carriers for chemical looping dry reforming of methane[J]. Catalysis Today, 2019, 333: 251-258.
[16] LOFBERG A, KANE T, GUERRERO-CABALLERO J, et al. Chemical looping dry reforming of methane: toward shale-gas and biogas valorization[J]. Chemical Engineering and Processing: Process Intensification, 2017, 122: 523-529.
[17] GE Yuanzheng, HE Tao, WANG Zhiqi, et al. Chemical looping oxidation of CH4 with 99.5% CO selectivity over V2O3�\based redox materials using CO2 for regeneration[J]. AIChE Journal, 2020, 66(1): e16772.
[18] LIM H S, KANG D, LEE J W. Phase transition of Fe2O3-NiO to NiFe2O4 in perovskite catalytic particles for enhanced methane chemical looping reforming-decomposition with CO2 conversion[J]. Applied Catalysis B: Environmental, 2017, 202: 175-183.
[19] SUN Zhao, CAI Tianyi, RUSSELL C K, et al. Highly efficient methane decomposition to H2 and CO2 reduction to CO via redox looping of Ca2FexAl2-xO5 supported NiyFe3-yO4 nanoparticles[J]. Applied Catalysis B: Environmental, 2020, 271: 118938.
[20] MORE A, HANSEN C J, VESER G. Production of inherently separated syngas streams via chemical looping methane cracking[J]. Catalysis Today, 2017, 298: 21-32.
[21] KIM Y, LIM H S, LEE M, et al. Ni-Fe-Al mixed oxide for combined dry reforming and decomposition of methane with CO2 utilization[EB/OL]. [2020-03-20]. https: //doi.org/10.1016/j.cattod.2020.02.030.
[22] KELLER M, MATSUZAKI Y, OTOMO J. CO2 activation by methane in a dual-bed configuration via methane cracking and iron oxide lattice oxygen transport: concept and materials development[J]. Chemical Engineering Journal, 2018, 349: 249-259.
[23] WARREN K J, CARRILLO R J, GREEK B, et al. Solar reactor demonstration of efficient and selective syngas production via chemical-looping dry reforming of methane over ceria[J]. Energy Technology, 2020, 8(6): 2000053.
[24] BHAVSAR S, NAJERA M, VESER G. Chemical looping dry reforming as novel, intensified process for CO2 activation[J]. Chemical Engineering & Technology, 2012, 35(7): 1281-1290.
[25] SHEN Qianqian, HUANG Fei, TIAN Ming, et al. Effect of regeneration period on the selectivity of synthesis gas of Ba-hexaaluminates in chemical looping partial oxidation of methane[J]. ACS Catalysis, 2019, 9(1): 722-731.
[26] KANG D, LEE M, LIM H S, et al. Chemical looping partial oxidation of methane with CO2 utilization on the ceria-enhanced mesoporous Fe2O3 oxygen carrier[J]. Fuel, 2018, 215: 787-798.
[27] KANG D, LIM H S, LEE M, et al. Syngas production on a Ni-enhanced Fe2O3/Al2O3 oxygen carrier via chemical looping partial oxidation with dry reforming of methane[J]. Applied Energy, 2018, 211: 174-186.
[28] KATHE M, EMPFIELD A, SANDVIK P, et al. Utilization of CO2 as a partial substitute for methane feedstock in chemical looping methane-steam redox processes for syngas production[J]. Energy & Environmental Science, 2017, 10(6): 1345-1349.
[29] GALVITA V V, POELMAN H, DETAVERNIER C, et al. Catalyst-assisted chemical looping for CO2 conversion to CO[J]. Applied Catalysis B: Environmental, 2015, 164: 184-191.
[30] HU Jiawei, GALVITA V V, POELMAN H, et al. Catalyst-assisted chemical looping auto-thermal dry reforming: spatial structuring effects on process efficiency[J]. Applied Catalysis B: Environmental, 2018, 231: 123-136.
[31] HU Jiawei, BUELENS L, THEOFANIDIS S A, et al. CO2 conversion to CO by auto-thermal catalyst-assisted chemical looping[J]. Journal of CO2 Utilization, 2016, 16: 8-16.
[32] HU Jiawei, GALVITA V V, POELMAN H, et al. Pressure-induced deactivation of core-shell nanomaterials for catalyst-assisted chemical looping[J]. Applied Catalysis B: Environmental, 2019, 247: 86-99.
[33] SPALLINA V, MARINELLO B, GALLUCCI F, et al. Chemical looping reforming in packed-bed reactors: modelling, experimental validation and large-scale reactor design[J]. Fuel Processing Technology, 2017, 156: 156-170.
[34] ZHU Min, SONG Yeheng, CHEN Shiyi, et al. Chemical looping dry reforming of methane with hydrogen generation on Fe2O3/Al2O3 oxygen carrier[J]. Chemical Engineering Journal, 2019, 368: 812-823.
[35] HU Jiawei, GALVITA V, POELMAN H, et al. Advanced chemical looping materials for CO2 utilization: a review[J]. Materials, 2018, 11(7): 1187.
[36] ADANEZ J, ABAD A, GARCIA-LABIANO F, et al. Progress in chemical-looping combustion and reforming technologies[J]. Progress in Energy and Combustion Science, 2012, 38(2): 215-282.
[37] LI Danyang, XU Ruidong, GU Zhenhua, et al. Chemical-looping conversion of methane: a review[J]. Energy Technology, 2020, 8(8): 1900925.
[38] TANG Mingchen, XU Long, FAN Maohong. Progress in oxygen carrier development of methane-based chemical-looping reforming: a review[J]. Applied Energy, 2015, 151: 143-156.
[39] ZENG Liang, CHENG Zhuo, Fan J A, et al. Metal oxide redox chemistry for chemical looping processes[J]. Nature Reviews Chemistry, 2018, 2: 349-364.
[40] KATHE M, FRYER C, SANDVIK P, et al. Modularization strategy for syngas generation in chemical looping methane reforming systems with CO2 as feedstock[J]. AIChE Journal, 2017, 63(8): 3343-3360.
[41] ����, ��ʱ��, ����, ��. ��ѧ��������������������ѧ������ʵ��[J]. ����ѧ��, 2019, 70(6): 2244-2251.
ZHU Min, CHEN Shiyi, LI Meng, et al. Thermodynamic and experimental analysis of chemical looping dry reforming with hydrogen production system[J]. CIESC Journal, 2019, 70(6): 2244-2251.
[42] DHARANIPRAGADA N V R A, MELEDINA M, GALVITA V V, et al. Deactivation study of Fe2O3-CeO2 during redox cycles for CO production from CO2[J]. Industrial & Engineering Chemistry Research, 2016, 55(20): 5911-5922.
[43] GALINSKY N L, SHAFIEFARHOOD A, CHEN Y, et al. Effect of support on redox stability of iron oxide for chemical looping conversion of methane[J]. Applied Catalysis B: Environmental, 2015, 164: 371-379.
[44] MA Shiwei, CHEN Shiyi, SOOMRO A, et al. Effects of CeO2, ZrO2, and Al2O3 supports on iron oxygen carrier for chemical looping hydrogen generation[J]. Energy & Fuels, 2017, 31(8): 8001-8013.
[45] MA Shiwei, CHEN Shiyi, SOOMRO A, et al. Effects of supports on hydrogen production and carbon deposition of Fe-based oxygen carriers in chemical looping hydrogen generation[J]. International Journal of Hydrogen Energy, 2017, 42(16): 11006-11016.
[46] ZHU Min, CHEN Shiyi, SOOMRO A, et al. Effects of supports on reduction activity and carbon deposition of iron oxide for methane chemical looping hydrogen generation[J]. Applied Energy, 2018, 225: 912-921.
[47] RIHKO-STRUCKMANN L K, DATTA P, WENZEL M, et al. Hydrogen and carbon monoxide production by chemical looping over iron-aluminium oxides[J]. Energy Technology, 2016, 4(2): 304-313.
[48] KIDAMBI P R, CLEETON J P E, SCOTT S A, et al. Interaction of iron oxide with alumina in a composite oxygen carrier during the production of hydrogen by chemical looping[J]. Energy & Fuels, 2012, 26(1): 603-617.
[49] ZHU Min, CHEN Shiyi, MA Shiwei, et al. Carbon formation on iron-based oxygen carriers during CH4 reduction period in chemical looping hydrogen generation process[J]. Chemical Engineering Journal, 2017, 325: 322-331.
[50] JOHANSSON M, MATTISSON T, LYNGFELT A. Investigation of Fe2O3 with MgAl2O4 for chemical-looping combustion[J]. Industrial & Engineering Chemistry Research, 2004, 43(22): 6978-6987.
[51] DHARANIPRAGADA N V R A, BUELENS L C, POELMAN H, et al. Mg-Fe-Al-O for advanced CO2 to CO conversion: carbon monoxide yield vs. oxygen storage capacity[J]. Journal of Materials Chemistry A, 2015, 3(31): 16251-16262.
[52] DE VOS Y, JACOBS M, VAN DRIESSCHE I, et al. Processing and characterization of Fe-based oxygen carriers for chemical looping for hydrogen production[J]. International Journal of Greenhouse Gas Control, 2018, 70: 12-21.
[53] DE VOS Y, JACOBS M, VAN DER VOORT P, et al. Sustainable iron-based oxygen carriers for chemical looping for hydrogen generation[J]. International Journal of Hydrogen Energy, 2019, 44(3): 1374-1391.
[54] CHEN Shiyi, SHI Qiliang, XUE Zhipng, et al. Experimental investigation of chemical-looping hydrogen generation using Al2O3 or TiO2-supported iron oxides in a batch fluidized bed[J]. International Journal of Hydrogen Energy, 2011, 36(15): 8915-8926.
[55] CHUNG C, QIN L, SHAH V, et al. Chemically and physically robust, commercially-viable iron-based composite oxygen carriers sustainable over 3000 redox cycles at high temperatures for chemical looping applications[J]. Energy & Environmental Science, 2017, 10(11): 2318-2323.
[56] LIU W, DENNIS J S, SCOTT S A. The effect of addition of ZrO2 to Fe2O3 for hydrogen production by chemical looping[J]. Industrial & Engineering Chemistry Research, 2012, 51(51): 16597-16609.
[57] YUZBASI N S, KIERZKOWSKA A M, IMTIAZ Q, et al. ZrO2-supported Fe2O3 for chemical-looping-based hydrogen production: effect of ph on its structure and performance as probed by X-ray absorption spectroscopy and electrical conductivity measurements[J]. The Journal of Physical Chemistry C, 2016, 120(34): 18977-18985.
[58] BUELENS L C, DHARANIPRAGADA A N V R, POELMAN H, et al. Exploring the stability of Fe2O3-MgAl2O4 oxygen storage materials for CO production from CO2[J]. Journal of CO2 Utilization, 2019, 29: 36-45.
[59] ZHU Xing, SUN Lingyue, ZHENG Yane, et al. CeO2 modified Fe2O3 for the chemical hydrogen storage and production via cyclic water splitting[J]. International Journal of Hydrogen Energy, 2014, 39(25): 13381-13388.
[60] ZHU Xing, ZHANG Mingyue, LI Kongzhai, et al. Chemical-looping water splitting over ceria-modified iron oxide: performance evolution and element migration during redox cycling[J]. Chemical Engineering Science, 2018, 179: 92-103.
[61] ZHU Xing, WEI Yonggang, WANG Hua, et al. Ce-Fe oxygen carriers for chemical-looping steam methane reforming[J]. International Journal of Hydrogen Energy, 2013, 38(11): 4492-4501.
[62] GALVITA V V, POELMAN H, BLIZNUK V, et al. CeO2-modified Fe2O3 for CO2 utilization via chemical looping[J]. Industrial & Engineering Chemistry Research, 2013, 52(25): 8416-8426.
[63] CHO W C, LEE J K, NAM G D, et al. Degradation analysis of mixed ionic-electronic conductor-supported iron-oxide oxygen carriers for chemical-looping conversion of methane[J]. Applied Energy, 2019, 239: 644-657.
[64] NEAL L M, SHAFIEFARHOOD A, LI F. Dynamic methane partial oxidation using a Fe2O3@La0.8Sr0.2FeO3-�� core-shell redox catalyst in the absence of gaseous oxygen[J]. ACS Catalysis, 2014, 4(10): 3560-3569.
[65] LIU Lu, ZACHARIAH M R. Enhanced performance of alkali metal doped Fe2O3 and Fe2O3/Al2O3 composites as oxygen carrier material in chemical looping combustion[J]. Energy & Fuels, 2013, 27(8): 4977-4983.
[66] KANG Yu, TIAN Ming, WANG Yuehan, et al. Silica modified alumina as supports of Fe2O3 with high performance in chemical looping combustion of methane[J]. ACS Sustainable Chemistry & Engineering, 2018, 6(10): 12884-12892.
[67] HAFIZI A, RAHIMPOUR M R, HASSANAJILI S. Calcium promoted Fe/Al2O3 oxygen carrier for hydrogen production via cyclic chemical looping steam methane reforming process[J]. International Journal of Hydrogen Energy, 2015, 40(46): 16159-16168.
[68] HAFIZI A, RAHIMPOUR M R, HASSANAJILI S. Hydrogen production via chemical looping steam methane reforming process: effect of cerium and calcium promoters on the performance of Fe2O3/Al2O3 oxygen carrier[J]. Applied Energy, 2016, 165: 685-694.
[69] IMTIAZ Q, S. YUZBASI N, M. ABDALA P, et al. Development of MgAl2O4 -stabilized, Cu-doped, Fe2O3-based oxygen carriers for thermochemical water-splitting[J]. Journal of Materials Chemistry A, 2016, 4(1): 113-123.
[70] YUZBASI N S, ABDALA P M, IMTIAZ Q, et al. The effect of copper on the redox behaviour of iron oxide for chemical-looping hydrogen production probed by in situ X-ray absorption spectroscopy[J]. Physical Chemistry Chemical Physics, 2018, 20(18): 12736-12745.
[71] QIN Lang, GUO Mengqing, LIU Yan, et al. Enhanced methane conversion in chemical looping partial oxidation systems using a copper doping modification[J]. Applied Catalysis B: Environmental, 2018, 235: 143-149.
[72] KANG Yu, TIAN Ming, HUANG Chuande, et al. Improving syngas selectivity of Fe2O3/Al2O3 with yttrium modification in chemical looping methane conversion[J]. ACS Catalysis, 2019, 9(9): 8373-8382.
[73] QIN Lang, CHENG Zhuo, GUO Mengqing, et al. Impact of 1% lanthanum dopant on carbonaceous fuel redox reactions with an iron-based oxygen carrier in chemical looping processes[J]. ACS Energy Letters, 2017, 2(1): 70-74.
[74] QIN Lang, GUO Mengqing, CHENG Zhuo, et al. Improved cyclic redox reactivity of lanthanum modified iron-based oxygen carriers in carbon monoxide chemical looping combustion[J]. Journal of Materials Chemistry A, 2017, 5(38): 20153-20160.
[75] TANG Mingchen, LIU Kuo, RODDICK D M, et al. Enhanced lattice oxygen reactivity over Fe2O3/Al2O3 redox catalyst for chemical-looping dry(CO2) reforming of CH4: synergistic La-Ce effect[J]. Journal of Catalysis, 2018, 368: 38-52.
[76] BHAVSAR S, VESER G. BIMETALLIC Fe-Ni oxygen carriers for chemical looping combustion[J]. Industrial & Engineering Chemistry Research, 2013, 52(44): 15342-15352.
[77] BHAVSAR S, VESER G. Chemical looping beyond combustion: production of synthesis gas via chemical looping partial oxidation of methane[J]. RSC Advances, 2014, 4(88): 47254-47267.
[78] MIHAI O, CHEN D, HOLMEN A. Catalytic consequence of oxygen of lanthanum ferrite perovskite in chemical looping reforming of methane[J]. Industrial & Engineering Chemistry Research, 2011, 50(5): 2613-2621.
[79] DAI Xiaoping, LI Ranjia, YU Changchun, et al. Unsteady-state direct partial oxidation of methane to synthesis gas in a fixed-bed reactor using AFeO3 (A=La, Nd, Eu) perovskite-type oxides as oxygen storage[J]. The Journal of Physical Chemistry B, 2006, 110(45): 22525-22531.
[80] MIHAI O, CHEN D, HOLMEN A. Chemical looping methane partial oxidation: the effect of the crystal size and O content of LaFeO3[J]. Journal of Catalysis, 2012, 293: 175-185.
[81] SHEN Yang, ZHAO Kun, HE Fang, et al. The structure-reactivity relationships of using three-dimensionally ordered macroporous LaFe1-xNixO3 perovskites for chemical-looping steam methane reforming[J]. Journal of the Energy Institute, 2019, 92(2): 239-246.
[82] ZHAO Kun, HE Fang, HUANG Zhen, et al. Perovskite-type oxides LaFe1-xCoxO3 for chemical looping steam methane reforming to syngas and hydrogen co-production[J]. Applied Energy, 2016, 168: 193-203.
[83] ZHAO Kun, ZHENG Anqing, LI Haibin, et al. Exploration of the mechanism of chemical looping steam methane reforming using double perovskite-type oxides La1.6Sr0.4FeCoO6[J]. Applied Catalysis B: Environmental, 2017, 219: 672-682.
[84] MAITI D, DAZA Y A. YUNG M M.et al. Oxygen vacancy formation characteristics in the bulk and across different surface terminations of La(1-x)SrxFe(1-y)CoyO(3-��) perovskite oxides for CO2 conversion[J]. Journal of Materials Chemistry A, 2016, 4(14): 5137-5148.
[85] SUN Zhao, CHEN Shiyi, HU Jun, et al. Ca2Fe2O5: a promising oxygen carrier for CO/CH4 conversion and almost-pure H2 production with inherent CO2 capture over a two-step chemical looping hydrogen generation process[J]. Applied Energy, 2018, 211: 431-442.
[86] CHAN M S C, LIU W, ISMAIL M, et al. Improving hydrogen yields, and hydrogen: steam ratio in the chemical looping production of hydrogen using Ca2Fe2O5[J]. Chemical Engineering Journal, 2016, 296: 406-411.
[87] HOSSEINI D, DONAT F, ABDALA P M, et al. Reversible exsolution of dopant improves the performance of Ca2Fe2O5 for chemical looping hydrogen production[J]. ACS Applied Materials & Interfaces, 2019, 11(20): 18276-18284.
[88] ISMAIL M, LIU W, CHAN M S C, et al. Synthesis, application, and carbonation behavior of Ca2Fe2O5 for chemical looping H2 production[J]. Energy & Fuels, 2016, 30(8): 6220-6232.
[89] SHAH V, MOHAPATRA P, FAN L S. Thermodynamic and process analyses of syngas production using chemical looping reforming assisted by flexible dicalcium ferrite-based oxygen carrier regeneration[J]. Energy & Fuels, 2020, 34(5): 6490-6500.
[90] SHAH V, JOSHI R, FAN L-S. Thermodynamic Investigation of process enhancement in chemical looping reforming of methane through modified Ca-Fe oxygen carrier utilization[J]. Industrial & Engineering Chemistry Research, 2020, 59(35): 15531-15541.
[91] MILLER D D, RILEY J, SIRIWARDANE R. Interaction of methane with calcium ferrite in the chemical looping partial oxidation application: experimental and DFT study[J]. Energy & Fuels, 2020, 34(2): 2193-2204.
[92] HUANG Zhen, JIANG Huanqi, HE Fang, et al. Evaluation of multi-cycle performance of chemical looping dry reforming using CO2 as an oxidant with Fe-Ni bimetallic oxides[J]. Journal of Energy Chemistry, 2016, 25(1): 62-70.
[93] HUANG Zhen, HE Fang, CHEN Dezhen, et al. Investigation on reactivity of iron nickel oxides in chemical looping dry reforming[J]. Energy, 2016, 116: 53-63.
[94] ZENG Dewang, QIU Yu, PENG Song, et al. Enhanced hydrogen production performance through controllable redox exsolution within CoFeAlOx spinel oxygen carrier materials[J]. Journal of Materials Chemistry A, 2018, 6(24): 11306-11316.
[95] QIU Yu, ZHANG Shuai, CUI Dongxu, et al. Enhanced hydrogen production performance at intermediate temperatures through the synergistic effects of binary oxygen carriers[J]. Applied Energy, 2019, 252: 113454.
[96] ZHU Yanyan, LIU Weiwei, SUN Xueyan, et al. La-hexaaluminate for synthesis gas generation by chemical looping partial oxidation of methane using CO2 as sole oxidant[J]. AIChE Journal, 2018, 64(2): 550-563.
[97] ZHU Yanyan, SUN Xueyan, LIU Weiwei, et al. Microstructure and reactivity evolution of LaFeAl oxygen carrier for syngas production via chemical looping CH4CO2 reforming[J]. International Journal of Hydrogen Energy, 2017, 42(52): 30509-30524.
[98] YU Zhongliang, YANG Yanyan, YANG Song, et al. Iron-based oxygen carriers in chemical looping conversions: a review[J]. Carbon Resources Conversion, 2019, 2(1): 23-34.
[99] SUN Zhenkun, LU D Y, SYMONDS R T, et al. Chemical looping reforming of CH4 in the presence of CO2 using ilmenite ore and NiO-modified ilmenite ore oxygen carriers[J]. Chemical Engineering Journal, 2020, 401: 123481.
[100] HU Jiawei, GALVITA V V, POELMAN H, et al. A core-shell structured Fe2O3/ZrO2@ZrO2 nanomaterial with enhanced redox activity and stability for CO2 conversion[J]. Journal of CO2 Utilization, 2017, 17: 20-31.
[101] MORE A, VESER G. Physical mixtures as simple and efficient alternative to alloy carriers in chemical looping processes[J]. AIChE Journal, 2017, 63(1): 51-59.
[102] NEAL L, SHAFIEFARHOOD A, LI F. Effect of core and shell compositions on MeOx@LaySr1-yFeO3 core-shell redox catalysts for chemical looping reforming of methane[J]. Applied Energy, 2015, 157: 391-398.
[103] SHAFIEFARHOOD A, HAMILL J C, NEAL L M, et al. Methane partial oxidation using FeOx@La0.8Sr0.2FeO3-�� core-shell catalyst-transient pulse studies[J]. Physical Chemistry Chemical Physics, 2015, 17(46): 31297-31307.
[104] MONAZAM E R, BREAULT R W, SIRIWARDANE R,et al. Kinetics of the reduction of hematite (Fe2O3) by methane (CH4) during chemical looping combustion: a global mechanism[J]. Chemical Engineering Journal, 2013, 232: 478-487.
[105] HUANG Liang, TANG Mingchen, FAN Maohong, et al. Density functional theory study on the reaction between hematite and methane during chemical looping process[J]. Applied Energy, 2015, 159: 132-144.
[106] CHENG Zhuo, QIN Lang, GUO Mengqing, et al. Oxygen vacancy promoted methane partial oxidation over iron oxide oxygen carriers in the chemical looping process[J]. Physical Chemistry Chemical Physics, 2016, 18(47): 32418-32428.
[107] BENNETT J W, HUANG X, FANG Y, et al. Methane dissociation on ��-Fe2O3(0001) and Fe3O4(111) surfaces: first-principles insights into chemical looping combustion[J]. The Journal of Physical Chemistry C, 2019, 123(11): 6450-6463.
[108] ARAMOUNI N A K, TOUMA J G, TARBOUSH B A, et al. Catalyst design for dry reforming of methane: analysis review[J]. Renewable and Sustainable Energy Reviews, 2018, 82: 2570-2585.
[109] LYNGFELT A, BRINK A, LANGORGEN O, et al. 11 000h of chemical-looping combustion operation: where are we and where do we want to go[J]. International Journal of Greenhouse Gas Control, 2019, 88: 38-56.
[110] SONG Tao, SHEN Laihong. Review of reactor for chemical looping combustion of solid fuels[J]. International Journal of Greenhouse Gas Control, 2018, 76: 92-110.
[111] THON A, KRAMP M, HARTGE E U, et al. Operational experience with a system of coupled fluidized beds for chemical looping combustion of solid fuels using ilmenite as oxygen carrier[J]. Applied Energy, 2014, 118: 309-317.
[112] MA Jinchen, ZHAO Haibo, TIAN Xin, et al. Chemical looping combustion of coal in a 5 kWth interconnected fluidized bed reactor using hematite as oxygen carrier[J]. Applied Energy, 2015, 157: 304-313.
[113] SHEN Laihong, WU Jiahua, XIAO Jun. Experiments on chemical looping combustion of coal with a NiO based oxygen carrier[J]. Combustion and Flame, 2009, 156(3): 721-728.
[114] SHEN Laihong, WU Jiahua, GAO Zhengping, et al. Reactivity deterioration of NiO/Al2O3 oxygen carrier for chemical looping combustion of coal in a 10 kWth reactor[J]. Combustion and Flame, 2009, 156(7): 1377-1385.
[115] STROHLE J, ORTH M, EPPLE B. Design and operation of a 1 MWth chemical looping plant[J]. Applied Energy, 2014, 113: 1490-1495.
[116] MARKSTROM P, LYNGFELT A. Designing and operating a cold-flow model of a 100 kW chemical-looping combustor[J]. Powder Technology, 2012, 222: 182-192.
[117] XUE Zhipeng, CHEN Shiyi, WANG Dong, et al. Design and fluid dynamic analysis of a three-fluidized-bed reactor system for chemical-looping hydrogen generation[J]. Industrial & Engineering Chemistry Research, 2012, 51(11): 4267-4278.
[118] FAN Liangshi, ZENG Liang, LUO Siwei. Chemical-looping technology platform[J]. AIChE Journal, 2015, 61(1): 2-22.
[119] SPALLINA V, GALLUCCI F, VAN SINT ANNALAND M . Chemical looping processes using packed bed reactors[J]. Handbook of Chemical Looping Technology, 2018: 61-92.
[120] BAYHAM S C, KIM H R, WANG D, et al. Iron-based coal direct chemical looping combustion process: 200-h continuous operation of a 25-kWth subpilot unit[J]. Energy & Fuels, 2013, 27(3): 1347-1356.
(�༭ ����ƽ)
�ո����ڣ� 2020 -09 -27; �����ڣ� 2020 -11 -11
������Ŀ(Foundation item)������ʡ������������ƻ���Ŀ(BM2019001) (Project(BM2019001) supported by the Innovation Ability Construction Plan of Jiangsu Province)
ͨ�����ߣ���������ʿ�о��������»�ѧ�������о���E-mail��hujun_seu@outlook.com
���ø�ʽ�� ������, ����, ��ʱ�ڵ�. ���������廯ѧ��CH4/CO2ת���о���չ[J]. ���ϴ�ѧѧ��(��Ȼ��ѧ��), 2021, 52(1): 70-85.
Citation: WANG Genbao, HU Jun, CHEN Shiyi, et al. Advances in Fe-based chemical looping technology for CH4/CO2 conversion[J]. Journal of Central South University(Science and Technology), 2021, 52(1): 70-85.

