
Establishment of water quality index (Na+, Ca2+) for purified water reused to zinc electrolysis process
CHAI Li-yuan(柴立元), XIAO Hai-juan(肖海娟), WANG Yun-yan(王云燕),
PEI Fei(裴 斐), SHU Yu-de(舒余德), ZHANG Jin-long(张金龙)
School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China
Received 12 December 2007; accepted 13 June 2008
Abstract: The effects of Na+ and Ca2+ in the purified water on the conductivity of zinc electrolyte and the current efficiency of zinc electrolysis were studied by the alternating current bridge method and the simulated electrolysis experiments, and the water quality index of reused water was established. The results show that the conductivity of the solution and the current efficiency decrease as these two kinds of positive ions are added in the electrolyte. The effect of Ca2+ is much more remarkable than that of Na+. ρ(Na+)≤ 8 g/L and ρ(Ca2+)≤20 mg/L are the quality indexes in the zinc electrolysis process and the concentrations of Na+ and Ca2+ in the purified water reused to the process should be less than the limited values, i.e. the water quality index of the purified water should be controlled by its reused amount.
Key words: water quality index; purified water; zinc electrolysis process; alternating current bridge method
1 Introduction
The nonferrous production is one of the high water consumption industries in China. Recently, although the water consumption per ton products has declined, the water recycling ratio is still lower than 85%, which lags behind 15-20 years compared with developed countries[1-2]. Additionally, the discharge of heavy metal-containing wastewater leads to lavishness of resource and the toxic heavy metals also seriously affect the safety of drinking water[3-4]. Therefore, it is very important to treat the wastewater and improve the recycling ratio of the purified water. At present, there are many treatment methods for the heavy metal- containing wastewater[5-7], however, whether the treated water can be reused to the smelting process is still a highly concerned problem today. For example, in the lead-zinc smelting system, zinc electrolyte is constituted of ZnSO4-H2SO4 solution with certain concentrations, but there are many other metallic and non-metallic impurity ions, such as Cu, Pb, Fe, Ni, Na, Mg, K, Ca, Cl, and F, which will inevitably enter into the electrolyte during the production process. The existence of these impurity ions not only affects the physical and chemical properties of zinc electrolyte[8-10], such as density, viscosity, conductivity, surface tension, specific heat and the diffusion coefficient of metal ions, but also influences the purity and surface conditions of the electrolytic sediment and the power consumption [11-13]. Consequently, it is of great significance to study the effect of impurity ions on zinc electrolysis and establish the water quality index of the purified water.
2 Establishment of quality index for purified water reused to zinc electrolysis process
Table 1 lists the permissible concentrations of main metallic impurity ions[11] in zinc electrolyte as well as that in the purified water treated by biologics.
Table 1 Concentrations of main cations in zinc electrolyte and that in purified water treated by biologics

From Table 1 it can be seen that the concentrations of Cu2+, Pb2+, Zn2+, Cd2+ and As3+ in the purified water are lower than the permissible values, respectively, but the permissible values of Na+ and Ca2+ in zinc electrolyte have not been identified for lacking of detailed investigations. Additionally, according to Ref.[12], the potential of such cations as Na+, Mg2+, K+ and Ca2+ are more negative than that of Zn2+ and they will not deposit on the cathode, but can absorb on the cathodic active point selectively as surfactants, resulting in the crystallization overpotential to some extent and affecting the surface properties. The hydrogen evolution and impurity precipitation will also be accelerated, in the end, the current efficiency will decrease. Moreover, when the quantity of these impurities is too high, the viscosity and resistance of the electrolyte will increase, the diffusion of Zn2+ will be obstructed, and then the power consumption will increase. Too high concentrations of Ca2+ and Mg2+ will easily from crystal and precipitate, which makes the pipelines block and operation trouble[8-9]. Therefore, Na+ and Ca2+ were chosen in this study as the main research objectives and the establishment of the indexes of Na+ and Ca2+ in purified water were carried out.
3 Experimental
The effects of Na+ and Ca2+ in the purified water on the conductivity of zinc electrolyte were studied by the alternating current bridge method[14] and the current efficiency was determined by the simulated electrolysis experiments to establish the water quality indexes of Na+ and Ca2+ in purified water finally.
3.1 Experiments of conductivity
The zinc electrolyte was sampled from Zhuzhou Smelter Group, which consisted of 4.071 g/L Na+ and 305 mg/L Ca2+. The chemicals were Na2SO4(AR), CaSO4?H2O(AR), ZnSO4?7H2O(AR) and the concentrated H2SO4(98%, AR). The temperature of water bath was controlled at 40 ℃. The schematic diagram of experimental equipments for the alternating current bridge method is shown in Fig.1.

Fig.1 Schematic diagram of experimental equipments for alternating current bridge method: 1―ZX3N4 AC resistance box; 2―ZX32 AC resistance box; 3―SBQ06-oscilloscope; 4―FMBa teaching denary capacitance boxes; 5―Audio signal generator; 6―Selfmade conductance pool
3.2 Simulated electrolysis experiments
The concentrations of Na+ and Ca2+ in zinc electrolyte used to the electrolysis experiments were 4.022 g/L and 323 mg/L, respectively. The chemicals were Na2SO4(AR) and CaSO4?H2O(AR). The schematic diagram of experimental equipments in the simulated electrolysis tests is shown in Fig.2.

Fig.2 Schematic diagram of experimental equipments for simulated electrolysis experiment: 1―Power supply; 2―DC ammeter; 3―Electrolytic cell; 4―Selfmade high groove; 5―Selfmade low groove; 6―Lead-silver anode plate (4 cm×4 cm); 7―Aluminum cathode plate (5 cm×4 cm)
The temperature of water bath was maintained within the range of 38-40 ℃; the current density was 500 A/m2, lasted for 8 h; the volume of bone glue was 300 mg/L; the electrolyte flow was controlled at 60 drop/min, and the distance between the anode and cathode was 58-62 mm.
4 Results and discussion
4.1 Effect of Na+ in purified water on conductivity of zinc electrolyte
The standard conductivity of KCl solution can be expressed as [15]
κKCl=0.065 340+1.739 72×10-3t+4.496 41×10-6t2 (1)
where κKCl is the standard conductivity of KCl solution in S/cm; t is the temperature in ℃.
The effects of Na+ on the conductivity of zinc electrolyte are listed in Table 2. It can be concluded from Table 2 that when the concentrations of Na+ and Zn2+ are fixed, the conductivity of the solution increases with the addition of H2SO4. But the conductivity declines when the concentration of Zn2+ increases under the same concentrations of Na+ and H2SO4. While the concentration of Na+ increases and the conductivity decreases. The Na+ ions in purified water influence the conductivity of zinc electrolyte.
Table 2 Conductivities of zinc electrolyte at different concentrations of Na+ (mS/cm)

The effect of Na+ in purified water on the conductivity of zinc electrolyte is shown in Fig.3, which illustrates that when the concentration of Na+ in purified water increases, the conductivity of zinc electrolyte decreases gradually. The relationship between the concentration of Na+ and the conductivity of zinc electrolyte can be linearly expressed as follows:
 =327.896 25-1.788 45 ρ(Na+) (2)
=327.896 25-1.788 45 ρ(Na+) (2)
It is known that the diffusion of metallic ions is obstructed due to the increase in the resistance of electrolyte, and the quantity of the metal depositing on the cathode decreases, finally, the current efficiency for electrolysis declines simultaneously. Therefore, the Na+ ions in purified water influence the current efficiency, too, when the Na+ ions enter into the zinc electrolysis.
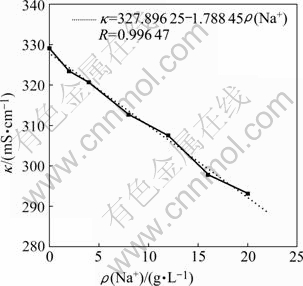
Fig.3 Effect of Na+ in purified water on conductivity of zinc electrolyte
4.2 Effect of Na+ in purified water on current efficiency of zinc electrolysis
The effect of Na+ in purified water on the current efficiency of zinc electrolysis process is listed in Table 3. It can be seen from Table 3 that the current efficiency decreases gradually from 88.28% to 87.41% as the concentration of Na+ increases from 0 to 8 g/L. While the concentration of Na+ is higher than 8 g/L, and the current efficiency is lower than 87%. This current efficiency can satisfy the main technical and economical index when the concentration of Na+ is less than 8 g/L (87% is a limited minimum value for the technical and economical index in Zhuzhou Smelter Group).
4.3 Effect of Ca2+ in purified water on conductivity of zinc electrolyte
The effect of Ca2+ on the conductivity of zinc electrolyte is listed in Table 4. The conductivity of zinc electrolyte increases with the increase of the H2SO4 concentration, but decreases with the decline of the Ca2+concentration. This also means that the Ca2+ ions in purified water affect the conductivity of zinc electrolyte.
Fig.4 shows the effect of Ca2+ in purified water on the conductivity of zinc electrolyte, which shows that the relationship between the conductivity of zinc electrolyte and the concentration of Ca2+ can be linearly fitted as the following equation:
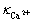 =327.437 14-0.315 94 ρ(Ca2+) (3)
=327.437 14-0.315 94 ρ(Ca2+) (3)
Eq.(3) obviously illustrates that the conductivity of zinc electrolyte decreases with the increase of the Ca2+ concentration. Comparing Eq.(2) with Eq.(3), it can be concluded that the effect of Ca2+ on the conductivity is much more remarkable than that of Na+.

Fig.4 Effect of Ca2+ in purified water on conductivity of zinc electrolyte
Table 3 Effect of Na+ on current efficiency of zinc electrolysis process

Table 4 Conductivity of zinc electrolyte at different concentrations of Ca2+ (mS/cm)

4.4 Effect of Ca2+ in purified water on current efficiency of zinc electrolysis
The effect of Ca2+ on the current efficiency of zinc electrolysis is listed in Table 5, which indicates that the current efficiency decreases gradually with the increase of the Ca2+ concentration in zinc electrolysis. The current efficiency decreases from 89.88% to 87.91% with the increase of the Ca2+concentration from 0 to 20 mg/L. The current efficiency is 86.86%, which is lower than the limited minimum of 87% in the Zn electrolysis process, when the Ca2+ concentration is 40 mg/L. Therefore, the current efficiency at the Ca2+concentration less than 20 mg/L can qualify the main technical and economical index in Zhuzhou Smelter Group.
Table 5 Effect of Ca2+ on current efficiency of zinc electrolysis process

4.5 Establishment of water quality index for purified water reused to zinc electrolysis
From the effects of Na+ and Ca2+ on conductivity of zinc electrolyte and current efficiency of zinc electrolysis, it can be concluded that the metal impurities in the purified water, even in low concentrations, make the conductivity and the current efficiency decrease. When the concentration of Na+ in zinc electrolyte is lower than 8 g/L or that of Ca2+ is below 20 mg/L, the current efficiency is higher than 87%, which satisfies the technical and economical index of Zhuzhou Smelter Group. Therefore, the concentrations of Na+ and Ca2+ in the purified water reused to the electrolysis process should be less than the limited values mentioned above. That is to say, the water quality index of the purified water should be controlled by the reused amount.
5 Conclusions
1) The conductivity of zinc electrolyte and the current efficiency of zinc electrolysis process decrease due to the presence of various metal impurities in purified water, even in low concentrations. The effect of Ca2+ on the conductivity of zinc electrolyte is much more remarkable than that of Na+.
2) ρ(Ca2+)≤8 g/L and ρ(Ca2+)≤20 mg/L are the quality indexes for the zinc electrolysis. The concentrations of Na+ and Ca2+ in the purified water reused to the process should be less than the limited values, i.e. the water quality index of the purified water should be controlled by the reused amount.
References
[1] RONG Qiang-tao, ZHANG Hong-guo, ZHOU Shao-qi. Water reuse and economical cost-benefit analysis [J]. Guangdong Chemical Industry, 2005(5): 36-38. (in Chinese)
[2] JIANG Pei-hua, LIU Zhen-hai, CHEN Ji-hua. Reusing technique of industry wastewater [J]. Shanghai Environment Science, 1995, 14(2): 33-37. (in Chinese)
[3] WANG Zhi-guo, LU An-huai. Analyzing about the advantages and disadvantages of mineral method in treating heavy metals pollution [J]. Environmental Science and Management, 2007, 32(5): 118-121. (in Chinese)
[4] VEEKEN A H M, RULKENS W H. Innovative developments in the selective removal and reuse of heavy metals from wastewaters [J]. Water Science and Technology, 2003, 47(10): 9-16.
[5] WANG Zhi-hui, YUAN Tian-jia, ZHOU Chun-li, TANG Chuang-lun. Comparison of the treatment methods of heavy metal-containing wastewater [J]. Journal of Dalian Nationalities University, 2007(3): 95. (in Chinese)
[6] HERRERO R, CORDERO B, LODEIRO P, REY-CASTRO C, de VICENTE S M E. Interactions of cadmium (II) and protons with dead biomass of marine algae Fucus sp [J]. Marine Chemistry, 2006, 99(1/4): 106-116.
[7] UTOMO H D, HUNTER K A. Adsorption of heavy metals by exhausted coffee grounds as a potential treatment method for waste waters [J]. E-Journal of Surface Science and Nanotechnology, 2006, 4: 504-506.
[8] LI Li-san. Investigation on the content of calcium and magnesium in zinc concentrate of Zhuzhou Smelter [J]. Science and Technology of Zhuzhou Smelter, 1995, 23: 35-38. (in Chinese)
[9] FU Yun-kang. Effect of Mg2+ on zinc electrolysis [J]. Nonferrous Metals (Extractive Metallurgy), 1998(1): 17-18. (in Chinese)
[10] WU Hui. Removing of Ca and Mg from zinc electrolysis system [J]. Nonferrous Metals (Extractive Metallurgy), 2006(4): 33-35. (in Chinese)
[11] ZHAO Tian-cong. Heavy metal metallurgy [M]. 2nd ed. Beijing: Metallurgical Industry Press, 1981: 57-70. (in Chinese)
[12] Editorial Board of Lead and Zinc Metallurgy. Lead and zinc metallurgy [M]. Beijing: Economic Science Press, 2003: 417-432. (in Chinese)
[13] CHEN Xin-feng. The effects of impurities on the current efficiency in zinc electro-winning [J]. Hunan Nonferrous Metals, 2006, 22(2): 24-26. (in Chinese)
[14] WANG Chang-zhen. Experimental approach to metallurgical physical chemistry [M]. Beijing: Metallurgical Industry Press, 2002: 333-347. (in Chinese)
[15] LI Jian, WANG Da-jian, PAN Xue-ping. One of the physical chemical property of copper electrolysis: conductivity of electrolyte [J]. Non-ferrous Mining and Metallurgy, 2003, 19(5): 30-33. (in Chinese)
Foundation item: Project(2007BAC25B01) supported by the National Key Project of Scientific and Technical Supporting Programs Funded by Ministry of Science and Technology of China during the 11th Five-Year Plan; Project(308019) supported by the Key Scientific and Technical Project, Ministry of Education of China; Project(08JJ3020) supported by Hunan Provincial Natural Science Foundation of China; Project (50830301) supported by the Key Project of the National Natural Science Foundation of China
Corresponding author: WANG Yun-yan; Tel: +86-731-8830875; Fax: +86-731-8710171; E-mail: wyy@mail.csu.edu.cn
DOI: 10.1016/S1003-6326(08)60300-3
(Edited by LI Xiang-qun)